兰科药用植物活性多糖研究进展
张明泽, 何春梅, 王浩斌, 俞振明, 司灿, 赵聪慧, 段俊*
兰科药用植物活性多糖研究进展
张明泽1,2, 何春梅1, 王浩斌1,2, 俞振明1, 司灿1,2, 赵聪慧1,2, 段俊1*
(1. 中国科学院华南植物园, 广东省应用植物学重点实验室, 中国科学院华南农业植物分子分析与遗传改良重点实验室, 广州 510650; 2. 中国科学院大学, 北京 100049)
兰科(Orchidaceae)是有花植物中第二大科,目前已确认的兰科植物有736属28 000种,其中有82属343种可作药用。常见的兰科药用植物有铁皮石斛、天麻、白及和金线莲等。兰科药用植物功效成分主要为糖类、茋类、酚类、萜类、生物碱类、黄酮类和甾醇等,其中主要由甘露糖和葡萄糖等单糖构成的水溶性多糖是其重要活性成分之一,具有增强免疫力、抗肿瘤、抗氧化、降血糖和改善记忆等多种药理功能。对兰科药用植物活性多糖的结构、药理作用和生物合成等方面的研究进展进行了综述,并提出今后需要重点研究的方向,为进一步推动兰科药用植物资源保护及合理开发利用提供参考。
兰科;多糖;药理活性;生物合成
兰科(Orchidaceae)植物是开花植物中除菊科(Asteraceae)外最兴盛且生态型最丰富的类群,主要生长于热带和亚热带地区,目前已确认的有736属28 000种[1]。兰科植物中有82属343种可作为药用[2]。2015年版《中华人民共和国药典》中收载的兰科药用植物有铁皮石斛()、金钗石斛()、鼓槌石斛()或流苏石斛()的栽培品及其同属植物近似种、天麻()、白及()、杜鹃兰()、独蒜兰()[3]。兰科药用植物功效成分较为丰富,主要包括糖类、茋类、酚类、萜类、生物碱类、黄酮类和甾醇等[4],其中水溶性多糖是其重要活性成分之一,具有增强免疫力、抗肿瘤、抗氧化、降血糖和改善记忆等多种药理功能。近年来,国内外对植物活性多糖的结构与药理功能研究较为深入和广泛,在活性多糖生物合成方面的研究也相继报道,本文主要对兰科药用植物活性多糖的单糖组分、药理作用和生物合成方面的研究进展进行综述。
1 活性多糖组成
多糖是指由10个以上单糖分子通过糖苷键连接形成的多聚体[5]。多糖生物活性与其分子量、单糖组分密切相关[6]。目前关于石斛、白及、金线莲()、天麻、毛唇芋兰()、石仙桃()、杜鹃兰、手参()等兰科药用植物活性多糖的研究报道较多。
石斛属为兰科第二大属,种类超过1 500种[7],据统计我国有80种[8],其中大约30种作为药用和功能性食品[9]。石斛多糖的结构类型主要有-乙酰化葡甘露聚糖、-乙酰化半乳甘露葡聚糖、甘露葡聚糖、半乳甘露葡聚糖、鼠李阿拉伯半乳聚糖、半乳木葡聚糖等[10]。目前对金钗石斛、铁皮石斛、霍山石斛()等少数种的多糖结构有研究报道(表1),石斛属不同种多糖的单糖组分有所差异, 但甘露糖和葡萄糖是多数种的共同组分,此外还含有阿拉伯糖、半乳糖醛酸、木糖、鼠李糖、半乳糖等。
兰科其他药用植物活性多糖结构也有报道(表1),白及块茎富含丰富的多糖胶,白及多糖是一种由甘露糖和葡萄糖组成的水溶性葡甘露聚糖。金线莲全草含有丰富的多糖,且具有多种生物活性,是一种高价值的食疗原料,对人体健康起着重要作用。天麻药用部位为块茎,天麻多糖主要由葡萄糖组成。石仙桃属植物石仙桃多糖存在于假鳞茎或全草,主要由甘露糖、木糖和岩藻糖组成。手参多糖主要由葡萄糖、阿拉伯糖、甘露糖和半乳糖组成。杜鹃兰多糖主要由甘露糖和葡萄糖组成。文心兰()多糖主要是一种水溶性的中性线性甘露聚糖。毛唇芋兰的茎叶具有药用和食用价值,其多糖主要由半乳糖、阿拉伯糖、鼠李糖和半乳糖醛酸组成。萼足兰属植物的多糖是一种具有抗炎功效的葡甘露聚糖。
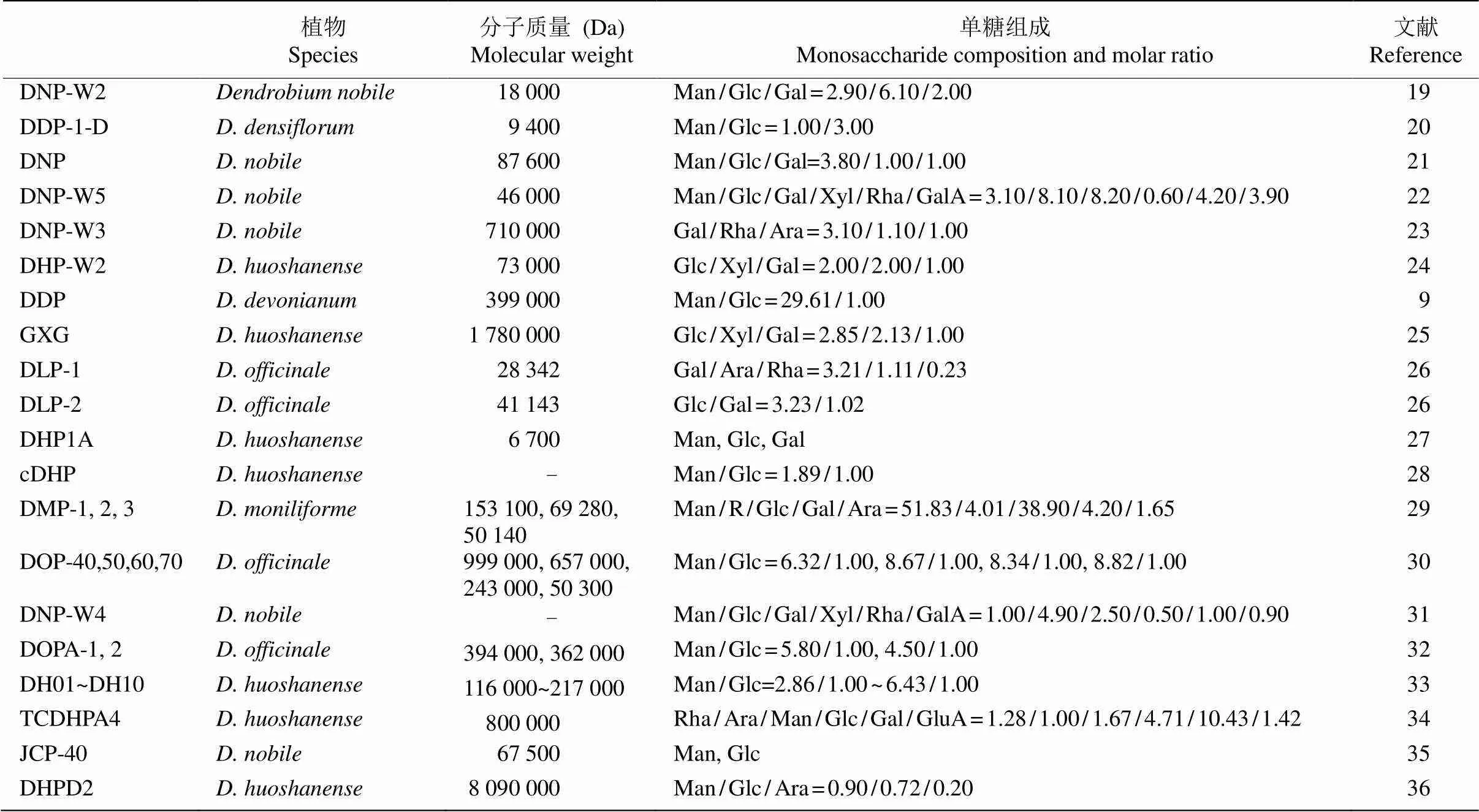
表1 石斛属植物的活性多糖组分
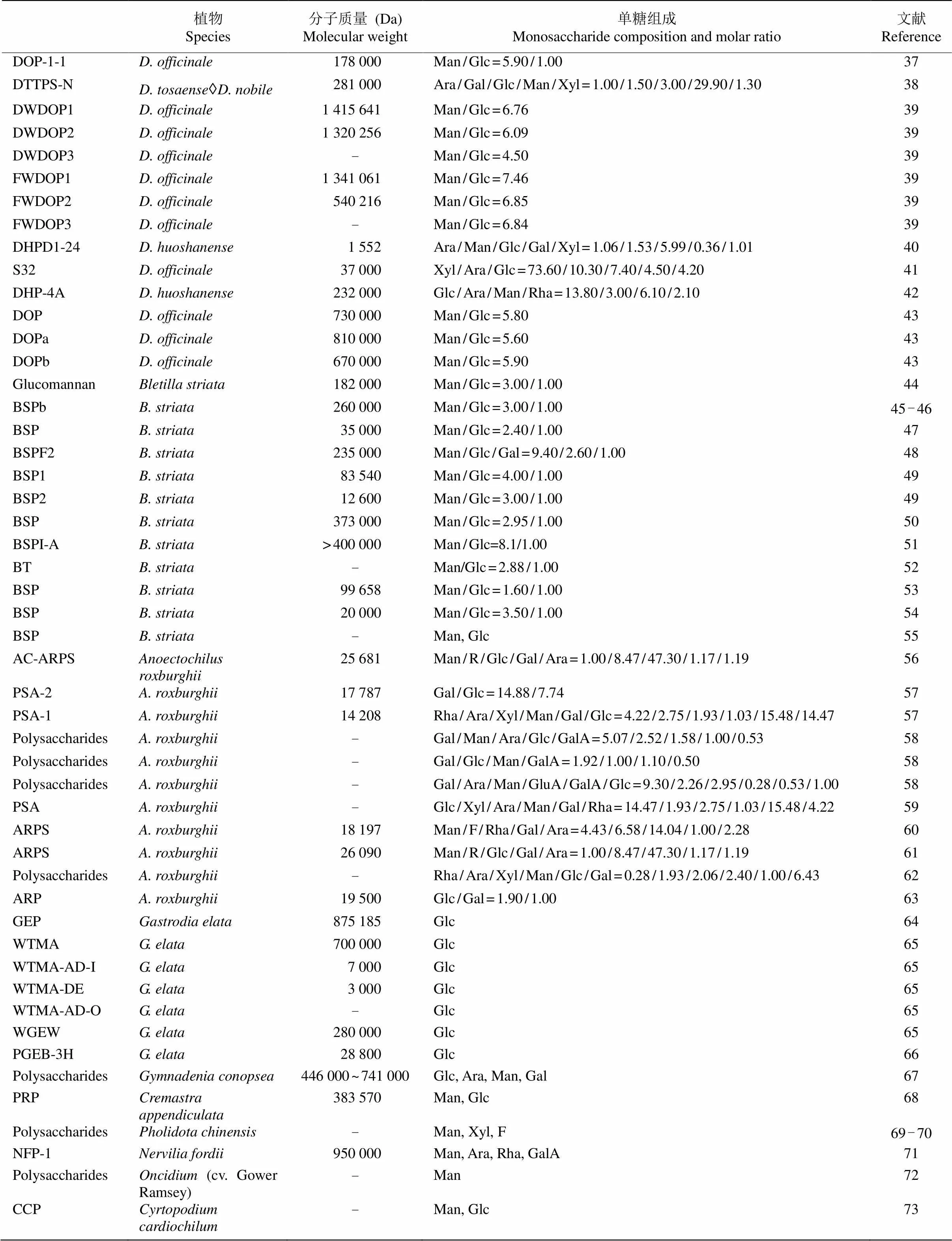
续表(Continued)
Glc: 葡萄糖; Man: 甘露糖; Gal: 半乳糖; Rha: 鼠李糖; Ara: 阿拉伯糖; Xyl: 木糖; R: 核糖; GalA: 半乳糖醛酸; GluA: 葡萄糖醛酸; F: 岩藻糖。
Glc: Glucose; Man: Mannose; Gal: Galactose; Rha: Rhamnose; Ara: Arabinose; Xyl: Xylose; R: Ribose; GalA: Galacturonic acid; GluA: Glucuronic acid; F: Fucose.
综上所述,兰科药用植物活性多糖多为中性多糖,由于部分结构中含有糖醛酸残基,因此也有少数酸性多糖存在。甘露糖和葡萄糖是兰科药用植物活性多糖的主要单糖组分,如铁皮石斛中的高分子水溶性多糖主要由甘露糖和葡萄糖构成的葡萄甘露聚糖[11]。不同属植物间的单糖组分有所差异,除甘露糖和葡萄糖外,部分兰科药用植物还含有半乳糖、鼠李糖、阿拉伯糖、木糖、核糖、岩藻糖等单糖组分。活性多糖中甘露糖残基的和位易发生乙酰化取代,这可能与增加多糖溶解度和生物活性有关[12–13]。兰科药用植物活性多糖的组成与其他科植物,如百合科(Liliaceae)植物芦荟(var.)[14]及天南星科(Araceae)植物魔芋()[15]的活性多糖结构极为相似,但兰科植物活性多糖中甘露糖与葡萄糖的比例较大,大于魔芋多糖和百合多糖中甘露糖与葡萄糖之比[16],而且甘露糖含量也较高,甘小娜等[17]的研究表明,铁皮石斛的甘露糖平均含量为32.33%,齿瓣石斛()为37.88%。此外,有研究表明,植物活性多糖的重要功能是调节机体的免疫功能,改善机体微循环,甘露糖具有抑制肿瘤生长的作用,甘露糖治疗癌症是一种简单、安全、有选择性的方法,适用于多种肿瘤类型[18]。因此,深入开展兰科药用植物活性多糖的研究在进一步推动兰科药用植物资源保护及合理开发利用等方面具有重要的意义。
2 活性多糖的药理功能研究
2.1 提高免疫力
