精子发生障碍的遗传学研究进展
张星雨,祝天喻,张清荣,郭雪江,王铖,靳光付,胡志斌
精子发生障碍的遗传学研究进展
张星雨,祝天喻,张清荣,郭雪江,王铖,靳光付,胡志斌
南京医科大学生殖医学国家重点实验室,南京 211166
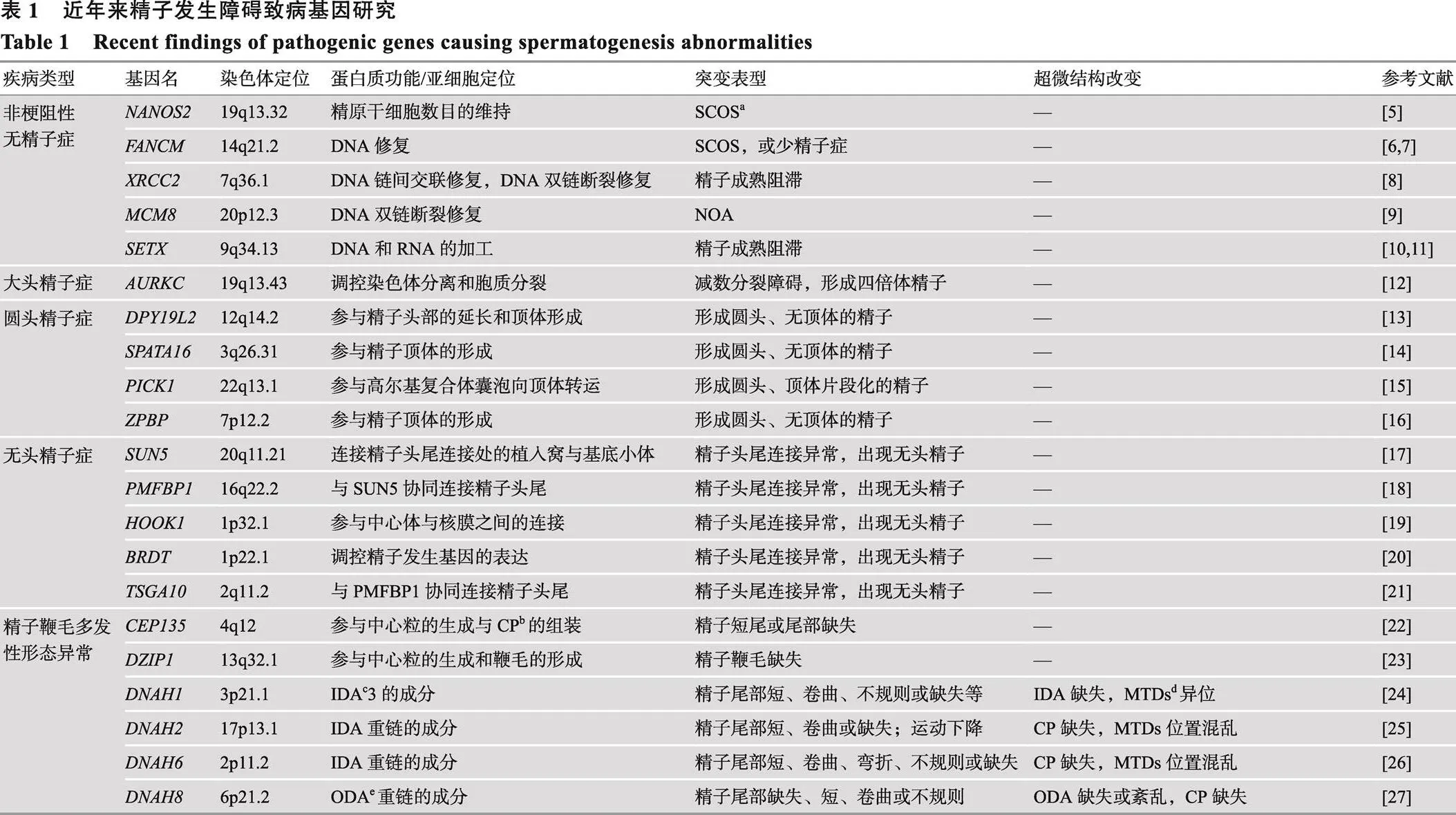
育龄人群中约15%的夫妻被不孕不育困扰,其中男方因素导致的不孕不育约占50%。男性不育通常由精子发生障碍导致,呈现为少、弱、畸形精子症,最严重的是无精子症。本文以精子发生障碍为主线,重点综述了非梗阻性无精子症和畸形精子症的遗传学病因研究。近年来,随着高通量芯片和测序技术的快速发展,无精子症和畸形精子症的遗传学因素得以深入的揭示与解析。围绕无精子症,全基因组关联研究与高通量测序研究揭示了一批非梗阻性无精子症的风险位点和致病基因;围绕畸形精子症,全外显子测序等研究鉴定了一系列致病基因,极大地丰富了精子鞭毛多发性形态异常等精子畸形的遗传学病因。大量致病基因的发现,促进了男性不育病理机制的阐明。全面而深入地了解精子发生障碍中的遗传因素,对男性不育的诊断、临床治疗和遗传咨询具有重要的意义。
精子发生;遗传因素;非梗阻性无精子症;精子畸形;鞭毛多发性形态异常
在全球范围内,约有15%的夫妇受不孕不育困扰,其中男性因素约占50%[1]。男性不育主要由于精子发生障碍导致。精子发生过程包括精原细胞有丝分裂、精母细胞减数分裂以及精子细胞变形形成蝌蚪状精子的过程,其中任一环节发生缺陷都会导致男性不育[2]。精子发生障碍在临床上主要体现为无精子症、少精子症、畸形精子症和/或弱精子症,其中最严重的是无精子症。无精子症是指在射出的精液中完全不存在精子,包括梗阻性无精子症(obstructive azoospermia, OA)和非梗阻性无精子症(nonobstructive azoospermia, NOA)[3]。OA患者的睾丸通常具有正常的精子产生能力,由于梗阻原因导致精子输送异常,从而表现为无精子症,而NOA则是由于睾丸内精子发生障碍导致。畸形精子症主要是精子的头部或尾部畸形,最近关注较多的精子鞭毛多发性形态异常(multiple morphological abnormalities of the flagella, MMAF)[4],主要是呈现为精子尾部畸形。引起精子发生障碍的因素主要包括环境因素和遗传因素。近年来基因组芯片和测序等高通量技术的快速发展,促进了复杂疾病的致病基因发现,精子发生障碍中非梗阻性无精子症与畸形精子症越来越多的相关基因被鉴定(表1)。本文主要围绕非梗阻性无精子症和畸形精子症的遗传学因素研究进展进行综述。
1 非梗阻性无精子症的遗传学研究
NOA是男性不育中精子发生障碍最严重的一种疾病,在男性不育患者中约占10%[3]。目前已知的NOA的遗传因素包括染色体数量和结构异常以及精子发生功能基因异常。
1.1 染色体数量/结构异常与NOA
染色体异常指染色体的数目或结构异常。在NOA患者中最常见染色体数目异常的是Klinefelter 综合征(47, XXY)[46]。Y染色体长臂上的微缺失会破坏精子发生关键基因的完整性,可解释约5~10%的NOA患者的病因[47,48]。此外,Lu等[49]通过将NOA患者与健康人对比发现Y染色体单倍型类群(Y chromosome haplogroups, Y-hgs) K*与O3e*两种类群在两组中的分布差异具有统计学意义:Y-hg K*人群中出现DAZ (Deleted in Azoospermia)多拷贝(>4)的频率显著高于Y-hg O3e*人群,提示DAZ的拷贝数过多可能是Y-hg K*人群导致NOA的原因之一。
1.2 NOA的致病突变和致病基因
目前人们对于NOA的致病突变/基因的认识十分局限,已确定的NOA致病基因较少[50]。近年来,高通量测序技术推动了NOA的致病突变的发现。
Laura 等[5]通过全外显子组测序(whole-exome sequencing, WES)技术在患有NOA的同胞兄弟中发现(nanos C2HC-type zinc finger 2)的突变,该基因可维持精原细胞数目,发生突变后则无法保证精原细胞的数目,最终导致SCOS。据报道,、和等基因在精母细胞减数分裂过程中参与了重组、染色体交叉互换、DNA双链断裂的修复,它们失去功能后会造成减数分裂阻滞,从而导致NOA的发生[5-11]。
已被发现和证实可导致NOA的基因尚十分局限,但是随着测序的发展与应用,将有更多NOA致病基因被逐渐解析。这对于生殖方向的基础研究和临床方向的基因诊断方法开发均意义重大。
1.3 NOA的易感基因与易感位点
全基因组关联研究(genome-wide association study, GWAS)可通过对比病例组和对照组全基因组单核苷酸多态性(single nucleotide polymorphism, SNP)发现疾病相关的SNP位点。比如Hu等[51]人首次开展NOA的多阶段GWAS研究,在2927名NOA患者和5734名健康对照中鉴定出3个NOA风险SNP位点:rs12097821、rs2477686和rs10842262;Zhao等[52]通过GWAS发现主要组织相容性复合体(major histocompatibility complex, MHC)基因座上的两个SNP位点rs3129878和rs498422与人群NOA的易感性存在显著关联。
随后,Hu等[53]又发现4个与NOA关联的新易感位点:rs7194、rs7099208、rs13206743和rs3000811。其中rs3000811附近易感基因(CDC42 binding protein kinase alpha)在果蝇中存在同源基因。将果蝇基因敲降后,雄性果蝇出现不育表型;rs7099208位于(family with sequence similarity 160 member B1)的末位内含子中。主要表达在精原细胞与圆形精子中,而在NOA患者睾丸中表达降低甚至缺失,因此,rs7099208位点可能通过影响的表达影响NOA发生的风险[54]。
在上述研究发现的易感位点中,rs7194、rs3129878和rs498422均位于MHC区域[51,52],于是Huang等[55]对MHC区域开展精细定位(fine mapping)分析,发现rs4997052与NOA易感性有关,这为揭示MHC区域与NOA易感性间的遗传机制提供了新的认识。此外,Qin等[56]结合精子发生关键基因对NOA-GWAS数据进行深入挖掘,发现了3个NOA的易感位点,定位至3个基因,其中(rs2126986)和(rs7226979)会增加人群对NOA的易感性,(rs1406714)则相反。
