Simulation and analysis of multi-stage centrifugal fractional extraction process of 4-nitrobenzene glycine enantiomers☆
Ping Wen ,Kewen Tang ,Jicheng Zhou ,*,Panliang Zhang ,*
1 Department of Chemical Engineering,Xiang Tan University,Xiangtan 411105,China
2 Department of Chemistry and Chemical Engineering,Hunan Institute of Science and Technology,Yueyang 414006,China
Keywords:Fractional extraction Multistage model Interfacial ligand exchange Chiral separation
ABSTRACT Based on the interfaciallig and exchange model and the law of conservation of mass,the multi-stage enantioselective liquid-liquid extraction model has been established to analyze and discuss on multi-stage centrifugal fractional extraction process of4-nitrobenzene glycine(PGL)enantiomers.The influence of phase ratio,extractant concentration,and PF6-concentration on the concentrations of enantiomers in the extract and raffinate was investigated by experiment and simulation.A good agreement between model and experiment was obtained.On this basis,the influence of many parameters such as location of stage,concentration levels,extractantexcess,and number of stages on the symmetric separation performance was simulated.The optimal location of feed stage is the middle of fractional extraction equipment.The feed flow must satisfy a restricted relationship on flow ratios and the liquid throughout of centrifugal device.For desired purity specification,the required flow ratios decrease with extractant concentration and increase with PF6-concentration.When the number of stages is 18 stages at extractant excess of 1.0 or 14 stages at extractant excess of 2.0,the ee eq(equal enantiomeric excess)can reach to 99%.
1.Introduction
Chirality is important in chemical research and chemical industry[1].When a molecule is chiral,the pharmacological activity and toxicity of the different enantiomers may be largely different[2].For example,(S),(S)-ethambutol is tuberculostatic,while(R),(R)-ethambutol causes blindness[3].The demand for enantiopure compounds is growing rapidly[4].The most common technique to obtain enantiopure compounds is the racemic resolution through crystallization[5].However,this technique is not always applicable because of excessive solid handling.With the development of separation technology,other techniques,such as simulated moving bed chromatography technique[6-10],enzymatic resolution or biocatalysis[11-14],liquid membrane technology[15-17],and enantioselective liquid-liquid extraction(ELLE)[18-35],have been developed.However,the high cost of SMB,the limitation of enzyme specificity and activity,and the limited transport rates in membrane technology,have impeded their industrial application,while ELLE has circumvented these imperfections and seems to be the most promising technology to separate the enantiomers,such as amino acid,amino alcohol,and aromatic acids.
Process intensification is a powerful concept to use smaller equipments that combine multiple operations in single highly integrated devices to replace large,energy intensive equipment or processes[36].The Centrifugal contactor separator(CCS)is a very attractive device to integrate dispersion of two immiscible liquids,chemical reaction,and subsequent phase separation of liquid-liquid systems,which is beneficial not only to energy and investments saving but also to improvement of the efficiency and the selectivity of the extraction processes.Therefore,the CCS is perfectly fit for PI on ELLE and bridges the gap between the typical ELLE laboratory experiments in batch to larger scale continuous operation.
In recent years,aromatic acid enantiomers have been successfully separated in CCS equipment in the operation conditions optimized by the multi-stage ELLE model on homogeneous reaction in our works[37,38].4-Nitrobenzene glycine(PGL)is an important pharmaceutical chemicals and can be used as D-serine transporter inhibitor for the treatment of nervous system disorders.Separation of PGL enantiomers is of much importance in its analysis and use.On this basis,the multistage ELLE model on interfacial reaction was established to investigate the multi-stage centrifugal fractional extraction process of 4-nitrobenzene glycine enantiomers in this paper.
2.Material and Methods
2.1.Material
Tetrakis(acetonitrile)copper(I)hexa fluorophosphate([(CH3CN)4Cu]PF6,purity>99%(w/w))was supplied by Hewei Chemical Co.,Ltd.(Guangzhou,China).(S)-(-)-2,2′-Bis(diphenylphosphino)-1,1′-binaphthalene((S)-BINAP)(purity > 99%(w/w))was obtained from Shengjia Chemical Co.,Ltd.(Hebei,China).4-nitrobenzene glycine(purity>98%(w/w))was purchased from HanHong Biochemical Co.,Ltd.(Jiangsu,China).Solvent for chromatography was of HPLC(Highperformance liquid chromatography)grade.Purified water was obtained by reverse osmosis followed by distillation.All other chemicals were of analytical-reagent grade and supplied by different suppliers.
