Chemical Modification of Cyclen to Raise the Phase Transition Temperature of Its Perrhenic Compounds
HE Wen-Hui YE Xing WANG Zi-Yu WEI Zhen-Hong RAO Wen-Jun CAI Hu
(School of Chemistry and Chemical Engineering,Nanchang University,Nanchang 330031,China)
Abstract:A methyl-substituted 1,4,7,10-tetraazacyclododecane(N-methyl-1,4,7,10-tetraazacyclododecane,Me-cyclen)was isolated by chemical modification of 1,4,7,10-tetraazacyclododecane(cyclen)with iodomethane.Reactions of cyclen and Me-cyclen with two equivalents of HReO4led to phase transition molecules(cyclen)(ReO4)2(1)and(Me-cyclen)(ReO4)2(2),respectively.Differential scanning calorimetry and dielectric studies revealed that compounds 1 and 2 show reversible phase transition with temperatures at 324 K for 1 and 384 K for 2.The research results demonstrate a way that the high-temperature phase transition organic-inorganic hybrid materials can be obtained by reducing molecular symmetry through chemical modification of organic amines.CCDC:2097827,1;2097829,2.
Keywords:cyclic organic amine;perrhenic acid;phase transition;dielectric;chemical modification
0 Introduction
Phase transition materials usually have high latent heat and can absorb or release a large amount of heat energy during the phase transition process.The phase transition materials have the advantages of small volume change,good energy-saving effect,and easily controlled,and can be widely used in new energy and energy storage fields[1-8].For phase transition materials,besides the traditional inorganic materials,organicinorganic hybrid materials have attracted more and more attention from researchers due to their significant structural variability and high tunability[9-12].
From a view of the application prospect,the aboveroom-temperature phase transition materials are more important due to the high phase transition temperature providing a wide operational temperature range and high stability for the production as key components in a variety of areas[13-15].At present,effective methods for the improvement of phase transition temperature have been extensively researched[16-18].On the one hand,the introduction of the deuterium effect or fluorine effect can greatly improve the phase transition temperature[19-24].On the other hand,reducing molecular symmetry by chemical substitution of organic templates can also achieve the purposes[25-29].However,the chemical modification method often requires us to deeply understand the relationship between structure and property.
In the past few decades,several popular molecules with high phase transition points and other reversible physical properties have been successfully obtained using chemical modification[30-35].For example,in 2020,Fu et al.designed two enantiomeric phase transition molecules,[R-2-Me-H2dabco][TFSA]2and[S-2-Me-H2dabco][TFSA]2(Dabco=1,4-diazabicyclo[2.2.2]octane),by introducing a methyl group on the Dabco molecule[36].The obtained two enantiomers showed phase transition temperatures at 405.8 and 415.8 K,higher than other hybrids based on Dabco itself.The key to success is that by introducing methyl into the spherical molecule Dabco can reduce the symmetry of the target molecule and finally lead to high-temperature phase transition materials.
On this basis,we envisioned adding methyl groups into the cyclic molecule 1,4,7,10-tetraazacyclododecane(cyclen)because cyclen has a crown etherlike ring structure and the four nitrogen atoms on the same side could be protonated.As shown in Scheme 1,the reactions of cyclen with methyl iodide produced an unsymmetric molecule Me-cyclen.Further treatment of cyclen and Me-cyclen with two equivalents of HReO4gave the corresponding compounds(cyclen)(ReO4)2(1)and(Me-cyclen)(ReO4)2(2)with high yields.Differential scanning calorimetry(DSC)measurement showed the phase transition temperatures of compounds 1 and 2 were at 324 and 384 K,respectively.Herein we report the synthesis,characterization,and phase transition of compounds 1 and 2.