对于多基因参与的复杂性疾病而言,低频遗传变异的数目远超过常见遗传变异,其遗传贡献尚不明确。Ni等[57]系统研究低频遗传变异与中国男性患NOA风险的关联,发现3个与NOA发病风险显著相关的低频遗传变异。其中rs2298090及rs200847762均显著降低了中国汉族男性患NOA的风险,而rs11754464 G>A与NOA发病风险的增加显著相关。该研究发现说明了低频变异在精子发生过程中的重要作用。
由此可见,通过大量样本的关联研究能够从人群层面系统鉴定NOA的易感位点和易感基因,有助于研究者更加深入而全面地认识NOA发生的遗传机制。
2 畸形精子症的遗传学研究
精子细胞经过精子形成过程产生精子的过程中,会经历一系列细胞生物学事件。如细胞核的浓缩、顶体的形成、组蛋白被鱼精蛋白的替换、胞质残余体的丢弃及鞭毛的发生,其中变化最大的是精子尾部。在精子变形期间,精子细胞中心体迁移到细胞表面,远端中心粒发出微管形成轴丝,导致精细胞质膜从细胞突出。然后形成鞭毛并延伸到生精小管管腔中,精子鞭毛的组装依赖鞭毛内转运(intraflagellar transport, IFT)机制,可选择性地将特定的分子转运到特定的位置,从而保证鞭毛的正常结构。
精子尾部可分为颈段、中段、主段和末段。颈段连接精子尾和头。中段位于颈段和主段之间,主要由轴丝、ODF、MS等组成。轴丝由周边的9对二联微管和一对中央微管组成,是典型的“9+2”结构,前接颈段,后向尾部延伸,几乎贯穿精子尾部。轴丝的每对二联微管由A型亚微管和B型亚微管组成,每个A型亚微管向下一个B型亚微管伸出2个短臂,称动力蛋白臂(dynein arm),包括IDA和ODA,均由重链(heavy chains, HC)、中链(intermediate chains, IC)、中轻链(light-intermediate chains, LIC)、轻链(light chains, LC)以及许多调节蛋白组成。ODA与精子运动密切相关。IDA影响到精子鞭毛拍击的力量和频率。同时A亚微管向中央微管发出丝状的结构,称放射辐(radial spokes, RS),起到连接中央微管和二联微管的作用(图1)。
2.1 大头精子症(macrozoospermia)
大头精子症在1977年被首次报道[58],是指精子头部尺寸大于正常精子头部,具有多条鞭毛,几乎全部为四倍体,通常伴随少精症出现。它是一种罕见的不育病因,在不育症患者中占比不到1%。目前,只有(aurora kinase C)基因被证实是大头精子症的遗传学病因[12]。编码减数分裂细胞中染色体分离所需的丝氨酸/苏氨酸蛋白激酶成分,对于减数分裂染色体分离和胞质分裂至关重要[12]。大多数大头精子症是由于基因3号外显子中胞嘧啶的纯合缺失(c.144delC)。该突变导致蛋白质的截短而缺少激酶结构域,从而影响减数分裂,并且当两次减数分裂都受到影响时,精子为四倍体[12]。
2.2 圆头精子症(globozoospermia)
圆头精子症特征是产生圆头、无顶体的精子。高尔基复合体产生前顶体囊泡,前顶体囊泡融合在核膜附近形成顶体。精子顶体含有的多种水解酶对于精卵结合及整个受精过程至关重要,因此当精子头部顶体缺陷时将导致严重的男性不育。圆头精子症非常少见,仅影响0.1%的不育男性[1]。在小鼠模型中,有50多个不同的基因突变会导致圆头精子症的发生。但在人类中,仅4个基因(、、和)的突变被证明与该疾病有关[13-15]。(dpy-19 like 2)突变是圆头精子症最常见(60%~80%)的遗传缺陷,该基因位于第12号染色体长臂,编码的蛋白是精子发生过程中头部延长和顶体形成所必需的,其功能缺失将导致这些过程的阻滞[59]。研究者发现在小鼠中敲除会导致类似的圆头精子症表型[60]。在患者中最常见的突变方式是整个基因的完全缺失。为了探究该基因缺失导致圆头精子症的分子机制,Guo等[61]通过高通量蛋白质组学分析缺失的圆头精子与正常精子之间的蛋白表达差异,发现SPACA1、IZUMO1、ZPBP和PLCZ1等多个参与精子变形与功能的蛋白表达异常,为人类圆头精子症的发生机制提供了线索。此外,(spermatogenesis associated 16)(protein interacting with PRKCA 1)与(zona pellucida binding protein)的突变也会导致人类圆头精子症[14-16]。
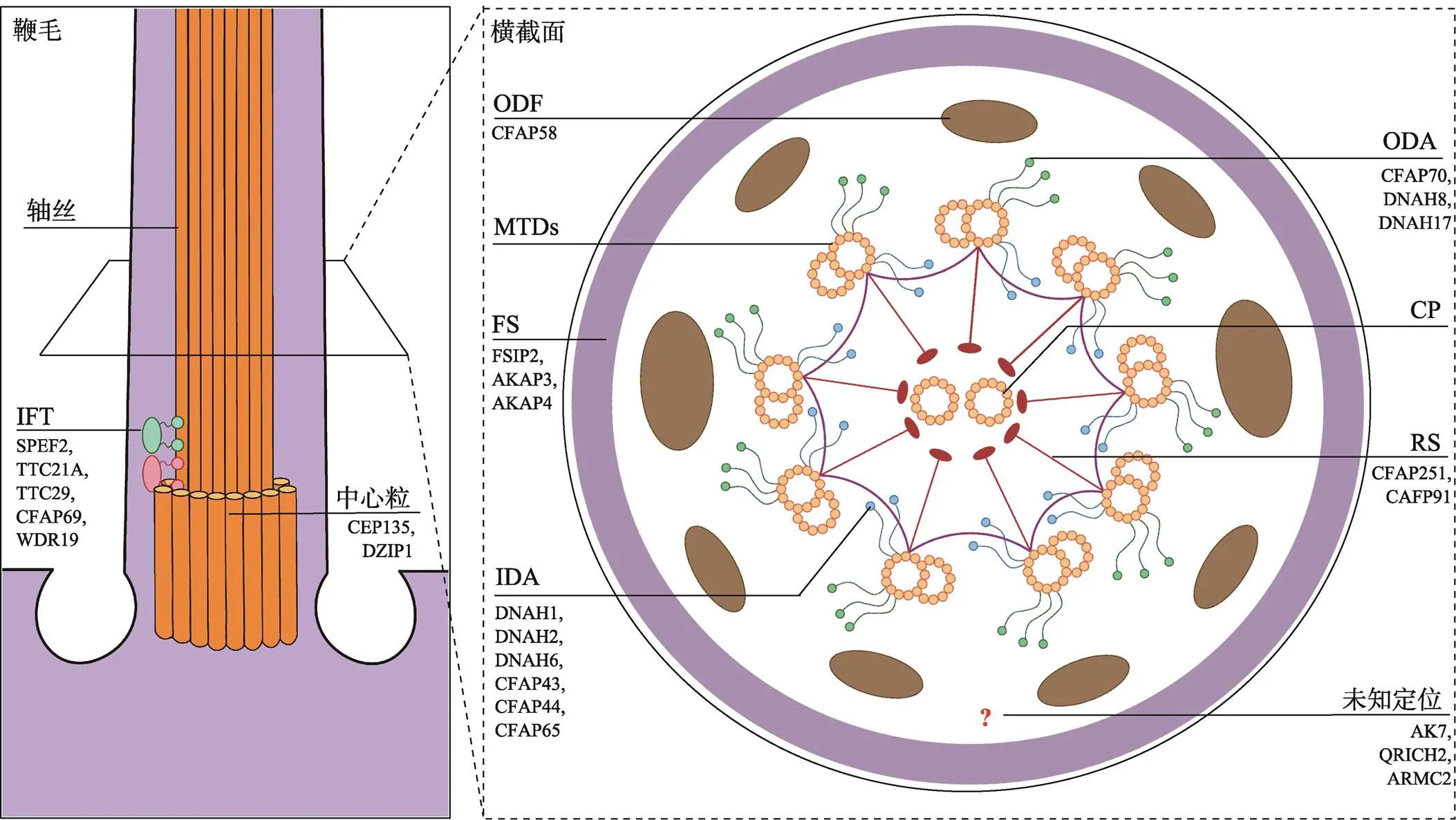
图1 精子鞭毛超微结构及主要的MMAF相关结构蛋白表达定位
FS:纤维鞘(fibrous sheath);ODA:外侧动力蛋白臂(outer dynein arm);IDA:内侧动力蛋白臂(inner dynein arm);CP:中央微管(central pair);ODF:外周致密纤维(outer dense fiber);MTDs:二联微管(microtubule doublets);RS:放射辐(radial spokes);IFT:鞭毛内转运(intraflagellar transport)。
2.3 无头精子症(acephalic spermatozoa, AS)
AS是男性不育症中最严重的类型之一,无头精子症患者的精液中大部分是无头的精子,还有少部分是头尾连接异常的精子。根据超微结构观察精子颈部的断裂位点,可分为Ⅰ、Ⅱ、Ⅲ三种亚型:Ⅰ型AS是在两个中心粒之间断开的,但相关基因尚未被报道,仍有待研究。Ⅱ型的断裂点在细胞核与近端的中心粒之间,目前已证实(SAD1 and UNC84 domain containing 5)、(polyamine modulated factor 1 binding protein 1)和(hook microtubule tethering protein 1)三个基因的突变与Ⅱ型AS的发生有关[17-19]。Ⅲ型无头精子的断裂点在远端中心粒和精子鞭毛中段之间,如(bromodomain testis associated)突变导致的AS[20]。