2.2.Analytical Method
The quantification of the PGL enantiomers in raffinate was performed by HPLC with a UV detector(Merck,Hitachi,Japan)operated at the UV wavelength 260 nm.The column was Lichrospher C18(250×4.6 mm i.d.,5 μm of the packing materials)(Hanbon Science&Technology,China).The mobile phase was a mixture of 30 mmol·L-1sodium acetate aqueous solution and methanol(80:20,V/V)containing 2.0×10-3mol·L-1L-phenylalanine and 0.5×10-3mol·L-1copper sulfate at pH=4.8(pH was measured with a pH electrode and a pH meter(Orion,model 720A)).The flow rate was set at 1 ml·min-1,and the column temperature was set at 27°C.
2.3.Extraction Experimental Method
The aqueous phase was prepared by dissolving NaPF6in 0.1 mol·L-1NaH2PO4/H3PO4buffer solution,and racemic PGL was dissolved in 0.1 mol·L-1NaH2PO4/H3PO4buffer solution to prepare the feeding phase.The organic phase was prepared by dissolving BINAP-Cu in 1,2-dichloroethane.Extraction experiment was performed by starting the engines of all CCSs and starting the extract(organic phase)pump.After starting the extract pump,the CCSs were filled up in the order from Stage 10 to Stage 1.After the organic phase out flow from Stage 1,the wash streams(aqueous phase)were started.When the aqueous phase ran from Stage 10,the feed pump was started.As soon as the feed pump started running,samples of 0.5 ml were taken every 15 min from raffinate.The concentrations of ADand AL(represent the enantiomers of D-PGL and L-PGL respectively)in raffinate were analyzed using HPLC.
3.Multi-stage Equilibrium ELLE Model Formulations
A typical flow scheme of a cascade for the separation of AD,L(represent the 4-nitrobenzene glycine raceme)into the enantiomers ADand ALis depicted in Fig.1.The fractional extractor consists of two sections,a stripping section and a wash section.The feed is entered at the feed stage and is mixed instantaneously with the aqueous phase that exits the wash section.The stage relations in the cascade follow Fig.2,in which a single stage from the cascade is displayed.The definition of parameters is listed in Table 1.The model parameter values were determined by the thermodynamic studies on the single-stage extraction of 4-nitrobenzene glycine enantiomers in our previous work[39].

Fig.2.Single extraction stage.i=D,L.

Table 1 Parameters of the equilibrium model
The component balances for Ai(i=D,L)and PF6-for each of the stages(j=1…N)are defined as
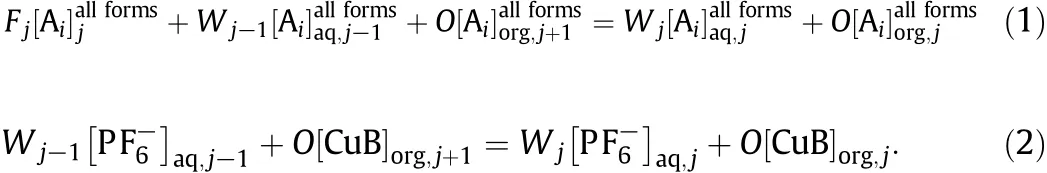
If j<f,then Fj=0 and Wj-1=Wj=W.If j=f,then Fj=F,Wj-1=W,and Wj=W+F.If j>f,then Fj=0 and Wj-1=Wj=W+F.
