Beneficial effects of protocatechuic acid on diabetic retinopathy in streptozocin-induced diabetic rats
Ling Wan, Jun Li, Wei-Bai Chen, Guo-Qiang Wu
1Department of Ophthalmology, Sichuan Provincial People’s Hospital, University of Electronic Science and Technology of China, Chengdu 610072, Sichuan Province, China
2Department of Ophthalmology, Chinese Academy of Sciences Sichuan Translational Medicine Research Hospital, Chengdu 610072, Sichuan Province, China
3Department of Ophthalmology, the First People’s Hospital of Lanzhou City, Lanzhou 730050, Gansu Province, China
Abstract● AIM: To determine the effects of protocatechuic acid (PCA)on streptozocin-induced diabetic retinopathy (DR) in rats.
● KEYWORDS: diabetic retinopathy; streptozocin;protocatechuic acid; oxidative stress; vascular endothelial growth factor
INTRODUCTION
Diabetic retinopathy (DR) is one of the majority of prevalent and severe microvascular complications of diabetes, leading to impaired vision and blindness[1].Approximately one-third of individuals with diabetes mellitus(DM) worldwide exhibit DR symptoms, particularly type 1 diabetic patients[2].The 35.4% of diabetics throughout the globe have DR, with one-third having vision-threatening DR and 7.6% having retinal edoema[3].The global yearly incidence of DR ranges from 2.2% to 12.7%, with progression ranging from 3.4% to 12.3%.Those with moderate disease are more likely to progress to proliferative DR than those with no disease at baseline[4].DR is the major cause of new instances of blindness in the Asia-Pacific region’s middle-aged and older populations.It is estimated that it causes 51% of blindness and 56% of visual impairment cases worldwide, yet knowledge of DR among DM patients ranges from 28% to 84%[5].
Clinically, DR is characterised by changes in retinal microvascular pathology that lead to severe vision loss and lifelong blindness.These changes include capillary obstruction,haemorrhages, microaneurysms, and neovascularization[6].To this day, the exact pathophysiological pathways that lead to DR remain unclear.The pathophysiology of DR is heavily influenced by oxidative stress and inflammation.Studies have demonstrated that sustained elevations in blood glucose levels enhance the retinal production of reactive oxygen species(ROS) and proinflammatory mediators[7].DR is exacerbated by macular edoema, tractional retinal detachment, and neovascular glaucoma, resulting in severe visual impairment[8].As a result of leaking blood vessels, the retinas of diabetes people and diabetic animals contain more proinflammatory factors such as tumor necrosis factor-alpha (TNF-α),interleukin (ⅠL)-1β, ⅠL-6, nuclear factor-κB (NF-κB),etc[9].Tight glycemic control may delay the start of DR because it is widely accepted that hyperglycemia is the key initial factor in the development of DR[10].Inflammation has been demonstrated to have a significant role in triggering DR in recent studies[11].By accelerating the death of capillary cells in the retinal microvasculature and inducing morphological and functional changes[12], oxidative stress is also suggested to play a significant role in developing DR.The cornerstone of treatment for diabetic vascular complications is the suppression of inflammatory and oxidative stress pathways.
The phenolic component protocatechuic acid (PCA) is present in olives, Eucommia ulmoides, Hibiscus sabdariffa,calamondin, and white grape wine[13].Harini and Pugalendi’s[14]study on diabetic rats found that PCA can potentially lower hyperlipidemia.Several studies suggested that PCA could protect against the progression of diabetes by acting as an antioxidant, an anti-inflammatory, and an anticoagulant[15].Even though PCA is an effective antidiabetic medicine in the past, it is also known to have antiglycative and retinolprotective characteristics.This study aimed to investigate the effects of PCA on rats that had developed DR due to being treated with streptozotocin.
MATERIALS AND METHODS
Ethical ApprovalThe study was conducted in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research.Chinese Academy of Sciences Sichuan Translational Medicine Research Hospital, Chengdu, Sichuan Province, China, oversaw the development of the experimental approach.
