Clinical assessment and management of liver fibrosis in non-alcoholic fatty liver disease
Alejandro Campos-Murguia, Astrid Ruiz-Margain, Jose A Gonzalez-Regueiro, Ricardo U Macias-Rodriguez
Abstract Non-alcoholic fatty liver disease (NAFLD) is among the most frequent etiologies of cirrhosis worldwide, and it is associated with features of metabolic syndrome; the key factor influencing its prognosis is the progression of liver fibrosis. This review aimed to propose a practical and stepwise approach to the evaluation and management of liver fibrosis in patients with NAFLD, analyzing the currently available literature. In the assessment of NAFLD patients, it is important to identify clinical, genetic, and environmental determinants of fibrosis development and its progression. To properly detect fibrosis, it is important to take into account the available methods and their supporting scientific evidence to guide the approach and the sequential selection of the best available biochemical scores, followed by a complementary imaging study (transient elastography, magnetic resonance elastography or acoustic radiation force impulse) and finally a liver biopsy, when needed. To help with the selection of the most appropriate method a Fagan′s nomogram analysis is provided in this review, describing the diagnostic yield of each method and their post-test probability of detecting liver fibrosis. Finally, treatment should always include diet and exercise, as well as controlling the components of the metabolic syndrome, +/- vitamin E, considering the presence of sleep apnea, and when available, allocate those patients with advanced fibrosis or high risk of progression into clinical trials. The final end of this approach should be to establish an opportune diagnosis and treatment of liver fibrosis in patients with NAFLD, aiming to decrease/stop its progression and improve their prognosis.
Key Words: Non-alcoholic fatty liver disease; Liver fibrosis; Clinical assessment; Diagnosis; Treatment; Test accuracy
INTRODUCTION
Non-alcoholic fatty liver disease (NAFLD) is one of the main etiologies of cirrhosis worldwide[1], given its close relationship with features of the metabolic syndrome, including obesity and insulin resistance, NAFLD is becoming one of the most frequent and the fastest-growing cause of chronic liver disease in the world, and it is expected to grow exponentially in the following years, thus increasing the health system and economic burden[2,3].
NAFLD is defined as the accumulation of fat in the liver (> 5%), after the exclusion of other potential causes such as alcohol, viral infections, and drugs, among others. The next entity in the spectrum of the disease is non-alcoholic steatohepatitis (NASH) which is defined as the presence of hepatocellular injury and cell death, with lobular and portal inflammation. The final stages, fibrosis and cirrhosis arise as a consequence of the deposition of collagen and subsequent vascular remodeling[4,5]. Finally, within the spectrum of the disease, hepatocellular carcinoma should be included as a complication after this series of pathophysiological events.
The frequency of hepatic steatosis varies significantly according to ethnicity, being more frequent in Hispanics (45%) than in Caucasians (10%-33%) and African Americans (24%), which is probably related to the higher prevalence of obesity and insulin resistance in this ethnic group, as well as the influence of genetic factors[3,6,7]. Genetic polymorphisms in NAFLD have been identified as associated with the presence of features of the metabolic syndrome (glucose and lipid metabolism, as well as hypertension) and inflammation[8]. The prevalence of NAFLD in high-risk groups, like type 2 diabetes mellitus (T2DM) is even higher, being present in almost 70% of this group[9]. Other high-risk populations include those with hypertension, obesity, and dyslipidemia[2,10]. In terms of fibrosis, according to the National Health and Nutrition Examination Survey up to 10.3% of the patients with NAFLD have advanced fibrosis[11].
The presence of fibrosis rather than the diagnosis of steatohepatitis is the most relevant feature associated with liver-related events and overall mortality. This effect is seen even in the early stages of fibrosis, showing a stepwise increase in adverse outcomes as the stage of fibrosis progresses[11-14]. Fibrosis parallels the development of the two major components of chronic liver diseases: Portal hypertension and functional hepatocyte insufficiency. However, the importance of liver fibrosis is beyond “liver prognosis” itself, as it is associated with other adverse clinical outcomes, including cardiovascular events[15], ischemic stroke[16], metabolic complications[17], and all-cause and cardiovascular mortality[18,19]. This could be explained by a more pronounced systemic inflammation profile influencing different organs and systems, and the interaction between them leading to further inflammation and activation of the immune response.
Therefore, early recognition and proper management of liver fibrosis in NAFLD are of major importance. In this review, we will address the risk factors for NAFLD and the risk of progression, as well as the currently available methods used to assess liver fibrosis and the new treatments available. This will help the clinicians to early recognize liver fibrosis in patients with NAFLD and use the best available methods for its evaluation and management.
DETERMINANTS OF FIBROSIS PROGRESSION
It is important to note that both biopsy-proven NASH and simple steatosis can progress to liver fibrosis, which contradicts the classic theory in which NAFLD has a benign curse while NASH has a more aggressive one[20]. Significant fibrosis can be observed in approximately one-third of patients with NAFLD in the absence of NASH[21]. The etiology of fibrosis in non-NASH patients is not entirely clear, although there are several theories; it has been hypothesized that these cases represent a form of NASH in remission as aminotransferase levels improve regardless of whether or not fibrosis progress or that T2DM by itself could be fibrogenic[22-24].
The progression of fibrosis in patients with NAFLD and NASH is variable, on average 40% have progression of fibrosis over a mean period of 3-6 years. Despite the incidence of fibrosis, the change is slow, being about 0.02 stages overall per year. There is considerable variability, with one out of six patients having a relatively rapid progression of more than 0.5 stages per year, and some patients progressing from no fibrosis to advanced fibrosis (F3-F4) on average in 12 years[23,24]. The determinants of fibrosis progression can be divided into genetic and clinical determinants.
Genetic determinants
Genetic factors are of major importance in the development[25]and the risk of fibrosis progression in NAFLD[26]. At least four genetic variants have been associated with fibrosis progression. The variant rs738409 in the human patatin-like phospholipase domain-containing 3 (PNPLA3) gene located in chromosome 22[25], is the best described and major genetic determinant of liver fibrosis development and progression in NAFLD[21,26-28]. The variant rs58542926, of the transmembrane 6 superfamily member 2 (TM6SF2) gene has also been implicated in de progression of fibrosis in NAFLD, however, the data is conflicting[28-30], probably due to the heterogeneity of the populations evaluated[27]. It is possible, however, that there is an additive effect of TM6SF2 and PNAPLA3 variants on the histological severity of NAFLD[27]. Finally, the rs641738 C>T genetic variant in Membrane-bound Oacyltransferase domain containing 7 (MBOAT7) and the variation in the glucokinase regulator have also been associated with higher severity of necroinflammation and fibrosis[31,32].
In a cohort of 515 patients with NAFLD, PCR-based assays were used to genotype the PNPLA3, TM6SF2, and MBOAT7 variants. The three variants were associated with increased liver injury. The TM6SF2 variant was associated predominantly with hepatic fat accumulation, whereas the MBOAT7 polymorphism was linked to fibrosis. The PNPLA3 polymorphism conferred a higher risk for both steatosis and fibrosis[33].
Clinical and environmental determinants
There are several clinical determinants of fibrosis progression in NAFLD and NASH, however, by far the presence of insulin resistance and T2DM are the major predictors of fibrosis progression[23]. Other clinical determinants of fibrosis progression rate are body mass index (BMI), sarcopenia, absence of treatment with renin-angiotensin system (RAS) inhibitors, non-obese NASH patients, alanine aminotransferase (ALT) levels above the upper limit of normal (ULN), and the severity of hepatic fat accumulation[22-24,34-36].