铁皮石斛多糖具有免疫调节功能,可以作为新型功能性食品的原料[74]。药理试验表明,铁皮石斛与金钗石斛杂交植株的石斛多糖(DTTPS)能显著增加脾脏自然杀伤细胞(NK)的数量、NK细胞毒性、巨噬细胞吞噬和细胞因子诱导,从而具有强烈的免疫调节作用[38]。铁皮石斛-乙酰化葡甘露聚糖DOP-1-1在低pH溶液中较为稳定,可通过TLR4介导的核转录因子κB (NF-κB)信号通路诱导免疫应答,巨噬细胞炎性蛋白-1(MIP-1/CCL4)和干扰素诱导蛋白-10 (IP10)可能是-乙酰化葡甘露聚糖刺激免疫应答的新靶点[75]。铁皮石斛多糖通过改变肠粘膜结构,促进肠道淋巴集结和肠系膜淋巴结分泌细胞因子,增加固有层分泌性免疫球蛋白A,调节肠粘膜免疫活性[76],还可以显著提高胃肠道传输率,恢复粪便排出特性,有助于缓解便秘[77]。铁皮石斛多糖(GXG)还可以改变肠道的生理状态,通过调节粘膜结构以及上调紧密连接蛋白的表达从而改善肠道的物理屏障,通过提升黏膜蛋白-2、-防御素和分泌型免疫球蛋白A的分泌而增强肠道生物屏障,并通过刺激产生细胞因子和促进免疫细胞发育来调节肠道免疫屏障[78]。鼓槌石斛多糖在免疫调节方面具有刺激小鼠脾脏细胞增殖的功效[79]。金钗石斛多糖增强T-淋巴细胞和B-淋巴细胞活性[22],提高机体免疫指数,促进小鼠体内细胞因子分泌[80]。齿瓣石斛多糖可促进巨噬细胞NO释放和吞噬等免疫功能,因此,可作为一种天然的免疫刺激剂[9]。白及多糖可促进药物输送和创伤修复[81],因其在巨噬细胞发炎和增生时期的调控作用而具有促进口腔溃疡愈合的功效[50,82]。研究表明,天麻多糖能显著刺激巨噬细胞释放NO,并以剂量依赖的方式增强细胞吞噬作用,表现免疫功能[83]。手参多糖具有免疫调节功能,其活性大小与其结构和组成有关[67]。体外研究表明,毛唇芋兰多糖(NFP-1)具有调节巨噬细胞的免疫功能[71]。巨噬细胞和树突细胞质膜上存在一些特定识别受体,如清道夫受体(SRs)、-d-葡聚糖受体树突细胞相关性C型植物血凝素1 (Dectin-1)、甘露糖受体(MR)、补体受体三型分子(CR3)及Toll样受体4 (TLR4)等,当多糖与这些受体结合后,触发多种信号通路,产生免疫应答[84]。从兰科植物中分离的多糖大部分是甘露聚糖,且可以调节人体免疫力。说明兰科植物中的甘露聚糖与巨噬细胞和树突细胞质膜上的甘露糖受体结合,刺激了人体内的免疫应答,从而具有提高人体免疫力的功效。
2.2 抗肿瘤
铁皮石斛多糖具有抗癌活性,通过Bcl-2和Bax依赖途径诱导人肝癌HepG2细胞凋亡[30],其活性与分子量和单糖组成有关,铁皮石斛粗多糖在HeLa细胞中均无明显活性,只有在降解为分子量较小的多糖后才表现出明显的抗肿瘤作用,降解的多糖通过丝裂原活化蛋白激酶(MAPK)信号通路诱导HeLa细胞凋亡,多糖中甘露糖相关组分含量越高,抗肿瘤活性越强[39]。白及多糖BSP-1能提高免疫缺陷模型小鼠的胸腺和脾脏指数,具有免疫调节作用[49]。从人工栽培的金线莲中提取的多糖(AC-ARPS)对体外肺癌A549、骨肉瘤143B、大鼠肾上腺嗜铬细胞瘤PC12、乳腺癌MCF-7、急性白血病HL60、慢性白血病K562、结肠癌SW620、食管癌OE19、肝癌HepG2和神经胶质瘤U251等肿瘤细胞具有抑制作用, AC-ARPS诱导OE19细胞凋亡和G2/M期阻滞[56]。从杜鹃兰假鳞茎中提取的多糖PRP具有明显的抗肿瘤特性,可以作为一种新的肿瘤治疗药物[68]。此外,天麻多糖也具有抗癌活性[65]。
2.3 抗氧化
研究表明,铁皮石斛多糖(DOP)对金属螯合活性较高[85],具有明显的1,1-二苯基-2-苦基肼(DPPH)自由基、2ʹ-联氨-双-3-乙基苯并噻唑啉-6-磺酸(ABTS)自由基和羟基自由基清除能力和还原能力,可作为天然抗氧化剂运用在食品或医药行业[11]。铁皮石斛多糖对脾脏细胞、T-淋巴细胞和B-淋巴细胞有刺激作用,能促进细胞活力,抑制凋亡,保护巨噬细胞免受过氧化氢诱导的氧化损伤[32]。天麻多糖具有较高的抗氧化活性,可以延缓因自由基引起的人体衰老[64]。石仙桃多糖具有抗氧化功能,对超氧阴离子和羟基自由基具有剂量依赖性的清除作用[69–70]。金线莲多糖(ARP)通过清除自由基从而对CCl4诱导的小鼠氧化性肝损伤具有保护作用[63,86]。
2.4 抗炎
铁皮石斛叶多糖具有保护THP-1细胞免受脂多糖激活的细胞毒性作用,抑制活性氧的形成,以及抑制脂多糖激活的THP-1细胞的受体蛋白(TLR- 4)、骨髓分化因子(MyD88)以及肿瘤坏死因子受体相关因子(TRAF-6)的mRNA和蛋白表达,因而具有抗炎功效[26]。通过研究铁皮石斛多糖(cDHP)对香烟烟雾(CS)诱导的小鼠肺炎的抑制作用,认为cDHP可增加肺泡数量,增厚肺泡壁,抑制肺大泡形成,减少炎症细胞浸润,还可以抑制CS诱导的血清和肺中TNF-、IL-1分泌,cDHP的抗炎作用是通过调节NF-κB和MAPK信号而介导的[28]。白及多糖通过在体外下调转化生长因子(TGF-RI、TGF-RII)及其下游-平滑肌肌动蛋白(SMA)的量,缓解肾脏炎症和纤维化[46]。
2.5 降血糖和保护神经系统
研究表明,从鼓槌石斛、霍山石斛和金钗石斛茎中提取的多糖能抑制血糖上升[87],具有抗糖化活性[24]和降血糖作用[88]。铁皮石斛多糖具有保护神经的作用,通过调节小胶质细胞的活性从而预防与阿尔茨海默病(AD)相关的认知衰退[89]。天麻多糖也具有保护神经的作用,能有效抑制皮质酮(CORT)诱导的PC12细胞凋亡[90]。
3 活性多糖生物合成研究
多糖是一类非常重要的生物大分子,但由于结构复杂和研究技术的滞后,造成多糖代谢研究较为缓慢。目前研究较为深入的是细胞壁多糖的合成。植物细胞壁由纤维素微纤丝组成,一些复杂的非纤维素多糖嵌入微纤丝中,非纤维素多糖通常大部分为果胶、木葡聚糖和非均一性木聚糖,少部分为非均一性甘露聚糖[91]。非均一性甘露聚糖类群因在一些藻类细胞壁中发现而被认为是一种最古老的植物细胞壁多糖[92],根据其主链组成和侧链取代基类型可分为4类:甘露聚糖、葡甘露聚糖、半乳甘露聚糖和半乳葡甘露聚糖。其中葡甘露聚糖与从石斛属植物中分离鉴定到的葡甘露聚糖结构较为相似,且研究最为深入。而兰科植物中鉴定的大部分为葡萄甘露聚糖,也是主要的活性多糖,其生物合成的研究主要集中在石斛葡甘露聚糖上。
