Therapeutic Effect of Mesenchymal Stem Cells Overexpressing Interleukin-10 on Inflammatory Bowel Disease
LU Xin, ZHAO Tao-Ran*, CHENG Hai-Qin, LI Xue-Wei, LIANG Ting, XU Xin-Rui,HU Zi-Xuan, MA Li-Hui, ZHANG Chun-Hong, CHEN Chun-Ting, WANG Lei, XIE Jun,LIU Zhi-Zhen, ZHAO Hong*
(1)Shanxi Key Laboratory of Birth Defect and Cell Regeneration, Department of Biochemistry and Molecular Biology, Shanxi Medical University,Taiyuan 030001, China; 2)Department of Rheumatology and Immunology, The First Affiliated Hospital of Hainan Medical College, Haikou 570100, China)
Abstract Inflammatory bowel disease (IBD) is an intractable inflammatory autoimmune disease characterized by T-cell infiltration to the colon. Mesenchymal stem cells (MSCs), owing to their immunosuppressive capabilities, have the potential to rescue IBD. But the therapeutic effectiveness of MSCs is sometime thwarted by their variable immunomodulatory ability in vivo. In the present study, we produced engineered MSCs that secrete interleukin10 (IL-10) and evaluated their therapeutic potential in IBD mouse model. The MSCs maintained the phenotype and cell proliferation rate after overexpression of IL-10 by lentivirus (LV) infection. Immune cells and MSCs in vitro co-culture systems exhibited that relative to unmodified MSCs, immune cells co-cultured with IL-10-overexpressing MSCs had significantly lower numbers of T helper 1 cells (Th1) and T helper 17 cells (Th17) (P<0.05), the content of TNF-α in the supernatant of macrophage cells co-cultured with MSCs overexpressing IL-10 was significantly decreased (P<0.0001). Tail vein injection of the IL-10 overexpressing MSCs achieved a better therapeutic effect in the dextran sodium sulfate (DSS) induced colitis mouse model than that of the unmodified MSCs, as indicated by colon length, disease activity index (DAI) and colonic cytokines expression. The experimental results were statistically different (P>0.05). Overall, LV induced MSCs overexpressing IL-10 might be a promising alternative therapeutic option for the treatment of IBD.
Key words inflammatory bowel disease (IBD); mesenchymal stem cells (MSCs); interleukin-10 (IL-10); cytokines secretion; T helper cells differentiation
Inflammatory bowel disease (IBD) is a set of multifactorial autoimmune disorders defined by persistent nonspecific inflammation in the intestines and includes two primary clinical subgroups, Crohn’s disease (CD) and ulcerative colitis (UC)[1]. It is usually a complex disorder with multiple factors such as genetic mutations, gut microbiome dysbiosis, psychological distress, autonomic dysfunction and hyperactivation of immunity[2].
Among those factors, recent literature showed that abnormal innate and adaptive immunity is the most essential pathogenesis in the development of UC and CD[3]. T helper 1 cells (Th1), showing increased interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α) expression, have been implicated in murine models and in patients with IBD[4]. T helper 17 cells (Th17) preferentially produce interleukin-17 (IL-17) and IL-22, leading to recruitment and activation of granulocytes and macrophages which are tissue-damaging[5]. IBD pathogenesis is also related to M1 macrophages that produce reactive oxygen and nitrogen species, as well as pro-inflammatory cytokines (TNF-α, IL-6). Uncontrolled M1 macrophage activity, on the one hand, might cause tissue damage and predispose the host to neoplastic tumors[6]. Macrophage-produced mediators, on the other hand, enhance the activation and differentiation of Th1 and Th17 cells[7].
Due to their anti-inflammation, multi-differentiation and tissue repair activities, mesenchymal stem cells (MSCs), particularly bone marrow-derived mesenchymal stem cells (BM-MSCs), are a good candidate for the therapy of IBD[8, 9]. MSCs possess immune suppressive effects by the inhibition of T cells and monocytes function via cell-to-cell contact or paracrine activity[10]. Immunomodulatory substances such as IL-10, transforming growth factor-β (TGF-β), and prostaglandin E2 (PGE2) are secreted by MSCs and play an important role in their therapeutic potential[9]. However, in the complicated pathogenic contexts, MSCs immunomodulatory function is very variable[11, 12]. Therefore, developing strategies that can improve and optimize the paracrine actions of MSCs then strengthen the effectiveness of MSC-based treatment is highly desired.
IL-10 is an anti-inflammatory cytokine which mainly regulates inflammatory reaction of immune cells. Specifically, IL-10 suppresses macrophage activation, T cell proliferation and pro-inflammatory cytokine (such as IFN-γ, IL-2 and TNF-α) secretion from Th1 cells[13, 14]. For the first time, a GWAS study was carried out to reveal the involvement between IL-10 and UC[15]. Polymorphisms in the IL-10 promoter have been connected to IBD and have been proven to modify IL-10 serum levels in studies[16-18]. Clinical studies have shown that IL-10 supplementation therapy is beneficial to the IBD patients, particularly those with low IL-10 serum levels or high disease activity[19, 20]. However, solely IL-10 is insufficient to inhibit all of the pro-inflammatory mediators involved in IBD. Furthermore, the dose used in past IL-10 treatments cannot results in a high enough IL-10 concentration in the intestine which can elicit a response[21].
