Clinical characteristics of pediatric cases infected with the SARS-CoV-2 Omicron variant in a tertiary children’s medical center in Shanghai,China
Nan Shen · Yu-Fen Wu · Yi-Wei Chen · Xiao-Yan Fang · Min Zhou · Wen-Yu Wang · Ming-Yu Tang ·Qiu-Hui Pan · Ji Ma · Hao Zhang · Qing Cao
Abstract Background The number of pediatric cases of infection with the severe acute respiratory syndrome coronavirus 2 (SARSCoV-2) Omicron variant has increased.Here,we describe the clinical characteristics of children in a tertiary children’s medical center in Shanghai.Methods A total of 676 pediatric coronavirus disease 2019 (COVID-19) cases caused by the Omicron variant who were admitted to the Shanghai Children's Medical Center from March 28 to April 30,2022 were enrolled in this single-center,prospective,observational real-world study.Patient demographics and clinical characteristics,especially COVID-19 vaccine status,were assessed.Results Children of all ages appeared susceptible to the SARS-CoV-2 Omicron variant,with no significant difference between sexes.A high SARS-CoV-2 viral load upon admission was associated with leukocytopenia,neutropenia,and thrombocytopenia (P =0.003,P =0.021,and P =0.017,respectively) but not with physical symptoms or radiographic chest abnormalities.Univariable linear regression models indicated that comorbidities (P =0.001) were associated with a longer time until viral clearance,and increasing age (P <0.001) and two doses of COVID-19 vaccine (P =0.001) were associated with a shorter time to viral clearance.Multivariable analysis revealed an independent effect of comorbidities (P <0.001) and age (P =0.003).The interaction effect between age and comorbidity showed that the negative association between age and time to virus clearance remained significant only in patients without underlying diseases (P <0.001).Conclusion This study describes the clinical characteristics of children infected with the Omicron variant of SARS-CoV-2 and calls for additional studies to evaluate the effectiveness and safety of vaccination against COVID-19 in children.
Keywords COVID-19 vaccine · Omicron variant · Pediatric COVID-19 · SARS-CoV-2
Introduction
At the beginning of 2022,a wave of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spread rapidly in Shanghai,China.The Shanghai Municipal Health Commission reported a total of 537,644 cases,including 53,355 confirmed cases as of April 30,2022.Some studies have suggested that this outbreak was caused by the BA.2.2 sublineage [1],which is derived from the Omicron variant[2].The Omicron variant has raised serious concerns among the global public health community and general public due to its increased transmissibility,severity,and capacity for immune escape [3].Promisingly,recent studies revealed that,as in adults,the severity of coronavirus disease 2019(COVID-19) in children infected with the Omicron variant was significantly less than that in those infected with the Delta variant [4,5].Moreover,as a severe complication of SARS-CoV-2 infection,a multisystem inflammatory syndrome in children during the Omicron wave was less frequent and severe than during the Alpha or Delta waves of the COVID-19 pandemic [6].However,notably,the presence of comorbidities was associated with moderate or severe outcomes among children with Omicron variant infection but not with Delta variant infection [4].To date,few studies have examined pediatric cases of Omicron infections in China.Therefore,an updated analysis of pediatric cases with confirmed SARS-CoV-2 Omicron variant infection could help define the scope of clinical characteristics and severity of this disease.
Shanghai Children's Medical Center (SCMC) bears more than a quarter of the medical demand of children in Shanghai,including the Pudong and Fengxian districts.As of May 31,2022,a total of 798 confirmed cases of COVID-19 have been admitted to SCMC.COVID-19 vaccines are highly effective against symptomatic forms of this disease,and inactivated vaccines are approved for use in children and adolescents aged 3–17 years in China [7].Modest reductions in mRNA vaccine effectiveness against infection and mild disease have been observed with the Beta and Delta variants[8–10].However,the effectiveness of inactivated vaccines against symptomatic COVID-19 caused by the Omicron variant has been investigated in only a few studies.In the present study,we summarized the clinical characteristics of the first 676 children who were admitted to SCMC from March 28 to April 30,2022,addressing the effectiveness of inactivated COVID-19 vaccines in children infected with the SARS-CoV-2 Omicron variant.
