Dry environment on the expression of lacrimal gland S100A9, Anxa1, and Clu in rats via proteomics
Yi-Lin Sun, A-Yuan Cui, Li-Xin Wang, Wang-Wang Zhang, Hong Shi,2
1College of Traditional Chinese Medicine, Xinjiang Medical University, Urumqi 830011, Xinjiang Uygur Autonomous Region, China
2Research on High Incidence of Diseases in Xinjiang Region,Key Laboratory of the Ministry of Education (Xinjiang Medical University), Urumqi 830011, Xinjiang Uygur Autonomous Region, China
Abstract
● KEYWORDS: dry eye; lacrimal gland; S100A9; Clu;Anxa1; environment; rats
INTRODUCTION
Dryness can be generalized as a climate environment with low relative humidity as the primary feature[1].Dryness is the main manifestation in autumn[2].In the traditional Chinese medicine (TCM) theory, records of the cause of dryness can be traced back to theInternal Medicine Classic of the Yellow Emperorwrote in the Chinese Western Han Dynasty.TCM believed that “dryness always feels astringent for people, is easy to hurt body fluid, and most likely to hurt lung”.When the body cannot tolerate dry environment, the“dryness” in the environment will cause diseases, which was named as “dryness evil” in TCM[3].
The organs exposed to the body surface are the most vulnerable influenced by external environment.As for the eyes and nose, the lacrimal gland secreted tears to keep the eyes surface moist[4].It also protects the eye surface exposed to the environment from the damage of different pathogens.Tears flow into the nasal cavity through the nasolacrimal duct, which can keep the nasal cavity moist and the humidity of the inhaled air.Tears also play a local immune role in the nasal mucosa to protect the airway[5].
It has reported that lung system (trachea, nose orifices,lung tissue) is influenced by dry environment.The lacrimal glands lobules of rats were slightly atrophic, the connective tissues between the lobules were proliferative, the glandular cavities were expanded, the columnar secretory cells were vacuolized, and the defensin beta-2 of lacrimal gland was“transient” enhancement[5].In order to further explored such above phenomenon, our present study screened the differential expressed proteins in lacrimal glands based on proteomics and verified the relevant target proteins.It provides a modern scientific evidence for finding the underlying mechanism of dry environment in TCM affecting the eyes symptom.
MATERIALS AND METHODS
Ethical ApprovalThe study was approved by the Ethics Committee in Animal Experimentation of Xinjiang Medical University [No.SYXK (New) 2011-0004], and protocol was reviewed and approved by Xinjiang Laboratory Animal Ethics Committee (IACUC20160616-05).
Animals Grouping and InterventionFemale SD rats weighing 140-160 g were randomly divided into two groups(n=10/group).The control group was fed in the specific pathogen free standard environment; the dryness group was placed in the artificial climate chamber and interfered with autumn dryness.
Artificial Simulation of Autumnal DrynessUsing the previous method[6](Table 1), artificial climate chamber (model PRX-1250B, Deyangyibang Company, Shanghai, China) in the laboratory was applied to create dry environment, by adjusting the relative humidity, temperature, wind speed, light intensity to simulate autumn dryness.The temperature parameters of Class I (4℃-24℃) and Class III (10℃-30℃) were set and used in the artificial climate chamber according to the time interval, the average daily temperature difference was ≥20℃;the relative humidity was 5%-45%, the average daily relative humidity was 24.33%±11.29%, the wind speed was 2.1 m/s, and the light intensity was 5-20 light intensity (lx; the ratio of day and night light duration was not less than 10:14).The starting and ending time of the 1stperiod is 23:00-01:00, and the 12thperiod is 21:00-23:00.The duration of each period is 2h.The numbered periods 1-12 correspond to 24thof a day.