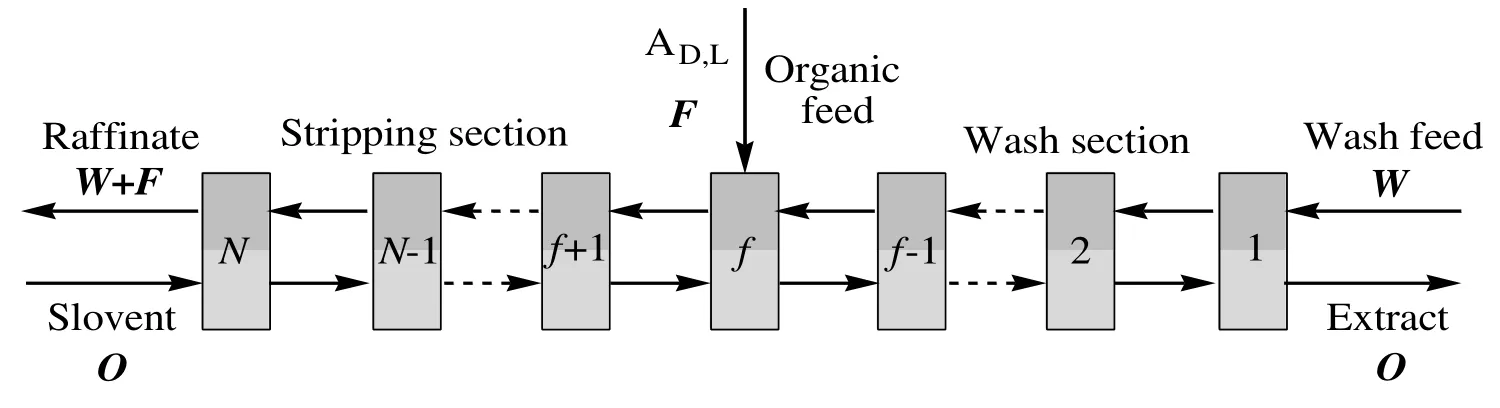
Fig.1.Flow scheme of the multi-stage centrifugal fractional extraction of PGL enantiomers.
The overall componentmass balances for CuB(represent CuPF6{(S)-BINAP})and the enantiomer Aiare defined as
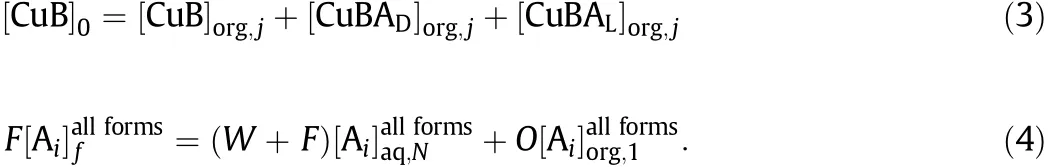
The model was solved using the software package Matlab.
4.Results and Discussion
The fractional extraction of 4-nitrobenzene glycine enantiomers is an extremely complicated process,which is mainly influenced by various process parameters,such as phase ratio(O/W),extractant concentration and PF6-concentration of aqueous phase.In this paragraph,a series of experiments and simulations are performed to reveal the objective laws of these process parameters on the concentrations of enantiomers in the extract and raffinate.
4.1.Influence of O/W Ratio
The influence of phase ratio on enantiomer concentration in the extract and raffinate was investigated at constant setting for concentration ofeach component,pHvalue,O/F ratio and feed flow(F).The results are given in Fig.3.At O/W ratio below 1.0,with the increase of organic phase flow,the L-PGL concentration in the raffinate decreases quickly and increases in the extract.But the D-PGL concentration in the raffinate and in the extractchange marginally.After O/W ratio of1.0,only a few LPGL exists in aqueous phase,and the L-PGL concentration in the extract decreases with the increase of organic phase flow.The reason for these may be that,L-PGL is preferentially distinguished by the extractant CuPF6{(S)-BINAP}.When O/W ratio is of 1.0 to 2.0,the extractant is present in excess with respectto L-PGL,most of D-PGL are distinguished and extracted into the organic phase,and itleads to D-PGL concentration in the raffinate decrease and increase quickly in the extract.When O/W ratio is higher than 2.0,nearly all the D-PGL and L-PGL enantiomers are extracted into organic phase,therefore,the concentrations of D-PGL and L-PGL in the extract is equal to zero.Thus,to get a better separation effect a suitable phase ratio is required.From Fig.3 O/W of 1.0 is applicable for separation ofPGL enantiomers.The mean relative error is 1.72%of

Fig.3.Influence of O/W ratio on the equilibrium concentrations of PGL enantiomers in the raffinate and extract.(Lines:model prediction.Symbols:experimental data.Conditions:O/F=2.0,[AD,L]=2.0×10-3 mol·L-1,[PF6-]=2.0×10-3 mol·L-1,[CuB]=1.0×10-3 mol·L-1,pH=7.0,f=6 and N=10).