Methods
Study design and participants
This study was a single-center,prospective,observational real-world study conducted at the Shanghai Children's Medical Center (SCMC) between March 28,2022,and April 30,2022.SCMC admits patients with various pediatric diseases up to the age of 18 years.In this study,pediatric patients infected with the SARS-CoV-2 Omicron variant were enrolled only if they volunteered to participate,and their electronic medical records were available and included demographic data,COVID-19 vaccination status,clinical symptoms,laboratory findings,and chest radiographic findings.The study was approved by the Institutional Review Board and the Ethics Committee of Shanghai Children's Medical Center (SCMCIRB-K2022053-3).Before inclusion,written informed consent was obtained from each patient and/or their parents.
SARS-CoV-2 nucleic acid detection
Oropharyngeal swabs were obtained upon admission and stored at 4 °C in inactivated virus preservation solution until RNA extraction.Simultaneous detection of three SARS-CoV-2 genes,ORF1ab(encoding RNA-dependent RNA polymerase),N(encoding nucleocapsid protein),andE(encoding envelope protein),was performed using SARSCoV-2 nucleic acid detection kits (Shanghai ZJ Bio-Tech Co.Ltd.,Liferiver,cat.No.Z-RR-0479-02-50) with preserved oropharyngeal swabs following the manufacturer's instructions.Briefly,we prepared 25 μL reaction mixtures containing 5 μL of purified RNA and 20 μL of polymerase chain reaction (PCR) master mix for each sample.The PCR cycling conditions were as follows: (1) 45 °C for 10 minutes;(2) 95 °C for 3 minutes;and (3) 45 cycles of 95 °C for 15 seconds and 58 °C for 30 seconds.A cycle threshold(Ct) ≤ 43 was set as a cutofffor SARS-CoV-2 positivity.
Clinical data collection
Electronic medical records were reviewed to obtain demographic and clinical data,including age,sex,comorbidities,clinical symptoms,laboratory findings,chest radiographic findings,and COVID-19 vaccination status.COVID-19-positive patients were then transferred to hospitals specializing in COVID-19 treatment,and follow-up interviews were conducted by phone every two weeks until viral clearance.The main purpose of the follow-up interview was to determine the time elapsed in hospital care until the patients were negative for SARS-CoV-2.
Several special clinical variables,including radiographic chest abnormalities,SARS-CoV-2 viral load upon admission,and time to SARS-CoV-2 clearance,were collected.The definitions of these variables are as follows.Radiographic chest abnormality referred to at least one abnormal radiographic feature,such as blurred lung texture,lung exudation,lung consolidation,and pleural effusion,based on chest X-ray or chest computed tomography (CT) at initial diagnosis of COVID-19.Moreover,the Ct value of quantitative PCR (qPCR) was used to indicate the SARS-CoV-2 viral load upon admission.A higher Ct value indicates a lower viral load.In addition,the time to SARS-CoV-2 clearance was taken from the date of qPCR positivity for SARSCoV-2 and ended on the first negative qPCR.
Statistical analysis
As appropriate,continuous variables were expressed as medians and interquartile ranges (IQRs) or means and standard deviations depending on the normality of the distribution of the variables.Categorical variables were summarized as counts and percentages.Comparisons of clinical characteristics among patients were conducted between those with normal and those with abnormal chest radiographic manifestations using either Student'sttest or the Wilcoxon ranksum test for continuous variables and the Chi-squared test or Fisher's exact test for categorical variables.Univariate logistic regression was used to evaluate the association between each COVID-19 symptom and the SARS-CoV-2 viral load upon admission.Univariate linear regression was further used to assess the association between routine blood test results and the viral load upon admission.In addition,baseline clinical impacts of the viral load and the time needed for viral clearance were estimated using univariate and multivariate linear regression models.Interactions between impacts were tested.If a significant interaction effect was detected,the multivariable model was further stratified.Pvalues <0.05 were considered statistically significant.All statistical analyses were performed using R software,version 4.0.3 (http:// www.R-proje ct.org).
Results
Demographic and clinical characteristics
A total of 676 pediatric cases of COVID-19 were enrolled in this study,of whom 611 (90.4%) completed followup interviews (Fig. 1).During this COVID-19 outbreak in Shanghai and in the Pudong District in particular,the number of admissions increased in our hospital in a manner consistent with the significant increase in the overall number of infections (Supplementary Fig.1).In the present study,474 of 676 patients underwent chest radiographic examination,including chest X-ray and chest CT.Eight of the 474 radiographic chest examinations showed abnormalities in both types of imaging,with 27.6% presenting with abnormalities in at least one type of imaging.The most common changes in chest imaging were blurred lung texture (89.3%) and exudation (13.7%).Supplementary Figure 2 shows the radiologic findings upon admission.
