Thermostable ethanol tolerant xylanase from a cold-adapted marine species Acinetobacter johnsonii☆
Dongsheng Xue,Xuhao Zeng,Dongqiang Lin,Shanjing Yao,*
1 Key Laboratory of Fermentation Engineering(Hubei Universty of Technology),Ministry of Education,Hubei Provincial Cooperative Innovation Center of Industrial Fermentation,Hubei Key Laboratory of Industrial Microbiology,Hubei University of Technology,Wuhan 430068,China
2 Department of Chemical and Bioengineering,Zhejiang University,Hangzhou 310027,China
Keywords:Xylanase Ethanol tolerant Thermostable Cold-adapted Acinetobacter Johnsonii
ABSTRACT A xylanase-producing bacterium,isolated from deep sea sediments,was identified as the cold-adapted marine species Acinetobacter Johnsonii.A cold-adapted marine species Acinetobacter Johnsonii could grow at 4°C.The optimum temperature and pH of xylanase from a cold-adapted marine species Acinetobacter Johnsonii were 55°C and pH 6.0.Xylanase from a cold-adapted marine species Acinetobacter Johnsonii remained at 80%activity after incubation for 1 h at 65 °C.The xylanase activity was 1.2-fold higher in 4%ethanol solution than in ethanol free solution.Gibbs free energy of denaturation,ΔG,was higher in 4%ethanol solution than in ethanol free solution.Thermostable ethanol tolerant xylanase was valuable for bioethanol production by simultaneous saccharification and fermentation process with xylan as a carbon source.
1.Introduction
Xylan,a major component of hemicellulose in plant cells,accounts for about 35%of the dry weight of plant cells.It is the most abundant polysaccharide in the earth except cellulose[1].Xylan consists of xylosyl residues.Xylosyl residues are substituted by α-L-arabinofuranose,α-(4-O-methyl)glucuronic acid and O-acetyl esters[2].Xylanase can hydrolyze xylan into xylo-oligosaccharide and pentose sugars,which are carbon sources used in brewing, feed, chemical industry [3-5]. Compared with chemical hydrolysis, xylan hydrolysis with xylanase has the advantages of high hydrolysis efficiency,low environmental pollution,and mild hydrolysis conditions[6,7].
There are abundant microbes in oceans [8]. The environment of deep sea has the characteristics of low organic content,low temperature,and high salinity [9, 10]. Diversified microorganisms in deep sea showed special metabolisms [11]. Some microorganisms in deep sea could produce hydrolase to hydrolyze macromolecule for their nutrition.Cold-adapted microorganisms can grow in deep sea environment.Common microorganisms produce enzymes at temperatures of 20°C-40°C.It consumes an amount of energy to maintain the temperature under low temperature condition. Producing enzymes with cold-adapted microorganisms can reduce energy consumption to maintain the temperature.
Simultaneous hydrolysis and fermentation process is a promising process to produce bioethanol with a low cost.Common xylanase loses the catalytic activity in high ethanol solution. Ethanol tolerant xylanase,which can show higher activity in high ethanol solution,is valuable for bioethanol production by simultaneous hydrolysis and fermentation process.Thermostable ethanol tolerant cellulase has been discovered[12].Thermostable ethanol tolerant xylanase has not been reported.
Thermostable ethanol tolerant xylanase is necessary for bioethanol production by simultaneous hydrolysis and fermentation process with a low cost.Production of thermostable xylanase from a cold-adapted microorganism can reduce xylanase cost to maintain temperature.
A xylanase producing bacterium,Acinetobacter Johnsonii was isolated from deep sea sediment. Acinetobacter Johnsonii could grow at a low temperature of 4 °C. The catalytic characteristics of the thermostable ethanol tolerant xylanase from a cold-adapted Acinetobacter Johnsonii were analyzed.
2.Experimental
2.1.Strain
2.2.Media