EVALUATION OF LIVER FIBROSIS IN NAFLD
Clinical evaluation
Most patients diagnosed with NAFLD are asymptomatic, with only a few of them complaining of mild upper quadrant pain related to fatty infiltration of the liver. Three general scenarios could arise the suspicion of NAFLD, including abnormalities on imaging performed for other reasons, abnormal liver enzymes, or based on high-risk features of NAFLD such as metabolic syndrome[37]. There are no specific signs or symptoms related to the early stages of NAFLD fibrosis, once the patient presents with advanced fibrosis, portal hypertension or/and liver dysfunction they will develop specific symptoms of hepatic decompensation (ascites, splenomegaly, spider angiomas, palmar erythema, caput medusae, hepatic encephalopathy, and jaundice). Since most of the cases will present as NAFLD alone or mild fibrosis the physician should have a high suspicion index in patients with high-risk factors such as insulin resistance, T2DM, hypertension, obesity, and dyslipidemia. Some of the clinical hallmarks the clinician should pay attention to include acanthosis nigricans and skin tags, usually located in the lateral area around the neck and axillae, as well as hirsutism, polycystic ovary syndrome, and a high waist/hip ratio[38,39]. In the case of obstructive sleep apnea (OSA), clinical evaluation includes the presence of fatigue, sleepiness and snoring, and the use of the Epworth Sleepiness scale, measurement of neck circumference, and Mallampati scale evaluation[40]. Although these features are not specific of fibrosis, their presence should raise suspicion of a high-risk phenotype, rendering further assessment mandatory.
Blood tests and scores
Serologic tests for fibrosis detection can be divided into direct and indirect markers. Indirect markers of fibrosis aim to obtain information from the overall liver function, whereas direct serologic markers are molecules that are obtained directly from byproducts or products related to collagen deposition[37].
Indirect markers, such as routine laboratory tests are unreliable to accurately and promptly detect liver fibrosis in NAFLD unless advanced fibrosis and subsequent portal hypertension exist when thrombocytopenia and high levels of aspartate aminotransferase (AST) are observed. However, these variables provide a rather overt diagnosis, relying on the personal experience of their interpretation.
Several biomarkers have been proposed as direct markers of liver fibrosis, one of the most described is cytokeratin 18 (CK-18). Cytokeratins are proteins of keratincontaining intermediate filaments located in the intracytoplasmic cytoskeleton of epithelial tissue, being CK-18 the predominant in the liver and released as a consequence of increased apoptosis and associated with fibrosis in NASH[41]. CK-18 has been investigated as a potential biomarker of severity and liver fibrosis in different etiologies[42-44]. In NAFLD, CK-18 increased significantly with steatosis and fibrosis stages, however, it has a low sensitivity and specificity, ranging from 54% to 62% and 69% to 85%, respectively, limiting its use as a reliable diagnostic tool[43,45].
Other direct markers of liver fibrosis are collagens and their fragments since they represent the principal component of the fibrotic scars. The most validated biomarkers for the measurement of type III collagen formation are the amino-terminal propeptide of procollagen type III (PIIINP) and N-terminal pro-collagen III peptide (PRO-C3) biomarkers[46]. PRO-C3 which is a collagen fragment is significantly higher in NASH patients with advanced fibrosis than those without advanced fibrosis. PRO-C3 levels have a direct correlation with worsening of liver fibrosis, in the same manner, PRO-C3 levels decrease with fibrosis improvement, identifying patients with active fibrogenesis. It is noteworthy that patients with advanced fibrosis can have an inactive disease, implying lower production of collagen and therefore having normal levels of PRO-C3[47]. The European Liver Fibrosis project developed the enhanced liver fibrosis (ELF™) test, a blood-based score that was comprised of ELISA measurements of hyaluronic acid, PIIINP, and tissue inhibitor of matrix metalloproteinases; this score has a sensitivity and specificity for severe liver fibrosis of 78% and 76%, respectively[48].
None of the currently available biomarkers by itself has sufficient accuracy for diagnosing fibrosis which is why predictive scores play an important role in providing a cutoff able to discern between no fibrosis or the presence of advanced fibrosis. Among the predictive scores fibrosis-4 index (FIB-4) index, NAFLD fibrosis score (NFS), the BARD score, FibroTest, HepatoScore, hepamet fibrosis score (HFS), and AST to platelets ratio index (APRI) score, are the most widely used (Table 1)[49,50].
FIB-4 index:FIB-4 index is a complex marker, based on age, platelet count, AST, and ALT, it was developed in 2006 as a non-invasive panel to stage liver disease in subjects with human immunodeficiency virus and hepatitis C virus co-infection[51]. In NAFLD, a cutoff value < 1.45 has a negative predictive value (NPV) of 90% and a sensitivity of 84% to exclude advanced fibrosis. A cutoff > 3.25, had a positive predictive value (PPV) of 65% and a specificity of 68%[50,52]. This indicates that 84% of patients with suspected NAFLD-related advanced fibrosis would be identified by the FIB-4 index and avoid a liver biopsy. However, more than 30% of NAFLD patients diagnosed as non-advanced fibrosis by the FIB-4 index may have advanced fibrosis in liver biopsy. Given that about a third of patients could be misdiagnosed as non-advanced fibrosis, FIB-4 cannot replace liver biopsy[53,54].

Table 1 Diagnostic performance of blood tests and scores for fibrosis assessment methods from studies made in patients with nonalcoholic fatty liver disease
NFS score:NFS score includes age, glycemia, BMI, platelet count, albumin, AST, and ALT; unlike other prognostic scores in NAFLD which were created for other etiologies, NFS was developed in 733 patients with biopsy-proven NAFLD[55]. NFS uses two diagnostic cutoffs, the low cutoff score (-1.455) to exclude advanced fibrosis (NPV 88%-93%), and the high cutoff score (0.676) to diagnose advanced fibrosis (PPV 82%-90%)[55], leaving one-third of patients in a “grey zone” where liver biopsy is still required[56]. In further validations, NFS has remained with good NPV (81%-98%) for advanced fibrosis (F3-F4), however, PPV has had more fluctuation (50%-100%)[54,57-59].
The BARD score:BARD score is a simple index defined by the presence of three clinical and laboratory parameters, BMI (> 28 kg/m2, 1 point), AST/ALT ratio (> 0.8, 2 points), and diabetes (1 point), ranging from 0 to 4. The BARD score was developed in 2008 in 827 patients with NAFLD. The result of the score is dichotomized as 0-1 and 2-4, for low and high risk of advanced fibrosis, respectively[60]. The PPV and NPV range from 26% to 68% and 81% to 96%, respectively[58-60]; while the sensitivity and specificity ranged from 51% to 88% and 66% to 88%, respectively[54,58,59]. The score can be easily derived from clinical data, however, as the BARD score takes into account the BMI, it may be less reliable for excluding the presence of advanced fibrosis in countries where subjects with NAFLD are not overweight or obese[61].
APRI score:This score is a simple ratio that takes into account the value of AST and platelets; it was developed in 2003 to predict liver fibrosis in patients with hepatitis C[62]. With an APRI threshold of 1.5, the sensitivity and specificity are 84.0% and 96.1%, respectively, for advanced fibrosis in NAFLD[63]. The score is a reliable tool to differentiate between patients with no fibrosis and patients with advanced fibrosis/cirrhosis, but it cannot reliably discriminate between intermediate stages of fibrosis. The area under the curve (AUROC) for the APRI score in patients with NADLF ranges from 0.8307 to 0.95[64,65].
FibroTest:FibroTest is a commercial algorithm that has shown good predictive values for diagnosing advanced fibrosis (AUROC = 0.81-0.88) in patients with NAFLD, however, its diagnostic accuracy may be affected by acute inflammation, sepsis or extrahepatic cholestasis[66,67]. This score integrates the value of five serum biomarkers (α-2-macroglobulin, haptoglobin, apolipoprotein A1, γ-glutamyltranspeptidase (GGT), and total bilirubin, adjusted for sex and age) for liver fibrosis (FibroTest), plus ALT for the necroinflammatory activity (ActiTest), into an equation-based algorithm, obtaining finally a result between 0 and 1, where higher values indicate a greater probability of liver fibrosis[68]. The AUROC to differentiate between fibrosis stages is lower, for intermediate stages, F1vsF2 is 0.66 and for advanced fibrosis, F3vsF4 is 0.69[67].