In this study, the potential enhancement of therapeutic efficiency of IL-10-overexpressing MSCs in a dextran sodium sulfate (DSS)-induced murine colitis model was investigated. The IL-10 transduction in BM-MSCs was achieved by lentivirus (LV) infection. Compared to the blank control group, the IL-10-overexpressing MSCs significantly improved the therapeutic effect. In addition, the IL-10-overexpressing MSCs significantly down-regulated the proportion of Th1 and Th17 cells in lymphocytes and the secretion of TNF-α from macrophages. Our results may provide helpful information for guiding the development of stem-cell-based therapies in treating IBD.
1 Materials and Methods
1.1 Reagents and animals
Dextran sodium sulfate (DSS, molecular weight, 36-50 kD) was obtained from MP Biomedicals (USA). Fluorescence-conjugated mAbs against rat CD11b, CD45, CD90 and CD29 (#201807, #202211, #202529, #17-0291-80) and against mouse CD4, CD25, IFN-γ and IL-17A antibody (#100431, #101903, #505841, #506915) were purchased from BioLegend (USA). Female C57BL/6 mice (6-8 weeks old) and male Sprague-Dawley (SD) rat (4 weeks old) were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd (China) and maintained in a specific pathogen-free (SPF) environment. All animal experiments were approved by and performed in compliance with the guidelines of the Ethics Committee of Animal Care and Use Committee of Shanxi Medical University.
1.2 Isolation and culture of MSCs
MSCs were isolated from the hindlimbs of SD rats under sterile circumstances and grown with complete culture medium [Modified Eagle Medium: F-12 (DMEM/F-12; Life Technologies, USA) containing 10% fetal bovine serum (FBS; Life Technologies), and an antibiotics mixture (100 U/mL penicillin and 100 mg/mL streptomycin (Beyotime Biotechnology, China)] at 37 ℃ in a humidified atmosphere with 5% CO2. The MSCs were passaged at a ratio of 1∶2 in triple flasks when cells reached 80%-90% confluence. All experiments used MSCs passaged no more than six times.
1.3 Construction and phenotypic determination of modified MSCs
LV vectors encoding mouse IL-10 (GenBank: NM_010548.2) and EGFP (IL-10-EGFP-LV) were generated by Cyagen (China). As a control, an empty EGFP expressing vector (EGFP-LV) was employed. MSCs at passage 1 were seeded into a 6-well plate for 24 h before being infected with the virus. When the cells were 30-40% confluent, the appropriate LV was injected to each well at a multiplicity of infection (MOI) of 10∶1 and incubated with the cells overnight. After being infected for 24 h, the medium was changed with fresh complete culture medium, and the MSCs were cultured for extra 48 h. Puromycin (2 μg/mL) was added to the infected MSCs and incubated for additional 48 h to remove uninfected MSCs. MSCs with both the IL-10 and EGFP genes (IL-10-LV-MSCs) as well as MSCs with only the EGFP gene (LV-MSCs) were then obtained. Infected MSCs (P5) were employed in subsequent experiments. Fluorescence microscopy and flow cytometry analyses were used to measure EGFP gene expression. Quantitative real-time PCR (qRT-PCR) and enzyme-linked immunosorbent assays (ELISA) were used to quantify IL-10 gene expression. The Cell Counting Kit-8 (CCK-8) (Absin, China) assay was used to create a growth curve. A microplate spectrometer was used to detect the absorbance at the wave length of 450 nm (SpectraMax190, Molecular Devices, USA). Cell surface markers were detected by flow cytometry.
1.4 MSCs-Lymphocyte co-culture reactions in vitro
Cell co-culture studies were used to examine the immunomodulatory effect of MSCs. As effector cells, lymphocytes were extracted from the spleens of mice with DSS-induced colitis, as follows. Spleens were dissected immediately after euthanasia and homogenized by grinding and passed through a 20G needle 3-4 times to obtain single-cell suspension. Cells were washed once with phosphate-buffered saline without calcium or magnesium (PBS) and red blood cells were lysed using RBC lysis buffer (Sigma-Aldrich, R7757). After neutralization with the complete medium [RPMI-1640 medium (Gibco, 22400089) supplemented with 10% heat-inactivated FBS and antibiotics mixture (100 U/mL penicillin and 100 mg/mL streptomycin (Beyotime Biotechnology, China)], cells were transferred to a culture dish and incubated at 37 ℃ with 5% CO2for 24 h to remove the adherent cells. The suspension cells were harvested as raw lymphocytes.
For MSCs-Lymphocyte co-culture, 1×106lymphocytes activated by 5 μg/mL of PHA (Gibco, USA) were added into 6-well plats pre-seeded 1×105per well MSCs one night before. After co-culture for 72 h, the cells were harvested for flow cytometry analysis.
1.5 MSCs-Macrophages co-culture reaction in vitro
The immunomodulatory function of MSCs was tested using the RAW 264.7 gamma NO (-) (ATCC, CRL-2278) as a macrophage model. Transwell co-culture system (LABSELECT, China) was assembled by using 1×105macrophages and 1×105MSCs (at the ratio of 1∶1). Macrophages were seeded in 6-well plates. Transwell inserts were placed in additional 6-well plate, and then the MSCs were seeded in the transwell inserts. To develop the MSCs-Macrophages co-culture systems, Transwell inserts containing MSCs were transferred into the equivalent 6-well plates containing macrophages after cells had adhered for ca. 12 h. To stimulate TNF-α release, LPS (10 ng/mL) was introduced to the co-culture system. The culture supernatants were obtained after the cells had been co-cultured for 48 h, then the culture supernatants were harvested to analysis the TNF-α concentration by ELISA.