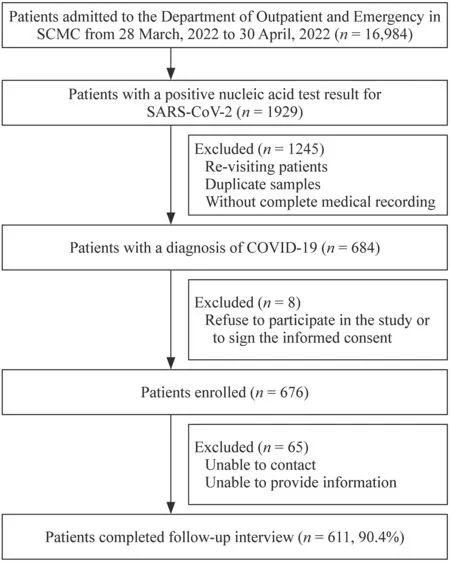
Fig.1 The flow chart of this study. SCMC Shanghai Children's Medical Center,COVID-19 coronavirus disease 2019,SARS-CoV-2 severe acute respiratory syndrome coronavirus 2
Notably,transcutaneous oxygen saturation remained normal in all patients at admission.Furthermore,no patient was admitted to an intensive care unit,required mechanical ventilation,or died in this study.Moreover,the median age of these patients was 2.9 (IQR=1.4–6.2)years,and 54.7% were male.The highest infection rate was observed in children aged 1–3 years (31.8%),followed by 4–6 years (22.2%),and 7–12 years (20.7%).Younger age was associated with a higher incidence of abnormal chest imaging (P=0.002).Fever was the most common symptom,present in 86.5% of the patients upon admission,followed by cough (35.8%),rhinorrhea (11.2%),and nausea and vomiting (9.9%).Among the overall children,10.5%had at least one underlying disease (e.g.,hematological malignancies,cardiovascular diseases,and chronic lung diseases).Children with comorbidities were more likely to present abnormal chest imaging than those without comorbidities,with borderline statistical significance (15.3% vs.8.7%,P=0.057).Vaccination status was known for 646(95.6%) patients,of whom 119 (18.4%) were vaccinated.Of the 119 vaccinated patients,22 (3.3%) were vaccinated once,and 97 (14.3%) were vaccinated twice.No children had ever received three doses of inactivated vaccines.Vaccination status was not associated with radiographic chest abnormalities in the patients.The demographic and clinical characteristics of the pediatric patients are presented in Table 1.
Detection assays performed at admission showed that the median Ct values of the SARS-CoV-2ORF1ab,N,andEgenes were 28.2 (IQR=21.1–32.4),27.6 (IQR=19.2–32.1),and 26.3 (IQR=19.4–30.7),respectively,with higher Ct values corresponding to lower viral RNA titers.No significant differences were found between the Ct values of theORF1ab,N,andEgenes in the abnormal chest imaging group and the normal chest imaging group(P=0.571,P=0.434,andP=0.550,respectively).In addition,there were no obvious differences between white blood cell,lymphocyte,and neutrophil counts or among the C-reactive protein values of these two groups (P=0.947,P=0.562,P=0.652,andP=0.185,respectively)(Table 1).The children diagnosed with COVID-19 with abnormal chest imaging did not have more prominent laboratory abnormalities than those with normal chest imaging.

Table 1 Clinical characteristics and laboratory findings of children with SARS-CoV-2 infection with normal or abnormal chest radiographic findings
Clinical characteristics and protective or impact factors for viral load associated with SARS-CoV-2 viral load upon admission
The associations between SARS-CoV-2 viral load upon admission and different clinical characteristics of these children were evaluated.First,univariate logistic regression was used to assess possible relationships between COVID-19 symptoms and SARS-CoV-2 viral load upon admission to our clinic.No significant association was observed between the prevalence of COVID-19-related symptoms and SARS-CoV-2 titer,using Ct values for theORF1abgene from qPCR-based detection tests as a marker for viral load in patients (Table 2).In addition,there was no association between chest radiographic results and SARSCoV-2 viral load upon admission [odds ratio (OR)=1.013,95% confidence interval (CI)=0.982–1.046,P=0.403].However,evaluation of potential links between routine blood tests and SARS-CoV-2 viral load upon admission by univariate linear regression revealed a significant inverse relationship between high viral load and several markers of infection,including low white blood cell counts (coefficient=0.056,95% CI=0.019–0.092,P=0.003),low neutrophil counts (coefficient=0.039,95% CI=0.006–0.072,P=0.021),and low platelet counts (coefficient=1.277,95% CI=0.233–2.322,P=0.017) (Table 3).These results thus indicated that SARS-CoV-2 viral load upon admission was associated with laboratory abnormalities rather than physical symptoms and radiographic chest abnormalities.In light of this association,we next investigated whether clinical features could be used to predict high SARS-CoV-2 viral loads at admission.Both univariate and multivariable linear regression analyses showed that age,sex,comorbidities,and COVID-19 vaccination status were not associated with Ct values from the initial SARS-CoV-2 detection assays(Table 4).