Enrichment medium(2216E seawater medium)was composed of 5.0 g peptone,1.0 g yeast extract,FePO40.1 g,and artifciial sea water 1.0 L,pH 7.0.Seed medium was same with the enrichment medium.
Isolation medium was composed of 15.0 g xylan, NH4NO35.0 g,K2HPO42.0 g,MgSO4·7H2O 5.0 g,agar 20.0 g,and artificial sea water 1.0 L,pH 7.0.
Enzyme producing medium was composed of 20.0 g bran,10.0 g yeast extract,and artificial sea water 1.0 L,pH 7.0.
2.3.Isolation of strains
10.0 g of deep sea sediment was added into a flask(250 ml)with 90.0 ml of artificial sea water. After being shaken for 30 min at 200 r·min-1, 1.0 ml of suspension was added into the enrichment medium.
After incubation for 2 d at 18 °C, the enrichment medium was diluted to 104cells·ml-1. The diluted liquid was smeared on the isolation medium.After cultivation for 48 h at 18°C,the single colonies were inoculated into the seed medium and cultured for 2 d at 18°C.
2.4.Construction of phylogenetic tree
27F primer:5′-AGAGTTT GATCCTGGCTCAG-3′and 1492R primer:5′-GGTTACCTTGTTA CGACTT-3′ were used to amplify the 16S rDNA sequence. The products of PCR were sequenced and blasted in NCBI.The similar sequences were chosen to construct a phylogenetic tree based on 16S rDNA homology[13].
2.5.Growth of Acinetobacter Johnsonii at different temperatures
1 ml of seed liquid was inoculated into 50 ml of the seed medium.
As a matter of fact, if anyone had a fancy for a town, or a province, he helped himself to it; but as long as the King had his horses and dogs, and the Queen her musicians and her actors, they did not trouble themselves about the matter
The mixture was cultured at 4 °C,18 °C and 30 °C,respectively.3 ml liquid samples were taken out each time and OD600of the samples was measured.
2.6.Xylanase purification
The fermentation broth was filtered through 8 layers of gauze and centrifuged at 10000 r·min-1for 10 min at 4 °C. Ammonium sulfate was added into the supernatant to saturation. Precipitation was collected and redissolved.The result solution was taken as the column sample and loaded onto a Sephadex G-100 column (26 mm× 700 mm).Elution was at a flow rate of 1.3 ml·min-1.Elution solution with the highest xylanase activity was loaded into a Sepharose 4FF gel column at a flow rate of 2 ml·min-1. Xylanase activity in collection tubes was measured.The solution in the tube with the xylanase activity was used as purified xylanase.The purification of xylanase was carried out by electrophoresis.
The amount of enzyme required to release 1 μmol reducing sugar(in terms of xylose)per minute is defined as one enzyme activity unit(U).
2.7.Optimal temperature and pH
0.1 ml of the purified xylanase solution was mixed with 0.9 ml of xylan solution (2 mg·ml-1). The mixture was adjusted to designed pH and incubated at different temperatures.The released reduce sugar was measured by the DNS method[14].
2.8.Thermostability of xylanase at different temperature and pH
The purified xylanase was incubated at different temperature and pH conditions.2 ml of samples was taken out at a 30 min interval.The remaining xylanase activity was analyzed.
2.9.Effect of metal ions and chemical reagents on xylanase
2 ml of metal ion solution (1 mmol·L-1) or 1 g·L-1solution of EDTA,polyethylene glycol 6000,urea,Tris,and Tween 80 was mixed into 2 ml of purified xylanase solution.The mixture was incubated at room temperature for 30 min. The remaining xylanase activity was analyzed.
2.10.Ethanol tolerance of xylanase
Ethanol was added into a purified xylanase solution.The mixture was incubated for 30 min at 30 °C. The remaining xylanase activity was analyzed.
2.11.Kinetics of thermal deactivation of xylanase
The purified xylanase solution was mixed with ethanol solution and incubated at 55 °C,60 °C, and 65 °C. The remaining xylanase activity was measured at a 10 min interval.The kinetic parameters of thermal deactivation were calculated out as the method described by Dongsheng Xue[15].