1.6 qRT-PCR
Excised colons and collected cells that were culturedinvitrowere homogenized to extract total RNA using the TRIzol (Solarbio Life Science, China). To acquire cDNA samples, reverse transcription was conducted using PrimeScript reverse transcriptase (TaKaRa, Japan). The qRT-PCR was carried out on an ABI Prism 7300 using the TB Green Premix Ex Taq kit (TaKaRa, Japan). The 2ΔCt method was used to compare relative amounts of target mRNA to GAPDH. All primers sequences were as follows:

Table 1 The sequences of PCR and quantitative real-time PCR primers
1.7 ELISAs
Concentrations of IL-10 in the culture supernatants from transduced MSCs and concentrations of TNF-α in the culture supernatants from the MSCs-macrophages co-culture system were measured using ELISA kits (RK00016, RK00027 respectively, Abclonal, China) according to the manufacturer’s instructions.
1.8 Flow cytometry
MSCs that had received various treatments were collected, then flow cytometry analysis was performed. A FITC cannel was used to detect EGFP expression. To detect the surface marker expression, the MSCs were stained with fluorescence-conjugated mAbs against rat CD11b, CD45, CD90 and CD29 respectively. A flow cytometer was then used to study the cells (BD FACSCelesta, BD Biosciences, USA).
ForinvitroT cell differentiation analysis, the cell surface was stained with anti-mouse CD4 and CD25 antibodies and the cells were fixed with the 1 × Fixation (#420801, BioLegend). The fixed cells were washed with 1×Staining Permeabilization Wash Buffer (10×) (#421002, BioLegend), followed by intracellular staining with anti-mouse IFN-γ and IL-17A antibodies. Then the cells were examined with a flow cytometer.
1.9 Induction of experimental colitis models and administration of MSCs
C57BL/6 mice were randomly split into five groups, including DSS+PBS, DSS+MSCs, DSS+LV-MSCs, DSS+IL-10-LV-MSCs and control groups (n=6 in each group). From day 0, the mice in DSS+PBS, DSS+MSCs, DSS+LV-MSCs and DSS+IL-10-LV-MSCs groups were given with 4 w/v% DSS in drinking water, while the control group received regular water. In DSS+MSCs, DSS+LV-MSCs and DSS+IL-10-LV-MSCs groups, the distinct populations of treated MSCs (1.0×106cells per mouse[22], pre-experiments shown that this dose can exert a stable therapeutic effect while avoiding cell aggregation and vascular embolism) were delivered by tail vein injection on day 3, 5 and 7 for a therapeutic research. Over the course of 11 days, the body weight, disease activity index (DAI) and survival rate were examined every day. The DAI was used to assess body weight loss, stool consistency, and rectal bleeding based on a previously published grading methodology[23]. On day 7, the colon was excised for macroscopic and histological investigation. Hematoxylin and eosin (H&E) staining and cytokine (IL-6, IL-1β and TNF-α) analyse using qRT-PCR were performed on colon tissues near rectum (about 3-5 cm).
1.10 Histopathology
The colons were embedded in paraffin after being treated with 4% paraformaldehyde. H&E was used to stain four-micrometer-thick slices. Under a light microscope (Olympus AX70, Japan), the degree of epithelial erosion, ulceration, vascular density and leukocyte infiltration were measured in the colon.
1.11 Statistical analysis
Data were presented as the mean ± standard error (S.D.). GraphPad Prism (v.7.00, La Jolla, CA, USA) was used to perform statistical analysis. One-way ANOVA with a post hoc Bonferroni correction was used to analyze differences across several groups.T-test was used to assess differences between two groups. A value ofP<0.05 was considered significant.
2 Results
2.1 Determination of IL-10 expression level in engineered MSCs
MSCs infected with IL-10-EGFP-LV (IL-10-LV-MSCs) or EGFP-LV (LV-MSCs) both exhibited EGFP expressions at 48 h after the infection (Fig.1A). By puromycin selection, IL-10-LV-MSCs and LV-MSCs reached the purity to 78% and 87%, respectively (Fig.1B). To verify the correct expression of IL-10, qPCR analysis and ELISA were conducted. Compared with the MSCs group, the expression of IL-10 gene mRNA in IL-10-LV-MSCs was significantly increased, and the difference was statistically significant (P<0.001). qPCR analysis demonstrated that IL-10-EGFP-LV infection effetely inducedIL-10 gene expression in MSCs (Fig.1C). ELISA analysis indicated IL-10 expression exist in both wild type MSCs and LV-MSCs, but the content of IL-10 in the culture supernatant of MSCs cells after IL-10-EGFP-LV infection exceeded 300 pg/mL (Fig.1D).
2.2 Phenotypic stability of MSCs after LV infection
Results using microscopy indicated that LV infection showed no effect on the morphology of MSCs (Fig.1A). In addition, the results of flow cytometry analysis indicated that the expression levels of CD11b (0.9%, 1.0%) and CD45 (0.5%, 0.4%) (leukocyte markers) were not elevated in the LV-MSCs group and the IL-10-LV-MSCs group. Therefore, both IL-10-EGFP-LV and EGFP-LV infection did not induce MSCs differentiate into leukocytes (Fig.2A and 2B). Furthermore, MSCs markers, CD29 (96.9%, 91.4%) and CD90 (88.3%, 90.1%), were not significantly declined among MSCs that received different treatments (Fig.2C and 2D). The results of the CCK-8 assay showed no significant change in the number of MSCs after LV infection (P>0.05) (Fig.2C).