3.1 植物葡甘露聚糖储存位置
植物细胞在一定生理发育阶段合成一些水溶性多糖,通常称为贮藏多糖,以固态、液态或溶胶状暂时贮存在细胞质体或液泡中,当生长需要时转化为单体重新进入细胞进行代谢。一些单子叶植物如兰科植物的营养器官就贮存了甘露聚糖这样的贮藏多糖[93],据早期研究报道,兰科红门兰属()植物块茎中含有由甘露糖和葡萄糖组成的葡甘露聚糖,具有很高的黏性[94],兰科其他药用植物水溶性多糖也主要是葡甘露聚糖。葡甘露聚糖主链由甘露糖残基和葡萄糖残基通过-1,4糖苷键连接[12]。葡甘露聚糖在植物进化过程中高度保守,经过多次独立进化后主要作为种子或茎等器官的储能物质[95–96]。葡甘露聚糖在高尔基体中合成后以水合态贮存于特化的薄壁细胞液泡中[97]。薄壁细胞是一种由初生细胞壁包围且具有分裂能力以及代谢活跃的细胞,典型的草本种子植物中90%的细胞是薄壁细胞[98],铁皮石斛茎中合成的多糖颗粒主要贮存于薄壁细胞的质体中[99]。
3.2 植物葡甘露聚糖生物合成过程
多糖合成过程包括核苷酸糖的合成,糖链的起始、延伸和终止[100]。合成非均一甘露聚糖的底物为活化的核苷酸糖,包括GDP-甘露糖、GDP-葡萄糖和UDP-半乳糖[101],在细胞质基质中,这些活化的核苷酸糖的合成途径为:蔗糖(蔗糖合酶或蔗糖转化酶和UDP-葡萄糖焦磷酸化酶催化)→UDP-葡萄糖(UDP-葡萄糖-4-差向异构酶催化)→UDP-半乳糖; 蔗糖(蔗糖转化酶催化)→果糖(果糖激酶催)→6-磷酸果糖(6-磷酸甘露糖异构酶催化)→6-磷酸甘露糖(甘露糖磷酸变位酶催化)→1-磷酸甘露糖(甘露糖- 1-磷酸尿苷转移酶催化)→GDP-甘露糖[102],但GDP-葡萄糖的合成途径尚不清楚。高尔基体膜上不同的核苷酸糖转运蛋白(nucleotide sugar transporters, NSTs)将细胞质基质中的核苷酸糖转运到高尔基体腔中[103],之后高尔基体中特异的糖基转移酶(GTs)催化核苷酸糖形成糖苷键,进而合成多糖[104],合成的多糖主要通过分泌面高尔基体网上的复合蛋白介导和随后的分泌囊泡穿梭这种常规分泌途径转运到贮存部位[102]。
3.3 多糖合成途径基因的鉴定与功能分析
GT2家族中纤维素合成酶类似物A (CslA), 如AtCslA9的活性域面向高尔基体腔[105],把GDP-甘露糖或GDP-葡萄糖添加到甘露聚糖主链上而延伸多糖[96]。合成核苷酸糖如UDP-半乳糖和GDP-葡萄糖转运蛋白基因已经鉴定[103]。在拟南芥() CslA家族基因中,已经证明多个基因, 如、、和具有调控合成-1,4-甘露聚糖的功能[106–107],参与了花序梗细胞壁大部分葡甘露聚糖的合成[108]。从瓜尔豆()种子中分离的-甘露聚糖合酶(ManS),与拟南芥AtCslA9同源[109]。从魔芋球茎中分离了AkCslA3,能利用GDP-甘露糖和GDP-葡萄糖合成葡甘露聚糖[110]。有研究表明, CslD (AtCslD2,3,5)在拟南芥根毛中也催化甘露聚糖的生物合成[111]。还有一些位于高尔基体上的酶辅助合成甘露聚糖,如敲除后突变体茎中葡甘露聚糖含量显著降低[112]。
石斛多糖合成的相关基因,如碱性/中性转化酶基因()[113],UDP-葡萄糖焦磷酸化酶基因()[114],GDP-甘露糖焦磷酸化酶基因()[115], 磷酸甘露糖合成酶基因()[116], UDP-葡萄糖-4-差向异构酶()基因[117]以及8种糖基转移酶基因()[118–119]已经克隆,部分基因的功能已被研究。结果表明,铁皮石斛基因组中涉及葡甘露聚糖合酶活性基因大量重复,可能与多糖的合成有关[120]。多糖生物合成相关候选基因的鉴定和克隆,为阐明多糖生物合成的分子机理奠定了基础[121],进一步可通过转基因等生物技术手段增加兰科药用植物活性多糖的含量[122–123]。
4 结语
我国拥有丰富的兰科植物资源,其药用价值还需进一步挖掘。兰科药用植物活性多糖的单糖组成不尽相同,但甘露糖和葡萄糖是多数兰科药用植物活性多糖的主要成分,多糖中甘露糖含量较高是多数兰科药用植物的一大特点。兰科药用植物是一种很好的葡甘露聚糖来源,据国外研究报道,用兰科植物块茎磨成的粉中葡甘露聚糖含量可达61.00%[124],而另一种重要的葡甘露聚糖来源植物——魔芋,其葡甘露聚糖含量为42.75%[125]。就商业用途而言,兰科植物活性多糖在国外已经用于制作饮料、可食用膜及冰淇淋等[126]。近年来,国内外学者对其结构和药理作用机制的研究越来越广泛,研究表明多糖具有增强免疫力、抗肿瘤、抗氧化、降血糖和改善记忆等多种药理作用,特别是甘露糖的药理作用研究报道较多,而且毒副作用较小甚至没有毒副作用,因此,兰科药用植物活性多糖在疾病预防及保健品开发等方面有广阔的应用前景。
在兰科药用植物活性多糖高级结构及其作用机理、生物合成等方面有待进一步深入研究。如多糖药理功能是由多糖中的某单一组分起作用还是由多种组分共同起作用尚不清楚。此外多糖支链的取代如乙酰化等是否影响其药理活性也需要进一步明确。由于兰科药用植物活性多糖生物合成方面的研究起步较晚,功能基因的相关研究报道大多仅限于基因的克隆和表达模式分析,基因功能验证方面的工作开展较少。因此,利用新一代高通量测序技术结合生物信息学解析兰科药用植物基因组和转录组信息,大规模发掘与活性多糖合成相关的基因,在全面揭示这些活性多糖在细胞中合成、运输和贮存的详细过程以及代谢调控分子机制的基础上,通过转基因或基因沉默等技术,对候选基因功能进行分析、验证,鉴定出一批在生产上能够提高多糖含量的关键调控基因。这些工作可促进兰科药用植物资源保护和可持续利用,因此将是未来研究的重要方向。
[1] CHASE M W, CAMERON K M, FREUDENSTEIN J V, et alAn updated classification of Orchidaceae [J]. Bot J Linn Soc, 2015, 177(2): 151–174. doi: 10.1111/boj.12234.