Hepascore:Hepascore is an algorithm to detect fibrosis in many chronic liver diseases, it combines clinical variables including age and sex with blood-based parameters such as bilirubin, gamma-glutamyl transferase, hyaluronic acid, and a-2-macroglobulin. In patients with NAFLD, a threshold of 0.37 helps to identify individuals with advanced fibrosis, with an AUROC of 0.778, NPV, and PPV of 79% and 63%, respectively[56].
Hepamet fibrosis scoring system:The HFS is the most recently developed score system, created from data of 2452 patients with biopsy proven-NAFLD at medical centers in Spain, Italy, France, Cuba, and China from the Hepamet registry. The HSF is calculated by a complex formula, using the following items: Patient sex, age, homeostatic model assessment score, presence of diabetes, AST levels, albumin, and platelet count; it is available online for public usage. HSF had an AUROC of 0.85 to discriminate between advanced fibrosis and no advanced fibrosis. In the validation cohort, a cut-off of 0.12 for low risk and 0.47 for high risk, identified patients with and without advanced fibrosis with 97.2% specificity, 74% sensitivity, a 92% NPV, and a 76.3% PPV. HFS is not affected by patient age, BMI, hypertransaminasemia, or the presence of T2DM. HFS was developed and validated in a large and heterogeneous population, giving it an advantage above other scores[69]. Recently, this score was evaluated in a cohort of 49 patients with morbid obesity undergoing bariatric surgery, a cut-off 0.47 for advanced fibrosis had a sensitivity of 11%, a specificity of 100%, a PPV of 100%, and an NPV of 83%[70].
After analyzing the data related to prediction scores, it is clear that overall, predictive scores for fibrosis have a good NPV for excluding advanced fibrosis with low PPV. Therefore, these scores may be confidently used for baseline risk stratification to exclude advanced fibrosis; however, due to their low specificity, a result of advanced fibrosis based in these scores should be further confirmed by other methods, such as imaging studies or liver biopsy depending upon availability[4].
Imaging studies
Conventional imaging studies such as ultrasonography (US), computed tomography (CT), and magnetic resonance imaging (MRI) are available techniques for the detection of fatty liver[71,72]. However, they have low to moderate accuracy to identify liver fibrosis[73,74].
Currently, elastography has become the non-invasive method of choice to quantify liver stiffness (elasticity). The basic principle is that fibrotic tissue is stiffer than normal tissue, therefore the waves spread faster in fibrotic tissue than in the normal liver. This technique uses a force to move the hepatic tissue, measuring this movement by US or MRI[75], giving the value of liver stiffness measurement (LSM) in kilopascal (kPa)[76]. The LSM obtained depends on the frequency of waves applied, therefore, it cannot be compared between different methods.
Transient elastography:One dimensional transient elastography (TE) is a noninvasive ultrasound-based method that uses shear wave velocity, providing LSM and controlled attenuation parameter (CAP) for the evaluation of steatosis. The benefit of TE compared with liver biopsy is that it measures a larger region, 100 times larger than the volume of the tissue obtained by biopsy[64]. The limitation is that occasionally measurements cannot be obtained in obese patients[49,77], reliable LSM measurements are obtained in about 80% to 90% of NASH/NAFLD patients[78,79]. It is noteworthy that at least three hours of fasting are necessary before evaluation with TE since a significant increase in LSM has been observed in patients with less than two hours of fasting[80]. In addition to this, there are quality standards that should be met to consider the result as adequate: At least 10 valid measurements (60% of success) and an IQR < 30%[81].
The cutoff value of CAP for differentiating hepatic steatosis is highly variable due to the heterogeneity of the populations evaluated, ranging from 214 to 289 dB/m, with moderate to high sensitivity and specificity (78%-91.9% and 79%-85.7%), as well as moderate to good predictive values (PPV of 77%-85.0% and NPV of 76%-92.3%)[78,82,83]. In one meta-analysis, in which 2735 patients were included, the optimal CAP cutoff was 248 dB/m, however, only 7% of the population had NAFLD diagnosis. Siddiquiet al[84], reported a prospective study of 393 patients with biopsy-proven NAFLD, in whom TE was performed, the median CAP scores for steatosis grades were 306 for S1 and, 340 for S2 and S3, the best cutoff, balancing sensitivity and specificity, to differentiate steatosis from no-steatosis was CAP of 285 dB/m, with a sensitivity of 80% and a specificity of 77%. It is noteworthy that the prevalence in the population, the etiology of liver disease, diabetes, and BMI deserve consideration when interpreting CAP, and perhaps lower thresholds should be applied to patients with high pretest probability, and higher thresholds in groups with low pretest probability[78].
For the detection of advanced fibrosis, TE has excellent accuracy (F3: Sensitivity 85%, specificity 82%, and F4: Sensitivity 92%, specificity 92%), whereas it has moderate accuracy for significant fibrosis (F2: Sensitivity 79%, specificity 75%)[61]. The diagnostic performance according to the AUROC values for the diagnosis of severe fibrosis and cirrhosis is good, ranging from 0.86 to 0.87; for the diagnosis of intermediate fibrosis (≥ F2), the AUROC is 0.82[85]. In general, the performance of TE is superior to prediction scores[63]. According to Siddiquiet al[84], a cutoff value of < 6.5 kPa excludes fibrosis with high accuracy (NPV 0.91), as well as a cutoff < 12.1 kPa excludes cirrhosis in NAFLD patients. Therefore we suggest that the cutoffs for the evaluation of steatosis and fibrosis should be the ones given by this work since the study evaluated only NAFLD patients with confirmed histology.
It is not clear if TE should be performed at defined intervals; recently a cohort of 611 patients with T2DM was followed with serial TE for 3.5 years, the majority of patients had NAFLD at baseline, and another 50% developed NAFLD during the period evaluated. Around 20% had advanced liver fibrosis at baseline, but only 4% developed advanced fibrosis in 3 years. Baseline BMI, ALT, and ∆ALT independently predicted LSM increase[80]. Based on this data, we could advise performing TE every three years only in patients with risk factors such as T2DM, elevated BMI, and ALT above the UNL, otherwise, TE could be performed every 5 years.
Acoustic radiation force impulse:Acoustic radiation force impulse (ARFI) is an elastography technique that uses modified commercially available ultrasound machines, combining both elastography and conventional B-mode US. The liver is mechanically excited using short-duration acoustic pulses with a frequency of 2.67 MHz to generate localized tissue displacements in tissue[86]. Mean normal values range from 0.8 to 1.7 m/s and mean values indicating advanced fibrosis range from 1 to 3.4 m/s, with a clear overlap of values[87]. According to Fierbinteanu-Braticeviciet al[88], the optimal cutoff value to identify advanced fibrosis is 1.54 m/s, with sensitivity and specificity of 97% and 100% respectively. In a meta-analysis of 13 studies, including 1163 patients, for detection of intermediate fibrosis (≥ F2), the summary sensitivity was 74% and the specificity was 83%, for the diagnosis of cirrhosis, the sensitivity was 87% and specificity was 87%. The diagnostic odds ratio of ARFI and TE did not differ significantly in the detection of significant fibrosis[89]. In NAFLD patients, several studies have investigated the best cutoff for the detection of advanced fibrosis, ranging from 1.15 m/s to 1.77 m/s with varying sensitivities (59%-90%) and specificities (63%-91%)[85,90-93].