4.2.Influence of Extractant Concentration
In the stripping section,the ionic PGL enantiomers in the aqueous phase react with CuPF6{(S)-BINAP}on two-phase boundary and the complexes transfer into the organic stream.To gain insights on the influence of extractant concentration on enantiomer concentration in the extract and raffinate,a series of experiments and simulations were carried out.As shown in Fig.4,the simulation results have a good agreement with the experimental data.Both enantiomers are extracted with a preference for L-PGL enantiomer,enantiomer concentration in the raffinate decreases with extractant concentration.At extractant concentration higher than 0.001 mol·L-1,D-PGL enantiomers begin to be largely extracted into the organic phase.After CuPF6{(S)-BINAP}concentration is higher than 0.0025 mol·L-1,all enantiomers in the aqueous phase are occupied by CuPF6{(S)-BINAP}and are extracted into the organic phase.Thus,enantiomer concentration in the raffinate drops to be zero and as a result both D-PGL and L-PGL enantiomer concentrations in the extract change to be constant.The mean relative error is 1.22%of[AD],0.73%of[AL],1.62%of[AD],and 1.14%of[AL],respectively.
4.3.Influence of PF6-Concentration
In the wash section,the molecular and ionic PGL enantiomers in the organic stream are redistributed over the aqueous and organic phase,a small percentage of them are washed back to the aqueous stream and then flow into the stripping section.Part of complex enantiomers(CuBADand CuBAL)from the stripping section are compelled to be decomposed at the interface with a precedence for CuBADbecause of the presence of PF6-in the aqueous