[2] LI J H, ZHANG L H. Application of tissue culture and mycorrhizal technology of medicinal orchid plants for seedling production [J]. Seed, 2015, 34(8): 133–134. doi: 10.16590/j.cnki.1001-4705.2015.08.133.李景蕻, 张丽华. 药用兰科植物组培苗菌根化技术在种苗生产中的应用[J]. 种子, 2015, 34(8): 133–134. doi: 10.16590/j.cnki.1001-4705. 2015.08.133.
[3] TANG H. DNA barcoding identification and ecological suitability of important medicinal plants of Orchidaceae [D]. Yaan: Sichuan Agricul- tural University, 2016: 9. 汤欢. 兰科重要药用植物DNA条形码鉴定及其生态适宜性 [D].雅安: 四川农业大学, 2016: 9.
[4] LIU H D, PAN L L, ZHOU X, et al. Research progress on chemical constituents and pharmacological activities of alkaloids in Orchidaceae plants [J]. Chin Trad Herb Drugs, 2019, 50(3): 731–744. doi: 10.7501/ j.issn.0253-2670.2019.03.029.刘宏栋, 潘玲玲, 周翔, 等. 兰科植物生物碱类化学成分及药理活性研究进展 [J]. 中草药, 2019, 50(3): 731–744. doi: 10.7501/j.issn. 0253-2670.2019.03.029.
[5] PRAJAPATI V D, JANI G K, MORADIYA N G, et alGalactomannan: A versatile biodegradable seed polysaccharide [J]. Int J Biol Macromol, 2013, 60: 83–92. doi: 10.1016/j.ijbiomac.2013.05.017.
[6] MA L S, CHEN H X, ZHU W C, et alEffect of different drying methods on physicochemical properties and antioxidant activities of polysaccharides extracted from mushroom[J]. Food Res Int, 2013, 50(2): 633–640. doi: 10.1016/j.foodres.2011.05.005.
[7] CAKOVA V, BONTE F, LOBSTEIN A.: Sources of active ingredients to treat age-related pathologies [J]. Aging Dis, 2017, 8(6): 827–849. doi: 10.14336/AD.2017.0214.
[8] DUAN J, DUAN Y P. High-efficient Cultivation Techniques for[M]. Fujian: Fujian Science and Technology Press, 2013: 1. 段俊, 段毅平. 铁皮石斛高效栽培技术 [M]. 福建: 福建科学技术出版社, 2013: 1.
[9] DENG Y, LI M, CHEN L X, et alChemical characterization and immunomodulatory activity of acetylated polysaccharides from[J]. Carbohydr Polym, 2018, 180: 238–245. doi: 10.1016/j.carbpol.2017.10.026.
[10] XING X H, CUI S W, NIE S P, et alA review of isolation process, structural characteristics, and bioactivities of water-soluble polysac- charides fromplants [J]. Bioact Carbohydr Diet Fibre, 2013, 1(2): 131–147. doi: 10.1016/j.bcdf.2013.04.001.
[11] FAN H R, MENG Q R, XIAO T C, et alPartial characterization and antioxidant activities of polysaccharides sequentially extracted from[J]. J Food Meas Charact, 2018, 12(2): 1054– 1064. doi: 10.1007/s11694-018-9721-8.
[12] PAULY M, GILLE S, LIU L F, et alHemicellulose biosynthesis [J]. Planta, 2013, 238(4): 627–642. doi: 10.1007/s00425-013-1921-1.
[13] HSIEH Y S Y, CHIEN C, LIAO S K S, et alStructure and bioactivity of the polysaccharides in medicinal plant[J]. Bioorg Med Chem, 2008, 16(11): 6054–6068. doi: 10.1016/j.bmc.2008. 04.042.
