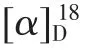割舌树中的两个新四降三萜walsurobustones F 和G
侯 丽,曹明明,赵 庆,何红平*,郝小江*
(1. 中国科学院昆明植物研究所,植物化学与西部植物资源持续利用国家重点实验室,云南 昆明 650201;2. 云南中医药大学,云南 昆明 650500;3. 湖南湘江新区管委会商务和市场监管局,湖南 长沙 410205)
1 Introduction
Limonoids are a famous family of plant secondary metabolites that are characterized by complex chemical structures and obvious bioactivities. They are usually isolated from the families of Meliaceae and Rutaceae[1-2]. The genus Walsura belongs to the family Meliaceae and is distributed in subtropical regions in Asia[3].It is commonly associated with producing limonoid chemical structures[4-11]. The structural diversity of limonoids is mainly due to their carbon skeleton and oxidations, or to the oxidation, degradation, and stereochemistry on the furan ring moiety[12-16]. Previously,walsurobustones A-E were isolated from Walsura robusta[17]. Herein, we report the isolation of walsurobustones F and G, with a focus on fixing their stereochemistry on the furan ring. Both walsurobustones F and G showed better antitumor activities than positive control cisplatin.
2 Results and discussion
Walsurobustone F (1), a white amorphous powder,showed the molecular formula of C27H34O8as established by the HRESIMS at m/z 509.2158 [M+Na]+(calcd. 509.2151). The 1D and 2D NMR spectra analysis of 1 revealed that 1 shared the same A, B, C and D rings in the limonoid core with walsurobustones (Table 1), while the different substitution on the furan ring was observed. Compared to walsurobustone C[17], the conjugated △20(22)double bond was substitute with the hydroxyl group at C-22 on structure of 1, which were supported by the HMBC correlation from H-20 (δH2.64) to C-22 (δC73.6). H-15 (δH3.74, s) and H-16β(δH2.85, dd, 14.0 and 6.4 Hz) (Figure 1) indicates JH16-17= 6.4 Hz which requires the cis relation with H-17, accordingly δH1.77 was assigned to H-16α. The ROESY correlations of H-20/H-16α, H-20/H3-18,H-22/H3-18 revealed that H-20 and H-22 were αoriented. No ROESY cross-peaks can be observed between H-20 and H-23 which infered H-23 was βoriented (Figure 2). According to the above spectral data, the planar structure of 1 was established as hydrate products of walsurobustone C.
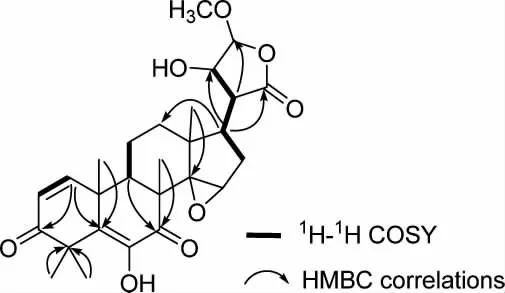
Figure 1. Key 1H-1H COSY, HMBC correlations of 1 and 2
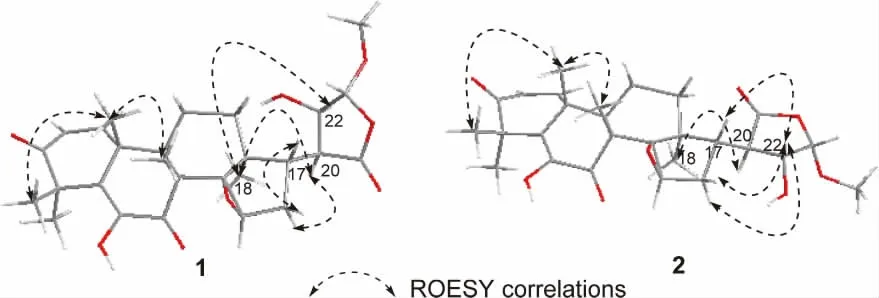
Figure 2. Key ROESY correlations of 1 and 2
Walsurobustone G (2) was assigned the molecular formula of C27H34O8by the HRESIMS at m/z 509.2137 [M + Na]+(calcd. 509.2151). By comparison of the1H and13C NMR data of 2 and 1 (Table 1), they showed high similarity. The 2D NMR spectra analysis of 2 revealed that 2 differed from 1 on the stereochemistry of furan ring. Regarding to the relative configuration of chiral centers of compound 2,H -22 was assigned as β -oriented based on the ROESY correlation of H-22/H-17 (Figure 2). The configuration of H-20 was assigned as α-oriented due to the significant NOE cross-peaks between H-20 and H3-18. H-23 was assigned as β-oriented since the absence of ROESY correlation between H-20 and H-23. Thus the planar structure of compound 2 was determined as shown.