stream,which will lead to a significant influence on the equilibrium concentrations of PGL enantiomers in the raffinate and extract.It can be seen in Fig.5 that the concentrations of PGL enantiomers in the extract decrease and the concentrations of PGL enantiomers in the raffinate increase at increasing PF6-concentration.Furthermore,in the PF6-concentration region between 0.0 and 0.002 mol·L-1,the influence of increasing PF6-concentration on DPGL enantiomer concentrations is far more significant than on L-PGL enantiomer concentrations.However,after PF6-concentration more than 0.002 mol·L-1,most CuBALcomplexes begin to be decomposed and it leads to the decrease of L-PGL enantiomer concentration in the extract and increase in the raffinate,whereas,D-PGL enantiomer concentration just changes slightly,which will largely cut down the separation performance.Thus,for a better separation performance,a properconcentration in wash feed is needed.The mean relative error is 0.64%of[AD],1.03%of[AL],2.14%of[AD],and 2.21%of[AL],respectively.
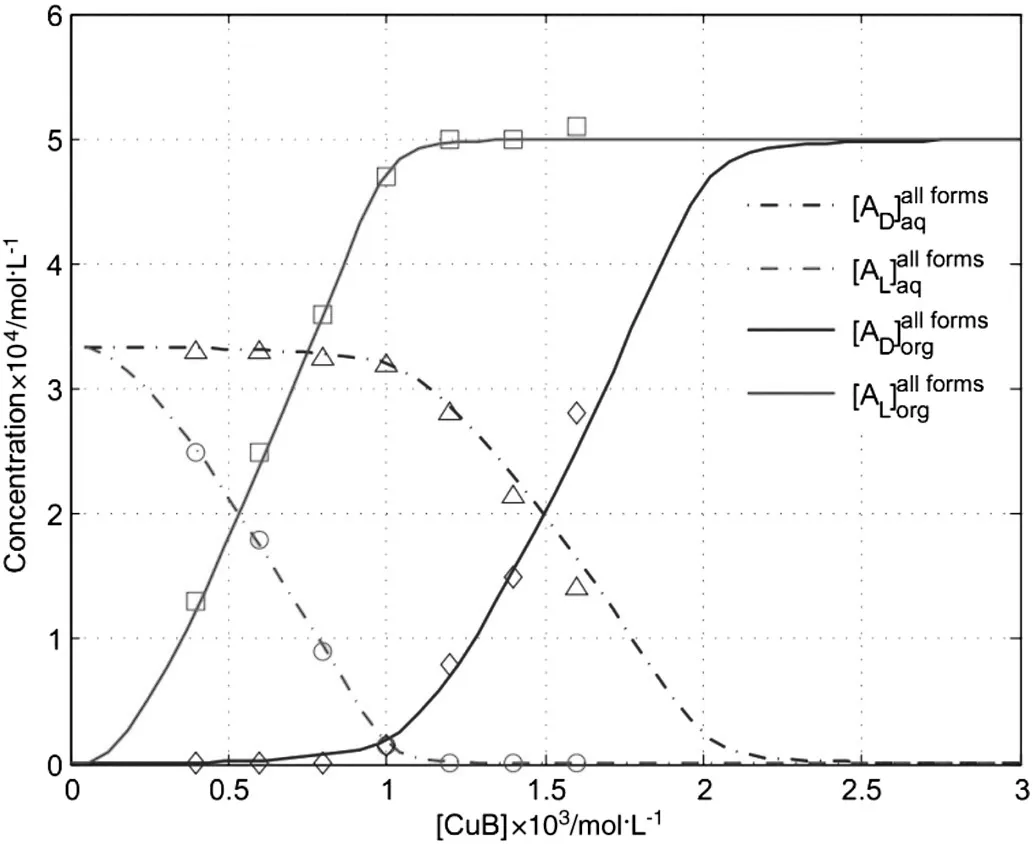
Fig.4.Influence of extractant concentration on the equilibrium concentrations of PGL enantiomers in the raffinate and extract.(Lines:model prediction.Symbols:experimental data.Conditions:O/F=2.0,O/W=1.0,[AD,L]=2.0×10-3 mol·L-1,[PF6-]=2.0×10-3 mol·L-1,pH=7.0,f=6 and N=10).
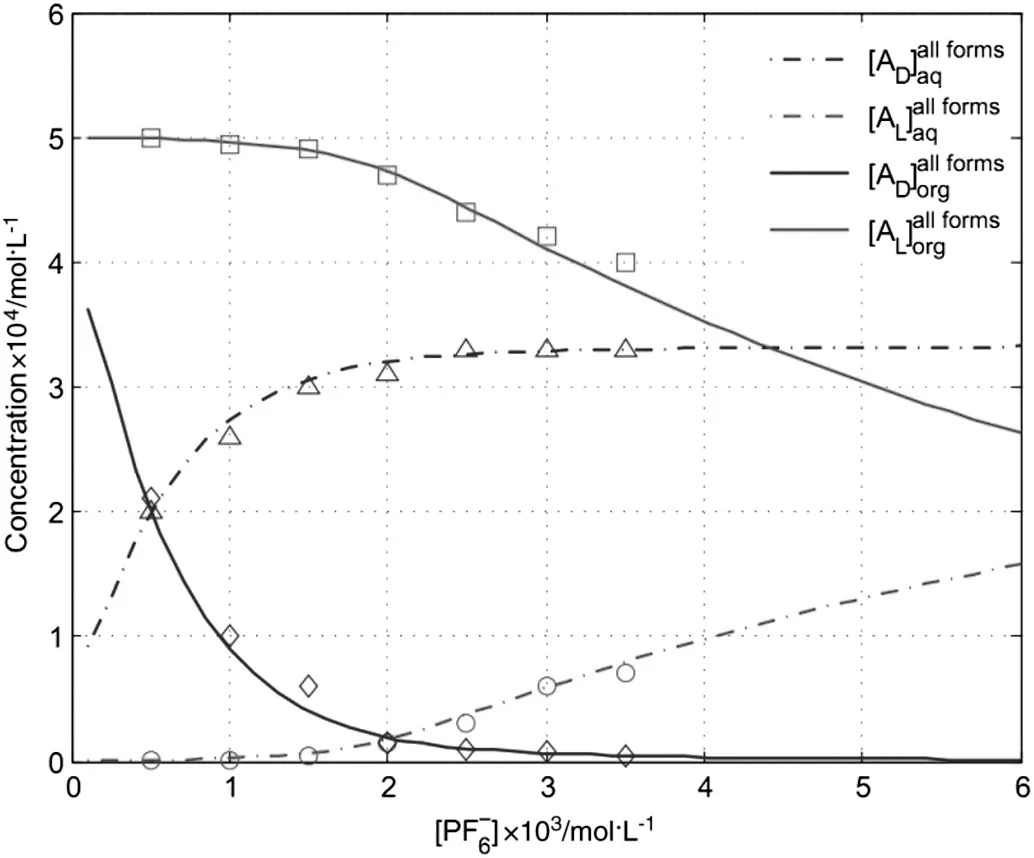
Fig.5.Influence of PF6-concentration on the equilibrium concentrations of PGL enantiomers in the raffinate and extract.(Lines:model prediction.Symbols:experimental data.Conditions:O/F=2.0,O/W=1.0,[A D,L]=2.0×10-3 mol·L-1,[CuB]=1.0×10-3 mol·L-1,pH=7.0,f=6 and N=10).