Magnetic resonance elastography:Magnetic resonance elastography (MRE) is a phase contrast-based MRI technique[94]in which a passive drive generates vibrations at 60 Hz by varying acoustic pressure waves transmitted from an active driver device[95]. LSM is performed by drawing region of interest (ROI) on the elastograms, which cover regions of the liver with sufficient wave amplitude. The mean ROIs from 4 slices are averaged and reported as the mean LSM of the liver[96]. An advantage of MRE is that the area measured in the liver is larger than in TE or liver biopsy, which can avoid the sampling variability caused by the heterogeneity of advanced fibrosis[77]. The cutoff LSM for the detection of advanced fibrosis in NAFLD patients varies according to the population evaluated, ranging from 3.6 to 4.8 kPa with high sensitivity and specificity[63,77,97-99]. The diagnostic accuracy of MRE for liver fibrosis in NAFLD is higher than that of clinical scoring systems and TE[77]. A 15% increase in liver stiffness on repeated MRE may be associated with histologic fibrosis progression (≥ 1 stage) and progression from early fibrosis to advanced fibrosis[100]. Magnetic resonance also quantifies hepatic steatosis with high accuracy by measuring the proton density fat fraction which is the fraction of MRI-visible protons bound to fat divided by all protons in the liver[101].
Combined methods
Recently there have been reports of test combinations for better detection of liver fibrosis in NAFLD. The serial combination of NFS or FIB-4 with TE improves the performance of the tests when applying the second test only in patients in the uncertainty area of the first test. This algorithm is a low-cost alternative that can be applied in daily practice allowing the correct classification of a high proportion of NAFLD patients[102].
The FibroScan-AST (FAST) score, was recently proposed as a reliable algorithm to identify among patients with NAFLD, those with NASH, intermediate liver fibrosis (≥ F2), and elevated NAFLD activity score (NAS ≥ 4) (i.e., those at high risk of progression of the disease). The results of the FAST score range from 0 to 1, and derive from a logarithm-based equation, considering 3 values, AST, CAP, and LSM, and are further divided into three zones, according to the value: Rule-out (≤ 0.35), gray (0.35-0.67) and rule-in (≥ 0.67). The performance of the test is good, with an AUROC of 0.85 (0.83-0.87), and a sensitivity, NPV, specificity, and PPV of 0.89, 0.94, 0.92, and 0.69, respectively[103].
Determining which method is best for the patient relies on several factors: The availability of the methods in a daily clinical care setting, the performance of the operator, the prevalence of the disease in the specific population, and the preference of the patient. As shown in some studies, a combination of non-invasive methods could help to improve the accuracy of the diagnosis.
Liver biopsy
Liver biopsy is the gold standard for the diagnosis and assessment of the severity of liver fibrosis in NAFLD[4]. In the context of NAFLD, liver biopsy is usually obtainedviaa percutaneous approach using ultrasound guidance. The use of a 16 gauge or wider needle is recommended for the biopsy. An adequate histology specimen should have at least 2 cm long and comprising 10 or more portal tracts, and the review of specimens should be carried out by two pathologists[104,105].
Specimens should be processed with hematoxylin and eosin staining and specifically with Masson’s trichrome or Sirius red staining to assess fibrosis. Liver fibrosis has a singular pattern in NAFLD, frequently beginning in the pericentral zone 3 and eventually progressing to bridging fibrosis and cirrhosis[106]. There are several systems to evaluate NAFLD biopsies. The NAFLD activity score was developed as a tool to measure changes in NAFLD during therapeutic trials, the maximum score is 8, comprises steatosis (0-3), ballooning (0-2) and lobular inflammation (0-3), with a major drawback as it does not take into account the amount of fibrosis[104]. For the evaluation of fibrosis in NAFLD, there are three scoring assessment systems, the Brunt system, the NASH Clinical Research Network (CRN) system, and the Steatosis, Activity, Fibrosis (SAF) system[107,108]. In the Brunt system, fibrosis stages are divided into four, stage 1, zone 3 perisinusoidal fibrosis; stage 2, portal fibrosis; stage 3, bridging fibrosis; and stage 4, liver cirrhosis[109]. The NASH CRN system is a modification of the Brunt system in which stage 1 is subdivided into three stages, to include a distinction between delicate (1a) and dense (1b) perisinusoidal fibrosis, and to detect portal-only fibrosis, without perisinusoidal fibrosis (stage 1c), showing reasonable interrater agreement among experienced pathologists[110]. The SAF system which includes the NASH CRN system was built from a cohort of morbidly obese patients undergoing bariatric surgery. This system separately assesses the grade of steatosis (S0 to S3), the grade of activity (A0 to A4) and the stage of fibrosis (F0 to F4), the latter according to the NASH-CRN staging system, with the single modification of pooling the three substages (1a, 1b, and 1c) into a single F1 score[111]. Some computerized techniques also have been developed for the quantification of fibrosis in NAFLD/NASH histology, representing promising and accurate methods for the evaluation of liver fibrosis in this group of patients[112,113].
Liver biopsy has some relevant inconveniences, including the fact that only 1/50000 of the whole liver tissue is sampled, therefore sampling error is a major concern. To prevent sampling error, it is important to collect a sufficient amount of tissue; the use of a thick needle and a collection of at least 2 samples are recommended[114]. Interobserver variability of the pathologist is another important concern, for the diagnosis of liver fibrosis in NAFLD, pathologists have a moderate inter-observer agreement (κ scores of 0.52-0.56) for fibrosis stage[115], therefore, at least two pathologists should review the specimens. Finally, complications associated with percutaneous liver biopsy are rare, only 1% to 3% of patients require hospitalization and the mortality rate is extremely low, 1 in 10000 to 1 in 12000 liver biopsies[114].
Due to the high prevalence of steatosis, it is not practical to perform a liver biopsy in every patient with NAFLD. The European Association for the Study of the Liver (EASL) guidelines recommend that for the identification of advanced fibrosis or cirrhosis, serum biomarkers/scores and/or TE are less accurate, and it is important to confirm these advanced stages by liver biopsy. According to the clinical context and in selected patients at high risk of liver disease progression, monitoring should include a repeat liver biopsy after at least a 5-year follow-up[4]. The American Association for the Study of Liver Disease (AASLD) guidelines recommend performing a liver biopsy when the diagnosis is not clear (i.e., there is a suspicion of another liver disease), or in those with a high probability of NASH/advanced fibrosis, especially in those considered for treatment with vitamin E or pioglitazone[116].
With the evidence above mentioned, it seems more practical to perform a liver biopsy in NAFLD only in patients with high suspicion of advanced fibrosis and/or rapid progression, when other causes of liver damage cannot be ruled out or for clinical trials exploring new-generation drugs.
Tables 1 and 2 summarize the diagnostic performance of the previously mentioned methods. It is of great importance to consider that some methods have different cutoff values, as can be seen in the table, and each cutoff holds different predictive values. This must be taken into account at the time of the patient's evaluation in the clinical practice and it is especially important to use the same cutoff if follow-up is to be done.
To further address this issue, we analyzed the diagnostic capabilities of several methods based on Fagan′s nomogram, which considers the pre-test probability, or prevalence, of the disease we are looking to diagnose and, uses the likelihood ratio that derives from the sensitivity and specificity reported in published studies, to estimate a final post-test probability, which simply represents the probability of a patient truly having the condition of interest if the test is positive or truly not having the disease if the test is negative.