Experimental Specimen CollectionRats lacrimal glands were collected 24d later, the left lacrimal gland was washed in 0.9% 4°C saline, then placed in lyophilization tubes for liquid nitrogen freezing and stored at -80℃ in the refrigerator for test.The right lacrimal gland was fixed with 4% paraformaldehyde,dehydrated routinely, embedded manually into the paraffin for test.

Table 1 The artificial climate chamber to simulate autumn dryness
Hematoxylin-eosin Staining for Lacrimal Gland TissueThe lacrimal gland sections were baked in a 63℃ oven,dewaxing routinely, hematoxylin solution (20031605,Zhongshan Company, Beijing, China) staining, washed with distilled water, then differentiation with ethanol hydrochloride for 1s, reverse blue with distilled water, eosin solution (20041701,Zhongshan Company, Beijing) staining, washed with distilled water, dehydration routinely, natural drying at room temperature (23℃-26℃), and then sealed with neutral gum.
Proteomic TestingThe protein concentration of lacrimal gland tissues were determined by bicinchronic acid (BCA)kit (P0011, Biyuntian Company, Shanghai).Pancreatin(SRT220102, Promega, Beijing) decomposed the protease into the peptide segments, and TMT kit (WD330302, Thermo Fisher Scientific, Shanghai) marked the segments.The labeled peptides were graded by applying high performance liquid chromatography (1260, Agilent, Shanghai, China), then mobile phase A [0.1% formic acid (0172192381, Fluka, Germany)and 2% acetonitrile (F22M6E201, Thermo Fisher Scientific,Shanghai) aqueous solution] and mobile phase B (0.1%formic acid and 90% acetonitrile aqueous solution) of high performance liquid chromatography were used for separating,and ionized in the protein electrophoresis instrument (EPS-300,Tianneng Company, Shanghai).Finally, the labeled peptides were analyzed by the mass spectrometer (Q Exactive HF-X,Thermo Fisher Scientific, Shanghai).Data were collected by data dependent acquisition program, and secondary mass spectrometry data were retrieved by Maxquant (v1.5.2.8).
Proteomic AnalysisProtein quantification repeatability was assessed by principal component analysis, relative standard deviation and Pearson’s correlation coefficient.The difference significance of the quantified proteins to obtain theP-value was used the two-tailt-test.WhenP<0.05, the fold change(FC) >1.2 and <1/1.2 were the change thresholds of significant up-regulated and significant down-regulated, respectively.Bioinformatics analysis was performed for differential expressed proteins in dryness group.UniProt-GOA database was used for Gene Ontology (GO) annotation classification and GO enrichment, and Kyoto Encyclopedia of Genes and Genomes (KEGG) database was used for KEGG pathway enrichment.
