RuP Nanoparticles Anchored on N-doped Graphene Aerogels for Hydrazine Oxidation-Boosted Hydrogen Production
Zheng-Min Wang ,Qing-Ling Hong ,Xiao-Hui Wang ,Hao Huang ,Yu Chen ,*,Shu-Ni Li ,*
1 Key Laboratory of Macromolecular Science of Shaanxi Province,School of Chemistry and Chemical Engineering,Shaanxi Normal University,Xi’an 710062,China.
2 School of Materials Science and Engineering,Shaanxi Normal University,Xi’an 710062,China.
3 Department of Microsystems,University of South-Eastern Norway,Borre 3184,Norway.
Abstract: ‘Green hydrogen’ is a promising clean energy carrier for use instead of traditional fuels.For obtaining ‘green hydrogen’,electrochemical water splitting has been receiving considerable attention due to its ecofriendly and low-cost properties.However,the sluggish kinetics of the anodic oxygen evolution reaction (OER) reduces the efficiency of hydrogen production.Accordingly,the hydrazine oxidation reaction (HzOR) with low theoretical potential (-0.33 V vs.RHE) has been proposed as a reasonable alternative for the OER.In this study,graphene aerogel (GA) was utilized as a conductive substrate with a 3D porous framework.RuIII-polyethyleneimine(RuIII-PEI) complexes were adsorbed on the GA surface.Phytic acid (PA)was further adsorbed to form RuIII-PEI-GA-PA hybrids through the hydrogen bond interaction between PA and PEI,which can serve as a precursor to synthesize RuP nanoparticles anchored on Ndoped GA (RuP/N-GA) through the phosphorization reaction.In the pyrolysis process,the ultra-small RuP was formed at the GA surface.Additionally,the decomposition of PEI and PA can introduce abundant N and P heteroatoms into the structure of GA.As a result,RuP/N-GA hybrids achieve efficient HzOR with a low working potential of -54 mV at 10 mA∙cm-2.Moreover,the novel RuP/N-GA hybrids with low Ru loading also exhibit a promising hydrogen evolution reaction(HER) activity with an overpotential of -19.6 mV at 10 mA∙cm-2.Among various RuP/N-GA hybrids,the Tafel plot of HER at RuP/N-GA-900 reveals the smallest value to be 37.03 mV∙dec-1,which affords the fastest HER kinetics.Meanwhile,the result suggests that the HER at RuP/N-GA-900 undergoes a Heyrovsky mechanism similar to that of Pt.The theoretical results revealed that the anchored structure and the presence of N heteroatoms can promote the HzOR on RuP nanoparticles.The free energy of hydrazine molecular adsorption on RuP/N-GA was -0.68 eV,indicating that N-doping in the RuP/N-GA structure can adjust the electronic structure of the Ru active site,which also contributes to the enhanced HzOR activity of the Ru site.Additionally,RuP/N-GA hybrids exhibited excellent cycling and long-term stability for both HER and HzOR,superior to those of commercial Pt/C.Based on the bifunctional activity of RuP/N-GA hybrids,the constructed two-electrode hydrazine split system exhibits an extremely low cell voltage of 41 mV at 10 mA∙cm-2 for the hydrogen production,which achieves the goal of energy-saved hydrogen production at low voltage.The excellent electrocatalytic activity of RuP/N-GA hybrids is attributed to the ultrasmall RuP nanoparticles for abundant Ru active sites.Meanwhile,the synergistic effect between N-doping in GA frameworks with RuP nanoparticles contributes to the activity enhancement of RuP/N-GA hybrids,in which the 3D porous N-GA with few-layer morphology accelerates the electron and mass transfer and the electron interaction between N-GA and RuP nanoparticles promotes the electrocatalytic activity of RuP nanoparticles for both HER and HzOR.This study extends the bifunctional electrocatalyst for the HER and HzOR to achieve energy-saved hydrogen production and sheds new light on the design and synthesis of advanced electrocatalysts via the adsorption-phosphatization method.
Key Words: Ruthenium phosphide; Graphene aerogel; Anchored structure; Hydrazine oxidation reaction;Hydrogen evolution reaction
1 Introduction
Owing to the energy crisis of fossil fuel consumption and environmental protection,the development and utilization of new clean energy sources is extremely urgent1–3.Hydrogen has become the most significant alternative fuel4.Electrochemical water splitting is a more suitable strategy to generate ‘green hydrogen’ without carbon emission and excessive consumption than stream reforming5–8.In overall water splitting,the cathodic hydrogen evolution reaction (HER) is the primary reaction that produces hydrogen whereas the anodic oxygen evolution reaction (OER) works as a counter-reaction for oxygen.Even now,the application of the HER is still limited by the overpotential,and there is an urgent need to exploit new electrocatalysts with higher efficiencies to further decrease the energy consumption for H2production.Because of their high price and low reserves,efficient Pt-based nanomaterials are not suitable for achieving industrialized electrochemical HER.Ru,at a price of 4.2% that of Pt,has received increasing attention as an alternative HER electrocatalyst9–13.Numerous efforts have been made to improve the activity of Ru sites in electrocatalysts for the HER,including engineering the electronic structure and modulating the morphology14–18.Several pioneering studies have reported that introducing highly electronegative elements into Ru-based nanomaterials is an effective strategy to adjust the electronic structure of Ru for the HER14–18.In contrast,highly conductive substrates with large surfaces,such as Ni foam and graphene,favor the electron and mass transfer of Ru nanostructures,resulting in excellent HER activity.Therefore,the synergism of introducing highly electronegative elements and employing a conductive substrate may impart outstanding HER properties to the Ru electrocatalyst,which may even exceed those of the Pt electrocatalyst.