Table 2 Effects of SARS-CoV-2 initial viral load on clinical symptoms

Table 3 Effects of SARS-CoV-2 initial viral load on laboratory findings
Factors associated with duration until SARS-CoV-2 viral clearance in children with COVID-19
We next investigated whether clinical characteristics were associated with the time to SARS-CoV-2 clearance.First,the examination of univariable linear regression models for SARS-CoV-2 viral clearance indicated that comorbidities (coefficient=2.07,95% CI=0.90–3.23,P=0.001) were associated with a longer time until viral clearance,and increasing age (coefficient=− 0.23,95% CI=− 0.33 to − 0.13,P<0.001) or two doses of a COVID-19 vaccine (coefficient=− 1.80,95%CI=− 2.86 to − 0.74,P=0.001) were associated with a shorter time to viral clearance.However,further examination revealed that only comorbidities (coefficient=2.77,95% CI=1.58–3.96,P<0.001) and increasing age (coefficient=− 0.33,95% CI=− 0.54 to − 0.11,P=0.003)remained significant in multivariable analysis (Table 5).Moreover,a significant interaction effect between age and comorbidity on time to virus clearance was detected(P<0.001) (Supplementary Table 1).Thus,regression analysis was further stratified by comorbidities.The subgroup analysis in Table 5 and along with Fig. 2 showed that the negative association between age and time to virus clearance remained significant only in patients without underlying diseases (coefficient=− 0.37,95%CI=− 0.46 to − 0.27,P<0.001 in the univariable model;coefficient=− 0.37,95% CI=− 0.50 to − 0.23,P<0.001 in the multivariable model) but not in patients with underlying diseases (coefficient=0.25,95% CI=− 0.12–0.63,P=0.184 in the univariable model;coefficient=0.18,95% CI=− 0.27–0.62,P=0.425 in the multivariable model) (Table 5).Furthermore,the influence of two doses of COVID-19 vaccine on time to SARS-CoV-2 clearance was not significant after adjustment for age,sex,and comorbidity status in multivariable analysis(coefficient=− 0.37,95% CI=− 1.71–0.98,P=0.594)(Table 5).In addition,we also noticed that only comorbidities (coefficient=3.58,95% CI=2.17–5.00,P<0.001)were associated with a longer time until viral clearance,and neither age nor vaccination status was associated with the time until viral clearance in the subgroup above three years of age (Supplementary Table 2).

Fig.2 The time until viral clearance among patients of different ages and vaccination statuses with (a) or without (b) underlying diseases

Table 4 Univariate and multivariable linear regression analyses of impact factors for initial viral load in the study patients

Table 5 Univariate and multivariable linear regression analyses of impact factors for time until viral clearance in all patients and the patients without or with underlying disease
Discussion
As the COVID-19 pandemic spread,so did its effects on the pediatric population.Although it was initially argued that children were less susceptible to SARS-CoV-2 infection,increasing evidence indicates that children have similar rates of SARS-CoV-2 infection as adults [11].Significant differences between COVID-19 in children and adults include a higher proportion of children with asymptomatic infection than adults,and even among children with symptomatic infection,the disease is most often mild [12,13].From the beginning of 2022,SARS-CoV-2 infections fueled by the Omicron variant have been predominantly responsible for outbreaks in China,especially in Shanghai.While the Omicron variant exhibits decreased levels of virulence,several reports have described a range of severe outcomes and considerable mortality rates among adults [14].The results of this study show that children of all ages are susceptible to the Omicron variant,with no significant difference between sexes.Although clinical manifestations in children infected with Omicron are not severe,younger children are more likely to display abnormalities on chest radiography.Moreover,a high SARS-CoV-2 viral load upon admission is associated with leukocytopenia,neutropenia,and thrombocytopenia but not with physical symptoms and radiographic chest abnormalities.In addition,the time until SARS-CoV-2 viral clearance is longer in younger children or those with underlying diseases.