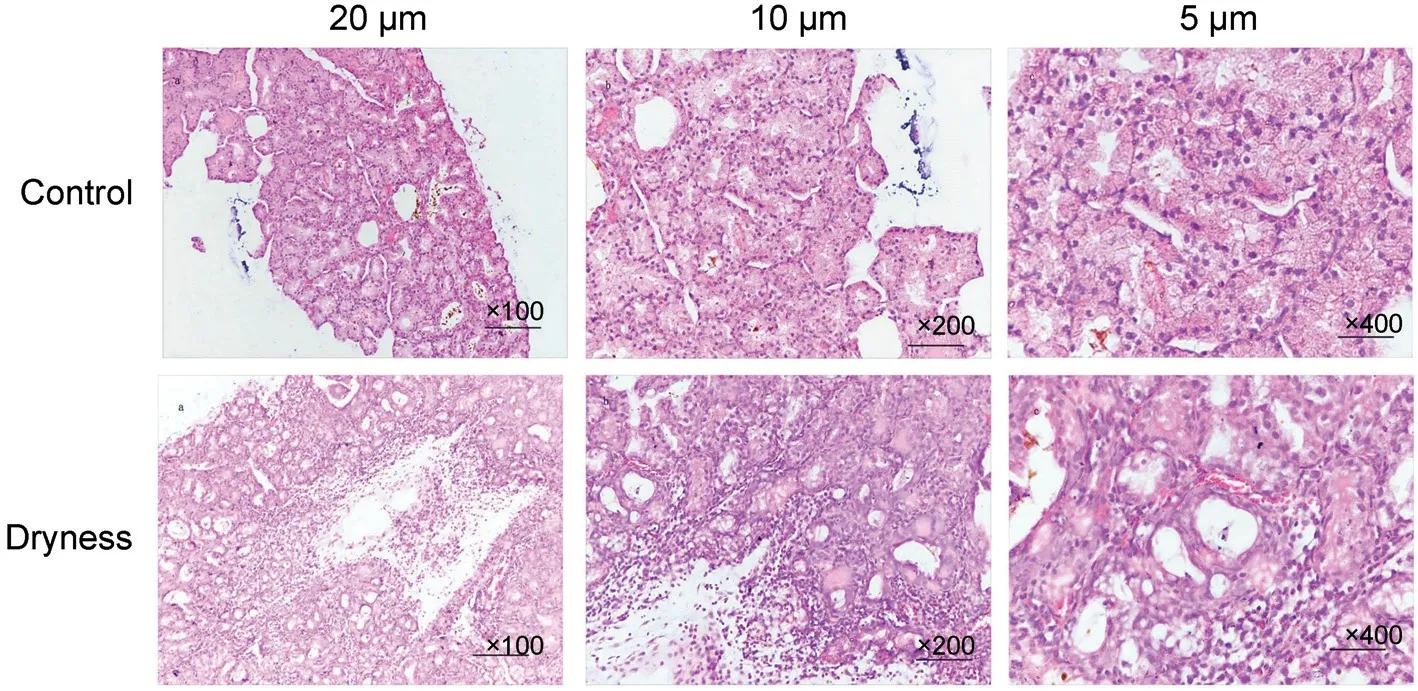
Figure 1 Hematoxylin-eosin staining of lacrimal gland in rats n=10.
Immunohistochemical AnalysisThe lacrimal gland sections were baked in the 63℃ oven, and dewaxing routinely.The sections were soaking in 3% H2O2and placed in citrate solution, heat them in microwave oven, and put them natural at room temperature.Put the sections into the wet box and drip goat serum solution (60 μL/sample; 20050901, Zhongshan Company, Beijing) to cover all tissues and seal at room temperature.The sections were shaken dry, and incubated with the primary antibodies as Abcam rabbit anti-S100A9 antibody(1:200; 26992-1-AP, Xiangtai Company, China), Abcam rabbit anti-Anxa1 antibody (1:200; Ab-AF5154, Xiangtai Company, China), and Abcam rabbit anti-Clu antibody (1:200;Ab-DF6431, Xiangtai Company, China), and then placed in the refrigerator at 4℃.The next day, the sections were rewarmed at room temperature, washed with phosphate buffer saline (PBS) buffer.The sections were dried with shaking,and added the second antibody (60 μL/sample; PV-9001,Zhongshan Company, Beijing), reacted at room temperature.The sections were developed with diaminobenzidine (DAB)chromogen substrate (K186916P, Zhongshan Company,Beijing, China), and color development was terminated by distilled water.Sections were counterstained with hematoxylin,differentiation in 1% hydrochloric acid ethanol for 1s, dried at room temperature after dehydrated routinely, and then sealed with neutral gum.Under light microscope (LEICA DM3000,Lecia Company, US), The photographs were randomly taken from each section.The integral optical density (IOD) value(IOD/area) was analyzed and calculated by Image-Pro Plus 6.0 software, and the average value of IOD value of six sections were used to represent the protein expression level of the section.The values were obtained as arbitrary units (a.u.).
Statistical AnalysisAll data were entered into SPSS 26.0 was used for statistical analysis.Origin Pro 8.5 was used for plotting.Means between the two groups were compared usingt-test when the variances were equal; approximatet-test when the variances were not equal.The test levelα=0.05, andP<0.05 as the difference was statistically significant.