As a counter-reaction to the HER,the anodic OER has four electrons with sluggish kinetics,which leads to a large input cell voltage for the overall water splitting device (up to 2.0 V).Exploiting new electrochemical technologies to replace the OER and saving energy in H2production has received significant attention.Hydrazine oxidation reaction (HzOR; N2H4+ 4HO-→N2(g) + 4H2O + 4e-) with an electrode potential of -0.33 Vvs.a reversible hydrogen electrode (RHE) is a perfect anodic substitutional reaction.Thus,highly efficient H2production can be achieved with a low cell voltage in the overall hydrazine splitting using the HER and HzOR as electrode reactions.In addition,owing to the inert N2being a by-product of the HzOR,the membrane-free electrolyzer can further reduce the cost.Unfortunately,despite significant progress in HER electrocatalysts,exploiting effective catalytic activity for the HzOR has been neglected,not to mention the bifunctional catalysts for overall hydrazine splitting.
Recently,metal phosphides have been confirmed as an effective precursor to realize highly active HzOR electrocatalysts because of the modulatory effect of highly electronegative P on the electronic structure of the active metal sites19,20.For instance,in alkaline media,doping with highly electronegative P in Ni foam resulted in exceptional HER and HzOR activities17.Thus,highly electronegative P can promote the electrocatalysis of the HzOR.Moreover,N heteroatoms generally provide a synergistic effect with metal sites and enhance hydrophilicity,endowing the catalysts with enhanced activity for electrochemical reactions.Interestingly,electrocatalysts with N heteroatoms also exhibit great potential to catalyze the HzOR21,22.Thus,we expect to explore a novel bifunctional electrocatalyst with N-doping to boost the development of hydrazine splitting,which has rarely been achieved.In addition,a synergetic mechanism of N heteroatoms and metal sites for HzOR catalysis has yet to be developed,which is of great significance for designing novel electrocatalysts for H2productionviahydrazine splitting.
Herein,we integrate the design concepts of both the HER and HzOR and propose a new bifunctional electrocatalyst strategy for efficient overall hydrazine splitting using ultrasmall RuP nanoparticles anchored on N-doped graphene aerogel (RuP/NGA).First,highly electronegative P was incorporated into the Ru nanostructure to modulate the electron structure and increase its activity for both the HER and HzOR.Second,the 3D porous structure of the N-doped graphene aerogel (GA) adjusts the electronic structure of the Ru sites and provides effective electron and mass transfer ability for electrocatalytic reactions.Finally,the synergistic effect between N doping and Ru sites contributes to an improvement in the electroactivity of the HzOR.As the result,RuP/N-GA exhibits excellent electrocatalytic activities with a low working potential of -19.6 mV for HER (at 10 mA∙cm-2) and 27 mV for HzOR (at 30 mA∙cm-2).Density functional theory (DFT) calculations confirmed that N doping in GA plays a significant role in the synergistic effect of the Ru sites on HzOR.Importantly,at 10 mA∙cm-2,the overall hydrazine-splitting electrolyzer utilizing the RuP/N-GA as a bifunctional electrocatalyst achieves a significantly low voltage (41 mV).Our work demonstrates that Ru can be used as an alternative to both HER and HzOR electrocatalysts.In addition,the highly electronegative P and Ndoping effect strengthen the bifunctional catalytic properties of hydrazine splitting to stimulate efficient H2production with low energy consumption.
2 Experimental section
2.1 Reagents and chemicals
Ruthenium trichloride hydrate (AR,99.9%,RuCl3),hydrazine hydrate (AR,80% in water,N2H4∙H2O) and ascorbic acid (AR,99.7%,AA) were obtained from Aladdin Industrial Co.,Ltd.Polyethyleneimine (AR,99%,PEI,Mw= 10000) was purchased from Nitto Boseki Co.,Ltd (Japan).Phytic acid (AR,70% in water,PA) was acquired from Sinopharm Chemical Reagent Co.,Ltd.Graphene oxide (AR,99.8%,GO) was purchased from JCNANO Technology Co.,Ltd.(Nanjing,China).The commercial 20% (mass fraction) Pt/C electrocatalyst was provided by Johnson Matthey Corporation.
2.2 Synthesis of RuP nanoparticles anchored on N-doped graphene aerogels (RuP/N-GA)
Typically,GA is synthesized according to a previously reported method using graphene oxide (GO) as the reaction precursor22.After adding 10 mg GA into the 6 mL mixture of 0.01 mol∙L-1RuCl3and 0.3 mol∙L-1PEI for 2 h,RuIII-PEI-GA hybrids (RuIII-PEI-GA) were obtained through centrifugation.Then,RuIII-PEI-PA-GA was obtained by mixing the RuIII-PEIGA in ethanol solution of PA for 2 h at 95 °C.After sufficient freeze-drying for 24 h,RuIII-PEI-GA-PA precursors were transferred as RuP/N-GAviapyrolysis at various temperatures for 2 h with a heating rate of 10 °C∙min-1under a N2atmosphere.The prepared RuP/N-GA samples were named RuP/N-GA-700,RuP/N-GA-800,RuP/N-GA-900,RuP/N-GA-1000,and RuP/NGA-1100 depending on the calcination temperature.