Figure 1A depicts the analysis of all TE studies presented in Table 2, and Figure 1B shows the MRE studies. As mentioned before, each study proposes different cutoffs, therefore it is of great importance to evaluate the behavior of each one by its post-test probability and not only by their sensitivity and specificity. In Figure 1A one can conclude that overall, the best cutoff to use is the one derived from the study by Imajoet al[77]where > 11.7 kPa has the highest probability to detect advance fibrosis if the patient actually has it, and the lowest probability of diagnosing it if the patient does not have advanced fibrosis. With respect to MRE studies in Figure 1B, two of the cutoffs have a great performance with a post-test probability > 80%; overall the best cutoff derives from the study of Kimet al[98]with a post-test probability of diagnosing advanced fibrosis of 84% in patients with the disease when the employed cutoff is >4.15 kPa. Interestingly once we compare both methods in Figure 1 it is apparent that MRE has better diagnostic capabilities, hence we created a Fagan′s nomogram using all the methods presented in Tables 1 and 2, to evaluate which one could diagnose or exclude AF better (Figure 2). Since the study by Boursieret al[56]evaluated most of the methods in their study we used those results to create the graph, for the remaining methods: MRE, HFS and AFRI, we selected the study by Xiaoet al[63], Ampueroet al[69], and Cassinottoet al[85]respectively. The analysis of this graph shows that the best methods with higher post-test probability to detect advanced fibrosis in patients with the disease in order of detection are MRE, Hepamet fibrosis scoring system, NFS score, APRI, TE, fibrometer and FIB-4, the best methods to exclude advanced fibrosis in patients without the disease are, MRE, AFRI, Fibrometer and TE.
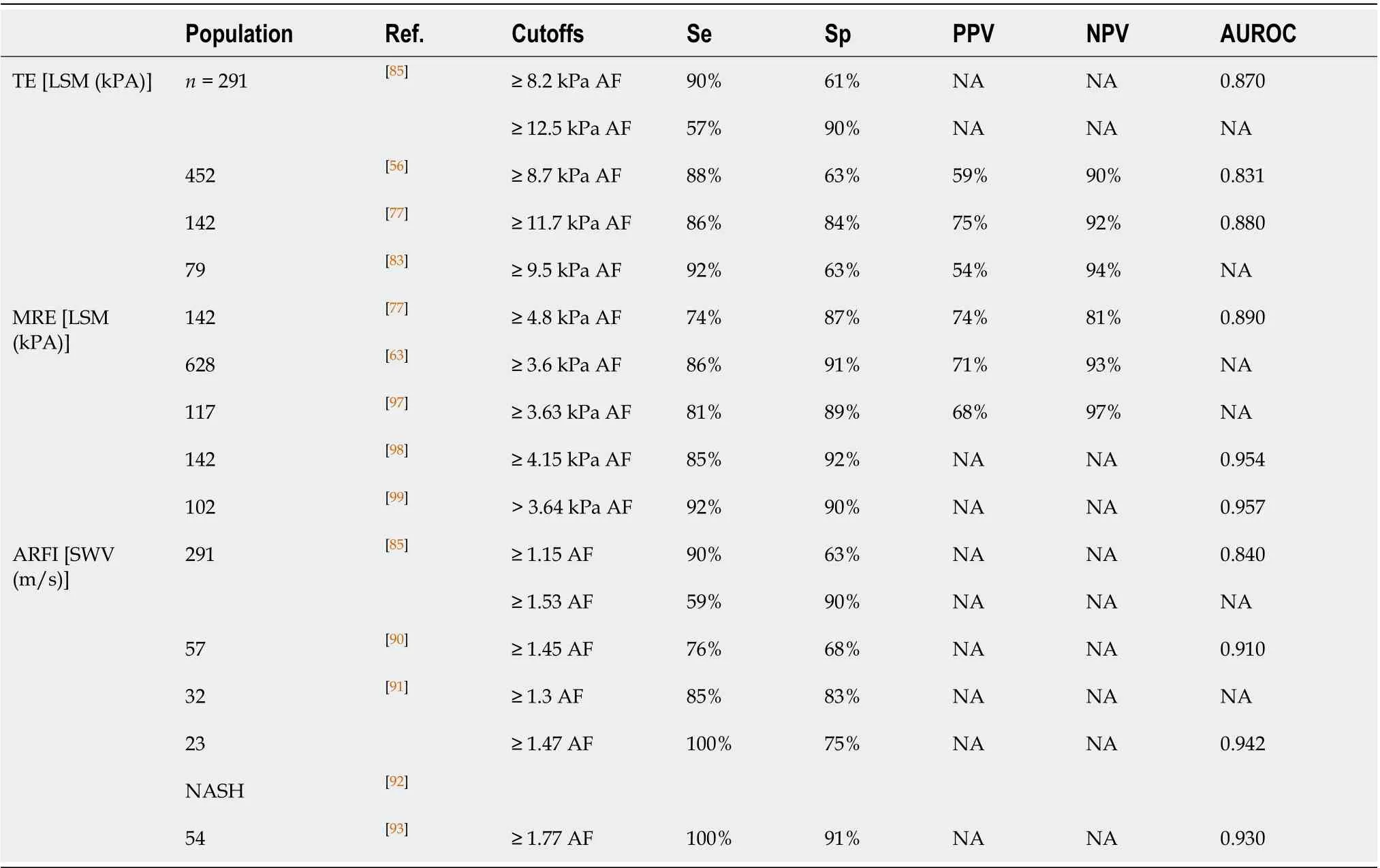
Table 2 Diagnostic performance of imaging studies for fibrosis assessment methods from studies made in patients with non-alcoholic fatty liver disease
Finally, after a comprehensive evaluation we propose a stepwise assessment of fibrosis in NAFLD patients, according to the available data, which can be found in Figure 3.
MANAGEMENT OF LIVER FIBROSIS IN NAFLD
The appropriate management of patients with NAFLD and fibrosis should be a comprehensive treatment that takes into account three major components: The treatment of underlying metabolic diseases, weight loss (WL), and pharmacological therapy (Figure 4).
Treatment of underlying metabolic diseases
As referred before, the importance of liver fibrosis is beyond the liver prognosis[18,19]. Therefore, to improve the prognosis of patients with NAFLD and fibrosis the treatment of concomitant diseases must be a priority.
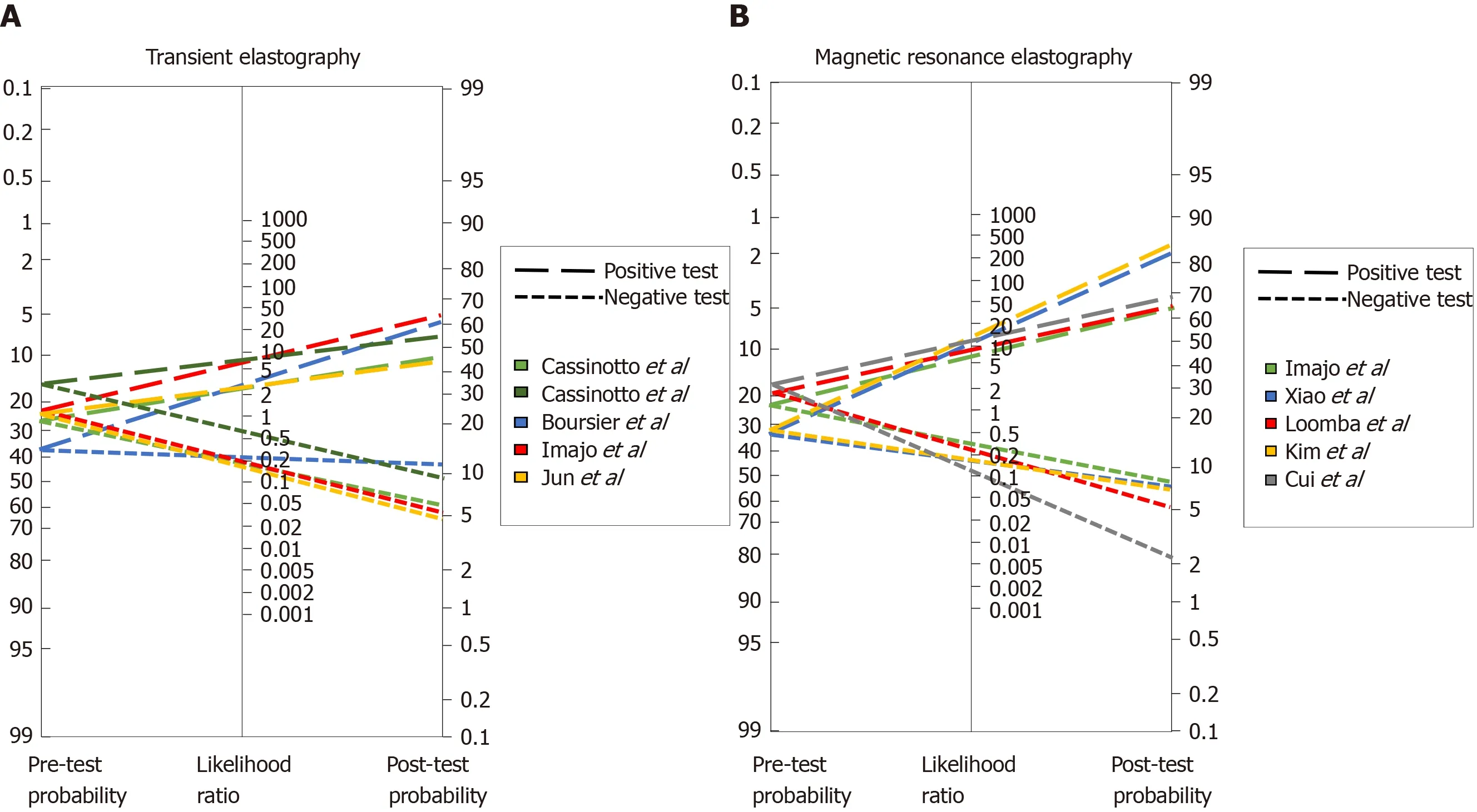
Figure 1 Fagan′s nomogram for (A) transient elastography and (B) magnetic resonance elastography studies.