[14] YAN C Q, ZOU K. Advances in research on plant polysaccharides of Liliaceae [J]. J China Three Gorges Univ (Nat Sci), 2009, 31(2): 96– 100. doi: 10.3969/j.issn.1672-948X.2009.02.025.晏传奇, 邹坤. 百合科植物多糖研究进展 [J]. 三峡大学学报(自然科学版), 2009, 31(2): 96–100. doi: 10.3969/j.issn.1672-948X.2009.02.025.
[15] SINGH S, SINGH G, ARYA S K. Mannans: An overview of properties and application in food products [J]. Int J Biol Macromol, 2018, 119: 79–95. doi: 10.1016/j.ijbiomac.2018.07.130.
[16] TESTER R F, AL-GHAZZEWI F H. Beneficial health characteristics of native and hydrolysed konjac () glucomannan[J]. J Sci Food Agric, 2016, 96(10): 3283–3291. doi: 10.1002/jsfa.7571.
[17] GAN X N, XU Y, XU H, et al. Improvement in determination for polysaccharide and mannose in dendrobii officinalis caulis and its comparation with dendrobii devoniani caulis [J]. Drug Stand China, 2014, 15(4): 276–279. doi: 10.19778/j.chp.2014.04.011.甘小娜, 徐英, 徐红, 等. 铁皮石斛中多糖和甘露糖含量测定方法的改进及与齿瓣石斛的比较研究 [J]. 中国药品标准, 2014, 15(4): 276–279. doi: 10.19778/j.chp.2014.04.011.
[18] GONZALEZ P S, O’PREY J, CARDACI S, et al. Mannose impairs tumour growth and enhances chemotherapy [J]. Nature, 2018, 563 (7733): 719–723. doi: 10.1038/s41586-018-0729-3.
[19] WANG J H, ZHA X Q, LUO J P, et alAn acetylated galactoman- noglucan from the stems ofLindl. [J]. Carbohydr Res, 2010, 345(8): 1023–1027. doi: 10.1016/j.carres.2010.03.005.
[20] LI Q, XIE Y, SU J W, et alIsolation and structural characterization of a neutral polysaccharide from the stems of[J]. Int J Biol Macromol, 2012, 50(5): 1207–1211. doi: 10.1016/j.ijbiomac. 2012.03.005.
[21] LUO A X, HE X J, ZHOU S D, et alantioxidant activities of a water-soluble polysaccharide derived fromLindl. extracts [J]. Int J Biol Macromol, 2009, 45(4): 359–363. doi: 10.1016/ j.ijbiomac.2009.07.008.
[22] WANG J H, LUO J P, ZHA X Q. Structural features of a pectic polysaccharide from the stems ofLindl. [J]. Carbo- hydr Polym, 2010, 81(1): 1–7. doi: 10.1016/j.carbpol.2010.01.040.
[23] WANG J H, LUO J P, YANG X F, et al. Structural analysis of a rham- noarabinogalactan from the stems ofLindl. [J]. Food Chem, 2010, 122(3): 572–576. doi: 10.1016/j.foodchem.2010.03.012.
[24] PAN L H, FENG B J, WANG J H, et al. Structural characterization and anti-glycation activity in vitro of a water-soluble polysaccharide from[J]. J Food Biochem, 2013, 37(3): 313–321. doi: 10.1111/j.1745-4514.2011.00633.x.
[25] XIE S Z, GE J C, LI F, et al. Digestive behavior ofpolysaccharides in the gastrointestinal tracts of mice [J]. Int J Biol Macromol, 2018, 107: 825–832. doi: 10.1016/j.ijbiomac.2017.09.047.
[26] ZHANG M, WU J W, HAN J J, et alIsolation of polysaccharides fromleaves and anti-inflammatory activity in LPS- stimulated THP-1 cells [J]. Chem Cent J, 2018, 12(1): 109. doi: 10.1186/ s13065-018-0480-8.
[27] TIAN C C, ZHA X Q, PAN L H, et alStructural characterization and antioxidant activity of a low-molecular polysaccharide from[J]. Fitoterapia, 2013, 91: 247–255. doi: 10.1016/j.fitote. 2013.09.018.
[28] GE J C, ZHA X Q, NIE C Y, et alPolysaccharides fromstems alleviates lung inflammation in cigarette smoke- induced mice [J]. Carbohydr Polym, 2018, 189: 289–295. doi: 10.1016/ j.carbpol.2018.02.054.
[29] XI X J, XIAO S Z, TANG F, et al. Monosaccharide analysis ofpolysaccharides by high performance liquid chromato-graphy/electrospray ionization mass spectrometry [J]. Amer J Agric For, 2016, 4(6): 156–162. doi: 10.11648/j.ajaf.20160406.13.
[30] XING S P, ZHANG X F, KE H N, et al. Physicochemical properties of polysaccharides fromby fractional precipitation and their preliminary antioxidant and anti-HepG2 cells activities[J]. Chem Cent J, 2018, 12(1): 100. doi: 10.1186/s13065-018-0468-4.
[31] WANG J H, ZUO S R, LUO J P. Structural analysis and immuno- stimulating activity of an acidic polysaccharide from the stems ofLindl. [J]. Molecules, 2017, 22(4): 611. doi: 10. 3390/molecules22040611.
[32] HUANG K W, LI Y R, TAO S C, et al. Purification, characterization and biological activity of polysaccharides from[J]. Molecules, 2016, 21(6): 701. doi: 10.3390/molecules21060701.
[33] DENG Y, CHEN L X, HAN B X, et alQualitative and quantitative analysis of specific polysaccharides inby using saccharide mapping and chromatographic methods [J]. J Pharm Biomed Anal, 2016, 129: 163–171. doi: 10.1016/j.jpba.2016.06.051.
[34] SI H Y, CHEN N F, CHEN N D, et al. Structural characterisation of a water-soluble polysaccharide from tissue-culturedC. Z. Tang et S. J. Cheng [J]. Nat Prod Res, 2018, 32(3): 252–260. doi: 10.1080/14786419.2017.1350670.
[35] ZHANG Y, WANG H X, GUO Q B, et al. Structural characterization and conformational properties of a polysaccharide isolated fromLindl. [J/OL]. Food Hydrocoll, 2019, (2019–01–22). doi: 10.1016/j.foodhyd.2019.01.044.
[36] LI X L, XIAO J J, ZHA X Q, et al. Structural identification and sulfated modification of an antiglycationpolysaccharide [J]. Carbohydr Polym, 2014, 106: 247–254. doi: 10. 1016/j.carbpol.2014.02.029.
[37] HE T B, HUANG Y P, YANG L, et al. Structural characterization and immunomodulating activity of polysaccharide from[J]. Int J Biol Macromol, 2016, 83: 34–41. doi: 10.1016/j. ijbiomac.2015.11.038.
[38] YANG L C, HSIEH C C, WEN C L, et al. Structural characterization of an immunostimulating polysaccharide from the stems of a new medicinalspecies:Taiseed Tosnobile [J]. Int J Biol Macromol, 2017, 103: 1185–1193. doi: 10.1016/j.ijbiomac.2017.05.185.
[39] YU W X, REN Z Y, ZHANG X F, et al. Structural characterization of polysaccharides fromand their effects on apoptosis of HeLa cell line [J]. Molecules, 2018, 23(10): 2484. doi: 10. 3390/molecules23102484.
[40] ZHA X Q, DENG Y Y, LI X L, et al. The core structure of apolysaccharide required for the inhibition of human lens epithelial cell apoptosis [J]. Carbohydr Polym, 2017, 155: 252– 260. doi: 10.1016/j.carbpol.2016.08.087.
[41] YUE H, LIU Y Q, QU H H, et al. Structure analysis of a novel hetero- xylan from the stem ofand anti angiogenesis activities of its sulfated derivative [J]. Int J Biol Macromol, 2017, 103: 533–542. doi: 10.1016/j.ijbiomac.2017.05.097.
[42] LI F, CUI S H, ZHA X Q, et al. Structure and bioactivity of a polysac- charide extracted from protocorm-like bodies of[J]. Int J Biol Macromol, 2015, 72: 664–672. doi: 10.1016/j.ijbio mac.2014.08.026.
[43] WEI W, FENG L, BAO W R, et alStructure characterization and immunomodulating effects of polysaccharides isolated from[J]. J Agric Food Chem, 2016, 64(4): 881–889. doi: 10. 1021/acs.jafc.5b05180.