Under the same synthetic conditions,a N-doped GA (N-GA)sample was synthesized without RuCl3or PA,a RuP/GA sample was synthesized without PEI,and a Ru/N-GA sample was synthesized without PA to compare the active sites of the catalytic materials.
2.3 Calculation and characterization
Details of the DFT calculations and physical and electrochemical characterizations are provided in the supporting information.
3 Results and discussion
3.1 Formation and characterization of RuP/N-GA
In this study,GA was utilized as a conductive substrate with a 3D porous framework formed through the reduction of GO by ascorbic acid24.The RuP/N-GA precursor was preparedviaa simple coordination-absorption method.Briefly,the RuIIIpolyethyleneimine (RuIII-PEI) complex is coordinated by the N sites of PEI,which are then adsorbed on the surface of GA.Ultraviolet and visible (UV-Vis) spectroscopy is carried out to verify the formation of the RuIII-PEI complex (Fig.S1,Supporting Information).Compared with RuCl3,there is an obvious broad peak at approximately 387 nm,indicating the presence of the RuIII-PEI complex.After adsorbing phytic acid(PA) in ethanolviathe hydrogen bond between PA and PEI,the RuIII-PEI-PA-GA hybrids (RuIII-PEI-GA-PA) work as a precursor to synthesize RuP nanoparticles anchored on N-doped GA (RuP/N-GA) through the phosphorization reaction.In this process,an ultrasmall RuP nanostructure is formed by the steric hindrance of the GA.The decomposition of PEI introduces abundant nitrogen heteroatoms into the GA structure (Fig.1a).

Fig.1 (a) Overall synthetic route of RuP/N-GA-900 hybrids.(b) XRD patterns of GO and RuP/N-GA.(c) SEM image of RuP/N-GA-900.
To clarify the elemental composition,energy dispersive X-ray(EDX) spectroscopy was performed for the RuP/N-GA sample.The spectrum shows that RuP/N-GA is composed of Ru,P,C,and N,and the atomic ratio of Ru : P is extremely close to 1 : 1(Fig.S2).Both X-ray powder diffractometer (XRD) patterns of RuP/N-GA show a wide diffraction peak at 2θ= 26.8° (Fig.1b)attributed to the graphitic structure.Obviously,GO precursor is successfully reduced by ascorbic acid (AA).Additionally,there is no characteristic peak of the RuP crystal in the XRD pattern of RuP/N-GA,which suggests that RuP nanoparticles occur in the form of ultrasmall or amorphous structures.The morphology of RuP/N-GA was investigated using scanning electron microscopy (SEM) and transmission electron microscopy(TEM).The SEM image shows the 3D porous framework structure of RuP/N-GA.RuP nanoparticles are not observed in the SEM images (Fig.1c).This phenomenon implies that RuP exists as ultrasmall nanoparticles,as indicated by the XRD pattern.The TEM images reveal the ultrathin layered structure of GA (Fig.2a) and ultrasmall RuP nanoparticles in the RuP/NGA sample (Fig.2b,c).The corresponding size statistics for RuP show that the size of most nanoparticles is < 5 nm.Such ultrasmall RuP nanoparticles can be one of the factors of the high electrochemical activity for bifunctional electrocatalysis (Fig.S3).The corresponding high-resolution transmission electron microscopy (HRTEM) image shows that the crystal spacings of the nanoparticles are approximately 0.251,0.267,and 0.276 nm(Fig.2d),corresponding to the (111),(102),and (200) planes of RuP (JCPDS No.64-8015),respectively.The selected-area electron diffraction (SAED) images show extremely weak diffraction rings of the RuP nanoparticles,which also illustrate the low crystallinity of the nanoparticles (Fig.2e).In addition,the mapping measurements on TEM are conducted to analyze the elemental distribution of RuP/N-GA.The uniform distribution of Ru,P,C,and N in RuP/N-GA indicated the presence of RuP nanoparticles and N-doped GA (Fig.2f).The thermogravimetric curve shows that the sample loses up to 89.8% of its weight,indicating that only trace Ru is present in the RuP/N-GA sample (Fig.S4).Raman spectra were recorded to investigate the graphitization of GA and RuP/N-GA.The Raman spectra of both RuP/N-GA and GA samples show two peaks (at approximately 1350 and 1580 cm-1),corresponding to the G and D peaks of the graphitic structure (Fig.S5).TheID/IGratio of RuP/N-GA was greater than that of GA,indicating the presence of more carbon defects in RuP/N-GA.

Fig.2 (a,b,c) TEM images,(d) HRTEM image,(e) SAED image,and (f) elemental mapping image with C,N,P,Ru of RuP/N-GA.