RESULTS
Effect of Autumn Dryness Intervention on the Histomorphology of Lacrimal Gland in RatsThe hematoxylin-eosin (HE) staining of rats lacrimal gland under autumn dryness intervention were shown in Figure 1.The lacrimal gland acini of rats in control were irregular round and shaped cell islands, with complete structure and tight arrangement, which were divided into lobule by connective tissues, glandular cavities were narrow, cytoplasm of secretory cells was uniform, nuclear distribution was uniform, but a few yellow and pink secretions appeared.In dryness, the connective tissue between the lobules of lacrimal gland proliferated significant, a large number of acini atrophied and fused, the expansion of glandular cavities were large, secretions (pink)appeared inside, a large number of macrophages appeared in the acinar cavity and interstitium, columnar secretory cells arranged closely and numerous vacuoles appeared, the nucleus were scattered and sparse, and local inflammatory infiltration was obvious.It is suggested that the fine structure of lacrimal gland in rats has pathological changes and local inflammatory reaction under the intervention of autumn dryness.
Differential Expressed Proteins in the Lacrimal Glands of Rats in DrynessAs shown in Figure 2, a total of 3375 proteins were identified, and 431 differential express proteins were screened in the lacrimal gland of rats from dryness group.There were 195 up-regulated proteins, 34 proteins with FC>1.5, and 236 down-regulated proteins, and 5 proteins with FC>0.83.
Major Differential Expressed Proteins in the Lacrimal Glands of Rats in DrynessThe proteomic result of the rat lacrimal gland differential proteins revealed (P<0.05; Table 2)that the higher FC proteins among the up-regulated differential proteins including Odorant-binding protein, S100A9,Reproductive homeobox on X chromosome 12,etc.,andthe higher FC proteins among the down-regulated differential proteins included isoform CRA_a, long-chain-fatty-acid-CoA ligase 5, Mannose receptor, C type 2,etc.
GO Functional Classification and Enrichment of Differential Expressed Proteins in the Lacrimal Gland of DrynessAs shown in Figures 3 and 4, GO functional classification and enrichment of differential expressed proteins in the lacrimal gland of dryness revealed (P<0.05) that the biological processes (BP) of differential expressed proteins were mainly included cellular process, single-organism process, biological regulation, metabolic process,etc.Upregulated differential expressed proteins were mainly enriched in positive regulation of cytokine production, regulation of angiogenesis, regulation of vasculature development,etc.;down-regulated differential expressed proteins were mainly enriched in peptide biosynthetic process, peptide metabolic process, amide biosynthetic process,etc.The CC of differential expressed proteins were mainly located in cell, organelle, cell membrane,etc.Up-regulated differential expressed proteins were mainly enriched in extracellular space, extracellular region, extracellular region part,etc.; down-regulated differential expressed proteins were mainly enriched in ribosome, ribosomal subunit, cytosolic ribosome,etc.The MF of differential expressed proteins were mainly included binding and catalytic activity.Up-regulated differential expressed proteins were mainly enriched in endopeptidase activity,peptidase activity, cysteine-type endopeptidase activity,etc.;down-regulated differential expressed proteins were mainly enriched in structural constituent of ribosome, structural molecule activity, extracellular matrix (ECM) structural constituent,etc.
KEGG Pathway Enrichment of Differential Expressed Proteins in the Lacrimal Gland of DrynessAs shown in Figure 5, KEGG pathway enrichment of lacrimal gland differential protein in dryness revealed (P<0.05) that the upregulated differential expressed proteins were mainly enriched in the lysosome, complement and coagulation cascades, and antigen processing and presentation and other pathways.The down-regulated differential proteins were mainly enriched in ribosome, ECM-receptor interaction, protein digestion and absorption and other pathways.