在动物与人类中突变均可导致Ⅱ型AS。SUN5是基底小体(basal body)对面核膜上的跨膜蛋白,具有促进植入窝与基底小体之间的连接和相互作用,确保头部锚定在精子尾。突发生变可造成无头精子的形成[17]。是另一个Ⅱ型AS基因,其蛋白在植入窝与基底小体上均有表达。PMFBP1蛋白定位于SUN5与TSGA10之间,三者形成的结构在精子颈部作为蛋白支架起到连接头尾的作用。当三者之间无法正常相互作用时,便会导致AS的发生[18,62]。此外,的突变也会导致Ⅱ型AS的发生[19]。
Ⅲ型AS基因也逐渐被发现。Li等[20]报道基因突变导致的无头精子尾部中段缺少MS,可能是人类无头精子症发生的致病基因。据报道在西方国家突变常造成严重的少精症甚至无精症。但是,已有报道称该基因的RNA剪接位点纯合突变与无头精子的发生有关[63]。与上述机制类似,在其他AS病例中,还发现了(testis specific 10)基因突变导致的无头精子伴随MS受损[21]。
2.4 精子鞭毛多发性形态异常
精子鞭毛多发性形态异常(MMAF)是最常见的精子畸形,它是精子鞭毛缺失、过短、不规则或卷曲等症状的总称。在电镜下,MMAF精子鞭毛的轴丝与轴丝旁结构显示出严重的错乱[64]。近几年,随着Khelifa等[24]在2014年报道突变可引起MMAF,关于MMAF的遗传学研究成果不断涌现,目前已经可以解释约60%的MMAF患者的遗传学病因[38,45]。精子鞭毛超微结构的稳定对维持鞭毛的形态和功能至关重要。研究表明,精子鞭毛的多个结构发生异常,均可导致MMAF[64](图1)。下面将按精子鞭毛的形成与结构,总结MMAF的遗传学研究进展。
2.4.1 中心体缺陷的相关基因
中心体对于精子鞭毛的发生起到至关重要的作用。中心体存在于所有的真核细胞中,由一对相互垂直的中心粒构成。中心体参与胞内许多的功能,比如形成细胞骨架、形成有丝分裂的纺锤体。在鞭毛或纤毛形成过程中,中心体迁移到细胞外围,以中心粒为基体启动轴丝的组装。
(centrosomal protein 135)编码一种中心体蛋白,目前仅报道一例MMAF病人与突变有关[22],并且该病人的精子无法通过ICSI与卵子形成正常受精卵,研究认为这与精子中心体参与调控受精卵第一次分裂有关。最近Lv等[23]在两名MMAF患者中发现(DAZ interacting zinc finger protein 1)突变也会导致中心粒功能障碍,造成精子尾部鞭毛的缺失,并在小鼠模型中得到验证。但是该报道并未提及此类精子是否能够成功ICSI,目前仍需更多的相关研究以阐明中心体功能障碍对于辅助生殖的影响。
2.4.2 动力蛋白臂蛋白缺陷的相关致病基因
近期研究发现了一些进化保守的动力蛋白臂相关基因及其蛋白的缺失可导致MMAF。Khelifa等[24]在20名北非MMAF患者中发现有6位患者的基因存在纯合突变。随后,多项研究相继报道MMAF患者存在突变[65-67]。内侧动力蛋白臂(IDA)由七分子复合物组成,以3-2-2的排列方式组合并对应于三种不同类型的球形头部(IDA1、IDA2和IDA3)。(在小鼠中的同源基因)敲除小鼠的IDA3头部缺失,表明DNAH1蛋白是IDA3的组成部分。研究者推测突变使IDA3的头部缺失,造成轮辐的锚定位点缺失,使中央微管的附着减弱,形成精子尾部的畸形[68]。后续研究表明,所在家族的多个成员突变均可导致MMAF,包括[25][26]DNAH8[27]以及[28]
2.4.3 动力蛋白臂关联蛋白缺陷的相关致病基因
在内侧动力蛋白臂中存在一个与之关联的有两个头部的动力蛋白,称作I1 dynein (dynein f),简称I1[69],而近年来在MMAF病人中报道的、和等基因与衣藻中编码内侧动力蛋白臂关联蛋白的基因是同源基因。
2017年,Zhang等[29]首次证实(cilia and flagella associated protein 43)和突变会导致精子MMAF表型,并怀疑具有相似的作用。2019年,接连两篇报道证实与缺陷是导致MMAF的原因之一[70,71]。另外基因突变的患者精子有MMAF的表现,并在敲除鼠中得到验证[30]。
CFAP70蛋白则属于外侧动力蛋白臂相关联的复合物,对于ODA的组装或活性至关重要[72]。2019年,Beurois等[31]在167名MMAF患者的队列中发现2例患者携带突变。
2.4.4 RS缺陷的相关致病基因
RS是二联微管的A型微管向中央微管发出的丝状结构。RS将中央微管与周围的二联管连接起来。(或)蛋白主要定位在RS,2018年Kherraf等[73]首次在78名MMAF患者的队列中鉴定到7人携带的突变。越来越多的证据表明突变是导致MMAF的原因之一:Auguste等[32]报道3名MMAF患者携带突变,伴随MS结构异常;Li等[74]在65名汉族MMAF患者中证实3人携带突变导致MMAF。此外,还有报道称基因突变会使中心微管与放射辐缺陷,导致MMAF[75]。
2.4.5 轴丝及其周围结构缺陷的相关致病基因
轴丝几乎贯穿精子尾部,其周围结构包括MS、ODF和FS。许多报道称MMAF的精子存在轴丝或其周围结构的结构紊乱,提示MMAF的发生可能与轴丝及其周围结构相关的基因缺陷有关。
早在2003年,Eddy等[76]阐述了两种A型激酶锚定蛋白AKAP3和AKAP4 (A-kinase anchoring protein 3, 4)作为结构蛋白在FS中含量最为丰富。其中AKAP3在圆形精子时期参与组成FS的基本结构,而AKAP4在精子发生的晚期表达参与FS的最终形成[77]。和基因突变会导致患者出现MMAF类似表型[34],并证实突变小鼠具有MMAF表型。
最近,He等[78]在90名MMAF患者中发现5人携带突变,并通过小鼠模型证实突变是导致MMAF的原因。此外(fibrous sheath interacting protein 2)基因突变会导致MMAF的发生[36,79]。上述基因的突变均会导致精子鞭毛的FS结构完全紊乱,其次轴丝也出现中心微管和动力蛋白缺失等缺陷。
2.4.6 鞭毛内转运失调的相关致病基因
IFT相关蛋白对于精子发生起重要作用,如小鼠突变模型显示其蛋白在精子的鞭毛形成过程中是必需的。近期,研究者发现人类IFT相关基因突变可导致MMAF的发生:
(tetratricopeptide repeat domain 21A),又称,编码的蛋白包含19个TPR结构域。2019年,Liu等[37]在两个不同的MMAF队列中共鉴定到5位患者携带的突变。突变鼠模型也具有MMAF表型,但机制尚不清楚。与类似。最近两组研究报道该基因突变与MMAF有关[38,80],突变患者的精子呈现明显的MMAF表型。