The cytotoxicity of 1 and 2 against the cancer cell lines (HL-60, SMMC-7721, A-549, MCF-7, and SW480) were evaluated by using the MTT method with taxol and cisplatin as positive controls. Both 1 and 2 showed obvious cytotoxicity, especially against SMMC-7721, A-549 and MCF-7 (Table 2).
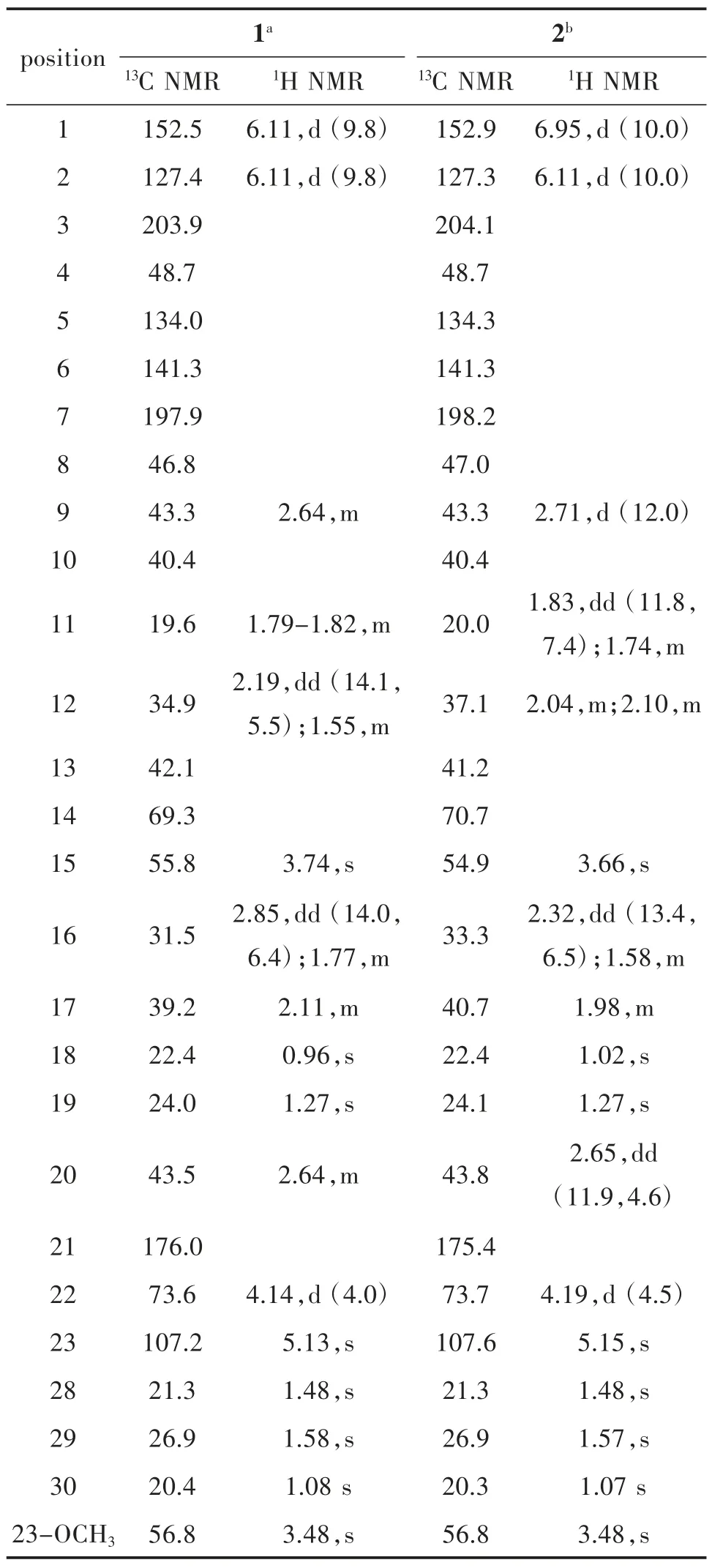
Tab.1 NMR spectroscopic data of 1 and 2 recorded in CDCl3
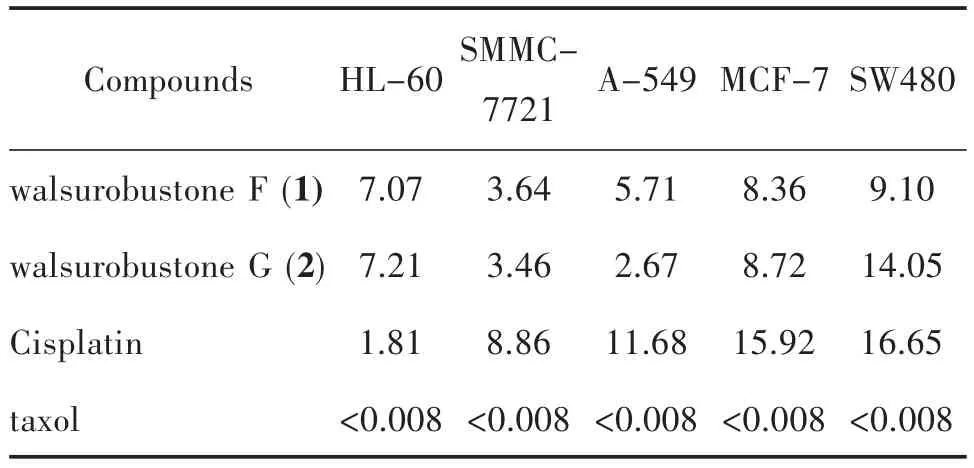
Tab.2 Cytotoxicity data for compounds 1 and 2 against the cancer cell lines
3 Experimental section
3.1 General experimental procedure Optical rotations were measured with a Horiba SEPA-300 polarimeter. UV spectra were detected on a ShimadzuUV-2401 spectrophotometer. IR spectra were determined on a Tenor 27 spectrophotometer with KBr pellets, whereas CD spectra were recorded with an Applied Photophysics Chirascan spectrometer. ESIMS and HRESIMS were measured on a Finnigan MAT 90 instrument and VG Auto Spec-3000 spectrometer, respectively. 1D and 2D NMR spectra were recorded on Bruker AM-400 and Bruker DRX-500 with TMS as internal standard. Semipreparative HPLC was performed on an Agilent 1100 liquid chromatographed with a Zorbax SB-C18 (9.4 mm 25 cm) column. Column chromatography was performed with silica gel(300 -400 mesh, Qingdao Marine Chemical, Inc.,Qingdao, People's Republic of China), and MCI gel(75-150 μM, Mitsubishi Chemical Corporation, Tokyo,Japan). Fractions were monitored by TLC, and spots were visualized by heating silica gel plates sprayed with 10% H2SO4in EtOH.