Correlation Pathway of Differential Expressed Proteins in the Lacrimal Gland of DrynessAs shown in Figure 6A, the up-regulated differential expressed proteins in the complement and coagulation cascades pathway with the high FC were complement C2, Clu, T-kininogen 2.The following up-regulated differential expressed proteins with the lower FC including C4b binding protein alpha chain,T-kininogen 1.Down-regulated differential expressed proteins in the complement and coagulation cascades pathway were complement C6, fibrinogen gamma chain, fibrinogen beta chain, fibrinogen alpha chain.As shown in Figure 6B, the up-regulated differential expressed proteins in the lysosome pathway with the high FC were Cathepsin S (CTSS) and Cathepsin H.The following up-regulated differential expressed proteins with the lower FC including cathepsin Z, carboxypeptidase, alpha-N-acetylgalactosaminidase,N-acetylglucosamine-6-sulfatase, dipeptidyl peptidase 1,clathrin light chain B,etc.Down-regulated differential expressed proteins in the lysosome pathway were prosaposin and V-type proton ATPase subunit.
Validation of Key ProteinsThrough bioinformatics analysis of differential expressed proteins, the study selected S100A9,Anxa1, and Clu as key targets for IHC verification.As shown in Figure 7, compared with control group, the expression of S100A9, Anxa1, and Clu were enhanced in the lacrimal glands of dryness group (P<0.05), which was consistent with the proteomic findings.
DISCUSSION

Figure 3 GO functional classification of differential expressed proteins A: Up-regulated differential proteins; B: Down-regulated differential proteins.GO: Gene Ontology.

Figure 4 Differential protein GO function enrichment A: Up-regulated differential proteins; B: Down-regulated differential proteins.The ordinate represents the P-value of the two-tailed t-test with a log-negative base of 10.GO: Gene Ontology.
When the “dryness evil” prevailed, people who were attacked by “dryness evil” often have sub-health symptoms such as dryness and discomfort in the mouth, eyes, nose, throat; dry cough, less phlegm, dry skin, astriction,etc[7].It has been found that indoor dry environment (microclimatic parameters)could cause dry eyes and visual fatigue[8], and at present people had bad living habits such as frequent use of air-conditioning and excessive use of eyes, which made increase the risk of developing the infectious diseases.In this study, the differential expressed proteins in lacrimal gland were analyzed by artificial simulation of autumn dryness in the laboratory, and S100A9,Clu and Anxa1 were selected as target proteins for verification,and hope to provide a scientific basis for dry environment affecting eyes symptom.S100A9 is found widely distributed in human epithelial tissues,interacted with enzymes, receptors, transcription factors,cytoskeletal proteins,etc[9].When S100A9 was released into the extracellular environment, it could mediate inflammation and prevent pathogen invasion, also be produced during tear oxidation change reaction.It plays a regulatory role in cell apoptosis, oxidative stress, inflammatory response, and repair processes[10].Numerous reports have indicated that S100A9 is up-regulated in immune dry eyes, ocular microbial infection,eye redness, corneal abrasion, transient blurred vision and other ocular diseases[9,11].Up-regulation of S100A9 may be associated with the dry environment-induced cell apoptosis,mediated ocular inflammation and caused local injury.Anxa1 refers to an effective anti-inflammatory medium, it can be secreted in different immune cells.For example, Anxa1 could block the activation of eye neutrophils and regulate the migration of leukocytes and the produced of pro-inflammatory mediators in neutrophils[12].Anxa1 could also mediate the adhesion, chemotaxis, aggregation, and other processes of inflammatory cells by activating multiple anti-inflammatory pathways, and inhibiting the production of inflammatory cytokines[13-14].It could activate the PLC pathway, stimulate mucin secretion from the conjunctival goblet cells, inhibite ERK1/2 phosphorylation, promote the homeostasis of the tear membrane mucin layer, and relieve the inflammatory cell infiltration of eye conjunctiva[15].In prsent study, the upregulated expression of Anxa1 demonstrated that there was a possibility of inflammation and ocular surface homeostasis imbalance caused by dry environment.