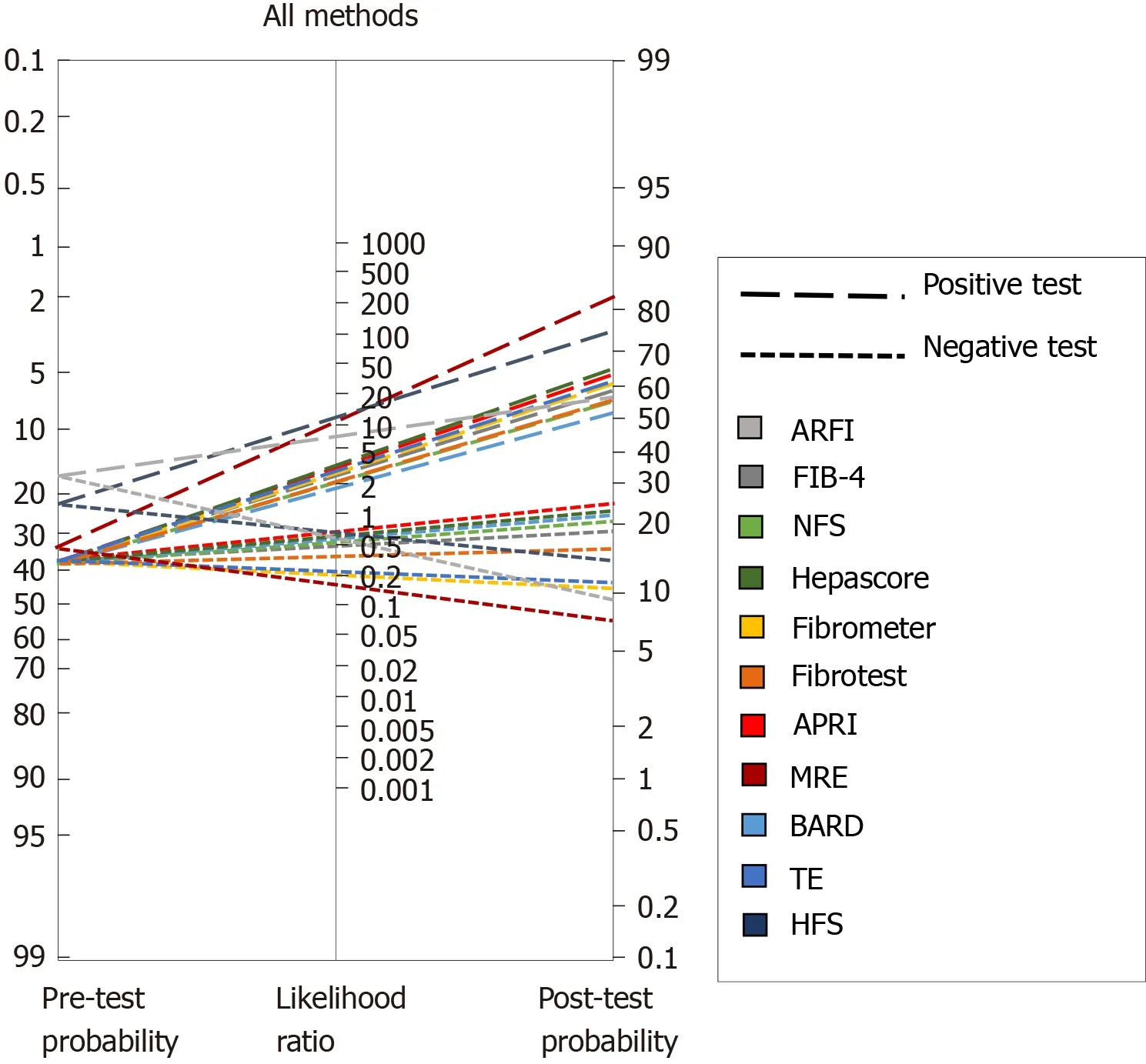
Figure 2 Fagan′s nomogram for all diagnostic methods. ARFI: Acoustic radiation force impulse; FIB-4: Fibrosis-4 index; NFS: Non-alcoholic fatty liver disease fibrosis score; APRI: Aspartate aminotransferase to platelet ratio index; MRE: Magnetic resonance elastography; TE: Transient elastography; HFS: Hepamet fibrosis score.
Nearly half of the patients with hypertension have concomitant NAFLD[117]. RAS seems to contribute to the development of liver fibrosis, interestingly, the administration of RAS inhibitors showed an improvement in liver histology and decrease in protein expression of alpha-smooth muscle actin and hepatic content of hydroxyproline in a murine model of NAFLD[118]. In a cross-sectional study in hypertensive patients with biopsy-proven NAFLD, the use of RAS blockers was associated with less advanced hepatic fibrosis suggesting a beneficial effect of RAS blockers in NAFLD[119]. A similar effect was found in a retrospective cohort of 118 NAFLD patients with paired liver biopsies, where the use of RAS inhibitors was associated with decreased fibrosis progression rate only in patients with T2DM[22]. Given these potential benefits, the high safety profile of the drugs, and the fact that RAS blockers are one of the main classes of drugs recommended as initial therapy by hypertension guidelines[120,121], we suggest that RAS blockers should be considered as first-line therapy in patients with NAFLD and hypertension, after assessing the potential contraindications.

Figure 3 Diagnostic flow-chart to assess liver fibrosis in patients with non-alcoholic fatty liver disease. NAFLD: Non-alcoholic fatty liver disease; T2DM: Type 2 diabetes mellitus; IR: Insulin resistance; OSA: Obstructive sleep apnoea; DLP: Dyslipidemia; HFS: Hepamet fibrosis score; NFS: Nonalcoholic fatty liver disease fibrosis score; FIB-4: Fibrosis-4 index; APRI: Aspartate aminotransferase to platelet ratio index; AF: Advanced fibrosis; TE: Transient elastography; MRE: Magnetic resonance elastography; BMI: Body mass index; ALT: Alanine aminotransferase.