最近研究发现,(sperm flagellar 2)突变与MMAF的发生有关[39,81,82]。雄性小鼠生殖细胞特异性敲除基因后具有MMAF表型,证明SPEF2蛋白为精子尾部的形成所需。此外研究者还发现该蛋白与细胞质动力蛋白1 (cytoplasmic Dynein1)相互作用,参与IFT过程[83]。突变也会造成人类的MMAF发生,并且在小鼠中得到了验证[40],伴有精子头部的异常。小鼠睾丸蛋白质组学分析显示CFAP69与SPEF2存在关联。此外,(WD repeat domain 19)是IFT复合物的一个核心成分,该基因突变的病人也表现出MMAF症状,主要为短尾和卷尾[41]。
2.4.7 其他鞭毛相关基因
一些具体功能定位尚不清晰的基因突变可导致MMAF,如(glutamine rich 2),(adenylate kinase 7)和(armadillo repeat containing 2)等。2019年两组研究共鉴定到4名MMAF患者的基因发生突变[42,43],突变小鼠模型具有MMAF表型[43]。在睾丸中特异表达,可以维持精子发生功能蛋白的稳定性。在成熟精子中,QRICH2蛋白与Tubulin一起沿着精子鞭毛分布。进一步研究发现,QRICH2蛋白可以通过抑制泛素化通路提高AKAP3、TTSK4等蛋白的稳定性,而这两种蛋白的缺失会导致精子尾部缺陷[77,84]。编码一种腺苷酸激酶,催化两分子ADP反应生成一分子ATP和一分子AMP。2018年的一项研究称人类突变会造成MMAF症状[44]。最近Coutton等[45]在168位MMAF病人的队列中发现5位患者的基因发生突变,之后在小鼠中敲除后,小鼠精子也表现出MMAF和雄性不育,证明了突变导致MMAF。
3 结语与展望
精子发生障碍的遗传病因研究是人类生殖健康的重要内容。得益于基因芯片和高通量测序等高通量技术的快速发展,精子发生障碍的遗传学研究取得了重大进展。如围绕精子发生障碍最严重的无精子症,利用GWAS和高通量测序等技术研究基于大样本人群揭示了多个疾病的风险位点和易感基因;畸形精子症若呈现为均一类型的精子畸形,通常由遗传学异常导致,深度测序的广泛使用,促进了一系列致病基因的发现,也是近期男性不育遗传学病因研究的热点领域。然而,已鉴定到的致病突变位点大多位于基因编码区,而基因的调控序列与非编码区域/基因的改变,以及环境基因交互作用对精子发生障碍的影响仍然需要进一步的研究。致病与易感基因的发现为临床男性不育病因学阐明、治疗方法的研发以及遗传咨询等均有着重要意义。
[1] Krausz C, Riera-Escamilla A. Genetics of male infertility, 2018, 15(6): 369–384.
[2] Neto FTL, Bach PV, Najari BB, Li PS, Goldstein M. Spermatogenesis in humans and its affecting factors, 2016, 59: 10–26.
[3] Cerván-Martín M, Castilla JA, Palomino-Morales RJ, Carmona FD. Genetic Landscape of Nonobstructive Azoospermia and New Perspectives for the Clinic, 2020, 9(2): 300.
[4] Coutton C, Escoffier J, Martinez G, Arnoult C, Ray PF. Teratozoospermia: spotlight on the main genetic actors in the human, 2015, 21(4): 455–485.
[5] Fakhro KA, Elbardisi H, Arafa M, Robay A, Rodriguez-Flores JL, Al-Shakaki A, Syed N, Mezey JG, Khalil CA, Malek JA, Al-Ansari A, Said SA, Crystal RG. Point-of-care whole-exome sequencing of idiopathic male infertility, 2018, 20(11): 1365–1373.
[6] Kasak L, Punab M, Nagirnaja L, Grigorova M, Minajeva A, Lopes AM, Punab AM, Aston KI, Carvalho F, Laasik E, Smith LB, GEMINI Consortium, Conrad DF, Laan M. Bi-allelic recessive loss-of-function variants in FANCM cause non-obstructive azoospermia, 2018, 103(2): 200–212.
[7] Yin H, Ma H, Hussain S, Zhang H, Xie XF, Jiang L, Jiang XH, Iqbal F, Bukhari I, Jiang HW, Ali A, Zhong LW, Li T, Fan SX, Zhang BB, Gao JN, Li Y, Nazish J, Khan T, Khan M, Zubair M, Hao QM, Fang H, Huang J, Huleihel M, Sha JH, Pandita TK, Zhang YW, Shi QH. A homozygous FANCM frameshift pathogenic variant causes male infertility, 2019, 21(1): 62–70.
[8] Yang YJ, Guo JH, Dai L, Zhu YM, Hu H, Tan LH, Chen WJ, Liang DS, He JL, Tu M, Wang KW, Wu LQ. XRCC2 mutation causes meiotic arrest, azoospermia and infertility, 2018, 55(9): 628–636.