4.4.Concentration Profile

Fig.6.Concentration profile in Stage 10 cascade at steady state.(O/F=2.0,O/W=1.0,[A D,L]=2.0×10-3 mol·L-1,[PF6-]=2.0×10-3 mol·L-1,[CuB]=1.0×10-3 mol·L-1,pH=7.0,f=6 and N=10).
For the in-depth knowledge on the multi-stage centrifugal fractional extraction process of PGL enantiomers,concentration profile of each component at steady state is given in Fig.6 through simulation.Note that the flow direction for the aqueous stream is from Stages 1 to 10,and for the organic stream counter-currently from Stages 10 to 1.Fig.6 shows that the concentrations of CuPF6{(S)-BINAP}and PF6-in the cascade are high at both ends and low in the middle of the cascade,but in the flow direction view,their tendencies are opposite.The concentrations of L-PGL enantiomers in the aqueous phase and organic phase are low at both ends and high in the middle.For D-PGL enantiomers,the concentrations are low between Stages 1 and 5 and high between Stages 6 and 10.The reason is that CuPF6{(S)-BINAP}has a preference for L-PGL enantiomers in the stripping section and PF6-has a preference for CuBADcomplex.In the experiments,the aqueous concentration of enantiomers in Stage 10,measured after 5 h at steady-state,HPLC chromatogram is depicted in Fig.7(b)and the original sample chromatogram is shown as Fig.7(a).
It can be concluded from the above that flow ratios,extractant concentration,and PF6-concentration have great effects on the extraction process.The concentrations of enantiomers in the extract and raffinate change regularly in the process of different extraction conditions.A high correlation between model and experiment was shown in the above studies.The model is satisfied for the multi-stage centrifugal fractional extraction process simulation and optimization of4-nitrobenzene glycine enantiomers.It is therefore the aim of below chapter to study with the model which different extraction conditions generate a specified yield and product optical purity in each stream,and the specifications of them are exchanged for ‘equal yield(yieldeq)’and ‘equal enantiomeric excess(eeeq)’.We call this operation as ‘symmetric separation’.The enantiomeric excess and yield are defined in equations as follows.
Enantiomer ALpredominates in extract stream,its ee is given in Eq.(5).Note that ‘AL’and ‘AD’encompass L-PGL and D-PGL in all forms(AL,AL-,CuBAL,etc.)in this equation.

Enantiomer ADpredominates in raffinate stream,its ee is defined by Eq.(6):

The yield of the enantiomer ALin extract stream is given in Eq.(7).Similarly,the yield of ADin raffinate stream can be defined as:

4.5.Symmetric Separation
4.5.1.Location of Feed Stage
The location of feed stage is a degree of freedom in the multi-stage centrifugal fractional extraction processing equipment.The amount of stages in the stripping section and the wash section will be altered with the location of feed stage,and it will lead to different performances of symmetric separation.It can be seen in Fig.8 that the feed stage located in the middle of the cascade equipment,a better symmetric separation performance can be obtained than at other locations.In this paper,the single L-PGL and D-PGL enantiomers have different medicinal value,at the same time the concentration of L-PGL and D-PGL enantiomers in the raw material is equal.Therefore,the feed stage located exactly in the middle is the most efficient solution for symmetrical separation of PGL enantiomers.

Fig.7.HPLC chromatogram of PGL enantiomers in the aqueous phase before(a)and after separation(b).
4.5.2.Concentration Level
In the cascade equipment of three solution inlets,concentration level encompasses enantiomer concentration of feed inlet([Ai]),extractant concentration of organic phase inlet([CuB])andconcentration of aqueous phase inlet([]).In this chapter,the performance for symmetrical separation of PGL enantiomers will be simulated under different concentration levels.