More than three-quarters of T2DM patients have coexistent NAFLD[9]; insulin resistance and hyperinsulinemia are the hallmarks of NAFLD and have an important role in stellate cell activation[122]. Therefore, treatment of patients with T2DM and NAFLD should be focused on improving insulin resistance. Even though metformin has not shown benefit in the histology of NAFLD, it remains the first-line drug for T2DM for its effects in the reduction of body weight, serum levels of lipids, and glucose[123,124]. Other benefits of metformin have been reported, such as the reduction in the risk of liver cancer[125,126]. Other insulin sensitizers such as dipeptidyl peptidase 4 inhibitors and glucagon-like peptide-1 analogs could also be good choices for the treatment of T2DM in patients with NAFLD[127]. Thiazolidinediones and Sodiumglucose co-transporter-2 (SGLT2) inhibitors are reviewed in the pharmacological management section[127]. Hence, insulin sensitizers should be the gold standard therapy for patients with T2DM and NAFLD.
OSA induces insulin resistance and systemic inflammation which as mentioned before are major features in NAFLD pathogenesis[128]. OSA patients should be screened for NAFLD and vice versa those with NAFLD for OSA[129]. The first-line treatment for OSA is continuous positive airway pressure (CPAP), which has several benefits, among which stand out the improvement of blood pressure and glucose resistance, and the reduction of overall and cardiovascular mortality[130-132]. The evidence of the benefit of CPAP on liver fibrosis in patients with NAFLD is scarce, at least three

Figure 4 Management flow-chart for patients with non-alcoholic fatty liver disease advanced fibrosis. We suggest vitamin E as it has demonstrated higher transplant-free survival and lower rates of hepatic decompensation. 1Other drugs such as cenicriviroc, obethicholic acid, dapagliflozin, and selonsertib have shown benefit in clinical trials and must be considered as well, especially as results continue to show beneficial results. NAFLD: Non-alcoholic fatty liver disease; RAS: Renin-angiotensin system; T2DM: Type 2 diabetes mellitus; CPAP: Continuous positive airway pressure; BW: Body weight.
studies have studied this, one showed improvement while the other two did not show this effect[132-134]. Longer trials may be needed to demonstrate a clear benefit. Although there is no clear evidence of a benefit to the liver in patients with OSA and NAFDL, CPAP should be encouraged in all patients with OSA.
Cardiovascular disease is one of the main causes of NAFLD-related deaths. Statins reduce NAFLD/NASH cardiovascular events, moreover, statins could reduce the risk of hepatocellular carcinoma related to NAFLD/NASH. Given these benefits and the fact that these medications have a high-security profile, they should be considered even in NAFLD patients without dyslipidemia[135,136].
Weight loss
To date, lifestyle modifications including diet and exercise are the first line and cornerstone of NAFLD/NASH treatment. Most studies have demonstrated a reduction of steatosis or/and steatohepatitis without improvement in liver fibrosis[137-143], nevertheless some have shown fibrosis regression, especially when a WL of 10% or more is achieved or when exercise therapy is included[144,145].
The Mediterranean diet (MD) with components such as fish, nuts, fruits, olive oil, whole grains, and vegetables is proposed by EASL guidelines for the treatment of NAFLD[4]. The MD has shown an inverse relationship with NAFLD prevalence and a reduction in liver steatosis[146-148]. In a non-randomized, open-label, 24-wk prospective study, 44 untreated NAFLD patients with non-significant fibrosis received nutritional counsel to increase adherence to MD with significant improvements in liver fibrosis at the end of the follow-up. It is noteworthy that the patients did not have significant fibrosis and that the technique for measurement was elastography ultrasound which could lead to unprecise data[149]. At the moment there is no evidence that dietper secould improve liver fibrosis, however, we must take into account that the diet that is going to have the maximum benefit is the one that will be followed by the patient in the long-term, therefore highly restrictive diets that induce a rapid WL should not be considered.
Regarding exercise, there is evidence that in selected patients with cirrhosis, moderate-intensity aerobic or resistance exercise, 4 d per week, 20 min, for at least 8 wk can have a positive physiological impact[150]. Moreover, it has been demonstrated that supervised exercise in cirrhotic patients can significantly lower the hepatic venous pressure gradient[151]. Exercise intervention studies in NAFLD are limited by low statistical power, and most have shown a reduction in intrahepatic triglyceride content without an improvement in liver fibrosis[152,153]. Few studies have shown a reduction in fibrosis, in particular, high-intensity interval training has recently been recognized as a novel exercise modality that demonstrated an improvement in liver stiffness (-16.8%), these benefits appeared to be independent of WL[154,155]. More evidence regarding the effect of exercise independently of the WL in fibrosis is required. As before concerning the diet, we consider that the exercise that is going to have the maximum impact is the one that can be maintained long-term, therefore the prescription of exercise should take into account the patients′ preferences and capabilities.
Bariatric surgery could be another approach in the management of NAFLD fibrosis, possibly through improvement of insulin resistance and hyperinsulinemia, which are related to the development of fibrosis in NAFLD[122]. In this context, bariatric surgery has proven to be an effective method for sustained WL in properly selected obese individuals[156-158], and importantly, in selected patients with T2DM, bariatric surgery is an effective therapy for glycemic control[159,160]. These changes have an additive effect considering that if 10% or more of WL is achieved with bariatric surgery, an improvement in fibrosis may be seen. There is some evidence of this benefit in retrospective and prospective cohorts with follow-up to 5 years[161-163]. Nevertheless, bariatric surgery should not be considered alone for the treatment of NAFLD fibrosis and the other indications of the procedure must be considered. The recommendation concerning this surgery in patients with advanced fibrosis must be individualized and evaluated by an experienced and interdisciplinary group[116].
Specific pharmacological management
Pharmacological management should be restricted to patients with NASH and/or advanced fibrosis. The current recommended drugs thus far in the guidelines are pioglitazone and vitamin E[4]. Although there are multiple treatments with different mechanisms of action under development for the treatment of NASH, and specifically aimed to reduce liver fibrosis, these drugs remain experimental and are considered for their use only in clinical trials.
Vitamin E is an antioxidant of polyunsaturated lipids and has a role in the treatment of NASH through antioxidant dependent and independent mechanisms[164]. In a NASH animal model, vitamin E supplementation decreased baseline levels of transforming growth factor beta 1 mRNA, suggesting a potential interference with both the initiation and progression of fibrosis[165]. Vitamin E improves transaminase activity, steatosis, inflammation, and ballooning in NASH patients[166-169]. Nevertheless, the data about its impact on liver fibrosis is controversial, several studies have shown an improvement in liver fibrosis[168-170], while others have not[124,169-172]. In the classical PIVENS study, 247 adults with NASH without T2DM were randomized to receive pioglitazone (30 mg/d), vitamin E (800 IU/d), or placebo, for 96 wk. Vitamin E therapy was associated with a significantly higher rate of improvement in NASH, with no reductions in fibrosis scores[167]. However, in a post hoc analysis, WL (≥ 2 kg) was associated with improvement on liver fibrosis scores, while weight gain (≥ 2 kg) was associated with worsening of fibrosis scores. These data reinforce the evidence-based recommendation for lifestyle modifications and WL as the basis of NASH/NAFLD treatment[116]. It is important to notice that since the PIVENS study did not include patients with T2DM[167], the evidence of Vitamin E in this group is not strong, and the few studies including T2DM have not shown benefit in liver fibrosis[173]. In a retrospective study, 90 patients with NASH and advanced fibrosis or cirrhosis consumed vitamin E (800 IU/d) for ≥ 2 years and were propensity-matched to 90 adults who did not take vitamin E, with a median follow-up was 5.6 years; vitamin E users had higher adjusted transplant-free survival, lower rates of hepatic decompensation than controls, and these benefits were evident in both, with and without T2DM patients[174]. There are some concerns with regard to the safety of vitamin E supplementation. A large long-term randomized trial (SELECT trial) showed a slight increase risk of prostate cancer in patients with vitamin E supplementation (Hazard ratio: 1.17; CI: 1.004-1.36)[175]. Furthermore, another large study associated vitamin E with an increased risk of hemorrhagic stroke, also with a marginal increment (Relative risk 1.22, 1.00-1.48)[176]. The significance of these side effects is not entirely clear since large-scale population studies tend to find statistical differences without clinical relevance, nevertheless, vitamin E must be prescribed with caution in high-risk groups.