AnimalsMale Albino Wistar rats between 180 and 220 g in weight and 2 months old were utilized for this study.The rats were housed in clean plastic cages, maintained at 23.2°C with a 12h light/dark cycle, and given unrestricted access to food and drink regularly.The animals were habituated to the facility for two weeks before the start of the experiment.The Shanghai Proton Biotechnology Co., Ltd., standard pellet contains 20%crude protein, 4% fat, 66% carbohydrates, and 10% water.
Induction of DiabetesA intraperitoneal injection of 50 mg/kg of streptozocin dissolved in freshly produced citrate buffer caused diabetes in rats that had fasted overnight(0.1 mol/L, pH 4.5).Rats were injected with the same volume of citrate buffer as the control group.The tail vein blood glucose level was measured three days after streptozocin injection using a glucose metre.If a rat’s blood glucose level was below 300 mg/dL, it was declared diabetic.As anticipated,all animals in groups 2, 3, and 4 developed diabetes.
Experimental DesignRandomly dividing 32 male Wistar albino rats into four groups (n=8), group 1 (Control) was considered a normal group devoid of diabetes, group 2(diabetes) was considered a diabetic group without treatment,group 3 (diabetes+PCA 25 mg/kg) was considered a diabetic group treated orally with 25 mg/kg PCA, and group 4(diabetes+PCA 50 mg/kg) was considered a diabetic group treated orally with 50 mg/kg PCA.After inducing diabetes,PCA treatment began from the conformation of diabetes and continued for eight weeks.Based on the previous literature support, 25 and 50 mg/kg•d[16]of PCA was used in the streptozocin rats.PCA was dissolved in saline and was given to rats by oral gavage technique for 8wk.After the trial, the animals were starved overnight, anaesthetized with ketamine hydrochloride (24 mg/kg, intramuscular injection),and decapitatedviathe neck.The retinas were taken for biochemical and molecular testing.
Determination of Serum Glucose and Glycosylated HemoglobinThe level of glucose in the blood was measured using the Trinder method[17]and a reagent kit bought from bioMerieux chemicals.A blood sample was drawn from each rat and placed in an ethylenediaminetetraacetic acid solution before calculating HbA1c using reagent kits acquired from Stanbio Corporation (Sigma-Aldrich, Cat.No.MAK115).
Retinal Advanced Glycosylated End Products DeterminationTo lyse and homogenize the retinal tissues, we utilized RIPA buffer containing a combination of protease inhibitors.The samples were centrifuged for 20min at 10 000×g and 4°C after homogenization.Using the rat advanced glycosylated end products (AGEs) ELISA reagent (Cat.No.MBS2000151) from MyBioSource, the AGEs content of the supernatant fraction of retinal tissue homogenates was determined.
Evaluation of ROSThe dichlorofluorescein technique was employed to measure the retinal ROS level[18].Different oxidants oxidise the dichlorofluorescein (a nonfluorescent fluorescein derivative) to produce dichlorofluorescein and emit fluorescence.The fluorescence intensity of dichlorofluorescein was measured using a fluorescent microplate reader at 485 nm for excitation and 528 nm for emission.The results were presented as a percentage of the shift seen in the comparison group.
Estimation of Oxidative Stress ParameterThe homogenate of retinal tissue (10% w/v) was produced using a trisphosphate buffer (50 mmol/L) with pH of 7.4.The homogenized tissue was centrifuged for 10min at 4°C.The resulting supernatant was analysed for malondialdehyde (MDA), superoxide dismutases (SOD), catalase (CAT) and reduced glutathione(GSH) activity.
Estimation of MalondialdehydeAs previously described[19], MDA levels in retinal tissue were assessed using thiobarbituric acid reactivity.For protein precipitation,500 µL of the supernatant was combined with 1500 µL of 10% trichloroacetic acid, centrifuged at 4000×g for 10min,and then recombined with 2 mL of 0.67 % thiobarbituric acid.After that, we put the mixture in a 100°C oven for half an hour.Supernatant absorbency was determined after chilling and centrifuging at 4000×g for 15min using spectrophotometryat 535 nm.Values were given in units of micromoles per milligram of protein relative to a standard curve derived from a standard MDA solution.