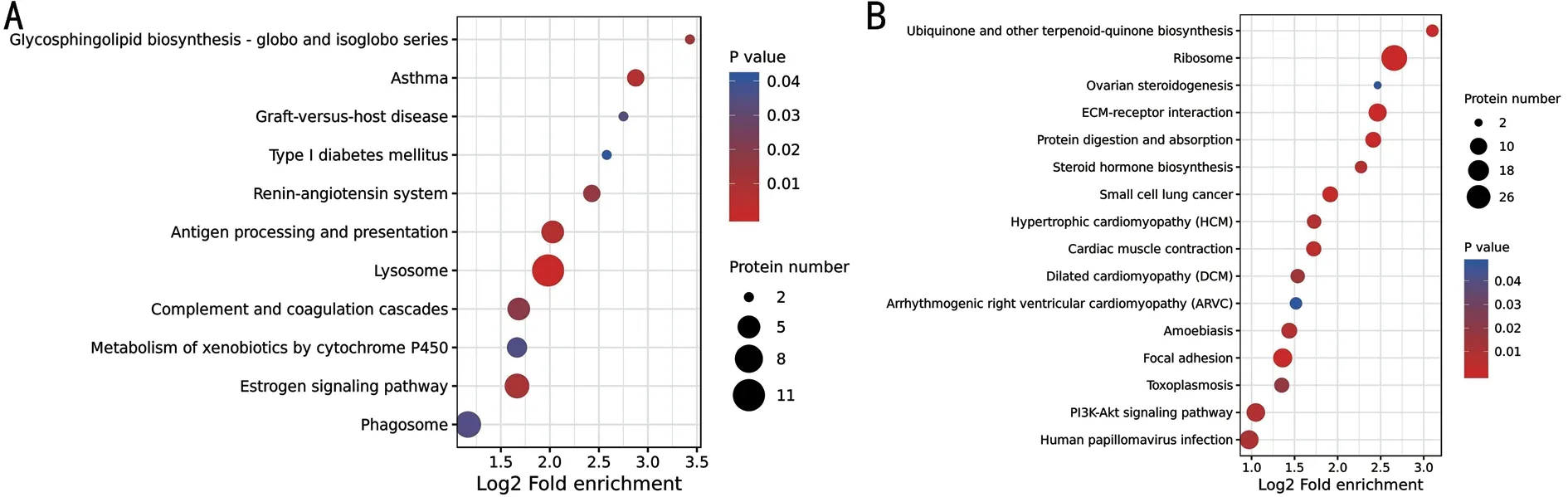
Figure 5 Differential protein KEGG pathway enrichment analysis A: Up-regulated differential proteins; B: Down-regulated differential proteins.The abscissa is the enrichment degree, and the ordinate is the KEGG path information.The size of the circle indicates the number of differential expressed proteins in the mapping pathway.The larger the circle, the more the number.The color of the circle represents the size of the P-value.The bluer the color is the larger P-value.KEGG: Kyoto Encyclopedia of Genes and Genomes.

Figure 6 Correlation pathway diagram The up-regulated differential expressed proteins are in red, down-regulated differential expressed proteins are in green.A: The complement and coagulation cascade pathway; B: The lysosome pathway.

Figure 7 Expression of S100A9, Anxa1, and Clu in lacrimal gland of rats A: IHC analysis the expression of S100A9, Anxa1, and Clu under light microscope ×400 Scale 5 μm; B: The results confirmed the expression of S100A9, Anxa1, and Clu in the control were significantly less than the dryness.The data are presented as the mean±SD.aP<0.01, n=10.S100A9: Protein S100-A9; Anxa1: Annexin A1; Clu: Clusterin; IHC:Immunohistochemical.