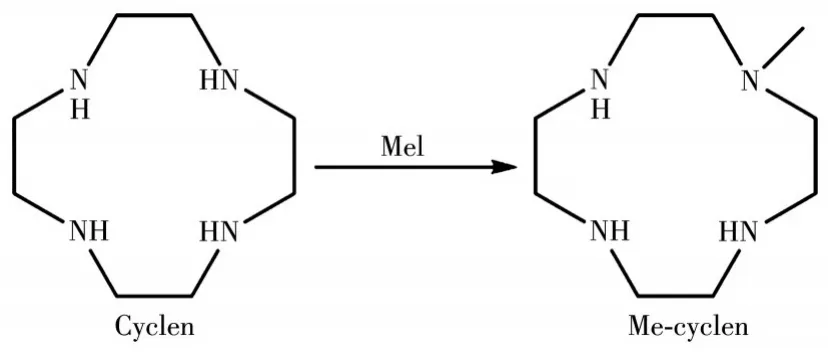
Scheme 1 Molecular structures of cyclen and Me-cyclen
1 Experimental
1.1 Instruments and materials
Me-cyclen was prepared according to the literature[37].Cyclen(98%)and perrhenic acid(75%in H2O)were purchased from the Aladdin company and used without further purification.FT-IR spectra were recorded using KBr pellets in a range of 4 000-400 cm-1on an ALPHA spectrometer.Powder X-ray diffraction(PXRD)analysis was performed using a D8 ADVANCE diffractometer(CuKαgraphite,λ=0.154 06 nm)operated at 40 kV and 15 mA with aKβfoil filter,collecting data in a 2θrange of 5°-52°with a step size of 0.02°.NETZSCH DSC 200 F3 instrument was used to perform DSC measurement at a heating/cooling rate of 10 K·min-1.The temperature-dependent dielectric constant of the crystal powder sample was measured on the Tonghui TH2828 analyzer at the frequency of 1 MHz.
1.2 Synthesis
1.2.1 Synthesis of compound 1
To a solution of cyclen(0.17 g,1.0 mmol)in 5 mL distilled water,perrhenic acid(0.72 g,2.0 mmol,75%)in 5 mL distilled water was added,and the mixture was stirred at room temperature(298 K)to give a clear solution.The colorless block crystals of 1 were finally obtained by evaporating the solution at room temperature for two days.Yield:0.53 g(59%).Anal.Calcd.for C8H22N4O8Re2(%):C 14.24,H 3.26,N 8.31;Found(%):C 14.44,H 3.36,N 8.67.IR(KBr,cm-1):3 893(m),3 736(w),3 427(w),3 305(s),3 097(s),2 914(s),2 863(s),2 631(m),2 358(m),2 121(m),1 863(m),1 712(m),1 575(s),1 445(s),1 267(m),1 088(m),911(vs),755(s),521(m).
1.2.2 Synthesis of compound 2
Me-cyclen(0.37 g,2.0 mmol)in 10 mL distilled water and HReO4(1.68 g,4.0 mmol,75%)in 5 mL distilled water were mixed and the obtained solution was being stirred at room temperature(298 K)for 15 min.The white columnar crystals of 2 were isolated by slowly evaporating the solvent at room temperature for two days.Yield:0.85 g(62%).Anal.Calcd.for C9H24N4O8Re2(%):C 15.70,H 3.51,N 8.14;Found(%):C 15.85,H 3.41,N 8.48.IR(KBr,cm-1):3 317(m),3 067(m),2 837(m),2 177(w),1 872(w),1 561(w),1 445(m),1 390(m),1 261(m),1 193(w),1 126(m),1 016(m),963(w),902(vs),867(vs),826(s),759(s).
1.3 X-ray crystallography
The single-crystal diffraction data of 1 and 2 were collected by MoKαradiation(λ=0.071 073 nm)using a Rigaku Saturn 924 diffractometer at room temperature.Empirical absorption correction was applied when the data processing was performed using the Crystalclear software package.The structures were solved by direct methods and refined with a full-matrix leastsquares technique(Olex2)[38-40].All nonhydrogen atoms were refined anisotropically,and the positions of all hydrogen atoms were generated geometrically.Crystallographic data and structure refinements are listed in Table 1.

Table 1 Crystallographic data for compounds 1 and 2
CCDC:2097827,1;2097829,2.