X-ray photoemission spectroscopy (XPS) technology is typically used to study the elemental composition and oxidation states on the surfaces of electrocatalysts.The overall XPS spectrum shows that Ru,P,C,and N are present in the structure of the RuP/N-GA sample (Fig.3a),which is consistent with the EDX results.For the C 1sorbital,the high-resolution spectrum exhibits a peak at approximately 284.5 eV (Fig.3b).Further analysis of the C 1sspectrum reveals peaks at 284.5,285.8,and 285.2 eV,which are assigned to the binding energy of C=C25,C―C26,and C―N25,respectively.The formation of C―N bonds revealed that N heteroatoms were successfully doped into the GA structure.The peak at approximately 280.4 eV is characteristic of Ru 3d5/225.Because of the very close peak positions of Ru 3d5/2and C 1s,it was difficult to investigate the oxidation state of Ru based on the Ru 3dorbital.Therefore,Ru 3pXPS is useful for studying the chemical states of carbonsupported Ru-based nanomaterials.The peaks at 461.6 and 483.9 eV belong to the binding energies of Ru 3p3/2and Ru 3p1/2,respectively.The sub-peaks at 464.1 and 486.8 eV indicate the existence of Ru―O on the surface of the sample14(Fig.3c).The P 2pXPS spectrum exhibits several characteristic peaks at 130.0,132.6,and 133.6 eV15(Fig.3d),which were identified as the metal phosphide (RuP),P―C,and P―O bonds,respectively.In particular,the presence of the P―C bond implies that some P heteroatoms were doped into the structure of GA.
Furthermore,the N2adsorption-desorption isotherm was used to analyze the specific surface area of the porous nanostructure.The Brunauer-Emmett-Teller (BET) surface area of RuP/N-GA is up to 200.4 m2∙g-1(Fig.S6a),which is mainly attributed to the 3D structure of N-GA.The pore volume of the sample is measured at 611.9 cm3∙g-1at a partial pressure (p/p0) of 1,reflected from the pore-size distribution diagram (Fig.S6b).In addition,no obvious adsorption isotherm plateau was observed under saturated vapor pressure,indicating that the pore-like structure exhibited by the GA mainly consists of narrow wedgeshaped pores.The adsorption-desorption curve indicated that slit pores were formed by the accumulation of layered materials,which was also confirmed by the TEM image (Fig.S7).The large surface area and pore volume of the RuP/N-GA sample were attributed to the ultrasmall nanoparticles,abundant nanoholes,and 3D structure of N-GA.These structural features are beneficial for mass and charge transfer in electrocatalytic reactions.
3.2 HER activity of RuP/N-GA
Linear sweep voltammetry (LSV) was performed to investigate the HER activity of the as-synthesized electrocatalysts.The LSV curve on RuP/N-GA is measured in the N2-saturated 1 mol∙L-1KOH electrolyte,which displays a better HER activity than the commercial Pt/C (Fig.4a).The overpotential is 19.6 and 26.6 mV at 10 mA∙cm-2for RuP/NGA-900 and commercial Pt/C,respectively.The enhanced HER activity of RuP/N-GA could be attributed to three factors: 1) the high-activity ultrasmall RuP nanoparticles,2) the excellent electrical conductivity of N-GA for high charge-transfer efficiency,and 3) the 3D porous structure of N-GA with abundant nanoholes and large specific area for effective mass transfer.
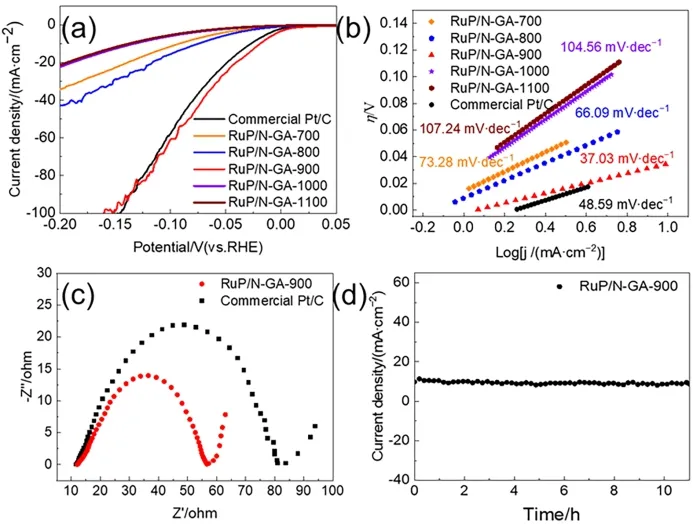
Fig.4 (a) LSV curves of RuP/N-GA and commercial Pt/C in 1 mol∙L-1 KOH solution at 5 mV s-1.(b) Tafel plots of RuP/N-GA and commercial Pt/C.(c) EIS of RuP/N-GA-900 and commercial Pt/C,at an overpotential of 20 mV.(d) Chronoamperometry curve of RuP/N-GA-900 at a 20 mV potential.
To further investigate the HER activity of RuP/N-GA,the LSV curves of GA,N-GA,RuP-GA,and Ru/N-GA were obtained under identical conditions (Fig.S8).Hardly any HER performance was observed for the GA and N-GA.Instead,the Ru/N-GA and RuP-GA samples have obvious electrocatalytic activities with overpotentials at 50.6 and 47.6 mV for HER,respectively.Compared to the GA structure,Ru is a more effective active species for HER electrocatalysis.The phosphatization of Ru can improve the HER activity,and the highly electronegative P can promote the further accumulation of electrons at the exposed Ru sites.Thus,RuP exhibits outstanding electrocatalytic activity for the HER than Ru14,27.The atomic radii of the N atoms were similar to those of the C atoms.The introduction of N can reduce damage to the GA structure.This can optimize the charge and spin densities of the adjacent C atoms,thereby optimizing the H2adsorption energy of the catalyst.In contrast,N doping and P incorporation in the RuP/N-GA structure can also synergize with Ru phosphide to enhance the electrical conductivity and improve the charge transfer efficiency between the electrocatalyst and electrolyte17.