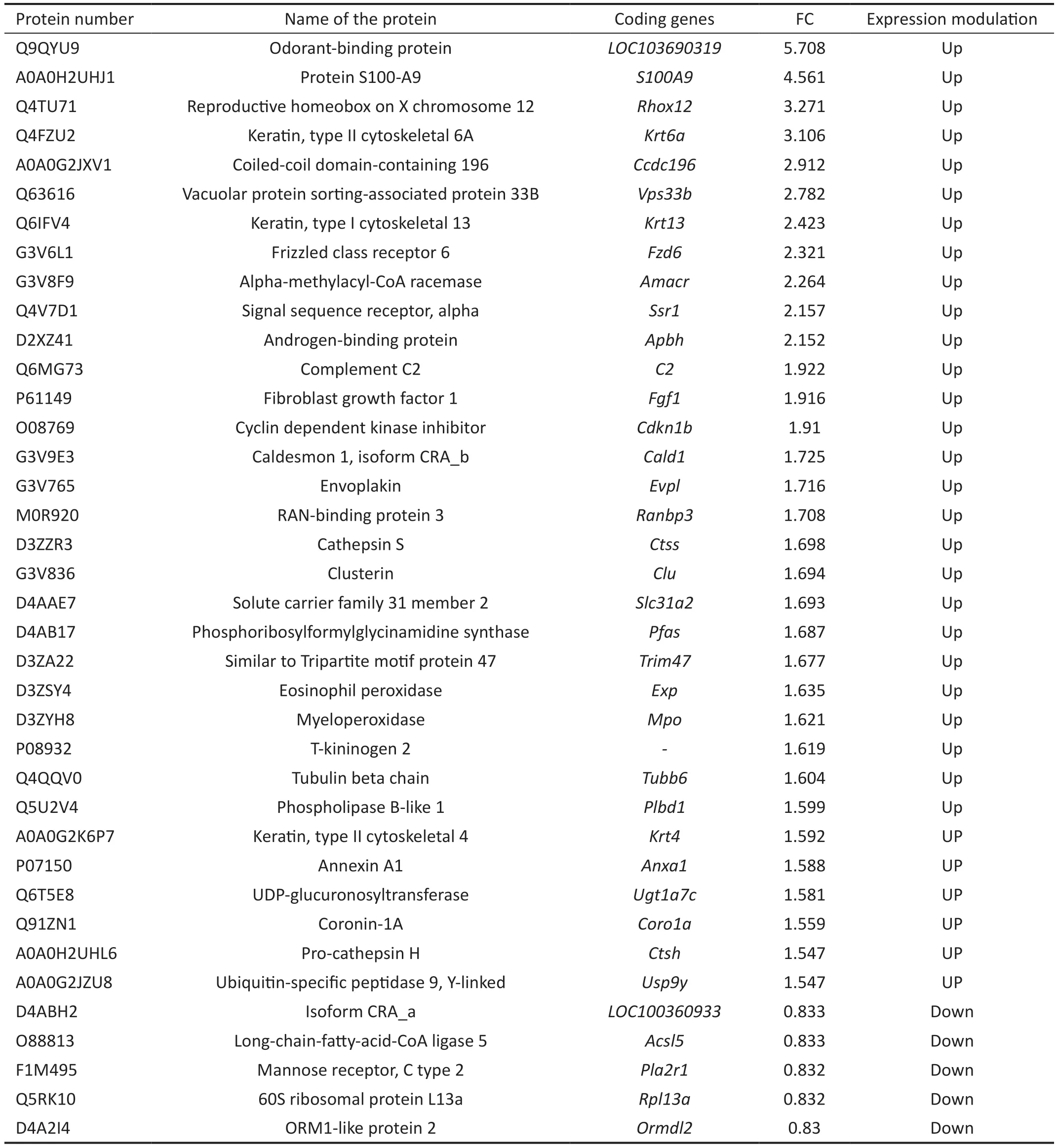
Table 2 Results of differential protein analysis in the lacrimal glands of rats in dryness
Complement and coagulation cascades pathway exerted a vital role in activating and maintaining natural immunity[16].Among them, the complement system, as an inherent component of innate immunity, is the first line of defense for exposing organs, such as eyes, nose, throat,etc.It has been reported that when the complement system was over-activated, it could directly affect the immune function of the eyes, caused local damage such as keratopathy, uveitis,etc[17-18].Clu could participate in the inflammatory reaction process by regulating the complement system.A large number of studies have shown that Clu could be expressed in various organization structures of the eyes, such as cornea, lens, uvea, sclera,etc[19].It was associated with a various of pathophysiological activities in the eye, including lipids transport, maintenance of membrane integrity, inhibition of cells apoptosis, neurodegenerative changes,etc[20].The present study speculated that the dry environment affected the lacrimal gland of rats and led a the abnormal expression of Clu, which might be related to the over-activation of complement and coagulation cascade pathway, and caused the inflammatory reaction and affected the immune function.