2.3 Immunomodulatory potential of IL-10 overexpressing MSCs in vitro
Engineered MScs were co-cultured with mice spleen lymphocytes or macrophages to determine their immunomodulatory potential. Lymphocytes were cultivated with or without MSCs for 3 days to investigate the effects of different populations of treated MSCs on Th cell differentiation. According to flow cytometry analyses (Fig.3A), IFN-γ+Th1 cells researched 34.2% of total Th cells without MSCs co-culture. The addition of the LV-MScs reduced the percentages of IFN-γ+Th1 cells to 26.5%. The lL-10-LV-MSCs reduced this percentages to 13.7%, indicating more effective inhibition on Th1 cells. In addition, the LV-MSCs lead an elevation on the percentages of IL-17A+Th17 cells (from 1.59% to 13.15%) IL-10-LV-MSCs, on the other hand, had no impact onTh17 cells (Fig.3B). Moreover, both kinds of MSCs could slightly but not significantly induce Treg cells differentiation (Fig.3C).
A large proportion of pro-inflammatory TNF-α was secreted by macrophages. To examine the influences of the differently modified MSCs on the secretion of TNF-α from macrophages, macrophages were seeded in a 6 well plates and co-cultured with MSCs by a Transwell insert (Fig.3D). LPS stimulation induced a very high TNF-α level, but the addition of the IL-10-LV-MSCs significantly reversed the TNF-α level rising (P<0.001) (Fig.3E). Unexpectedly, the MSCs without IL-10 overexpression even improved TNF-α secretion (P<0.05).
2.4 IL-10 overexpressing MSCs protected against DSS-induced colitis
Mice were given a 4% DSS solution experienced rapid-onset colitis, which included weight loss, diarrhea, and bloody stool, and could not survive more than 8 days (Fig.4B, 4C and 4D). The DSS+IL-10-LV-MSCs group lost less weight and had lower DAI ratings than the DSS+PBS group (Fig.4C and 4D). A 7-day macroscopic examination of colons revealed a shorter colon length (no more than 5 cm) as well as significant hyperemia and inflammation in the DSS+PBS group (Fig.4E and 4F). Compared with the mice in the DSS+PBS group, colon length in mice treated with IL-10-LV-MSCs was longer without hyperemia, and the colon length has a statistically significant difference (P<0.01) (Fig.4E and 4F). Histological examination sections indicated that, though wild type of MSCs treatment assisted colon tissue repair to a certain extent, IL-10-LV-MSCs injection more significantly reduced submucosal edema, epithelium loss and inflammatory cell infiltration (Fig.4G).
The impact of IL-10-LV-MSCs on colonic cytokines was also studied. The IL-10-LV-MSCs treatment group had lower mRNA levels of pro-inflammatory cytokines (IL-1β, IL-6 and TNF-α) in the inflamed colon than the wild type MSCs and blank LV vector treated MSCs group (P<0.001) (Fig.4H), indicating that the IL-10 overexpressing MSCs also reduced innate responsesinvivo.
3 Discussion
Despite recent advances in clinical therapies aimed at reducing inflammation, around 30% of IBD patients may not respond to biological therapy, and others may experience severe side effects[24]. A large number of preclinical studies have reported that MSCs can decrease intestinal inflammation without causing side effects[25], but these promising outcomes in animal models of IBD have yet to be duplicated in phase I/II clinical trials[12]. Therefore, several approaches have been developed to enhance the therapeutic properties of MSCs, including culture media optimization, combination with other molecules or other cell therapy, scaffolding method and genetic modification[22]. In this study, for the first time, the effects ofIL-10 gene edited BM-MSCs in an IBD mouse model were reported.