[44] TOMODA M, NAKATSUKA S, TAMAI M, et al. Plant mucilages. VIII. Isolation and characterization of a mucous polysaccharide, “- glucomannan,” fromTubers [J]. Chem Pharm Bull, 1973, 21(12): 2667–2671. doi: 10.1248/cpb.21.2667.
[45] YUE L, WANG W, WANG Y, et alpolysaccharide inhibits angiotensin II-induced ROS and inflammation via NOX4 and TLR2 pathways [J]. Int J Biol Macromol, 2016, 89: 376–388. doi: 10. 1016/j.ijbiomac.2016.05.002.
[46] WANG Y, LIU D, CHEN S J, et al. A new glucomannan from: Structural and anti-fibrosis effects [J]. Fitoterapia, 2014, 92: 72–78. doi: 10.1016/j.fitote.2013.10.008.
[47] DONG L, XIA S H, LUO Y, et al. Targeting delivery oligonucleotide into macrophages by cationic polysaccharide fromsuccessfully inhibited the expression of TNF-[J]. J Control Release, 2009, 134(3): 214–220. doi: 10.1016/j.jconrel.2008.11.013.
[48] PENG Q, LI M, XUE F, et al. Structure and immunobiological activity of a new polysaccharide from[J]. Carbohydr Polym, 2014, 107: 119–123. doi: 10.1016/j.carbpol.2014.02.042.
[49] WANG Y R, HAN S W, LI R F, et al. Structural characterization and immunological activity of polysaccharides from the tuber of[J]. Int J Biol Macromol, 2019, 122: 628–635. doi: 10.1016/j. ijbiomac.2018.10.201.
[50] LIAO Z C, ZENG R, HU L L, et al. Polysaccharides from tubers of: Physicochemical characterization, formulation of buccoadhesive wafers and preliminary study on treating oral ulcer [J]. Int J Biol Macromol, 2019, 122: 1035–1045. doi: 10.1016/j.ijbiomac. 2018.09.050.
[51] WANG B, XU S, HUANG L J, et al. Isolation, purification and structural characterization of a polysaccharide fraction from stem tuber of, named BSPI-A [J]. Food Sci, 2010, 31(17): 120–123. 王博, 徐莎, 黄琳娟, 等. 白芨多糖BSPI-A的分离纯化及结构研究 [J]. 食品科学, 2010, 31(17): 120–123.
[52] LIU J Y, WANG H C, YIN Y, et al. Controlled acetylation of water- soluble glucomannan from[J]. Carbohydr Polym, 2012, 89(1): 158–162. doi: 10.1016/j.carbpol.2012.02.065.
[53] WU X G, XIN M, CHEN H, et al. Novel mucoadhesive polysaccharide isolated fromimproves the intraocular penetration and efficacy of levofloxacin in the topical treatment of experimental bacterial keratitis [J]. J Pharm Pharmacol, 2010, 62(9): 1152–1157. doi: 10.1111/j.2042-7158.2010.01137.x.
[54] ZHANG M S, SUN L, ZHAO W C, et al. Cholesteryl-modification of a glucomannan fromand its hydrogel properties [J]. Molecules, 2014, 19(7): 9089–9100. doi: 10.3390/molecules19079089.
[55] WANG C, LUO W F, LI P W, et al. Preparation and evaluation of chitosan/alginate porous microspheres/polysaccharide composite hemostatic sponges [J]. Carbohydr Polym, 2017, 174: 432– 442. doi: 10.1016/j.carbpol.2017.06.112.
[56] YU X L, LIN S E, ZHANG J Q, et al. Purification of polysaccharide from artificially cultivated(Wall.) Lindl. by high-speed counter current chromatography and its antitumor activity [J]. J Sep Sci, 2017, 40(22): 4338–4346. doi: 10.1002/jssc.201700340.
[57] ZHANG X H. Structural analysis and antidiabetic activities of polysac- charides from[D]. Shantou: Shantou University, 2011: 21–27. 张晓辉. 金线莲多糖结构分析及抗糖尿病活性研究 [D]. 汕头: 汕头大学, 2011: 21–27.
[58] WU Y B, ZHANG X C, YI J, et al. Determination of the monosaccharide composition inpolysaccharide from different origins by pre-column derivatization HPLC method [J]. China Pharm, 2015, 26(15): 2116–2119. doi: 10.6039/j.issn.1001-0408.2015.15.37.吴岩斌, 张秀才, 易骏, 等. 柱前衍生化HPLC法测定不同基源金线莲多糖的单糖组成 [J]. 中国药房, 2015, 26(15): 2116–2119. doi: 10.6039/j.issn.1001-0408.2015.15.37.
[59] YANG Z G, ZHANG X H, YU J. Primary study on the characterization of polysaccharides from[J]. China J Pharm Econ, 2015, 10(S1): 36–37. 杨振国, 张晓辉, 余杰. 金线莲多糖结构的初步分析 [J]. 中国药物经济学, 2015, 10(S1): 36–37.
[60] ZHANG J Q. The purification and the structure representation of polysac- charides from(Wall) Lindl. and the anti-tumor activities research [D]. Fuzhou: Fujian Medical University, 2010: 4.张锦雀. 金线莲多糖的分离纯化、结构表征及其抗肿瘤活性 [D]. 福州: 福建医科大学, 2010: 4.
[61] LIN S E, HUANG L Y, YU X L. Analyze monosaccharide composition of polysaccharide in artificial cultivatedby pre-column derivatization HPLC [J]. J Fujian Med Univ, 2016, 50(3): 148–154. 林守二, 黄丽英, 俞晓玲. 柱前衍生化HPLC法分析人工栽培金线莲中多糖的单糖组成 [J]. 福建医科大学学报, 2016, 50(3): 148–154.
[62] LI S L. Studies on the content changes, structural characteristics and pharmacological activities ofpolysaccharides [D]. Hangzhou: Zhejiang Agricultural and Forestry University, 2018: 31. 李帅玲. 金线莲多糖的含量变化、结构表征及药理活性研究 [D]. 杭州: 浙江农林大学, 2018: 31.
[63] YANG Z G, ZHANG X H, YANG L W, et al. Protective effect ofpolysaccharide against CCl4-induced oxidative liver damage in mice [J]. Int J Biol Macromol, 2017, 96: 442–450. doi: 10.1016/j.ijbiomac.2016.12.039.
[64] CHEN L, ZHANG Y P, JIN L X. Preparation, characterization and anti- ageing activity ofblume polysaccharide [J]. Acta Aliment, 2018, 47(2): 210–219. doi: 10.1556/066.2018.47.2.10.
[65] CHEN X, CAO D X, ZHOU L, et al. Structure of a polysaccharide fromBl., and oligosaccharides prepared thereof with anti-pancreatic cancer cell growth activities [J]. Carbohydr Polym, 2011, 86(3): 1300–1305. doi: 10.1016/j.carbpol.2011.06.029.
[66] MING J, LIU J, WU S R, et al. Structural characterization and hypolipidemic activity of a polysaccharide PGEB-3H from the fruiting bodies ofBlume [J]. Procedia Eng, 2012, 37: 169–173. doi: 10.1016/j.proeng.2012.04.221.
[67] LIN P C, WU D T, XIE J, et al. Characterization and comparison of bioactive polysaccharides from the tubers of[J]. Food Hydrocoll, 2015, 43: 199–206. doi: 10.1016/j.foodhyd.2014.05.015.
[68] FANG Y K, NING A H, LI S, et al. Polysaccharides extracted fromhave antitumor propertiesand in an H22 mouse hepatoma ascites model[J]. Int J Mol Sci, 2018, 19(5): 1386. doi: 10.3390/ijms19051386.
[69] YANG H H, WU Y J, GAN C J, et al. Characterization and antioxidant activity of a novel polysaccharide fromLindl. [J]. Carbohydr Polym, 2016, 138: 327–334. doi: 10.1016/j.carbpol.2015.11. 071.
[70] LUO D H, WANG Z J, LI Z M, et al. Structure of an entangled heteropolysaccharide fromLindl. and its anti- oxidant and anti-cancer properties [J]. Int J Biol Macromol, 2018, 112: 921–928. doi: 10.1016/j.ijbiomac.2018.02.051.