To investigate the effect of the pyrolysis temperature on the bifunctional electroactivity,various RuP/N-GA samples were prepared,denoted as RuP/N-GA-X(whereXis the pyrolysis temperature).The LSV curves show that the RuP/N-GA-900 sample exhibits the best electrocatalytic behavior for the HER(Fig.4a and S9).With an increased calcination temperature,GA can obtain a better graphitization degree and more defects.Therefore,the enriched defects of RuP/N-GA-900,rather than those of RuP/N-GA-700 and RuP/N-GA-800,can be confirmed by the Raman spectra (Fig.S10).On the other hand,increasing the temperature to 1000 or 1100 °C can lead to severe agglomeration of RuP (Fig.S11).As a result,RuP/N-GA-1000 and RuP/N-GA-1100 showed lower HER activities.The Tafel slope (Fig.4b) was used to assess the HER kinetics of the RuP/N-GA-Xcatalysts.The Tafel plot of RuP/N-GA-900 reveals the smallest value of 37.03 mV∙dec-1,which is considerably lower than that of RuP/N-GA-700 (73.28 mV∙dec-1),RuP/N-GA-800 (66.09 mV∙dec-1),RuP/N-GA-1000(104.56 mV∙dec-1),RuP/N-GA-1100 (107.24 mV∙dec-1),and even commercial Pt/C (48.59 mV∙dec-1).The lowest Tafel slope indicates the fastest HER kinetics on the surface of RuP/N-GA-900,and the HER route of RuP/N-GA-900 follows a Heyrovsky mechanism similar to that of Pt.Generally,the HER rate is determined by the recombination rate of H* on the surface of RuP/N-GA-90028.Because of the low Ru loading,the RuP/NGA-900 sample has a greater advantage in HER electrocatalysis than commercial Pt/C and other Ru phosphide nanocatalysts(Table 1).
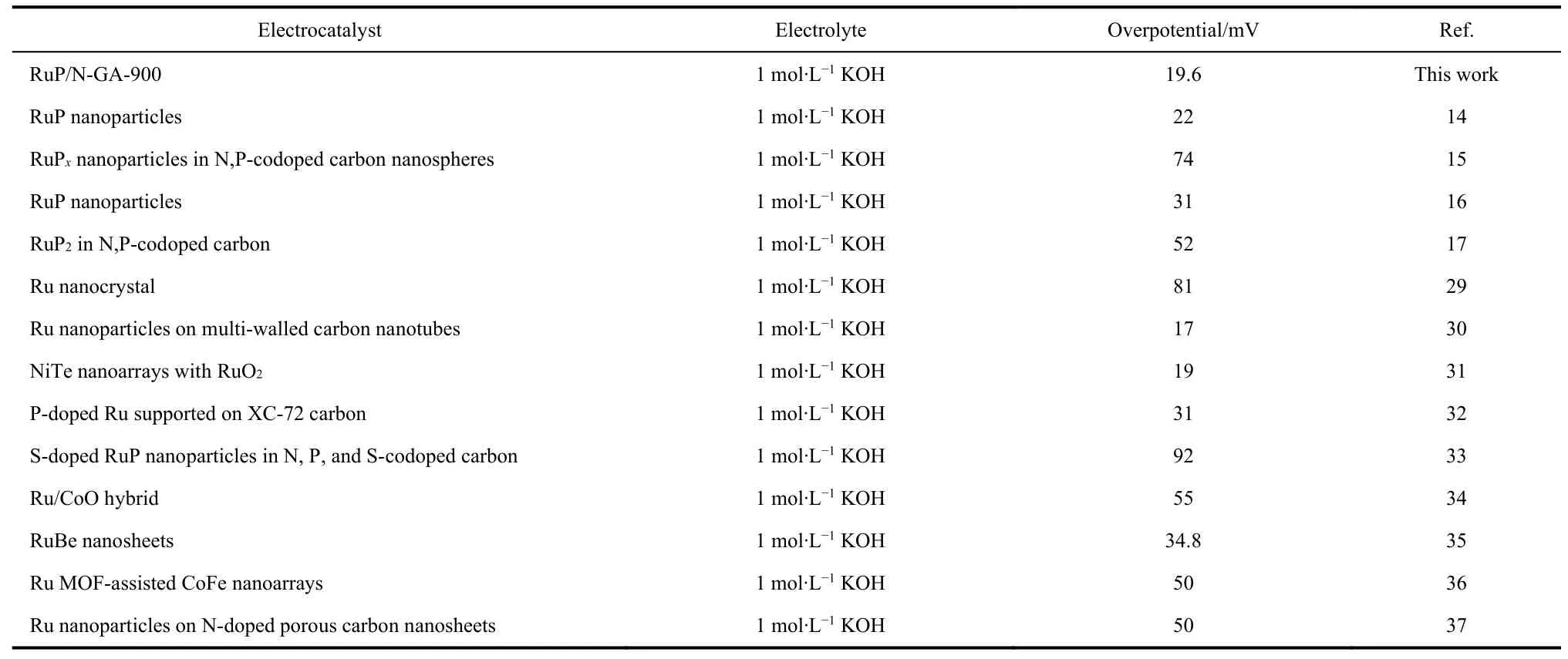
Table 1 Summary of HER activities of Ru-based electrocatalysts at 10 mA∙cm-2.