3.2 Plant material The leaves of W. robusta collected in Hainan Province, People's Republic of China in December 2010. The plant was authenticated by Dr.Guangwan Hu, Kunming Institute of Botany, Chinese Academy of Sciences. A voucher specimen (No.H20101202) was deposited in the State Key Laboratory of Photochemistry and Plant Resources in West China, Kunming Institute of Botany, Chinese Academy of Sciences.
3.3 Extraction and isolation The dried and powdered leaves (12 kg) of W. robusta were extracted with MeOH three times under reflux, and the solvent was evaporated in vacuo. The residue was partitioned in H2O and extracted successively with petroleum ether and EtOAc. The EtOAc fraction (200 g) was separated by silica gel column chromatography (CC) eluted with a gradient of petroleum ether/Me2CO (50 ∶1 to 1 ∶1) and CHCl3/MeOH in a gradient (15 ∶1 to 3 ∶1). Eight fractions (Fr. A-H) were obtained according to TLC monitor. Fr. C (17 g) was subjected to MCI-gel column(MeOH/H2O, 6 ∶4 to 9 ∶1) to give sixteen sub-fractions(C1-C16). C7 (4.1 g) was then chromatographed on a column of reversed-phase C18 silica gel eluted with MeOH/H2O (5 ∶5 to 9 ∶1) to get eight parts (C7a-C7h).Fr. C7h was purified by CC over silica gel and then applied to a Sephadex LH-20 column using solvent system acetone to provide 1 (11 mg) and 2 (15 mg).