Table 1 Primer list for RT-PCR analysis
Determination of Antioxidant Enzyme ActivityThe Sigma-Alrich kit (Cat.CS0009) measured SOD activity.The amount of SOD necessary to slow the rate of 2-(4-iodophenyl)-3-(4-nitrophenol)-5-phenyltetrazolium chloride (INT) decline by 50% was designated as one unit.Units per milligram of protein are used to evaluate SOD activity.
CAT activity was assessed using the previously established method of Aebi[20].In a cuvette, 2.9 mL of H2O2in phosphate buffer (50 mmol/L, pH 7.0) was inserted, and the reaction began with the addition of supernatant (100 µmL).At 240 nm,the variations in absorbance per minute were measured.CAT activity was expressed as U/mg protein.
The Beutler[21]method was used to calculate the GSH concentration.NaOH (0.04 mL), DTNB (0.1 mL), Naphosphate puffer (0.2 mol/L, pH 7.4), and supernatant(0.5 mL) were employed in the operation.After 15min at room temperature, the solution was analyzed using a spectrophotometer set at 412 nm.The quantity of GSH present was expressed as micromoles per grams of protein.
Real-time Polymerase Chain ReactionTotal RNA was taken from retinal tissue by following the directions on the RNeasy Mini kit.Real-time polymerase chain reaction (PCR,Eppendorf, Thermocycler, USA) was used to examine the mRNA expression of TNF-α, ⅠL-1β, ⅠL-6, vascular endothelial growth factor (VEGF), and receptor for AGEs (RAGE) in retinal tissue.Table 1 displays the primer sequences for the various target genes.
Western Blot AnalysesRetinal tissues were extracted and mixed at 4°C in 1 mL of RIPA buffer carrying protease inhibitor.After attaining the supernatants, the total quantity of protein was calculated.The total quantity of protein was evaluated after obtaining the supernatants.Proteins were separated using a 12% sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) loaded with 30 µg of sample and transferred to a polyvinylidene difluoride membrane.After blocking the membranes with a 5% nonfat dry milk solution,they were incubated overnight at 4°C with primary antibodies against RAGE (1:1200, Cat.ab37647, Abcam) and anti-NFκB p65 (1:1100, Cat.ab16502).The blots were inspected with a Bio-Rad imaging system equipped with an improved chemiluminescence detection method, and the data was then imported into Image J for additional analysis.

Figure 1 The effect of PCA on the blood HbA1c and serum glucose levels of diabetic rats The data are presented as mean±SD.For this experiment, there were eight animals per group.aP<0.05 compared to the control group; bP<0.05 compared to the diabetic group.HbA1c: Glycated hemoglobin; PCA: Protocatechuic acid.
Statistical AnalysisData were presented as means±standard deviations, and one-way analysis of variance (ANOVA) was performed with the Statistical Package for the Social Sciences version 17.0, and Duncan’s multiple range test (DMRT) was used to generate individual comparisons.WhenP<0.05, values are considered statistically significant.
RESULTS
Effect of PCA on Glucose and HbA1c LevelFigure 1 shows the impact of PCA administration on serum glucose levels and blood HbA1c.Streptozocin-induced diabetic rats had higher(P<0.05) serum glucose levels and HbA1c than the control group.The glucose and HbA1c were significantly lowered by administration of PCA at doses of 25 and 50 mg/kg.
Effect of PCA on AGEs LevelAccording to the results of our investigation, there were more AGEs in the diabetic group’s retina than in control group (Figure 2A).However, these levels were significantly reduced (P<0.05) following treatment with 25 and 50 mg/kg of PCA.RAGE mRNA and protein levels are substantially higher in the diabetic group’s retina, according to RT-PCR (Figure 2B) and Western blot analyses (Figure 2C and 2D;P<0.05).These alterations were reduced after an 8-week PCA therapy (P<0.05).