2 Results and discussion
2.1 Synthesis and characterization
Compounds 1 and 2 are prepared according to the strict stoichiometry of cyclic amine with perrhenic acid of 1∶2.Excess or less perrhenic acid is easy to give impure products.The crystals of 1 and 2 with high purity were obtained by evaporating solvent at room temperature(298 K)and then washed with petroleum ether.The purity of compounds 1 and 2 was verified by PXRD,microanalysis,and IR.As shown in Fig.S1(Supporting information),it can be seen that the experimentally measured PXRD data of 1 and 2 were completely consistent with the simulated data obtained by their single-crystal X-ray diffraction,confirming the bulk phase purity.Besides,it can be seen from the IR spectra(Fig.S2)that the absorptive peaks at 3 097 and 3 305 cm-1belong to the N—H bonds stretching vibration,the absorption at 1 445 cm-1belongs to the C—N bonds stretching vibration,the absorption peaks at 2 914 and 2 863 cm-1belong to the C—H stretching vibration of methylene and methyl groups,and the absorptions near 911 and 755 cm-1are ascribed to the characteristic absorption peaks of ReO4-[41-43].
2.2 Phase transition
The DSC test can be used to determine whether the compound has a reversible phase transition during the heating and cooling processes[44-45].As shown in Fig.1,compounds 1 and 2 both exhibited a pair of reversible endothermic and exothermic peaks.Compound 1 had an endothermic peak at 324 K and a corresponding exothermic peak at 313 K;compound 2 had an endothermic peak at 384 K and a corresponding exothermic peak at 361 K.It can be seen that the chemical modification of cyclen with one methyl group on nitrogen atom could greatly increase the phase transition temperature of 60 K.This may be an effective way to improve the phase transition temperature of organic-inorganic hybrid compounds.

Fig.1 DSC curves of 1(red)and 2(blue)in heating and cooling runs
As shown in Table 2,to further study the thermal abnormal behavior of compounds 1 and 2,we have calculated the transformation enthalpy(H)and entropy(S)change of the corresponding compounds.According to the Boltzmann equation ΔS=RlnN(Ris the molar gas constant,Nis the ratio of the number of possible orientations of the entire disordered system),theNvalues of compounds 1 and 2 could be calculated to be 15.79 and 53.45,respectively.Generally speaking,the larger theNvalue of a substance is,the more serious order and disorder phase transition occurred in the substance[46-47].So,compound 2 proceeded a more serious order and disorder characteristics than compound 1 during the phase transition.
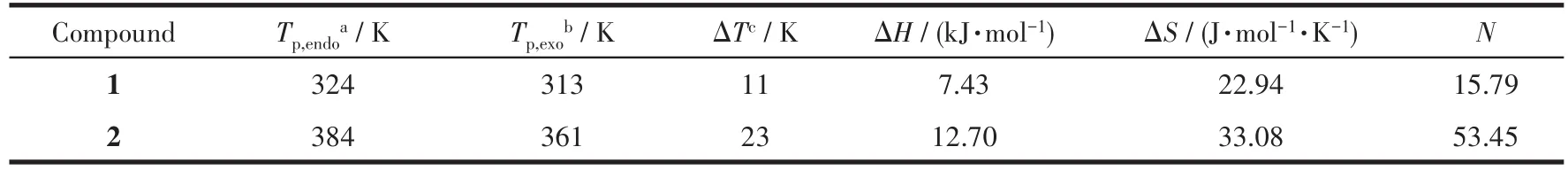
Table 2 Thermal values in the phase transition of 1 and 2
2.3 Dielectric properties
Dielectric measurement is a simple and effective method for finding new phase transition materials by studying the dynamic behavior of polar molecules in these structures.The dielectric constant is generally closely related to the molecular structure of compounds themselves[48-50].It can be seen from Fig.2 that compounds 1 and 2 exhibited dielectric anomalies near the phase transition points,which were consistent with the thermal anomaly curves of DSC on heating and cooling.The dielectric anomaly of compound 1 was more obvious than that of compound 2.During the heating process,the real part of the dielectric constant of compound 1 gradually increased with the increase of temperature.When the temperature was close to 311 K,the dielectric constant showed a significant increase,and the value ofε'at 1 MHz gradually increased from 5.36 at 289 K to a maximum of 23.72 near the phase transition.Compared with 1,theε'value of 2 at 1 MHz increased by about 0.35 times from 335 K to the phase transition point,which is much lower than the 3.4-fold increase ofε'value of 1 at 1 MHz.During the cooling process,dielectric anomalies were also found in compounds 1 and 2 near the phase transition temperatures,which further indicates both compounds have undergone a reversible dielectric transition behavior.