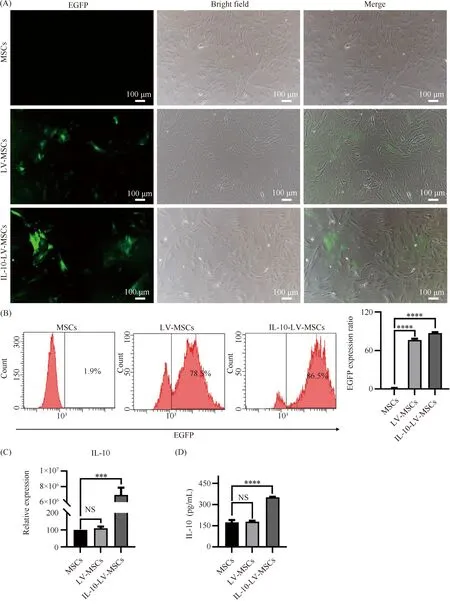
Fig.1 MSCs expression of EGFP and IL-10 after LV infection (A) The fluorescence micrograph (left) and light micrograph (middle) of MSCs and MSCs infected with different types of LVs after 48 hours, green indicates EGFP. Scale bars, 100 μm. (B) Representative flow cytometry pictures of EGFP expression in wild type MSCs and MSCs infected with the indicated LV, as well as the percentage of EGFP positive cells in MSCs (n=3,****P<0.0001). (C) The amount of IL-10 mRNA in wild type MSCs and MSCs infected with the indicated LV (n=3, NS: no significant difference,***P<0.001). (D) The protein level of IL-10 in the culture supernatant of MSCs infected with various kinds of LV (n=3, NS: no significant difference,****P<0.0001). MSCs represents wild type MSCs, IL-10-LV-MSCs and LV-MSCs represents MSCs infected with IL-10-EGFP-LV or EGFP-LV
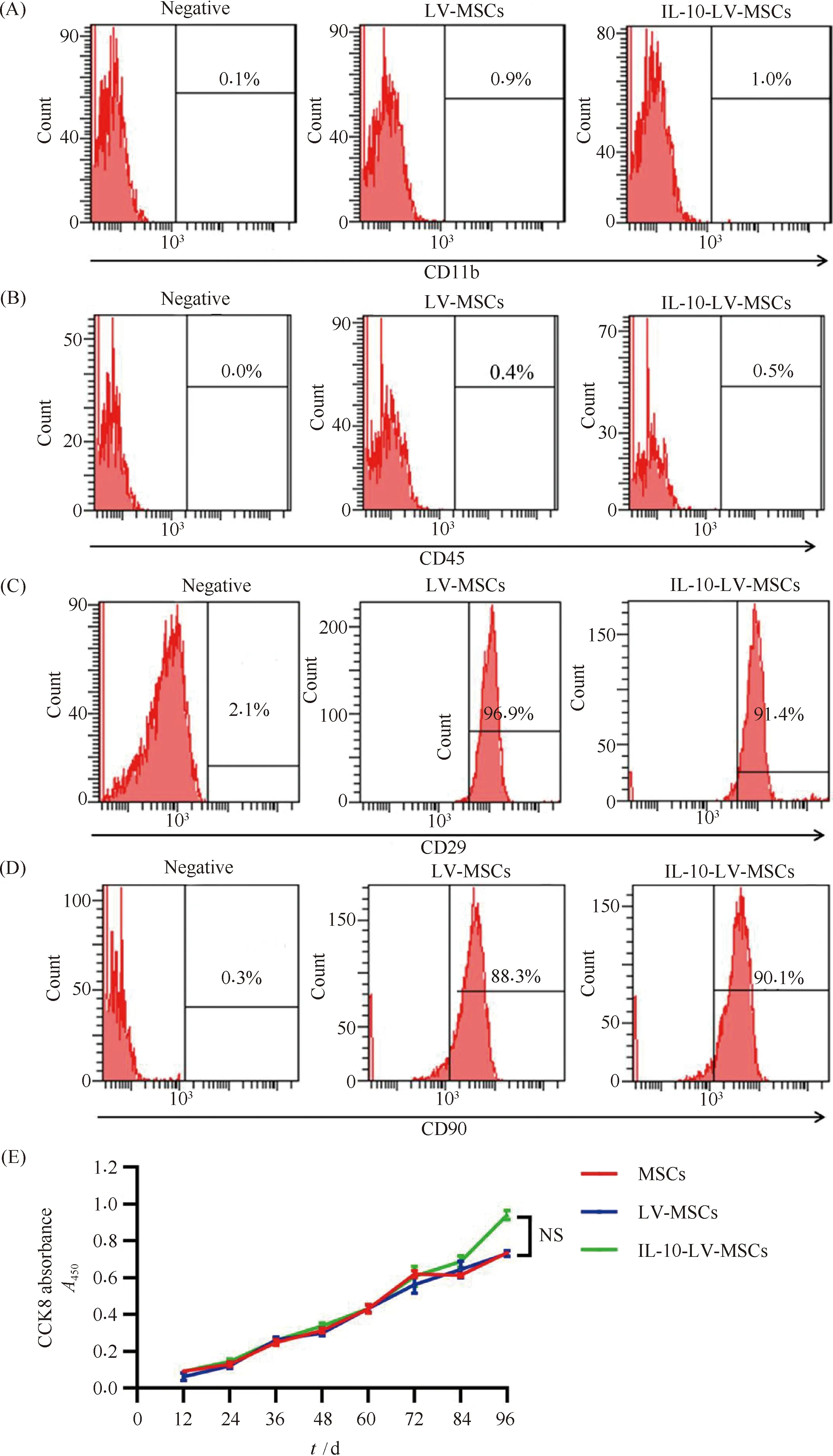
Fig.2 Phenotypic characterization of MSCs after LV infection (A) Flow cytometry analysis of CD11b in MSCs had undergone the indicated treatments. (B) Flow cytometry analysis of CD45 in MSCs had undergone the indicated treatments. (C) Flow cytometry analysis of CD29 in MSCs had undergone the indicated treatments. (D) Flow cytometry analysis of CD90 in MSCs had undergone the indicated treatments. (E) Cell proliferation rates of MSCs given the indicated treatments, CCK-8 assay was used to determine (n=3, NS: no significant difference)
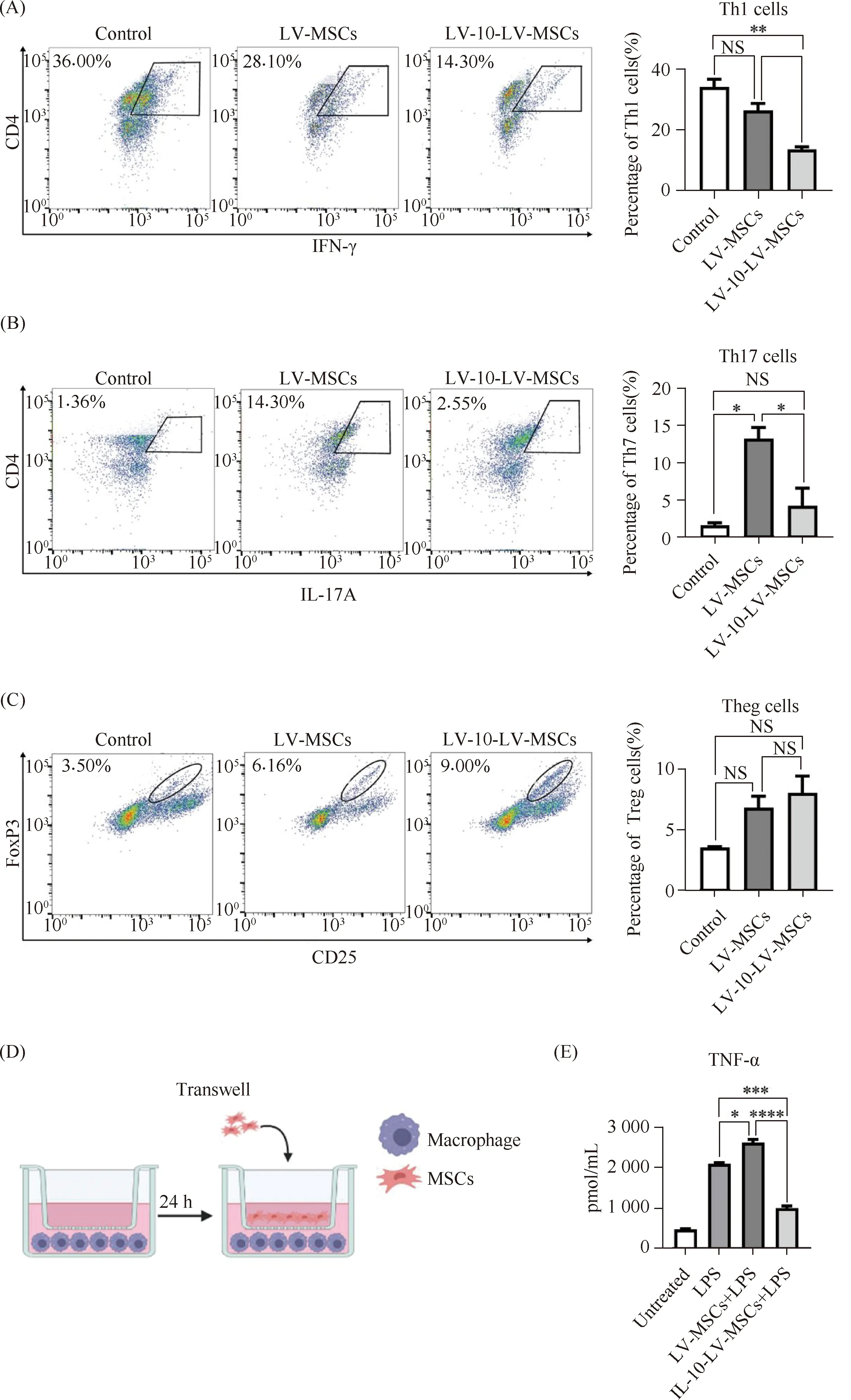