[9] Tenenbaum-Rakover Y, Weinberg-Shukron A, Renbaum P, Lobel O, Eideh H, Gulsuner S, Dahary D, Abu-Rayyan A, Kanaan M, Levy-Lahad E, Bercovich D, Zangen D. Minichromosome maintenance complex component 8 (MCM8) gene mutations result in primary gonadal failure, 2015, 52(6): 391–399.
[10] Catford SR, O'Bryan MK, McLachlan RI, Delatycki MB, Rombauts L. Germ cell arrest associated with aSETX mutation in ataxia oculomotor apraxia type 2, 2019, 38(6): 961–965.
[11] Becherel OJ, Fogel BL, Zeitlin SI, Samaratunga H, Greaney J, Homer H, Lavin MF. Disruption of spermatogenesis and infertility in ataxia with oculomotor Apraxia Type 2 (AOA2), 2019, 18(3): 448–456.
[12] Dieterich K, Rifo RS, Faure AK, Hennebicq S, Amar BB, Zahi M, Perrin J, Martinez D, Sèle B, Jouk PS, Ohlmann T, Rousseaux S, Lunardi J, Ray PF. Homozygous mutation of AURKC yields large-headed polyploid spermatozoa and causes male infertility, 2007, 39(5): 661–665.
[13] Koscinski I, Elinati E, Fossard C, Redin C, Muller J, de la Calle JV, Schmitt F, Khelifa MB, Ray PF, Kilani Z, Barratt CLR, Viville S. DPY19L2 deletion as a major cause of globozoospermia, 2011, 88(3): 344–350.
[14] Dam AHDM, Koscinski I, Kremer JAM, Moutou C, Jaeger AS, Oudakker AR, Tournaye H, Charlet N, Lagier-Tourenne C, van Bokhoven H, Viville S. Homozygous mutation in SPATA16 is associated with male infertility in human globozoospermia, 2007, 81(4): 813–820.
[15] Xiao N, Kam C, Shen C, Jin WY, Wang JQ, Lee KM, Jiang LW, Xia J. PICK1 deficiency causes male infertility in mice by disrupting acrosome formation, 2009, 119(4): 802–812.
[16] Oud MS, Okutman Ö, Hendricks LAJ, de Vries PF, Houston BJ, Vissers LELM, O'Bryan MK, Ramos L, Chemes HE, Viville S, Veltman JA. Exome sequencing reveals novel causes as well as new candidate genes for human globozoospermia, 2020, 35(1): 240–252.
[17] Zhu FX, Wang FS, Yang XY, Zhang JJ, Wu H, Zhang Z, Zhang ZG, He XJ, Zhou P, Wei ZL, Gecz J, Cao YX. Biallelic SUN5 mutations cause autosomal-recessive acephalic spermatozoa syndrome, 2016, 99(4): 942–949.
[18] Zhu FX, Liu C, Wang FS, Yang XY, Zhang JJ, Wu H, Zhang ZG, He XJ, Zhang Z, Zhou P, Wei ZL, Shang YL, Wang L, Zhang RD, Ouyang YC, Sun QY, Cao YX, Li W. Mutations in PMFBP1 cause acephalic spermatozoa syndrome, 2018, 103(2): 188–199.
[19] Chen HX, Zhu Y, Zhu ZJ, Zhi E, Lu KM, Wang XB, Liu F, Li Z, Xia WL. Detection of heterozygous mutation in hook microtubule-tethering protein 1 in three patients with decapitated and decaudated spermatozoa syndrome, 2018, 55(3): 150–157.
[20] Li L, Sha YW, Wang X, Li P, Wang J, Kee K, Wang BB. Whole-exome sequencing identified a homozygous BRDT mutation in a patient with acephalic spermatozoa, 2017, 8(12): 19914–19922.
[21] Sha YW, Sha YK, Ji ZY, Mei LB, Ding L, Zhang Q, Qiu PP, Lin SB, Wang X, Li P, Xu X, Li L. TSGA10 is a novel candidate gene associated with acephalic spermatozoa, 2018, 93(4): 776–783.
[22] Sha YW, Xu XH, Mei LB, Li P, Su ZY, He XQ, Li L. A homozygous CEP135 mutation is associated with multiple morphological abnormalities of the sperm flagella (MMAF), 2017, 633: 48–53.
[23] Lv MR, Liu WJ, Chi WF, Ni XQ, Wang JJ, Cheng HR, Li WY, Yang SM, Wu H, Zhang JQ, Gao Y, Liu CY, Li CH, Yang CY, Tan Q, Tang DD, Zhang JJ, Song B, Chen YJ, Li Q, Zhong YD, Zhang ZH, Saiyin H, Jin L, Xu YP, Zhou P, Wei ZL, Zhang CM, He XJ, Zhang F, Cao YX. Homozygous mutations in DZIP1 can induce asthenoteratospermia with severe MMAF, 2020, 57(7): 445–453.
[24] Khelifa MB, Coutton C, Zouari R, Karaouzène T, Rendu J, Bidart M, Yassine S, Pierre V, Delaroche J, Hennebicq S, Grunwald D, Escalier D, Pernet-Gallay K, Jouk PS, Thierry-Mieg N, TouréA, Arnoult C, Ray PF. Mutations in DNAH1, which encodes an inner arm heavy chain dynein, lead to male infertility from multiple morphological abnormalities of the sperm flagella, 2014, 94(1): 95–104.
[25] Li Y, Sha YW, Wang X, Ding L, Liu WS, Ji ZY, Mei LB, Huang XJ, Lin SB, Kong SB, Lu JH, Qin WB, Zhang XZ, Zhuang JM, Tang YG, Lu ZX. DNAH2 is a novel candidate gene associated with multiple morphological abnormalities of the sperm flagella, 2019, 95(5): 590–600.
[26] Tu CF, Nie HC, Meng LL, Yuan SM, He WB, Luo AX, Li HY, Li W, Du J, Lu GX, Lin G, Tan YQ. Identification of DNAH6 mutations in infertile men with multiple morphological abnormalities of the sperm flagella, 2019, 9(1): 15864.
[27] Liu CY, Miyata H, Gao Y, Sha YW, Tang SY, Xu ZL, Whitfield M, Patrat C, Wu H, Dulioust E, Tian SX, Shimada K, Cong JS, Noda T, Li H, Morohoshi A, Cazin C, Kherraf ZE, Arnoult C, Jin L, He XJ, Ray PF, Cao YX, TouréA, Zhang F, Ikawa M. Bi-allelic DNAH8 variants lead to multiple morphological abnormalities of the sperm flagella and primary male infertility, 2020, 107(2): 330–341.
[28] Whitfield M, Thomas L, Bequignon E, Schmitt A, Stouvenel L, Montantin G, Tissier S, Duquesnoy P, Copin B, Chantot S, Dastot F, Faucon C, Barbotin AL, Loyens A, Siffroi JP, Papon JF, Escudier E, Amselem S, Mitchell V, Touré A, Legendre M. Mutations in DNAH17, encoding a Sperm-Specific axonemal outer dynein arm heavy chain, cause isolated male infertility due to asthenozoospermia, 2019, 105(1): 198–212.
[29] Tang SY, Wang X, Li WY, Yang XY, Z Li, Liu WJ, Li CH, Zhu ZJ, Wang LX, Wang JX, Zhang L, Sun XL, Zhi E, Wang HY, Li H, Jin L, Luo Y, Wang J, Yang SM, Zhang F. Biallelic mutations in CFAP43 and CFAP44 cause male infertility with multiple morphological abnormalities of the sperm flagella, 2017, 100(6): 854–864.
[30] Li WY, Wu H, Li FP, Tian SX, Kherraf ZE, Zhang JT, Ni XQ, Lv MR, Liu CY, Tan Q, Shen Y, Amiri-Yekta A, Cazin C, Zhang JJ, Liu WJ, Zheng Y, Cheng HR, Wu YB, Wang JJ, Gao Y, Chen YJ, Zha XM, Jin L, Liu MX, He XJ, Ray PF, Cao YX, Zhang F. Biallelic mutations in CFAP65 cause male infertility with multiple morphological abnormalities of the sperm flagella in humans and mice, 2020, 57(2): 89–95.