Figure 2 PCA reduces the levels of AGEs (A) and RAGE (B–D) in the retina of diabetic rats A: Effect of PCA on AGE levels in diabetic rat retinas.B: The histogram represents the quantification of three separate investigations (mean±SD).The expression level of RAGE genes was normalized to the expression level of β-actin mRNA.C: Using Western blotting, the effects of PCA on RAGE protein expression were measured.D: Effect of PCA on densitometrically scanned RAGE protein band intensities.The results have been normalized by designating the RAGE control group as 1 unit, and the histogram shows the quantification of three separate experiments(mean±SD).Three animals were utilized in each cohort for this investigation.aP<0.05 compared to the control group; bP<0.05 compared to the diabetic group.AGEs: Advanced glycosylated end products; RAGE: Receptor for advanced glycation end products; PCA:Protocatechuic acid.
Effect of PCA on ROS and Oxidative StressThe retinal tissue’s ROS and MDA levels were assessed eight weeks following the onset of diabetes (Figures 3 and 4).The results showed that the diabetic retina had considerably greater ROS and MDA levels than the control group (P<0.05) compared to that group.Compared to the diabetic group, PCA administration decreased retinal ROS and MDA levels (P<0.05).
In the retina of streptozocin-induced rats, GSH levels and SOD and CAT activity significantly decreased (P<0.05; Figure 4).Compared to the diabetic group, the administration of PCA raised retinal GSH levels and SOD and CAT activity.
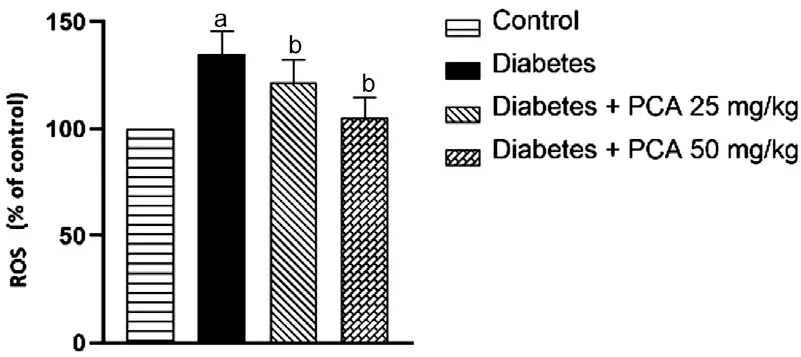
Figure 3 PCA attenuates ROS levels in the retina of diabetic rats The data are presented as mean±SD.For this experiment, there were eight animals per group.aP<0.05 compared to the control group;bP<0.05 compared to the diabetic group.ROS: Reactive oxygen species; PCA: Protocatechuic acid.

Figure 4 Effect of PCA on streptozocin-induced oxidative stress and antioxidant status in retinal tissue of control and experimental rats The data are presented as mean±SD.For this experiment, there were eight animals per group.aP<0.05 compared to the control group;bP<0.05 compared to the diabetic group.MDA: Malondialdehyde;SOD: Superoxide dismutases; CAT: Catalase; GSH: Reduced glutathione; PCA: Protocatechuic acid.
PCA Inhibits the Increase of Inflammatory Cytokines in Retinas of Diabetic RatsWhen compared to normal controls,inflammatory cytokines such as TNF-α, ⅠL-6, ⅠL-1β, and VEGF were produced considerably higher in diabetic rats (P<0.05;Figure 5).PCA (25 and 50 mg/kg) therapy substantially decreased their expression (P<0.05) in diabetic rats.

Figure 5 PCA attenuates TNF-α, Il-6, Il-1β and VEGF levels in the retina of diabetic rats The gene expression levels of TNF-α, IL-6, IL-1β, and VEGF were normalized to the expression level of β-actin mRNA in each sample.aP<0.05 compared to the control group;bP<0.05 compared to the diabetic group.TNF: Tumor necrosis factor;IL: Interleukin; VEGF: Vascular endothelial growth factor; PCA:Protocatechuic acid.