Fig.3 Immunomodulatory potential assays of engineered MSCs (A) Quantitative analysis of Th1 cells in spleen lymphocytes of DSS mice co-cultured with engineered MSCs (n=3, NS: no significant difference,*P<0.05,**P<0.01). (B) Quantitative analysis of Th17 cells in spleen lymphocytes of DSS mice co-cultured with engineered MSCs (n=3, NS: no significant difference,*P<0.05). (C) Quantitative analysis of Treg cells in spleen lymphocytes of DSS mice co-cultured with engineered MSCs (n=3, NS: no significant difference). (D) Transwell co-culture model of macrophages with Engineered MSCs. (E) ELISA analysis of TNF-α in supernatants of co-cultured macrophages and MSCs (n=3,*P<0.05,***P<0.001,****P<0.0001)
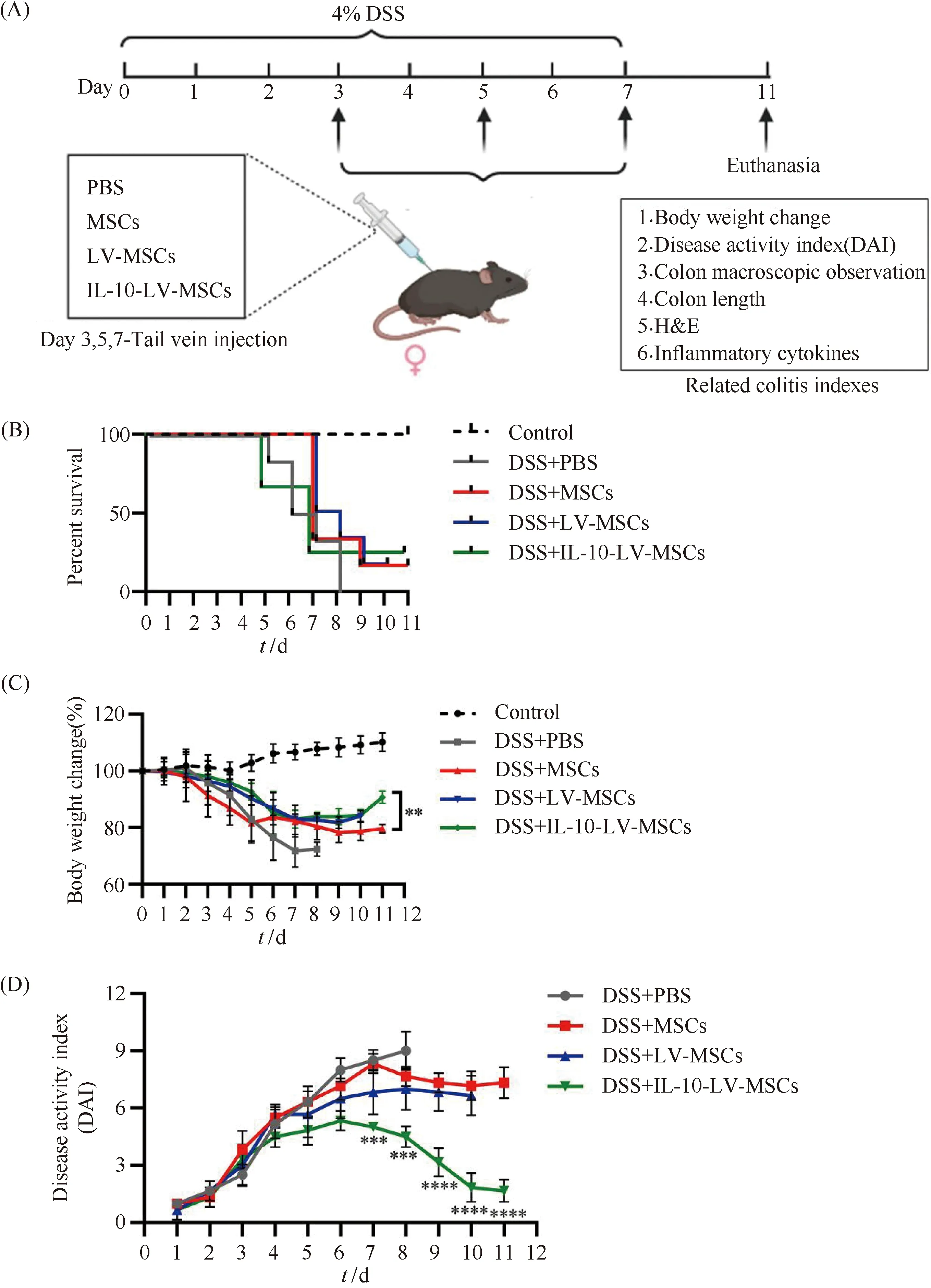
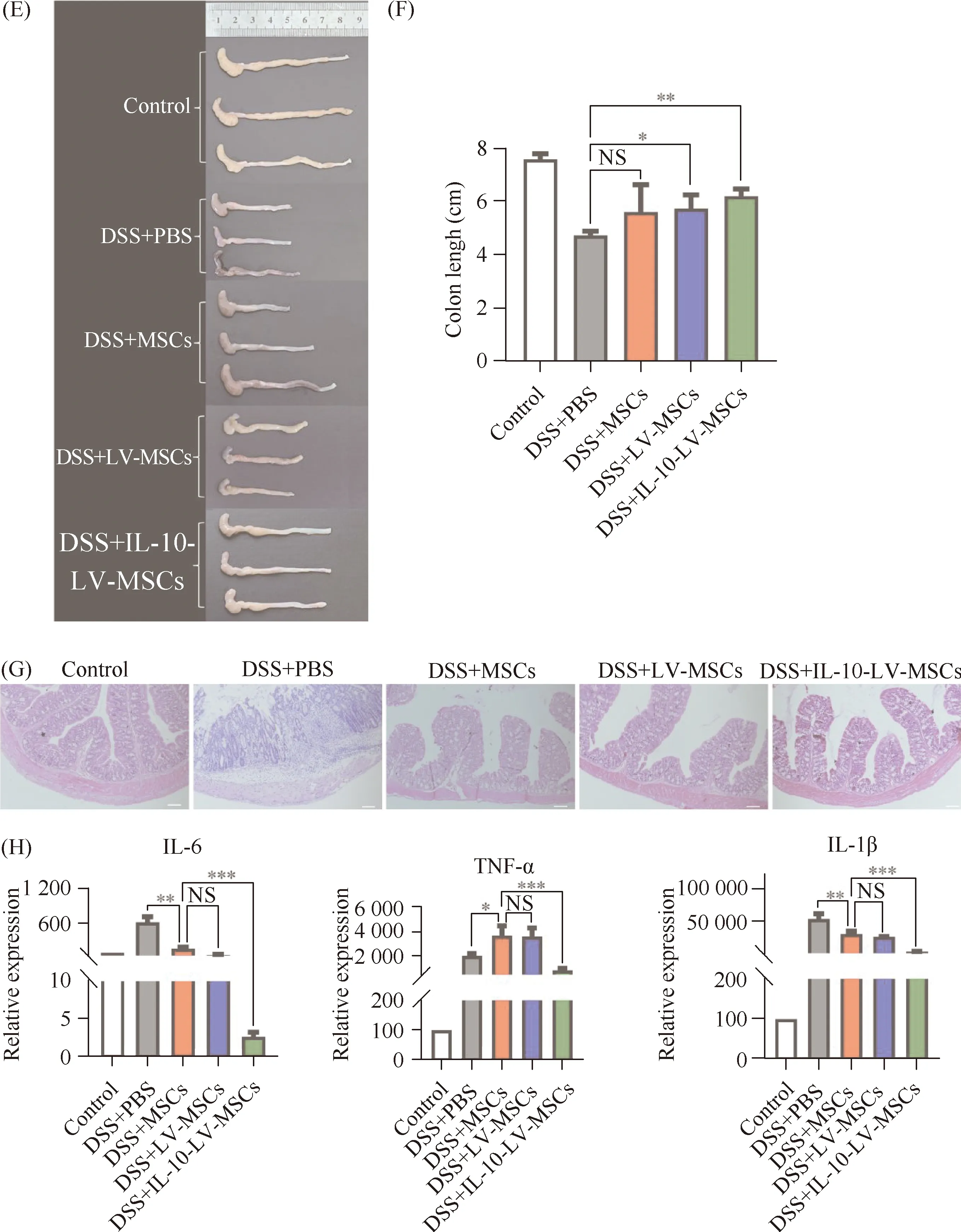
Fig.4 Evaluation for the protective effects of engineered MSCs on DSS-induced colitis mice (A) Schematic diagram of the treatment of IBD with engineered mesenchymal stem cells overexpressing IL-10. (B) Survival analysis of mice in each group was performed (n=6). (C) Body weight of mice in each group was recorded daily (n=6,**P<0.01). (D) DAI of mice in each group was recorded daily (n=6,***P<0.001,****P<0.0001). (E) Colons excised from mice received three times different treatments were photographed. (F) Colon length statistical analysis (n=6, NS: no significant difference,*P<0.05;**P<0.01). (G) H&E staining was used to evaluate colon sections from mice that had received various treatments. Scale bars, 100 μm. (H) Expression of inflammatory factors in mice from each group, IL-6 (left), TNF-α (middle), IL-1β (right) (*P<0.05,**P<0.01,***P<0.001)
In this study, the LV vectors were used to deliver potent therapeutic genes to MSCs. In clinical research, however, problems have been found for the safety of MSCs genetically engineered with stable DNA[26, 27]. In the current work, phenotypic characterization analysis was used to assess the safety of modified MSCs. The results of this work elucidated that LV infection had no impact on MSCs phenotype or cell proliferation rate.