Pioglitazone has a role in the treatment of NASH through modulation by an adiponectin-mediated effect on insulin sensitivity and hepatic fatty acid metabolism, as well as acting as a PPAR (gamma with Greek letter)[177]. Like vitamin E, pioglitazone has shown conflicting evidence regarding the reduction of fibrosis. The PIVENS study did not show an improvement[167], while two other randomized studies in diabetic and non-diabetic patients showed a reduction in liver fibrosis[171,172]. Cusiet al[172], studied 101 patients with prediabetes or T2DM and NASH, all patients were prescribed a hypocaloric diet and then randomly assigned to pioglitazone (45 mg/d), or placebo for 18 mo, followed by an 18-mo open-label phase with pioglitazone treatment. The pioglitazone group had improvement in the mean fibrosis score, however, this treatment was associated with significant weight gain, suggesting that pioglitazone could alter the natural history of the disease.
Cenicriviroc (CVC) is a dual antagonist of CCR2 and CCR5. CVC significantly reduced monocyte/macrophage recruitment and collagen deposition in animal models of fibrosis[178]. The CENTAUR study, a multicenter phase 2b clinical trial, randomized 289 patients to receive CVC (150 mg/d) or placebo for 12 mo, showed no differences in NAS score between the CVC and placebo group, however, twice as many subjects on CVC achieved a reduction in the fibrosis stage (1 stage) with no worsening of steatohepatitis compared to those on placebo; this benefit was seen across all stages of fibrosis, particularly for stage 2 and 3[179]. Based on the prior results, a large phase 3 trial, the AURORA study, is currently recruiting patients with NASH and histopathological evidence of stage 2 or 3 liver fibrosis to evaluate the benefit of CVC on the improvement in fibrosis and determine long-term clinical outcomes[180].
Obeticholic acid (OCA), is a farnesoid-X receptor (FXR) agonist[181]. FXR is a nuclear hormone receptor that regulates glucose and lipid metabolism. In a double-blind, placebo-controlled, proof-of-concept study in patients with NAFLD and T2DM, OCA increased insulin sensitivity and decreased significantly markers of liver fibrosis, nevertheless, no histopathology was obtained[182]. The FLINT study, a multicenter, randomized, placebo-controlled trial in non-cirrhotic NASH patients to assess treatment with OCA (25 mg/d) or placebo for 72 wk. OCA improved fibrosis, hepatocellular ballooning, steatosis, and lobular inflammation when compared with placebo[183]. Finally, in a recent multicenter double-blind, placebo-controlled study, 1968 patients with stage F1-F3 fibrosis (931 with F2-F3) were randomized to receive oral placebo, OCA 10 mg, or OCA 25 mg daily for 18 mo; an improvement of at least 1 stage of liver fibrosis was observed in 18% of the 10 mg group, and 23% in the 25 mg group, however, mild to moderate pruritus was reported in half of the patients with 25 mg of OCA[184]. A baseline NAS > 5, baseline triglyceride level 154 mg/dL, baseline INR 1, baseline AST level 49 U/L, and decrease in ALT level at week 24, were significant predictors of histologic response in NASH patients treated with OCA[185]. Tropifexor is another FXR agonist[186]. In a murine model, tropifexor reversed established fibrosis and reduced the NAFLD activity score, hepatic triglycerides, and profibrogenic gene expression[187]. Currently, at least three clinical trials are evaluating the safety, tolerability, and efficacy of different doses of tropifexor in NASH patients[188-190].
Elafibranor is an agonist of the peroxisome proliferator-activated receptor-α and peroxisome proliferator-activated receptor-δ. In a randomized, double-blind placebocontrolled trial, including 276 patients for 52 wk, NASH resolved without fibrosis worsening in a higher proportion of patients in the Elafibranor group (120 mg/d), and NASH resolution was associated with a reduction in liver fibrosis stage[191].
SGLT2 inhibitors, a relatively novel class of oral antidiabetic drugs that reduce hyperglycemia by promoting the urinary excretion of glucose[192]. Dapagliflozin, an SGLT2 inhibitor, has proven beneficial effects other than lower glucose levels in T2DM, such as a significant reduction in total body weight, predominantly by reducing total body fat mass and visceral fat[193]. In an open-label trial, 57 patients with T2DM and NAFLD were randomized to dapagliflozin (5 mg/d) (n= 33) or standard treatment (n= 24) for 24 wk. In 14 patients from the dapagliflozin group who had significant fibrosis (≥ 8.0 kPa), LSM decreased significantly from 14.7 ± 5.7 to 11.0 ± 7.3 kPa[194]. Licoglicoflzin, a dual sodium-glucose co-transporter 1/2 inhibitor which has proven positive effects on body weight in obese patients[195,196], in combination with tropifexor is currently under recruitment to evaluate the efficacy, safety, and tolerability in patients with NASH and significant fibrosis (F2 and F3)[189].
Finally, selonsertib, an inhibitor of apoptosis signal-regulating kinase 1, a serine/threonine signaling kinase, that can lead to fibrosis was evaluated alone or in combination with simtuzumab, in NAFLD patients with stage 2 or 3 liver fibrosis for 24 wk. Reductions in LSM on MRE and collagen content and lobular inflammation on liver biopsy were observed[197].
It is expected that shortly evidence from these and new drugs will increase exponentially, however, given the complex physiopathology of NAFLD fibrosis, it is unlikely that one drug by itself will deliver significant clinical outcomes, and perhaps the combinations of drugs with different targets will be necessary to obtain better results. Additionally, it is important to mention that until now, there is no evidence showing a clear effect of the “hepatoprotectors” on liver fibrosis (ursodeoxycholic acid, pentoxifylline, antioxidants among others), and therefore should not be used indiscriminately.
CONCLUSION
The main concern in NAFLD/NASH patients is the presence and progression of liver fibrosis, therefore all the efforts should center on it. There are several available scores to predict and stratify the risk of advanced fibrosis although the accuracy is limited in intermediate stages. To improve the accuracy of the diagnosis a combination of methods such as TE or US may be used, while MRE or liver biopsy alone are considered as the best options to accurately diagnose fibrosis. To select a diagnostic test or tests the prevalence of the disease in the specific center should be considered, as well as the experience of the center and the observers. The treatment should always take into account the presence of comorbidities such as the features of metabolic syndrome and should always include lifestyle modifications considering the preferences of the patient to ensure long-term adherence. The only approved pharmacological treatments so far are Vitamin E and pioglitazone, however, they have shown conflicting results on liver fibrosis improvement. Therefore, several new drugs and trials are being created and conducted aiming to improve both steatosis and liver fibrosis with very promising results thus far.
 World Journal of Gastroenterology2020年39期
World Journal of Gastroenterology2020年39期
- World Journal of Gastroenterology的其它文章
- Use of artificial intelligence in improving adenoma detection rate during colonoscopy: Might both endoscopists and pathologists be further helped
- Real-world treatment attrition rates in advanced esophagogastric cancer
- Metastatic pattern in esophageal and gastric cancer: Influenced by site and histology
- Relationships of early esophageal cancer with human papillomavirus and alcohol metabolism
- Dynamic contrast-enhanced magnetic resonance imaging and diffusion-weighted imaging in the activity staging of terminal ileum Crohn's disease
- Enteroscopy in children and adults with inflammatory bowel disease