To investigate the charge-transfer properties,electrochemical impedance spectroscopy (EIS) tests were performed on RuP/NGA-900 and commercial Pt/C (Fig.4c).In 1 mol∙L-1KOH,theRctof RuP/N-GA-900 is 47.3 Ω,smaller than that of commercial Pt/C (67.7 Ω).A smallerRctindicates faster charge transfer at the electrode surface of RuP/N-GA-900.The long-term stability of electrocatalysts is another significant factor affecting their successful application.The LSV curve after 5000 cycles displays a 10.5 mV increase shift to the first LSV curve on RuP/N-GA-900 at 10 mA∙cm-2(Fig.S12).In contrast,the commercial Pt/C catalyst shows an approximate 16.5 mV shift between the first and 5000thcycle,revealing relatively better stability on RuP/NGA-900 for HER.After 5000 CV cycles,RuP does not significantly aggregate (Fig.S13).Similarly,the chronoamperometry curve only displays a slightly decreased current after 10 h of electrocatalysis (Fig.4d).Anchoring on N-GA effectively prevented the agglomeration of the ultrasmall RuP structure during the electrochemical process.The TEM image of RuP/N-GA-900 after a 10-h chronoamperometry test confirms the uniform dispersion of the RuP nanoparticles (Fig.S14).Therefore,RuP/N-GA-900 showed high stability in the long-term electrocatalytic process,suggesting its promising application for H2production under alkaline conditions.
3.3 HzOR performance of RuP/N-GA-900
The HzOR behaviors of RuP/N-GA-Xwere investigated in 1 mol∙L-1KOH with 0.4 mol∙L-1N2H4(Fig.5a).The LSV curves show that all the RuP/N-GA-Xcatalysts have obvious electrochemical activities toward the HzOR.In particular,the RuP/N-GA-900 has the best HzOR performance with a potential of -54 and 24 mV at 10 and 50 mA∙cm-2,respectively.Compared with current HzOR electrocatalysts,RuP/N-GA-900 is still a very competitive catalyst in alkaline electrolytes (Table 2).Such high activity originates from the structural advantages of RuP/N-GA-900,as demonstrated by HER electrocatalysis: 1)ultrasmall RuP nanoparticles,2) N-doping in GA frameworks,and 3) few-layer N-GA morphology.Moreover,LSV measurement with different hydrazine concentrations in 1 mol∙L-1KOH was conducted.A larger concentration of N2H4(0.1–0.4 mol∙L-1) can lead to an increased current density of hydrazine oxidation (Fig.5b).After the hydrazine concentration reaches 0.5 mol∙L-1,the LSV curve keeps a similar catalytic current as that of 0.4 mol∙L-1N2H4.This phenomenon indicates that higher hydrazine concentration can not improve the efficiency of HzOR.The electrocatalytic activity of RuP/N-GA-900 is already saturated when hydrazine concentration reaches 0.4 mol∙L-1.Chronoamperometry was used to investigate the stability of RuP/N-GA-900 in the HzOR.In the chronoamperometry curves,RuP/N-GA-900 maintains a stable current density for 5 h (Fig.5c),indicating that RuP/N-GA-900 is more stable than commercial Pt/C for HzOR.