In the present study, the BM-MSCs were armed with overexpressingIL-10 gene to construct a bioreactor which can continuously produce high level IL-10. IL-10R includes two distinct chains: IL-10R1 expressed mostly on monocytes and macrophages, and IL-10R2 could be found in most cell types[28]. The immunosuppressive IL-10 was originally reported to affect many important functions of macrophages from phagocytosis to the production of cytokines[28]. IL-10 also blocks the proliferation of Th17 cells via the directly binding on IL-10 receptor which is highly expressed on Th17 cells. Meanwhile, the immunomodulatory capacity of MSCs could be altered by environmental signals[29], possibly including IL-10. Because IL-10 was found to be necessary for MSCs maintenance in bone marrow[30]and to facilitate MSCs osteogenesis under an osteogenic condition[31]. An in-depth transcriptome and proteomic analysis is needed to clarify whether overexpressing IL-10 can improve the immunomodulatory effects of MSCs themselves.
The immunomodulatory effects ofIL-10 gene-modified BM-MSCs was explored via immune cells and MSCs co-culture systemsinvitro, and the treatment potential on IBD of the genetically-engineered BM-MSCs was on DSS induced colitis mouse model. When compared to wild type MSCs, MSCs overexpressing IL-10 showed an elevated ability to attenuate Th1 and Th17 responses, as well as a strengthened potential in inhibiting TNF-α secretion from activated macrophages. Furthermore, we demonstrated that systemic administration of IL-10-LV-MSCs substantially ameliorated the colon length, DAI and colonic cytokine expression in the DSS induced colitis mouse model.