[31] Beurois J, Martinez G, Cazin C, Kherraf ZE, Amiri-Yekta A, Thierry-Mieg N, Bidart M, Petre G, Satre V, Brouillet S, Touré A, Arnoult C, Ray PF, Coutton C. CFAP70 mutations lead to male infertility due to severe astheno-teratozoospermia. A case report, 2019, 34(10): 2071–2079.
[32] Auguste Y, Delague V, Desvignes JP, Longepied G, Gnisci A, Besnier P, Levy N, Beroud C, Megarbane A, Metzler-Guillemain C, Mitchell MJ. Loss of calmodulin- and Radial-Spoke-Associated complex protein CFAP251 leads to immotile spermatozoa lacking mitochondria and infertility in men, 2018, 103(3): 413–420.
[33] Martinez G, Beurois J, Dacheux D, Cazin C, Bidart M, Kherraf ZE, Robinson DR, Satre V, Le Gac G, Ka C, Gourlaouen I, Fichou Y, Petre G, Dulioust E, Zouari R, Thierry-Mieg N, Touré A, Arnoult C, Bonhivers M, Ray P, Coutton C. Biallelic variants in MAATS1 encoding CFAP91, a calmodulin-associated and spoke-associated complex protein, cause severe astheno-teratozoospermia and male infertility, 2020, 57(10): 708–716.
[34] Baccetti B, Collodel G, Estenoz M, Manca D, Moretti E, Piomboni P. Gene deletions in an infertile man with sperm fibrous sheath dysplasia, 2005, 20(10): 2790–2794.
[35] He XJ, Liu CY, Yang XY, Lv MR, Ni XQ, Li Q, Cheng HR, Liu WJ, Tian SX, Wu H, Gao Y, Yang CY, Tan Q, Cong JS, Tang DD, Zhang JJ, Song B, Zhong YD, Li H, Zhi WW, Mao XH, Fu FF, Ge L, Shen QS, Zhang MY, Saiyin H, Jin L, Xu YP, Zhou P, Wei ZL, Zhang F, Cao YX. Bi-allelic loss-of-function variants in CFAP58 cause flagellar axoneme and mitochondrial sheath defects and asthenoteratozoospermia in humans and mice, 2020, 107(3): 514–526.
[36] Martinez G, Kherraf ZE, Zouari R, Mustapha SFB, Saut A, Pernet-Gallay K, Bertrand A, Bidart M, Hograindleur JP, Amiri-Yekta A, Kharouf M, Karaouzène T, Thierry-Mieg N, Dacheux-Deschamps D, Satre V, Bonhivers M, Touré A, Arnoult C, Ray PF, Coutton C. Whole-exome sequencing identifies mutations in FSIP2 as a recurrent cause of multiple morphological abnormalities of the sperm flagella, 2018, 33(10): 1973–1984.
[37] Liu WJ, He XJ, Yang SM, Zouari R, Wang JX, Wu H, Kherraf ZE, Liu CY, Coutton C, Zhao R, Tang DD, Tang SY, Lv MR, Fang YY, Li WY, Li H, Zhao JY, Wang X, Zhao SM, Zhang JJ, Arnoult C, Jin L, Zhang ZG, Ray PF, Cao YX, Zhang F. Bi-allelic mutations in TTC21A induce asthenoteratospermia in humans and mice, 2019, 104(4): 738–748.
[38] Liu CY, He XJ, Liu WJ, Yang SM, Wang LB, Li WY, Wu H, Tang SY, Ni XQ, Wang JX, Gao Y, Tian SX, Zhang L, Cong JS, Zhang ZH, Tan Q, Zhang JJ, Li H, Zhong YD, Lv MR, Li JS, Jin L, Cao YX, Zhang F. Bi-allelic Mutations in TTC29 Cause Male Subfertility with Asthenoteratospermia in Humans and Mice, 2019, 105(6): 1168–1181.
[39] Liu CY, Lv MR, He XJ, Zhu Y, Amiri-Yekta A, Li WY, Wu H, Kherraf ZE, Liu WJ, Zhang JJ, Tan Q, Tang SY, Zhu YJ, Zhong YD, Li CH, Tian SX, Zhang ZG, Jin L, Ray P, Zhang F, Cao YX. Homozygous mutations in SPEF2 induce multiple morphological abnormalities of the sperm flagella and male infertility, 2020, 57(1): 31–37.
[40] Dong FN, Amiri-Yekta A, Martinez G, Saut A, Tek J, Stouvenel L, Lorès P, Karaouzène T, Thierry-Mieg N, Satre V, Brouillet S, Daneshipour A, Hosseini SH, Bonhivers M, Gourabi H, Dulioust E, Arnoult C, Touré A, Ray PF, Zhao HQ, Coutton C. Absence of CFAP69 causes male infertility due to multiple morphological abnormalities of the flagella in human and mouse, 2018, 102(4): 636–648.
[41] Ni XQ, Wang JJ, Lv MR, Liu CY, Zhong YD, Tian SX, Wu H, Cheng HR, Gao Y, Tan Q, Chen BL, Li Q, Song B, Wei ZL, Zhou P, He XJ, Zhang F, Cao YX. A novel homozygous mutation in WDR19 induces disorganization of microtubules in sperm flagella and nonsyndromic asthenoteratospermia, 2020, 37(6): 1431–1439.
[42] Kherraf ZE, Cazin C, Coutton C, Amiri-Yekta A, Martinez G, Boguenet M, Mustapha SFB, Kharouf M, Gourabi H, Hosseini SH, Daneshipour A, Touré A, Thierry-Mieg N, Zouari R, Arnoult C, Ray PF. Whole exome sequencing of men with multiple morphological abnormalities of the sperm flagella reveals novel homozygous QRICH2 mutations, 2019, 96(5): 394–401.
[43] Shen Y, Zhang F, Li FP, Jiang XH, Yang YH, Li XL, Li WY, Wang X, Cheng J, Liu MH, Zhang XG, Yuan GP, Pei X, Cai KL, Hu FY, Sun JF, Yan LZ, Tang L, Jiang C, Tu WL, Xu JY, Wu HJ, Kong WQ, Li SY, Wang K, Sheng K, Zhao XD, Yue HX, Yang XY, Xu WM. Loss-of-function mutations in QRICH2 cause male infertility with multiple morphological abnormalities of the sperm flagella, 2019, 10(1): 433.
[44] Lorès P, Coutton C, Khouri EE, Stouvenel L, Givelet M, Thomas L, Rode B, Schmitt A, Louis B, Sakheli Z, Chaudhry M, Fernandez-Gonzales A, Mitsialis A, Dacheux D, Wolf JP, Papon JF, Gacon G, Escudier E, Arnoult C, Bonhivers M, Savinov SN, Amselem S, Ray PF, Dulioust E, Touré A. Homozygous missense mutation L673P in adenylate kinase 7 (AK7) leads to primary male infertility and multiple morphological anomalies of the flagella but not to primary ciliary dyskinesia, 2018, 27(7): 1196–1211.
[45] Coutton C, Martinez G, Kherraf ZE, Amiri-Yekta A, Boguenet M, Saut A, He XJ, Zhang F, Cristou-Kent M, Escoffier J, Bidart M, Satre V, Conne B, Mustapha SFB, Halouani L, Marrakchi O, Makni M, Latrous H, Kharouf M, Pernet-Gallay K, Bonhivers M, Hennebicq S, Rives N, Dulioust E, Touré A, Gourabi H, Cao YX, Zouari R, Hosseini SH, Nef S, Thierry-Mieg N, Arnoult C, Ray PF. Bi-allelic mutations in ARMC2 lead to severe Astheno-Teratozoospermia due to sperm flagellum malformations in humans and mice, 2019, 104(2): 331–340.
[46] Bojesen A, Gravholt CH. Klinefelter syndrome in clinical practice, 2007, 4(4): 192–204.
[47] Krausz C, Casamonti E. Spermatogenic failure and the Y chromosome, 2017, 136(5): 637–655.
[48] Vorona E, Zitzmann M, Gromoll J, Schüring AN, Nieschlag E. Clinical, endocrinological, and epigenetic features of the 46,XX male syndrome, compared with 47,XXY Klinefelter patients, 2007, 92(9): 3458–3465.