4.5.2.1.Enantiomer Concentration.PGL enantiomer concentration and feed flow get together to determine the production rate.There is no doubt that the bigger the values of enantiomer concentration and feed flow are,the better will be in the industrial production.The enantiomer concentration is limited by the solubility of PGL enantiomers in the feed stream,which can be determined experimentally.Therefore no further simulations were carried out on the enantiomer concentration level.

Fig.8.Influence of the location of feed stage on the performance for symmetrical separation of PGL enantiomers.(O/F=2.0,O/W=1.0,[AD,L]=2.0×10-3 mol·L-1,[PF6-]=2.0×10-3 mol·L-1,[CuB]=1.0×10-3 mol·L-1,pH=7.0,N=10).
In addition,the feed flow is determined by the flow ratios(O/W=a,O/F=b)and the minimum and maximum liquid throughput(Vminand Vmax,L·min-1)of the real centrifugal device.The three inlet flows must satisfy the restriction on ‘Vmin≤ W+O’and ‘W+O+F ≤ Vmax’.Then the value of the feed flow can be deduced to:

Furthermore,for a fixed equal enantiomeric excess of products in the process design(e.g.0.90 eeeq)the flow ratios are bound up with the selected concentration levels of extractant and PF6-.Thus,it is necessary to investigate the influence of extractant and PF6-concentration level on the performance for symmetrical separation of PGL enantiomers.
4.5.2.2.Extractant Concentration.Extractant concentration is an important degree of freedom in the multi-stage extraction system.The eeeqis simulated as a function of flow ratios at different extractant concentration settings.It can be seen in Fig.9(a),(b),(c)that eeeqdecreases to the minimum with O/W and increases to the maximum with O/F at a fixed extractant concentration settings.Furthermore,by comparative analysis,it can be found that extractant concentration level has a significant influence on the flow ratios for a desired purity specification(e.g.0.90 eeeq).With the increasing of extractant concentration,the required lf ow ratios decrease.
concentration as a degree of freedom plays an important role in improving the washing effect of washing section.The eeeqis simulated as a function of flow ratios at differentconcentration settings in Figs.10(a),(b)and 9(b).The eeeqgets to the minimum with O/W and drops to the maximum with O/F at a fixedconcentration settings.For a desired purity specification(e.g.0.90 eeeq),with the increasing ofconcentration,the required flow ratios increase.
It can be seen from the above investigations that the mutual influences of concentration levels and flow ratios conclude that there are many combinations of them to obtain desired production purity.Thus,without a detailed economical analysis,it cannot be decided which way is most efficient.However,the simulation can provide a scientific method for comparative analysis in the real industrial production.

Fig.9.Influence of extractant concentration on the symmetrical separation of PGL enantiomers.((a)[CuB]=0.0001 mol·L-1,(b)[CuB]=0.001 mol·L-1,(c)[CuB]=0.001 mol·L-1,[PGL]=2.0×10-3 mol·L-1,[PF6-]=2.0×10-3 mol·L-1,pH=7.0,f=6 and N=10).
4.5.3.Number of Stages
In industrial production,it is necessary to operate not only at higher enantiomer concentration but also at minimal extractant concentration and number of stages to obtain higher purity and higher yield and to cut down the production cost.The concentrations of extractant and enantiomers are expressed as ‘extractant excess’(Eq.(9)).
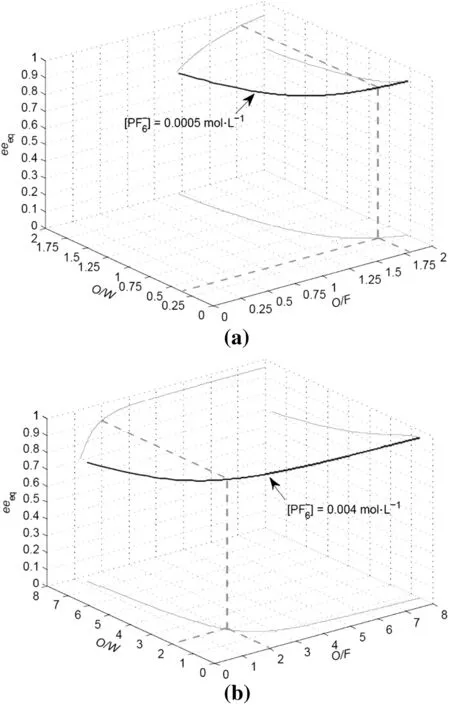
Fig.10.Influence ofPF6-concentration on the symmetrical separation of PGL enantiomers.((a)[PF6-]=0.0005 mol·L-1,(b)[PF6-]=0.004 mol·L-1,[PGL]=2.0×10-3 mol·L-1,[CuB]=1.0×10-3 mol·L-1,pH=7.0,f=6 and N=10).