Effects of PCA on the Protein Expression of NF-κBWhen diabetic retinas were compared to controls, NF-κB expression levels increased considerably (P<0.05; Figure 6).However,when diabetic retinas were treated with PCA at doses of 25 and 50 mg/kg, the elevated (P<0.05) level of NF-κB was dramatically reduced.
DISCUSSION
In traditional medicine, herbal treatments have been used as a safe source of therapeutic ingredients to treat various ailments[22].PCA is extensively dispersed and found in most food plants used in traditional Chinese medicine[23].Several protective mechanisms against diabetes[24]have been associated to antioxidant and anti-inflammatory properties of PCA.The results of this study show that PCA therapy dramatically reduced DR in a diabetic rat model by suppressing inflammatory markers such as AGEs, RAGEs, and ROS levels as well as NF-κB p65 phosphorylation.

Figure 6 The effects of PCA on the expression of the NF-κB p65 protein measured using Western blotting (A) and the effect of PCA on the intensities of NF-κB p65 protein bands measured by a densitometer (B) The histogram illustrates the quantification of three independent experiments (mean±SD), with data normalized by defining the control group with NF-κB p65 as 1 unit.aP<0.05 compared to the control group; bP<0.05 compared to the diabetic group.NF-κB: Nuclear factor-kappa B; PCA: Protocatechuic acid.
Streptozocin is typically employed to cause diabetes in rats.streptozocin kills specifically the pancreatic cells that release insulin, resulting in reduced active pancreatic cells and diabetes[25].In accordance with earlier research, the present study demonstrates that streptozocin induces diabetes through increasing blood glucose levels.PCA oral therapy dramatically decreased blood glucose levels, indicating the well-known antihyperglycemic properties of PCA[14].HbA1c levels are elevated in diabetic rats due to prolonged hyperglycemia,which causes haemoglobin glycation.HbA1c concentration is correlated with DR, nephropathy, and neuropathy and is regarded as a diagnostic and prognostic tool for diabetesrelated problems[26].In our work, the treatment of PCA in diabetic rats dramatically lowered HbA1c.PCA reducing HbA1c levels in diabetic rats demonstrates its potential to avoid problems connected with diabetes.
The progress of DR is linked to the accumulation of AGEs in retinal tissue, which is directly associated with the pathophysiology of DR[27].Several studies have shown that people with DR had greater amounts of AGEs in their blood and retinal vasculature, linking increased AGEs levels to the onset of DR.Evidence is mounting suggesting the buildup of AGEs and their receptor, RAGE, causes alterations in the expression of many downstream target proteins in retinal microvascular endothelial cells.In addition, it was shown that AGEs might amplify the neuronal cell death seen in the retina[28].Therefore, complications associated with diabetes may be efficiently reversed by regulating the AGE-RAGE axis.Researchers have shown that treating streptozocin-induced diabetic rats with phytochemicals may reduce their excessive production of AGEs and RAGE[29].Our results showed that AGEs levels in the retina were significantly lower in the diabetic group following treatment with 25 and 50 mg/kg of PCA.Based on these results, it seems that PCA treatment of diabetic rats may lower AGE levels and that RAGE may ameliorate complications produced by AGEs.
Hyperglycemia is widely known to induce alterations in several metabolic processes that generate ROS.DR is one of the most generally acknowledged complications of DM[30].A significant element in DR has increased ROS production caused by high blood glucose levels in diabetic patients[31].Retinal cells are vulnerable to oxidative attacks because they contain several polyunsaturated fatty acids.High glucose oxidation levels in retinal cells also contribute to the aetiology of DR[32].Ⅰn this regard, we have detected a significant increase in MDA and ROS levels and a significant drop in endogenous antioxidants (CAT, GPx, and SOD).PCA supplementation dramatically decreased MDA and ROS levels and restored the activity of endogenous antioxidants (GPx, CAT, and SOD).In DR pathogenesis, ROS are mainly involved[33], which seems suited for therapeutic approaches, such as using antioxidants derived from natural sources.PCA is a dietary phenolic compound shown to have significant antioxidant effects in several diabetic animal models and other studies[14].The renoprotective effect of PCA in DR may thus be mediated by an increase in antioxidative defence and a decrease in oxidative stress markers.