Fig.1.Phylogenetic tree based on 16S rDNA sequences.
3.Results and Discussion
3.1.Identification of the isolated bacteria
The sequence of 16S rDNA of the isolated bacteria was 1427 bp.The sequence was blasted in NCBI.The similar sequences were chosen to construct a phylogenetic tree(Fig.1).
Based on the phylogenetic tree,the isolated bacteria were identified as Acinetobacter johnsonii.
As shown in Fig.2,the marine species Acinetobacter johnsonii could grow fast at 4 °C. It is a cold-adapted bacterium. An Acinetobacter johnsonii strain was isolated from city sewage.It was not a cold-adapted strain[16].A cold-adapted Acinetobacter johnsonii strain has not been reported.A cold-adapted bacterium could produce enzymes at low temperature. Producing xylanase with a cold-adapted Acinetobacter Johnsonii had the advantage of reducing energy consumption.

Fig.2.Growth of Acinetobacter johnsonii at different temperatures.
3.2.Optimum temperature and pH of xylanase activity
SDS PAGE of the purified xylanase was shown in Fig.3.The molecular weight of the xylanase was about 14.5 kDa.
As shown in Fig.4,the optimal temperature of xylanase activity was 55°C.Xylanase from a cold-adapted Acinetobacter Johnsonii was thermostable.Thermostable xylanase could efficiently hydrolyze xylan at high temperature.
As shown in Fig.4,the optimal pH of xylanase from a cold-adapted Acinetobacter Johnsonii is pH 6.0. The xylanase had a high activity in the wide range of pH 4 to 7.The xylanase maintained above 69%activity at pH 4.The xylanase belonged to acidic xylanase.In the food industry,most reactions are reacted in an acidic environment,especially in the brewing industry[17].Xylanase from a cold-adapted marine species Acinetobacter Johnsonii was valuable for the food industry.
As shown in Fig.5,the xylanase solution was incubated at 55°C for 2 h and xylanase retained more than 65%of the original activity.The optimum temperature and pH of xylanase from Aspergillus terreus were 50 °C and pH 6.0 [18]. The optimum temperature and pH of xylanase from Penicillium occitanis Pol6 and Penicillium funiculosum GH11 were at 45°C and pH 4.0 and 40°C and pH 3.0[19].
As shown in Fig.6,the xylanase remained at above 50%activity after incubation for 96 h at pH 4.Incubated in the environment of pH 4-5 for 10 d, xylanase retained some activities. The results showed that the xylanase was thermostable at an acidic environment. Immobilized xylanase showed stability at a wide range of pH and temperature too[20]. Immobilization technology probably was used to increase the thermostability of xylanase from a cold-adapted Acinetobacter Johnsonii.

Fig.3.SDS PAGE of the purified xylanase from Acinetobacter johnsonii.A.Protein marker;B.purified protein.

Fig.4.Effect of temperature and pH on the xylanase activity.
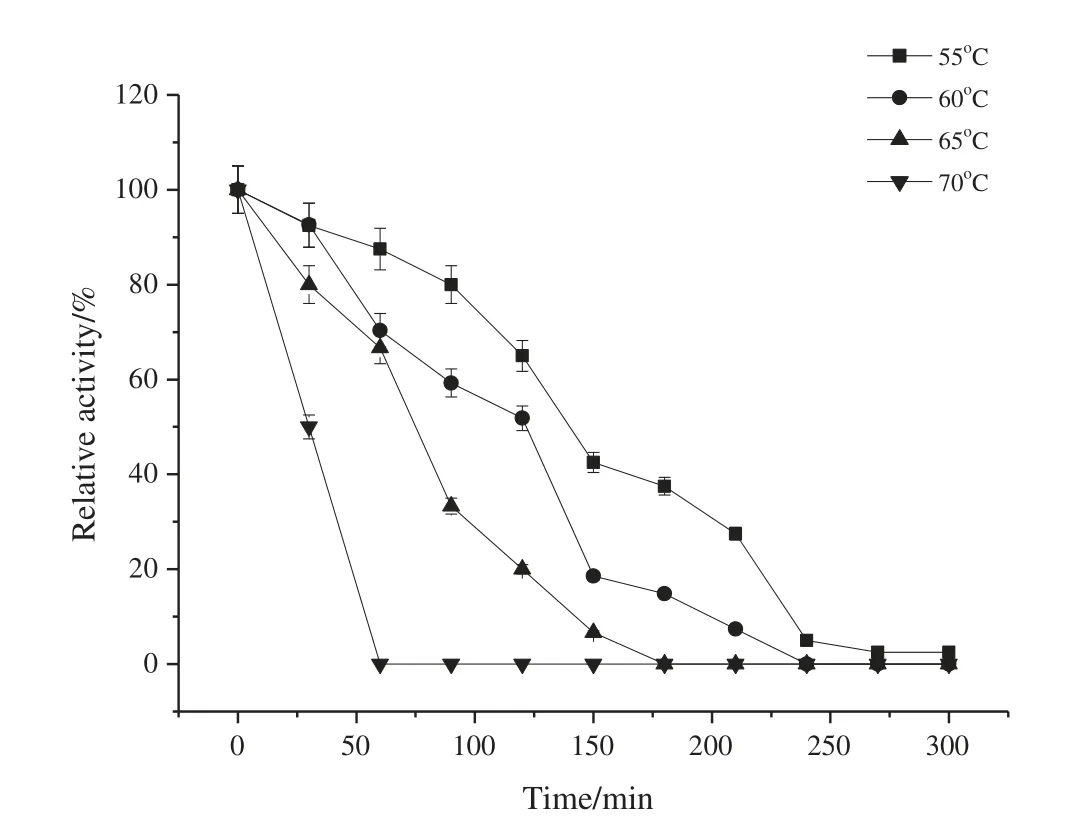
Fig.5.Thermostability of the xylanase at different temperatures.