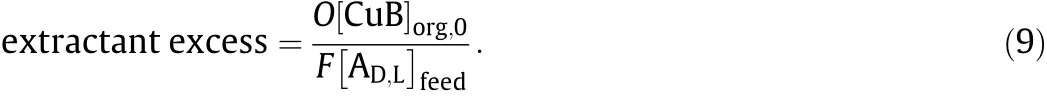
The eeeqis simulated as a function of number of stages at different extractant excess via changing extractant.It can be seen in Fig.11 that the eeeqincreases with the increasing of number of stages and then finally reaches a plateau.It was also observed in the simulation results that at lower extractant excess,no matter how many stages increase to,a full separation can never be obtained in the multi-stage centrifugal fractional extraction process.In these conditions,the extractant will become fully loaded,and increasing the number of stages will not increase the optical purity any further.Therefore,in order to make good use of the centrifugal contactors,a reasonable extractant excess is necessary.As shown in Fig.11,when the number of stages is 18 stages at extractant excess of 1.0 or 14 stages at extractant excess of 2.0,the eeeqcan reach to 99%.The settings for the two cases are listed in Table 2.
5.Conclusions

Fig.11.Influence of number of stages on the symmetrical separation of PGL enantiomers at different extractant excess by increased[CuB].(O/F=2.0,[AD,L]=2.0×10-3 mol·L-1,[]=2.0×10-3 mol·L-1,pH=7.0,f=2/N+1).

Table 2 Settings for symmetrical separations with extractant excess=1.0 or 2.0,[AR,S]=2.0×10-3 mol·L-1,pH=7.0
The multi-stage ELLE(enantioselective liquid-liquid extraction)model has been successfully developed to simulate and analysis of multi-stage centrifugal fractional extraction process of 4-nitrobenzene glycine enantiomers.Influence of process parameters such as phase ratio,extractant concentration,andconcentration on the concentrations of enantiomers in the extract and raffinate is investigated by a series of experiments and simulations.Furthermore,by simulation and analysis on the symmetric separation performance,many important conclusions have been made to instruct process design.When the number of stages is 18 stages atextractantexcess of1.0 or 14 stages atextractant excess of 2.0,the eeeq(equal enantiomeric excess)can reach to 99%.
Nomenclature
CuB CuPF6{(S)-BINAP}
ee enantiomeric excess
K complexation constants
Kaamino acid dissociation constant,mol·L-1
PGL 4-nitrobenzene glycine
Piphysical partition coefficient for ionic PGL
P0physical partition coefficient for molecular PGL
Vmaxmaximum liquid throughput
Vminminimum liquid throughput
Subscripts
aq aqueous phase
eq equal value
i index for D,L
j stage index
org organic phase
0 initial value
 Chinese Journal of Chemical Engineering2015年11期
Chinese Journal of Chemical Engineering2015年11期
- Chinese Journal of Chemical Engineering的其它文章
- N-methyl-2-(2-nitrobenzylidene)hydrazine carbothioamide—A new corrosion inhibitor for mild steel in 1 mol·L-1 hydrochloric acid
- A dual-scale turbulence model for gas-liquid bubbly flows☆
- Gas-liquid hydrodynamics in a vessel stirred by dual dislocated-blade Rushton impellers☆
- Convective mass transfer enhancement in a membrane channel by delta winglets and their comparison with rectangular winglets☆
- Cobalt-free gadolinium-doped perovskite Gd x Ba1-x FeO3-δas high-performance materials for oxygen separation☆
- Synthesis and adsorption property of zeolite FAU/LTA from lithium slag with utilization of mother liquid☆