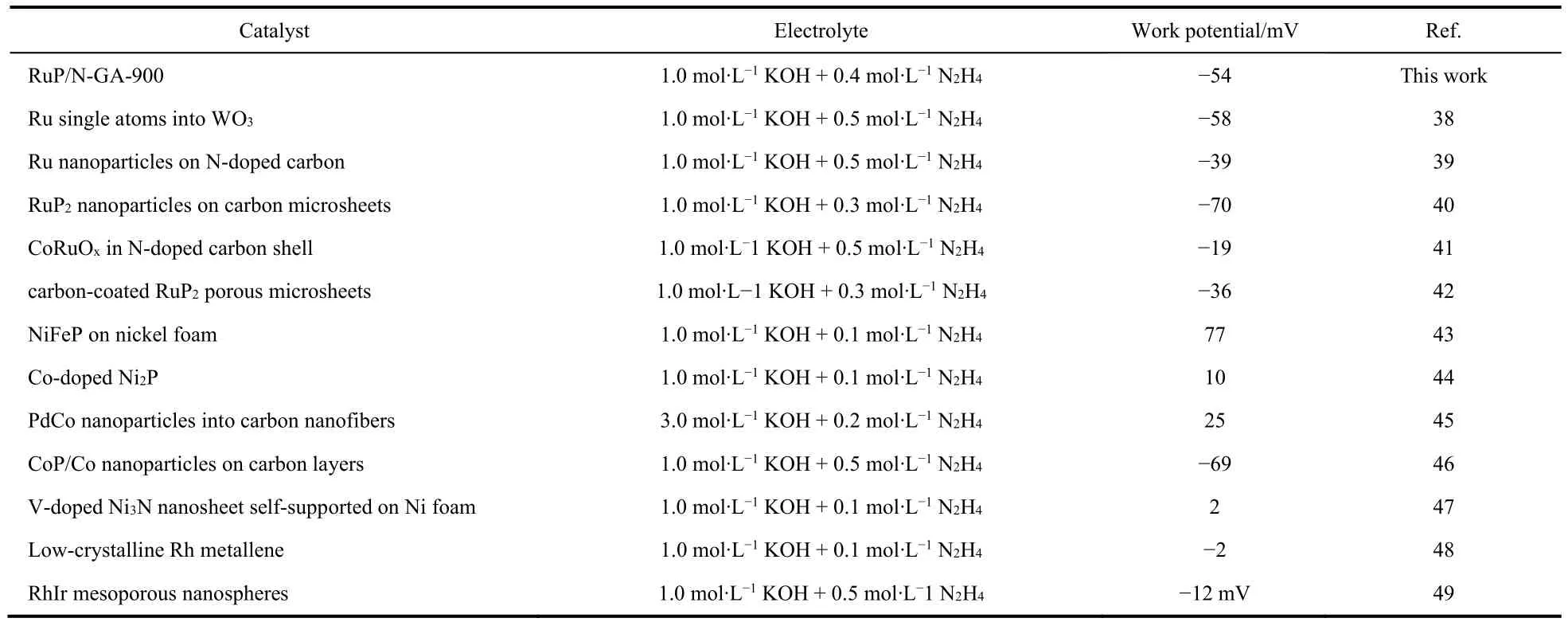
Table 2 Summary of HzOR performance of Ru-based and other electrocatalysts at 10 mA∙cm-2.
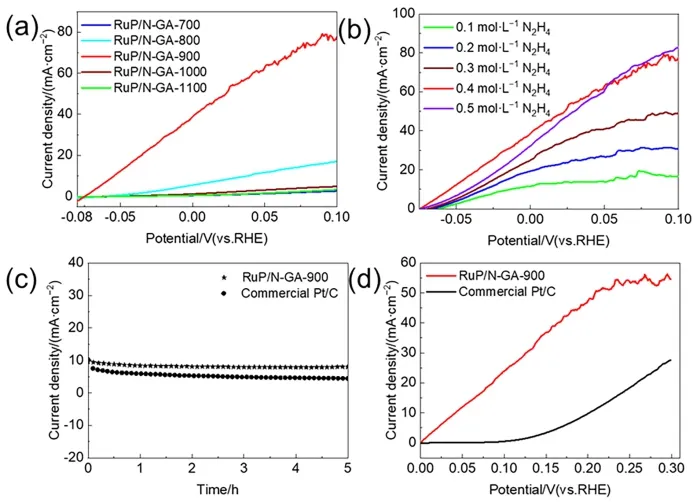
Fig.5 (a) LSV curves of RuP/N-GA-700,RuP/N-GA-800,RuP/N-GA-900,RuP/N-GA-1000 and RuP/N-GA-1100 in 1 mol∙L-1 KOH + 0.4 mol∙L-1 N2H4 electrolyte.(b) LSV curves of RuP/N-GA-900 in 1 mol∙L-1 KOH electrolyte with N2H4 concentrations from 0.1 to 0.5 mol∙L-1.(c) Chronoamperometry curves of RuP/N-GA-900 and commercial Pt/C in 1 mol∙L-1 KOH + 0.4 mol∙L-1 N2H4 electrolyte.(d) Comparison of LSV curves for OHzS in 1.0 mol∙L-1 KOH +0.4 mol∙L-1 N2H4 electrolyte and OWS in 1 mol∙L-1 KOH between RuP/N-GA-900 and commercial Pt/C.
RuP/N-GA-900 is a promising bifunctional electrocatalyst with excellent hydrazine oxidation-assisted H2production.Considering the large voltage required in the overall water splitting (OWS) system,a bifunctional two-electrode overall hydrazine splitting (OHzS) system was designed using RuP/NGA-900 as an electrocatalyst for efficient HzOR and HER.Fortunately,the OHzS system with RuP/N-GA-900 can achieve a very low voltage of 41 mV at 10 mA∙cm-2.In contrast,the OHzS system based on the commercial Pt/C electrocatalyst requires 202.0 mV to drive H2production (Fig.5d).This result indicates that RuP/N-GA-900 is a better alternative to the commercial Pt/C electrocatalyst for hydrazine splitting.