In addition, we believed that lysosome pathway was also worthy of attention[21].As known, lysosome is the site of autophagy and endocytosis degradation.Autophagy can clean up the invading pathogens, denatured proteins and damaged organelles in the lysosomal degradation pathway,then maintain cells stability[22].The autophagy could regulate ocular surface inflammation and reduce the damage of lacrimal gland secretion function and corneal epithelium[23].Moreover,cathepsin was the main acid hydrolase in lysosomal, it devotes a crucial effect in the degradation of proteins in lysosomal and is inseparable from autophagy[24].And CTSS is regarded as a ties to regulate the dissolution and remodeling of connective tissue and basement membrane to affect the inflammation and immune response.Up-regulation of CTSS expression will destroy the ocular surface homeostasis, and inhibiting its activity could reduce the inflammatory reaction of lacrimal gland and improve the secretion of tear[25-26].Therefore, it is reasonable to speculate that the activation of lysosome pathway may affect the autophagy level of lacrimal gland tissue, then the related proteins is regulated and it participates in local inflammation and immune response.The pathway and related proteins should be further verified and explored in subsequent studies.
In conclusion, the intervention of dry environment (autumn dryness) caused the tissue morphological changes, local inflammatory infiltration and enhanced the expression of S100A9, Anxa1, and Clu in rats lacrimal gland.It might be related to the low relative humidity of the environment.Such a dry environment affected the micro-environment of the eyes,leading to abnormal pathological links such as inflammation and immune response.Therefore, this study believes that the pathological changes in the fine structure of lacrimal gland in rats and the abnormal expression of S100A9, Anxa1, and Clu proteins are one of the underlying mechanisms of the influence of dry environment on eyes symptoms.It is speculated that long-term exposure to dry environment may lead to xerophthalmia, eye infection, Sjögren’s syndrome,etc.
ACKNOWLEDGEMENTS
Foundations:Supported by Regional Science Foundation Project of the National Natural Science Foundation of China (No.82060827; No.82260891); The Key Discipline of Universities in the “14th Five-Year Plan” Autonomous Region-Traditional Chinese Medicine at Xinjiang Medical University.
Conflicts of Interest: Sun YL,None;Cui AY,None;Wang LX,None;Zhang WW,None;Shi H,None.
 International Journal of Ophthalmology2024年3期
International Journal of Ophthalmology2024年3期
- International Journal of Ophthalmology的其它文章
- Meibomian glands segmentation in infrared images with limited annotation
- Artificial intelligence for the detection of glaucoma with SD-OCT images: a systematic review and Meta-analysis
- Overexpression of TRPV1 activates autophagy in human lens epithelial cells under hyperosmotic stress through Ca2+-dependent AMPK/mTOR pathway
- Semaphorin 7A impairs barrier function in cultured human corneal epithelial cells in a manner dependent on nuclear factor-kappa B
- Novel MIP gene mutation causes autosomal-dominant congenital cataract
- A rare missense PAX6 mutation causes atypical aniridia in a three-generation Chinese family