VEGF is considered to be responsible for the increased permeability and angiogenesis associated with DR[34].Diabetesinduced retinal VEGF levels are thought to be regulated by oxidative stress[35].In the last several decades, many clinical studies have demonstrated that the expression of inflammatory markers is elevated in the retina of DR rats[36].The disruption of blood-retinal barrier (BRB) homeostasis caused by the overexpression of inflammatory markers such as VEGF,TNF-α, and ⅠL-1β may result in hypoxia and ischemia[37].Due to the role of VEGF and inflammatory responses in the pathophysiology of DR, pharmacological medications with anti-VEGF and anti-inflammatory actions are given special consideration for treating DR[38].In the present study, PCA treatment reduced the expression of retinal VEGF, ⅠL-1β,ⅠL-6, and TNF-α in diabetic rats.Due to its anti-VEGF and anti-inflammatory characteristics, PCA may thereby protect against DR.Many studies have shown that p38 MAP kinase regulates NF-κB activity in various tissues[39].Previous research found that DR in rats was associated with elevated NF-κB activity in the retinas[40].Anti-RAGE treatment for DR rats decreased NF-κB expression and retinal inflammation, according to a separate study by Aminet al[41].Proinflammatory cytokines including ⅠL-1β, ⅠL-6, and TNF-α are often produced in response to NF-κB activation and are considered key mediators of inflammatory processes.The increase in intercellular adhesion molecule-1 (ICAM-1), leukocyte adhesion, and BRB leakage that is seen in diabetic retinas can be mitigated by inhibiting NF-κB[42].Diabetic mice showed increased phosphorylation of NF-κB p65 compared to healthy controls.As a result, our findings are consistent with previous research that suggests elevated NF-κB activation in DR.PCA therapy significantly reduced this.Treatment of diabetic rats with PCA indicates a considerable reduction in the high levels of NF-κB in the retinal extracts.The decrease of NF-κB levels generated by PCA injection suggests that it can minimize retinal inflammation and consequential damage caused by DR.In conclusion, the current study proved that PCA treatment might ameliorate DR in streptozocin-induced diabetic rats.The antioxidant and anti-inflammatory characteristics of PCA, as well as its inhibition of AGEs and RAGE, may be responsible for its protective effects against DR.We found that PCA had positive effects on many DR targets.This discovery might be helpful in alternative therapy in DR.However, more research is advised to verify the current results.
The principal constraint of our study resides in the unavailability of electroretinography data regarding the experimental animals.Electroretinography stands as a crucial diagnostic modality to appraise the sensitivity of the retina to light stimuli.
ACKNOWLEDGEMENTS
Authors’contributions:Wan L and Li J performed the experiments; Chen WB did statistical analysis; Wu GQ designed the experiments and wrote the paper.
Foundation:Supported by Model Construction of Regional Prevention and Treatment of Diabetic Retinopathy, Sichuan Science and Technology Support Program (No.2016FZ0091).
Conflicts of Interest:Wan L, None; Li J, None; Chen WB,None;Wu GQ, None.
 International Journal of Ophthalmology2023年6期
International Journal of Ophthalmology2023年6期
- International Journal of Ophthalmology的其它文章
- Preliminary proteomic analysis of human tears in lacrimal adenoid cystic carcinoma and pleomorphic adenoma
- Assessment of the effects of intrastromal injection of adipose-derived stem cells in keratoconus patients
- Evaluation of optic nerve head vessels density changes after phacoemulsification cataract surgery using optical coherence tomography angiography
- Stability of neodymium:YAG laser posterior capsulotomy in eyes with capsular tension rings
- Comparison of the efficacy and safety of ultrasonic cycloplasty vs valve implantation and anti-VEGF for the treatment of fundus disease-related neovascular glaucoma
- Volumetric fluid analysis of fixed monthly anti-VEGF treatment in patients with neovascular age-related macular degeneration