3.4 Electrocatalytic mechanism for HzOR
DFT calculations were performed to investigate the possible electrocatalytic mechanism of HzOR.RuP/N-GA and RuP on GA (RuP/GA) slabs were prepared to elucidate the synergistic effect between N doping and Ru sites.The electronic propertieswere clarified from the total and partial density of states (PDOS)results (Fig.6a,b).The electron distribution of both slabs was continuous near the Fermi level,indicating that the slabs had metallicity and excellent conductivity for electrocatalysis.Meantime,for the Ru sites,thed-band center of RuP/N-GA is-1.62 eV,more positive than that of RuP/GA (-1.76 eV).This indicates that N doping can effectively adjust the electronic structure of the Ru active sites,and the moderate value of the dband center could achieve a balance between adsorption and desorption50.Thus,the N heteroatoms in the RuP/N-GA slab can modulate the electronic structure of the Ru sites,which could be advantageous for bifunctional electrocatalytic activity.However,the study of HzOR is in its fledgling stages,thus,DFT calculations on the electrochemical reaction mechanisms are meaningful for further studies.According to the limited previous work,the HzOR process is composed of the adsorption and dehydrogenation steps (*N2H4→ *N2H3→ *N2H2→ *N2H →*N2)51.The free energies and number of hydrazine molecules adsorbed on the RuP/N-GA and RuP/GA slabs (Fig.7) were calculated (Fig.6c).The free energy on RuP/N-GA is -0.68 eV,much lower than that of RuP/GA (-0.59 eV),suggesting that Ndoping makes it easier for RuP/N-GA to adsorb hydrazine molecule on Ru sites.In the dehydrogenation steps,for both slabs,the dehydrogenation of *N2H2to *N2H is the ratedetermining step (RDS).On RuP/N-GA,the free energy difference value is 0.79 eV,which is smaller than the dehydrogenation energy on RuP/GA.This result shows the Ndoping in the structure of RuP/N-Ga can modulate the electronic structure of Ru active sites,which also contributes to HzOR activity enhancement of Ru sites.
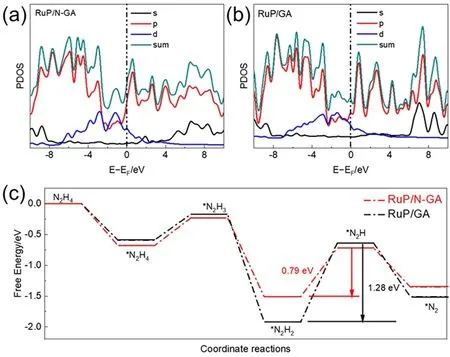
Fig.6 PDOS of (a) RuP/N-GA and (b) RuP/GA slabs.(c) Free-energy profiles for HzOR on RuP/N-GA and RuP/GA slabs.
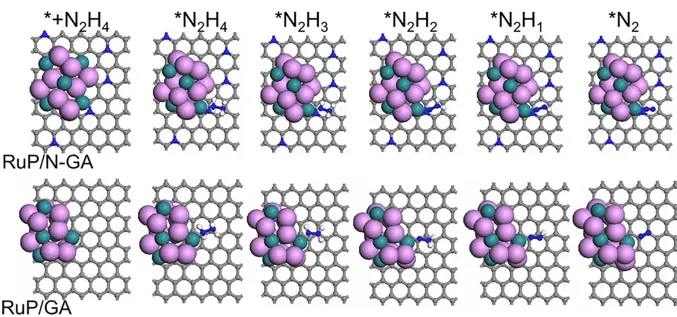
Fig.7 Adsorption slabs of HzOR on RuP/N-GA and RuP/GA slabs.(grey atoms: carbon,blue atoms: nitrogen,purple atoms: ruthenium).
4 Conclusions
To achieve an effective bifunctional electrocatalyst for overall hydrazine splitting,ultrasmall RuP nanoparticles anchored on Ndoped graphene aerogels were preparedviaa facile adsorptionphosphatization method.During pyrolysis,PEI can release N active atoms as heteroatoms for doping into the 3D structure of the GA framework,which simultaneously introduces abundant defects in RuP/N-GA to increase both the HER and HzOR activities.As a result,RuP/N-GA exhibits better performance with a low overpotential (19.6 mV) for the HER than a commercial Pt/C catalyst.Additionally,RuP/N-GA exhibits an excellent HzOR electrocatalytic activity,which only requires a very low potential (-54 mV) for 10 mA∙cm-2.Such high activity can be attributed to the structural advantages of RuP/N-GA-900:1) ultrasmall RuP nanoparticles for abundant Ru active sites,2)the synergistic effect of N-doping in GA frameworks with Ru phosphide for better electrocatalytic activity,and 3) 3D porous N-GA with a few-layer morphology for superior electron and mass transfer.The DFT calculation results confirm that N doping can effectively enhance the PDS activity of the HzOR.Impressively,the OHzS system utilizing the RuP/N-GA-900 as a bifunctional electrocatalyst achieves a very low voltage of 41 mV at 10 mA∙cm-2.Thus,the HzOR-assisted H2production was successfully achieved at low voltages.This work extends the bifunctional electrocatalyst for the HER and HzOR and sheds new light on the design and synthesis of advanced electrocatalystsviathe adsorption-phosphatization method.
Supporting Information: available free of chargeviathe internet at http://www.whxb.pku.edu.cn.
- 物理化学学报的其它文章
- Recent Advances in Self-Supported Transition-Metal-Based Electrocatalysts for Seawater Oxidation
- 用于高灵敏快速核酸检测的荧光碳点
- Introducing Novel,Multiple Cd Coordination Modes into Gold Nanoclusters by Combined Doping for Enhancing Electrocatalytic Performance
- 电催化二氧化碳还原催化剂、电解液、反应器和隔膜研究进展
- 利用多氟丙烯酸酯添加剂提升准二维钙钛矿发光二极管性能
- 电子自旋效应在电催化剂中的作用