A complex relationship between Th17 cells and IBD has been reported in recent literatures. Increased activation of Th17 cells and expression of Th17-related cytokines (such as IL-17 and IL-22) were present in IBD patients[3]. From an evolutionary perspective, Th17 cells play an important role in host defense against bacterial and fungal infection[32]. However,under disease condition especially sustained exposure to IL-1 and IL-23,Th17 cells can cause severe local tissue damage by recruit macrophages and granulocytes[33]. One aspect of the variability of the immunomodulatory function of MSCs is related to their regulatory effects on Th17 cells: MSCs inhibit Th17 cells differentiation and function[34], or induce Th17 differentiation[35]under different research background. In our study, the Th17 induction effect of MSCs was abolished by IL-10-overexpressing modification. This change may contribute to the improvement of therapeutic effect on IBD.
In summary, BM-MSCs modified by LV-mediated IL-10 overexpression showed enhanced immunomodulatory potentials without affection on the phenotypic characterization. Systemically administration of the engineered MSCs remarkably protected mice suffering from experimental colitis. This technique of using engineered MSCs to relieve mucosal inflammatory responses is a potential therapeutic approach that might be useful in the treatment of IBD.
Acknowledgments
We thank Dr.Sheng He for his kinlly sharing of the cell strain KAW 264.