Fig.2 Temperature dependence of the real parts(ε')of 1 and 2 at 1 MHz upon heating and cooling
2.4 Crystal structure analysis
At room temperature,compound 1 crystallizes in the triclinic system,the space group isP,and the asymmetric unit of compound 1 is composed of one(cyclen)+cation and two perrhenate anions.While compound 2 crystallizes in the monoclinic system with the centrosymmetricP21/nspace group,the asymmetric unit is composed of one(Me-cyclen)+cation and two perrhenate anions.In both structures,two hydrogen atoms from perchloric acid have transferred to the nitrogen of cyclen or Me-cyclen.But due to the protonated hydrogen atoms being in a free state,two secondary nitrogen atoms in compounds 1 and 2 were randomly added with hydrogen atoms by the theoretical calculations.
As shown in Fig.3,in both structures,the perrhenic anions ReO4-are located outside the cyclen ring and interlinked with cations by the classic intermolecular hydrogen bonds(N—H…O).The distances between the donor N atoms and acceptor O atoms are in a range of 0.296 9-0.323 1 nm for 1 and 0.287 4-0.314 7 nm for 2(Table S1 and S2).Besides the intermolecular hydrogen bonds,there are four intramolecular hydrogen bonds(N—H…N)that can be found in compounds 1 and 2.The distances between the donor N atoms and the acceptor N atoms are in a range of 0.279 7-0.290 1 nm for 1 and 0.281 9-0.294 4 nm for 2.To be noted,the four intramolecular hydrogen bonds force the nitrogen atoms in the cyclen ligand to be located on the same side of the ring(Table S1 and S2).As shown in Table S3 and S4,the bond angles in the cyclen and Me-cyclen rings are as large as possible,which may reduce the tension of the macrocycle to make the ring not fold and keep all atoms in one plane as can as possible.The average bond lengths of C—N in 1(0.147 7 nm)are longer than those in 2(0.146 8 nm),but the average bond lengths of C—C in 1(0.150 7 nm)are shorter than those in 2(0.152 7 nm).In both crystals,the Re—O bond lengths are about 0.171 0 nm,and the O—Re—O bond angles are near 109°28',indicating the ReO4-anions choose a slightly irregular tetrahedral structure.

Fig.3 Crystal structures of 1(a)and 2(b)at room temperature,where the dashed lines represent the hydrogen bonds
3 Conclusions
In summary,adding a methyl group on the nitrogen atom of cyclen gave a low symmetric ligand Me-cyclen.Reactions of the twelve membered cyclic amine cyclen and Me-cyclen with two equivalents of perrhenic acid in an aqueous solution led to two-phase transition materials(cyclen)(ReO4)2(1)and(Me-cyclen)(ReO4)2(2).The IR,element analysis,and PXRD were carried out to prove their good purity.The DSC and dielectric measures showed the phase transition temperature of 2 at 384 K was much higher than that of compound 1 at 324 K.The analysis of the single-crystal structure reveals that the increase of the phase transition point in 2 is caused by the lower symmetric structure of Me-cyclen because the introduction of a methyl group on cyclen increases the internal steric hindrance of a new molecule.This discovery reveals that chemical modification is an effective way to improve the phase transition temperature.
Acknowledgments:We thank the National Natural Science Foundation of China(Grants No.21865015,22071094,22075123)for financial support.
Supporting information is available at http://www.wjhxxb.cn