[49] Lu CC, Wang Y, Zhang F, Lu F, Xu MF, Qin YF, Wu W, Li SL, Song L, Yang SP, Wu D, Jin L, Shen HB, Sha JH, Xia YK, Hu ZB, Wang XR. DAZ duplications confer the predisposition of Y chromosome haplogroup K* to non-obstructive azoospermia in Han Chinese populations, 2013, 28(9): 2440–2449.
[50] Kasak L, Laan M. Monogenic causes of non-obstructive azoospermia: challenges, established knowledge, limitations and perspectives, 2021, 140(1): 135–154.
[51] Hu ZB, Xia YK, Guo XJ, Dai JC, Li HG, Hu HL, Jiang Y, Lu F, Wu YB, Yang XY, Li HZ, Yao B, Lu CC, Xiong CL, Li Z, Gui YT, Liu JY, Zhou ZM, Shen HB, Wang XR, Sha JH. A genome-wide association study in Chinese men identifies three risk loci for non-obstructive azoospermia, 2011, 44(2): 183–186.
[52] Zhao H, Xu JF, Zhang HB, Sun JL, Sun YP, Wang Z, Liu JY, Ding Q, Lu SM, Shi R, You L, Qin YY, Zhao XM, Lin XL, Li X, Feng JJ, Wang L, Trent JM, Xu CY, Gao Y, Zhang B, Gao X, Hu JM, Chen H, Li GY, Zhao JZ, Zou SH, Jiang H, Hao CF, Zhao YR, Ma JL, Zheng SL, Chen ZJ. A genome-wide association study reveals that variants within the HLA region are associated with risk for nonobstructive azoospermia, 2012, 90(5): 900–906.
[53] Hu ZB, Li Z, Yu J, Tong C, Lin Y, Guo XJ, Lu F, Dong J, Xia YK, Wen Y, Wu H, Li HG, Zhu Y, Ping P, Chen XF, Dai JC, Jiang Y, Pan SD, Xu P, Luo KL, Du Q, Yao B, Liang M, Gui YT, Weng N, Lu H, Wang ZQ, Zhang FB, Zhu XB, Yang XY, Zhang Z, Zhao H, Xiong CL, Ma HX, Jin GF, Chen F, Xu JF, Wang XR, Zhou ZM, Chen ZJ, Liu JY, Shen HB, Sha JH. Association analysis identifies new risk loci for non-obstructive azoospermia in Chinese men, 2014, 5: 3857.
[54] Zhang Y, Qian J, Wu MH, Liu MX, Zhang K, Lin Y, Guo XY, Zhou ZM, Hu ZB, Sha JH. A susceptibility locus rs7099208 is associated with non-obstructive azoospermia via reduction in the expression of FAM160B1, 2015, 29(6): 491–500.
[55] Huang MT, Zhu M, Jiang TT, Wang YF, Wang C, Jin GF, Guo XJ, Sha JH, Dai JC, Wang XM, Hu ZB. Fine mapping the MHC region identified rs4997052 as a new variant associated with nonobstructive azoospermia in Han Chinese males, 2019, 111(1): 61–68.
[56] Qin YF, Ji J, Du GZ, Wu W, Dai JC, Hu ZB, Sha JH, Hang B, Lu CC, Xia YK, Wang CR. Comprehensive pathway-basedanalysis identifies associations of BCL2, GNAO1 and CHD2with non-obstructive azoospermia risk, 2014, 29(4): 860–866.
[57] Ni BX, Lin Y, Sun LD, Zhu M, Li Z, Wang H, Yu J, Guo XJ, Zuo XB, Dong J, Xia YK, Wen Y, Wu H, Li HG, Zhu Y, Ping P, Chen XF, Dai JC, Y Jiang, Xu P, Du Q, Yao B, Weng N, Lu H, Wang ZQ, Zhu XB, Yang XY, Xiong CL, Ma HX, Jin GF, Xu JF, Wang XR, Zhou ZM, Liu JY, Zhang XJ, Conrad DF, Hu ZB, Sha JH. Low-frequency germline variants across 6p22.2-6p21.33 are associated with non-obstructive azoospermia in Han Chinese men, 2015, 24(19): 5628–5636.
[58] Nistal M, Paniagua R, Herruzo A. Multi-tailed spermatozoa in a case with asthenospermia and teratospermia, 1977, 26(2): 111–1118.
[59] Harbuz R, Zouari R, Pierre V, Khelifa MB, Kharouf M, Coutton C, Merdassi G, Abada F, Escoffier J, Nikas Y, Vialard F, Koscinski I, Triki C, Sermondade N, Schweitzer T, Zhioua A, Zhioua F, Latrous H, Halouani L, Ouafi M, Makni M, Jouk PS, Sèle B, Hennebicq S, Satre V, Viville S, Arnoult C, Lunardi J, Ray PF. A recurrent deletion of DPY19L2 causes infertility in man by blocking sperm head elongation and acrosome formation, 2011, 88(3): 351–361.
[60] Pierre V, Martinez G, Coutton C, Delaroche J, Yassine S, Novella C, Pernet-Gallay K, Hennebicq S, Ray PF, Arnoult C. Absence of Dpy19l2, a new inner nuclear membrane protein, causes globozoospermia in mice by preventing the anchoring of the acrosome to the nucleus, 2012, 139(16): 2955–2965.
[61] Guo YS, Jiang JY, Zhang HT, Wen Y, Zhang H, Cui YQ, Tian JY, Jiang M, Liu XF, Wang GG, Li Y, Hu ZB, Zhou ZM, Sha JH, Chen DZ, Yang XY, Guo XJ. Proteomic analysis of Dpy19l2-Deficient human globozoospermia reveals multiple molecular defects, 2019, 13(6): e1900007.
[62] Sha YW, Wang X, Xu XH, Ding L, Liu WS, Li P, Su ZY, Chen J, Mei LB, Zheng LK, Wang HL, Kong SB, You M, Wu JF. Biallelic mutations in PMFBP1 cause acephalic spermatozoa, 2019, 95(2): 277–286.
[63] Barda S, Yogev L, Paz G, Yavetz H, Lehavi O, Hauser R, Doniger T, Breitbart H, Kleiman SE. BRDT gene sequence in human testicular pathologies and the implication of its single nucleotide polymorphism (rs3088232) on fertility, 2014, 2(4): 641–647.
[64] Mbango JFN, Coutton C, Arnoult C, Ray PF, Touré A. Genetic causes of male infertility: snapshot on morphological abnormalities of the sperm flagellum, 2019, 29: 2.
[65] Amiri-Yekta A, Coutton C, Kherraf ZE, Karaouzène T, Tanno PL, Sanati MH, Sabbaghian M, Almadani N, Gilani MAS, Hosseini SH, Bahrami SH, Daneshipour A, Bini M, Arnoult C, Colombo R, Gourabi H, Ray PF. Whole-exome sequencing of familial cases of multiple morphological abnormalities of the sperm flagella (MMAF) reveals new DNAH1 mutations, 2016, 31(12): 2872–2880.
[66] Wang X, Jin H, Han F, Cui Y, Chen J, Yang C, Zhu P, Wang W, Jiao G, Wang W, Hao C, Gao Z. Homozygous DNAH1 frameshift mutation causes multiple morphological anomalies of the sperm flagella in Chinese, 2017, 91(2): 313–321.
[67] Hu JJ, Lessard C, Longstaff C, O'Brien M, Palmer K, Reinholdt L, Eppig J, Schimenti J, Handel MA. ENU-induced mutant allele of Dnah1, ferf1, causes abnormal sperm behavior and fertilization failure in mice, 2019, 86(4): 416–425.
[68] Vernon GG, Neesen J, Woolley DM. Further studies on knockout mice lacking a functional dynein heavy chain (MDHC7). 1. Evidence for a structural deficit in the axoneme, 2005, 61(2): 65–73.
[69] Springer AL, Bruhn DF, Kinzel KW, Rosenthal NF, Zukas R, Klingbeil MM. Silencing of a putative inner arm dynein heavy chain results in flagellar immotility in Trypanosoma brucei, 2011, 175(1): 68–75.