[71] XIE J Z, ZOU L H, LUO X, et al. Structural characterization and immunomodulating activities of a novel polysaccharide from[J]. Int J Biol Macromol, 2018, 114: 520–528. doi: 10.1016/j. ijbiomac.2018.03.124.
[72] WANG H L, YEH K W, CHEN P R, et al. Isolation and characteri- zation of a pure mannan from(cv. Gower Ramsey) current pseudobulb during initial inflorescence development [J]. Biosci Biotechnol Biochem, 2006, 70(2): 551–553. doi: 10.1271/bbb.70.551.
[73] BARRETO D W, PARENTE J P. Chemical properties and biological activity of a polysaccharide from[J]. Carbohydr Polym, 2006, 64(2): 287–291. doi: 10.1016/j.carbpol.2005. 11.038.
[74] MENG L Z, LV G P, HU D J, et al. Effects of polysaccharides from different species of(Shihu) on macrophage function [J]. Molecules, 2013, 18(5): 5779–5791. doi: 10.3390/molecules18055779.
[75] HUANG Y P, HE T B, CUAN X D, et al. 1,4--d-glucomannan fromactivates NF-кBTLR4 to regulate the immune response [J]. Molecules, 2018, 23(10): 2658. doi: 10.3390/molecules 23102658.
[76] XIE S Z, LIU B, ZHANG D D, et al. Intestinal immunomodulating activity and structural characterization of a new polysaccharide from stems of[J]. Food Funct, 2016, 7(6): 2789– 2799. doi: 10.1039/C6FO00172F.
[77] LUO D D, QU C, LIN G S, et al. Character and laxative activity of polysaccharides isolated from[J]. J Funct Foods, 2017, 34: 106–117. doi: 10.1016/j.jff.2017.04.024.
[78] XIE S Z, LIU B, YE H Y, et al.polysac- charide regionally regulates intestinal mucosal barrier function and intestinal microbiota in mice [J]. Carbohydr Polym, 2019, 206: 149– 162. doi: 10.1016/j.carbpol.2018.11.002.
[79] PAN L H, WANG J, YE X Q, et al. Enzyme-assisted extraction of polysaccharides fromand its functional properties and immunomodulatory activity [J]. LWT-Food Sci Technol, 2015, 60(2): 1149–1154. doi: 10.1016/j.lwt.2014.10.004.
[80] LUO A X, FAN Y J. Immune stimulating activity of water-soluble polysaccharide fractions fromLindl. [J]. Afr J Pharm Pharmacol, 2011, 5(5): 625–631. doi: 10.5897/AJPP11.169.
[81] WANG Y, LIU J J, LI Q, et al. Two natural glucomannan polymers, from Konjac and, as bioactive materials for pharmaceutical applications [J]. Biotechnol Lett, 2015, 37(1): 1–8. doi: 10.1007/s 10529-014-1647-6.
[82] CHEN Z Y, CHENG L Z, HE Y C, et al. Extraction, characterization, utilization as wound dressing and drug delivery ofpolysaccharide: A review [J]. Int J Biol Macromol, 2018, 120: 2076– 2085. doi: 10.1016/j.ijbiomac.2018.09.028.
[83] CHEN J C, TIAN S, SHU X Y, et al. Extraction, characterization and immunological activity of polysaccharides from[J]. Int J Mol Sci, 2016, 17(7): 1011. doi: 10.3390/ijms17071011.
[84] FERREIRA S S, PASSOS C P, MADUREIRA P, et al. Structure- function relationships of immunostimulatory polysaccharides: A review [J]. Carbohydr Polym, 2015, 132: 378–396. doi: 10.1016/j.carbpol. 2015.05.079.
[85] LUO Q L, TANG Z H, ZHANG X F, et al. Chemical properties and antioxidant activity of a water-soluble polysaccharide from[J]. Int J Biol Macromol, 2016, 89: 219–227. doi: 10.1016/j. ijbiomac.2016.04.067.
[86] ZENG B Y, SU M H, CHEN Q X, et alAntioxidant and hepato- protective activities of polysaccharides from[J]. Carbohydr Polym, 2016, 153: 391–398. doi: 10.1016/j.carbpol.2016. 07.067.
[87] ZHAO Y P, SON Y O, KIM S S, et al. Antioxidant and anti-hypergly- cemic activity of polysaccharide isolated fromLindl. [J]. J Biochem Mol Biol, 2007, 40(5): 670–677. doi: 10.5483/ BMBRep.2007.40.5.670.
[88] PAN L H, LI X F, WANG M N, et al. Comparison of hypoglycemic and antioxidative effects of polysaccharides from four differentspecies [J]. Int J Biol Macromol, 2014, 64: 420–427. doi: 10.1016/j.ijbiomac.2013.12.024.
[89] FENG C Z, CAO L, LUO D, et al.polysaccharides atte- nuate cognitive impairment in senescence-accelerated mouse prone 8 micemodulation of microglial activation [J]. Brain Res, 2019, 1704: 1–10. doi: 10.1016/j.brainres.2018.09.030.
[90] ZHOU B H, TAN J, ZHANG C, et al. Neuroprotective effect of polysaccharides fromBlume against corticosterone- induced apoptosis in PC12 cellsinhibition of the endoplasmic reticulum stress-mediated pathway [J]. Mol Med Rep, 2018, 17(1): 1182–1190. doi: 10.3892/mmr.2017.7948.
[91] BURTON R A, FARROKHI N, BACIC A, et al. Plant cell wall polysaccharide biosynthesis: Real progress in the identification of participating genes [J]. Planta, 2005, 221(3): 309–312. doi: 10.1007/ s00425-005-1495-7.
[92] DOMOZYCH D S, CIANCIA M, FANGEL J U, et al. The cell walls of green algae: A journey through evolution and diversity [J]. Front Plant Sci, 2012, 3: 82. doi: 10.3389/fpls.2012.00082.
[93] HOCH G. Cell wall hemicelluloses as mobile carbon stores in non- reproductive plant tissues [J]. Funct Ecol, 2007, 21(5): 823–834. doi: 10.1111/j.1365-2435.2007.01305.x.
[94] FRANZ G. Metabolism of reserve polysaccharides in tubers ofL. [J]. Planta Med, 1979, 36(5): 68–73. doi: 10.1055/s-0028- 1097242.
[95] SCHELLER H V, ULVSKOV P. Hemicelluloses [J]. Annu Rev Plant Biol, 2010, 61: 263–289. doi: 10.1146/annurev-arplant-042809-112315.
[96] VOINICIUC C, DAMA M, GAWENDA N, et al. Mechanistic insights from plant heteromannan synthesis in yeast [J]. Proc Natl Acad Sci USA, 2019, 116(2): 522–527. doi: 10.1073/pnas.1814003116.
[97] CHUA M, HOCKING T J, CHAN K, et al. Temporal and spatial regulation of glucomannan deposition and mobilization in corms of(Araceae) [J]. Amer J Bot, 2013, 100(2): 337– 345. doi: 10.3732/ajb.1200547.
[98] CRANG R, LYONS-SOBASKI S, WISE R. Plant Anatomy: A Concept- based Approach to the Structure of Seed Plants [M]. Cham: Springer, 2018: 182. doi: 10.1007/978-3-319-77315-5.
[99] HE C M, WU K L, ZHANG J X, et al. Cytochemical localization of polysaccharides inand the involvement ofin the synthesis of mannan polysaccharides [J]. Front Plant Sci, 2017, 8: 173. doi: 10.3389/fpls.2017.00173.
[100]DOBLIN M S, PETTOLINO F, BACIC A. Plant cell walls: The skeleton of the plant world [J]. Funct Plant Biol, 2010, 37(5): 357– 381. doi: 10.1071/FP09279.
[101]LIEPMAN A H, WILKERSON C G, KEEGSTRA K. Expression of cellulose synthase-like () genes in insect cells reveals that CslA family members encode mannan synthases [J]. Proc Natl Acad Sci USA, 2005, 102(6): 2221–2226. doi: 10.1073/pnas.0409179102.
[102]VERBANČIČ J, LUNN J E, STITT M, et al. Carbon supply and the regulation of cell wall synthesis [J]. Mol Plant, 2018, 11(1): 75–94. doi: 10.1016/j.molp.2017.10.004.