Fig.6.Thermostability of the xylanase at different pH.
3.3.Effect of metal ions and chemical reagents on xylanase activity
The effect of metal ions on the xylanase activity is shown in Fig.7.Fe2+,Fe3+and Zn2+could increase the xylanase activation.Cu2+and Mg2+decreased the activity. Previous studies reported that heavy metal ions,such as Cu2+,Mn2+,and Co2+,could strongly inhibit the xylanase activity.The reason was that heavy metal ions could bind irreversibly to the main polypeptide or side chains[21,22].Xylanase from a cold-adapted Acinetobacter Johnsonii showed excellent tolerance to the metal ions.It was only slightly inhibited by K+,Al3+and Pb2+.

Fig.7.Effect of metal ions on the xylanase activity.
As showed in Fig.8,polyethylene glycol 6000,urea,Tris,Tween 80 and EDTA decreased the xylanase activity.EDTA is a chelating agent[23] and its inhibition ability indicated that specific ions might be actively involved in the catalytic reaction of the xylanase[24].
3.4.Ethanol tolerance of xylanase

Fig.8.Effect of chemical reagents on the xylanase activity.
As shown in Fig. 9, the activity of xylanase from a cold-adapted Acinetobacter Johnsonii increased in ethanol solution. The xylanase remained at about 50%activity in 16%ethanol solution.The xylanase was ethanol tolerant. Xylanases from Aspergillus oryzae showed also ethanol tolerance[25].The excellent ethanol tolerance means that it was valuable for the production of bioethanol by a simultaneous hydrolysis and fermentation process[26].

Fig.9.Effect of ethanol concentration of the xylanase activity.
3.5.Thermal deactivation kinetics of xylanase
Gibbs free energy,ΔG,was an effective parameter to anal yze enzyme thermostability.The higher the ΔG was,the more thermostable the enzymes were[27].Gibbs free energy,ΔG was higher in 4%ethanol solution than ethanol free solution (Table 1). The xylanase was more stable in 4%ethanol solution than in ethanol free solution.The xylanase probably had a more stable structure in 4%ethanol solution.Ethanol is apolar compound.An oxhydryl of ethanol probably interacted with basic amino acids.The interaction probably made the xylanase have a different structure or molecular bridges[15].Structure or molecular bridge change probably increased the stability.

Table 1 Gibbs free energy of xylanase in different concentrations of ethanol solution.
4.Conclusions
The xylanas from a cold-adapted Acinetobacter johnsonii was ethanol tolerant and thermostable. The xylanase from a cold-adapted Acinetobacter johnsonii displayed higher activity and better thermostability in 4%ethanol solution.The xylanase from a cold-adapted Acinetobacter Johnsonii was valuable for bioethanol production by simultaneous hydrolysis and fermentation.
 Chinese Journal of Chemical Engineering2019年5期
Chinese Journal of Chemical Engineering2019年5期
- Chinese Journal of Chemical Engineering的其它文章
- Assessment of the TFM in predicting the onset of turbulent fluidization☆
- CFD study on double-to single-loop flow pattern transition and its influence on macro mixing efficiency in fully baffled tank stirred by a Rushton turbine☆
- Simulation of drop breakage in liquid-liquid system by coupling of CFD and PBM:Comparison of breakage kernels and effects of agitator configurations☆
- Heat transfer characteristics of molten plastics in a vertical falling film reactor☆
- Stabilizing silica nanoparticles in high saline water by using polyvinylpyrrolidone for reduction of asphaltene precipitation damage under dynamic condition
- Numerical simulation and experimental study on dissolving characteristics of layered salt rocks