[70] Sha YW, X Wang, Xu XH, Su ZY, Cui YQ, Mei LB, Huang XJ, Chen J, He XM, Ji ZY, Bao HC, Yang XY, Li P, Li L. Novel mutations in CFAP44 and CFAP43 cause multiple morphological abnormalities of the sperm flagella (MMAF), 2019, 26(1): 26–34.
[71] Wu H, Li WY, He XJ, Liu CY, Fang YY, Zhu FX, Jiang HH, Liu WJ, Song B, Wang X, Zhou P, Wei ZL, Zhang F, Cao YX. NovelCFAP43 andCFAP44 mutations cause male infertility with multiple morphological abnormalities of the sperm flagella (MMAF), 2019, 38(5): 769–778.
[72] Shamoto N, Narita K, Kubo T, Oda T, Takeda S. CFAP70 is a novel axoneme-binding protein that localizes at the base of the outer dynein arm and regulates ciliary motility, 2018, 7(9): 124.
[73] Kherraf ZE, Amiri-Yekta A, Dacheux D, Karaouzène T, Coutton C, Christou-Kent M, Martinez G, Landrein N, Tanno PL, Mustapha SFB, Halouani L, Marrakchi O, Makni M, Latrous H, Kharouf M, Pernet-Gallay K, Gourabi H, Robinson DR, Crouzy S, Blum M, Thierry-Mieg N, TouréA, Zouari R, Arnoult C, Bonhivers M, Ray PF. A homozygous ancestral SVA-Insertion-Mediated deletion in WDR66 induces multiple morphological abnormalities of the sperm flagellum and male infertility, 2018, 103(3): 400–412.
[74] Li WY, He XJ, Yang SM, Liu CY, Wu H, Liu WJ, Lv MR, Tang DD, Tan J, Tang SY, Chen YJ, Wang JJ, Zhang ZG, Wang HY, Jin L, Zhang F, Cao YX. Biallelic mutations of CFAP251 cause sperm flagellar defects and human male infertility, 2019, 64(1): 49–54.
[75] Martinez G, Beurois J, Dacheux D, Cazin C, Bidart M, Kherraf ZE, Robinson DR, Satre V, Le Gac G, Ka C, Gourlaouen I, Fichou Y, Petre G, Dulioust E, Zouari R, Thierry-Mieg N, Touré A, Arnoult C, Bonhivers M, Ray P, Coutton C. Biallelic variants in MAATS1 encoding CFAP91, a calmodulin-associated and spoke-associated complex protein, cause severe astheno-teratozoospermia and male infertility, 2020, 57(10): 708–716.
[76] Eddy EM, Toshimori K, O'Brien DA. Fibrous sheath of mammalian spermatozoa, 2003, 61(1): 103–115.
[77] Brown PR, Miki K, Harper DB, Eddy EM. A-kinase anchoring protein 4 binding proteins in the fibrous sheath of the sperm flagellum, 2003, 68(6): 2241–2248.
[78] He XJ, Liu CY, Yang XY, Lv MR, Ni XQ, Li Q, Cheng HR, Liu WJ, Tian SX, Wu H, Y Gao, Yang CY, Tan Q, Cong JS, Tang DD, Zhang JJ, Song B, Zhong YD, Li H, Zhi WW, Mao XH, Fu FF, Ge L, Shen QS, Zhang MY, Saiyin H, Jin L, Xu YP, Zhou P, Wei ZL, Zhang F, Cao YX. Bi-allelic loss-of-function variants in CFAP58 cause flagellar axoneme and mitochondrial sheath defects and asthenoteratozoospermia in humans and mice, 2020, 107(3): 514–526.
[79] Liu WJ, Wu H, Wang L, Yang XY, Liu CY, He XJ, Li WY, Wang JJ, Chen YJ, Wang HY, Gao Y, Tang SY, Yang SM, Jin L, Zhang F, Cao YX. Homozygous loss-of-function mutations in FSIP2 cause male infertility with asthenoteratospermia, 2019, 46(1): 53–56.
[80] Lorès P, Dacheux D, Kherraf ZE, Nsota Mbango JF, Coutton C, Stouvenel L, Ialy-Radio C, Amiri-Yekta A, Whitfield M, Schmitt A, Cazin C, Givelet M, Ferreux L, Mustapha SFB, Halouani L, Marrakchi O, Daneshipour A, Khouri EE, Do Cruzeiro M, Favier M, Guillonneau F, Chaudhry M, Sakheli Z, Wolf JP, Patrat C, Gacon G, Savinov SN, Hosseini SH, Robinson DR, Zouari R, Ziyyat A, Arnoult C, Dulioust E, Bonhivers M, Ray PF, Touré A. Mutations in TTC29, encoding an evolutionarily conserved axonemal protein, result in asthenozoospermia and male infertility, 2019, 105(6): 1148–1167.
[81] Sha YW, Liu WS, Wei XL, Zhu XS, Luo XM, Liang L, Guo TH. Biallelic mutations in Sperm flagellum 2 cause human multiple morphological abnormalities of the sperm flagella (MMAF) phenotype, 2019, 96(5): 385–393.
[82] Liu WS, Sha YW, Li Y, Mei LB, Lin SB, Huang XJ, Lu JH, Ding L, Kong SB, Lu ZX. Loss-of-function mutations in SPEF2 cause multiple morphological abnormalities of the sperm flagella (MMAF), 2019, 56(10): 678–684.
[83] Lehti MS, Sironen A. Formation and function of sperm tail structures in association with sperm motility defects, 2017, 97(4): 522–536.
[84] Li Y, Zhao SG, Yu YH, Ma CL, Zheng Y, Niu Y, Wei DM, Ma JL. Risk factors associated with pre-eclampsia in pregnancies conceived by ART, 2019, 39(6): 969–975.
[85] Wang XL, Wei YH, Fu GL, Li HT, Saiyin H, Lin G, Wang ZG, Chen S, Yu L. Tssk4 is essential for maintaining the structural integrity of sperm flagellum, 2015, 21(2): 136–145.
Progress in the genetic studies of spermatogenesis abnormalities
Xingyu Zhang, Tianyu Zhu, Qingrong Zhang, Xuejiang Guo, Cheng Wang, Guangfu Jin, Zhibin Hu
,,
About 15% couples suffer from infertility, half of which are caused by male factors. Male infertility usually manifests as teratozoospermia, oligospermia and/or asthenospermia, of which the most severe form is azoospermia. In this review, we summarize the recent progress in the study of genetic factors involved in nonobstructive azoospermia and teratozoospermia, Recently, with the rapid development of high-throughput chips and sequencing technologies, many genetic factors of spermatogenesis have been discovered and analyzed. For the nonobstructive azoospermia, genome-wide association studies (GWAS) and high-throughput sequencing revealed many risk loci of nonobstructive azoospermia. For the teratozoospermia, the application of whole-exome sequencing (WES) revealed a series of disease-causing genes, greatly enriching our knowledge of teratozoospermia including multiple morphological abnormalities of the flagella (MMAF). The discovery of lots of disease genes helped the characterization of the pathological mechanisms of male infertility. Therefore, a comprehensive and in-depth understanding of genetic factors in spermatogenesis abnormalities will play important roles in the clinical diagnosis, treatment and genetic counseling of male infertility.
spermatogenesis; genetics; nonobstructive azoospermia (NOA); teratozoospermia; multiple morphological abnormalities of the flagella (MMAF)
2020-12-17;
2021-03-16
国家重点研发计划项目(编号:2016YFA0503300)[supported by the National Key R&D Program of China (No. 2016YFA0503300)]
张星雨,在读本科生,专业方向:预防医学。E-mail: xingyuzhang@njmu.edu.cn
胡志斌,教授,研究方向:生殖医学。E-mail: zhibin_hu@njmu.edu.cn
10.16288/j.yczz.20-343
2021/4/8 10:42:52
URI: https://kns.cnki.net/kcms/detail/11.1913.R.20210407.1424.004.html
(责任编委: 刘默芳)
——水芹主要害虫识别与为害症状