[103]ORELLANA A, MORAGA C, ARAYA M, et al. Overview of nucleo- tide sugar transporter gene family functions across multiple species [J]. J Mol Biol, 2016, 428(16): 3150–3165. doi: 10.1016/j.jmb.2016. 05.021.
[104]BRETON C, ŠNAJDROVÁ L, JEANNEAU C, et al. Structures and mechanisms of glycosyltransferases [J]. Glycobiology, 2006, 16(2): 29R–37R. doi: 10.1093/glycob/cwj016.
[105]DAVIS J, BRANDIZZI F, LIEPMAN A H, et al.mannan synthase CSLA9 and glucan synthase CSLC4 have opposite orien- tations in the golgi membrane [J]. Plant J, 2010, 64(6): 1028–1037. doi: 10.1111/j.1365-313X.2010.04392.x.
[106]BUCHANAN M, BURTON R A, DHUGGA K S, et al. Endo-(1,4)--glucanase gene families in the grasses: Temporal and spatial co- transcription of orthologous genes [J/OL]. BMC Plant Biol, 2012, 12: 235. doi: 10.1186/1471-2229-12-235.
[107]SANDHU A P S, RANDHAWA G S, DHUGGA K S. Plant cell wall matrix polysaccharide biosynthesis [J]. Mol Plant, 2009, 2(5): 840– 850. doi: 10.1093/mp/ssp056.
[108]GOUBET F, BARTON C J, MORTIMER J C, et al. Cell wall gluco- mannan inis synthesised by CSLA glycosyltransferases, and influences the progression of embryogenesis [J]. Plant J, 2009, 60(3): 527–538. doi: 10.1111/j.1365-313X.2009.03977.x.
[109]DHUGGA K S, BARREIRO R, WHITTEN B, et alGuar seed- mannan synthase is a member of the cellulose synthase super gene family [J]. Science, 2004, 303(5656): 363–366. doi: 10.1126/science. 1090908.
[110]GILLE S, CHENG K, SKINNER M E, et al. Deep sequencing of voodoo lily (): An approach to identify rele- vant genes involved in the synthesis of the hemicellulose glucomannan [J]. Planta, 2011, 234(3): 515–526. doi: 10.1007/s00425-011-1422-z.
[111]VERHERTBRUGGEN Y, YIN L, OIKAWA A, et al. Mannan synthase activity in the CSLD family [J]. Plant Sign Behav, 2011, 6(10): 1620– 1623. doi: 10.4161/psb.6.10.17989.
[112]WANG Y, MORTIMER J C, DAVIS J, et al. Identification of an additional protein involved in mannan biosynthesis [J]. Plant J, 2013, 73(1): 105–117. doi: 10.1111/tpj.12019.
[113]GAO F, CAO X F, SI J P, et al. Characterization of the alkaline/neutral invertase gene inand its relationship with polysaccharide accumulation [J/OL]. Genet Mol Res, 2016, 15(2): gmr. 15027647. doi: 10.4238/gmr.15027647.
[114]WAN R L, SUN J, HE T, et al. Cloning cDNA and functional charac- terization of UDP-glucose pyrophosphorylase in[J]. Biol Plant, 2016, 61(1): 147–154. doi: 10.1007/s10535-016-0645-z.
[115]HE C M, YU Z M, TEIXEIRA DA SILVA J A, et al.fromcontributes to mannose content of water- soluble polysaccharides and plays a role in salt stress response [J/OL]. Sci Rep, 2017, 7: 41010. doi: 10.1038/srep41010.
[116]HE C M, ZENG S J, TEIXEIRA da S J A, et al. Molecular cloning and functional analysis of the phosphomannomutase () gene fromand evidence for the involvement of an abiotic stress response during germination [J]. Protoplasma, 2017, 254(4): 1693–1704. doi: 10.1007/s00709-016-1044-1.
[117]YU Z M, HE C M, TEIXEIRA da S J A, et al. Molecular cloning and functional analysis ofrelated to water-soluble polysac- charides fromwith enhanced abiotic stress tolerance [J]. Plant Cell Tiss Org Cult, 2017, 131(3): 579–599. doi: 10. 1007/s11240-017-1308-2.
[118]HE C M, ZHANG J X, LIU X C, et al. Identification of genes involved in biosynthesis of mannan polysaccharides inby RNA-seq analysis [J]. Plant Mol Biol, 2015, 88(3): 219– 231. doi: 10.1007/s11103-015-0316-z.
[119]HE C M, WU K L, ZHANG J X, et al. Cytochemical localization of polysaccharides inand the involvement ofin the synthesis of mannan polysaccharides [J]. Front Plant Sci, 2017, 8: 173. doi: 10.3389/fpls.2017.00173.
[120]ZHANG G Q, XU Q, BIAN C, et al. TheLindl. genome sequence provides insights into polysaccharide synthase, floral development and adaptive evolution [J]. Sci Rep, 2016, 6: 19029. doi: 10.1038/srep19029.
[121]ZHANG G Y, NIE S P, HUANG X J, et alStudy on-acetyl-glucomannan (Dendronan): 7. Improving effects on colonic health of mice [J]. J Agric Food Chem, 2016, 64(12): 2485– 2491. doi: 10.1021/acs.jafc.5b03117.
[122]TEIXEIRA da S J A, DOBRÁNSZKI J, CARDOSO J C, et al. Methods for genetic transformation in[J]. Plant Cell Rep, 2016, 35 (3): 483–504. doi: 10.1007/s00299-015-1917-3.
[123]TEIXEIRA da S J A, JIN X H, DOBRÁNSZKI J, et al. Advances inmolecular research: Applications in genetic variation, identification and breeding [J]. Mol Phylogenet Evol, 2016, 95: 196– 216. doi: 10.1016/j.ympev.2015.10.012.
[124]TEKINŞEN K K, GÜNER A. Chemical composition and physico- chemical properties of tubera salep produced from some Orchidaceae species [J]. Food Chem, 2010, 121(2): 468–471. doi: 10.1016/j.food chem.2009.12.066.
[125]HARIJATI N, MASTUTI R, CHAIRIYAH N, et al. Effects of seeding material age, storage time, and tuber tissue zone on glucomannan content ofBlume [J]. Int J Plant Biol, 2018, 9(1): 34–38. doi: 10.4081/pb.2018.7626.
[126]SING H S, SINGH G, ARYA S K. Mannans: An overview of properties and application in food products [J]. Int J Biol Macromol, 2018, 119: 79–95. doi: 10.1016/j.ijbiomac.2018.07.130.
Advances in Active Polysaccharides in Medicinal Plants of Orchidaceae
ZHANG Ming-ze1,2, HE Chun-mei1, WANG Hao-bin1,2, YU Zhen-ming1, SI Can1,2, ZHAO Cong-hui1,2, DUAN Jun1*
(1. Guangdong Provincial Key Laboratory of Applied Botany, Key Laboratory of South China Agricultural Plant Molecular Analysis and Gene Improvement, South China Botanical Garden, Chinese Academy of Sciences,Guangzhou 510650, China; 2. University of Chinese Academy of Sciences,Beijing 100049, China)
Orchidaceae is the second largest family of flowering plants. There are 28 000 species in 736 genera identified, among them, 343 species in 82 genera are used as medicine, such as,,,, and so on. The main functional constituents in medicinal Orchidaceae plants are polysaccharides, stilbene derivatives, phenolics, terpenoids, alkaloids, flavones and sterols. Water-soluble polysaccharide in orchid plant are mainly composed of mannose and glucose, which is one of important pharmacological active constituents with many pharmacological functions, such as enhancing immunity, anti-tumor, anti-oxidation, lowering blood glucose and improving memory. The structure, pharmacological function and biosynthesis of active polysaccharide in medicinal Orchidaceae plants are reviewed, and some key problems were proposed for further research, which would provide references for further promoting the protection and rational exploitation of medicinal resources of Orchidaceae.
Orchidaceae; Polysaccharide; Pharmacological activity; Biosynthesis
10.11926/jtsb.4073
2019-04-02
2019-06-15
国家自然科学基金项目(31871547)资助
This work was supported by the National Natural Science Foundation of China (Grant No. 31871547).
张明泽(1986~ ), 男, 博士研究生, 研究方向为铁皮石斛生物技术育种。E-mail: zhangmingze@scib.ac.cn
E-mail: duanj@scib.ac.cn