More than half a million cases of SARS-CoV-2 virus were identified in Shanghai in March and April of 2022.The infection rate in children was also much higher than that reported for previous outbreaks.However,this unfortunate outcome for children could be expected,in light of data showing that the Omicron variant has higher transmissibility than any of the previous ten SARS-CoV-2 variants [15].The high number of mutations in the spike protein facilitates at least partial immune evasion for Omicron,even from polyclonal antibody responses,frequently facilitating reinfection and vaccine breakthroughs [16].One recent study revealed that Omicron can replicate quickly in primary cultures of human airway cells at rates faster than those reported for the Delta variant,which was previously the dominant variant of concern.The Omicron spike protein also binds to its primary receptor,human angiotensin-converting enzyme 2,with higher affinity than other variants.These results show that Omicron can infect more cells in the respiratory epithelium than other variants,increasing its infectiousness at lower exposure doses and enhancing its intrinsic transmissibility [17].
Fortunately,no seriously ill pediatric patients infected with the SARS-CoV-2 Omicron variant required mechanical ventilation in this study,regardless of whether they had underlying diseases.COVID-19 disease severity appeared to be lower during the Omicron period than during other previous periods of high transmission [18].In a recent in vivo study,the SARS-CoV-2 Omicron variants exhibited less pathogenicity than other earlier SARS-CoV-2 strains [19].Additionally,consistent with previous reports in children,we found that the SARS-CoV-2 viral load at the time of hospital admission was not associated with disease severity.Asymptomatic children can also carry high viral loads and shed culturable viruses [20].Therefore,although COVID-19 severity is milder in pediatric populations than in adult populations,children can still potentially serve as community reservoirs for actively replicating the virus.
This study showed that increasing age or two doses of a COVID-19 vaccine were associated with a shorter time to viral clearance in a univariable model.However,only increasing age remained significant in multivariable analysis.In China,inactivated COVID-19 vaccines are available for children older than three years [7].In the subgroup above three years of age,there was no correlation between age or vaccination status and the time to viral clearance.The difference between these results may be related to the high correlation between COVID-19 vaccination status and age.Given that Shanghai successively launched vaccination programs for children aged 12–17,6–11 and 3–5 years old from August to December 2021,101 (36.2%) patients above three years of age without underlying diseases were vaccinated in the present study.Among them,the vaccination coverage rate in patients aged seven years or above reached 65.8%.Additionally,18 (39.1%) children over three years of age with comorbidities were vaccinated,of whom 15 were over seven years old.Obviously,older age was related to a high vaccination rate.Thus,determining which factor played a vital role in shortening viral clearance when both were associated was difficult in this study.
The widespread COVID-19 vaccine is a critical tool to combat COVID-19 in children.One recent study showed that two doses of the COVID-19 mRNA vaccine could provide limited protection against symptomatic disease caused by the Omicron variant,but that protection waned over time [21].Data from an in vitro study indicated that this highly infectious variant can be neutralized after a boost with immune sera elicited by inactive COVID-19 vaccines[22].A recent study demonstrated that a three-dose inactivated vaccine is a protective factor for severe COVID-19 in adults infected with the Omicron variant;however,no significant correlation was observed between vaccination and disease severity in children.Furthermore,the duration of hospitalization and recovery was shorter for those who had received two or three doses of inactivated vaccine than for unvaccinated patients for patients across all age groups,but no difference was observed in pediatric patients [5].Nevertheless,the sample size of this study was too small to be conclusive.Our findings also suggest that COVID-19 disease severity is not associated with vaccination status in children.Two doses of the COVID-19 vaccine may be beneficial in shortening the time to viral clearance.Whether two doses of COVID-19 vaccine can be used as an independent protective factor in shortening the time to viral clearance remains incompletely defined.Thus,additional well-designed cohort studies are required to explore the safety and efficacy of inactivated vaccines in children to improve current vaccination policies.
This study has several other limitations.First,this was a single-center study;a larger cohort including different regional populations in a multicenter study could provide more robust statistical support for our conclusions.Second,chest CT scans were unavailable for the large majority of cases in this study.Because the amount of radiation populations received in CT examination was higher than that in X-rays,and the patients in this study were nonsevere at the time of admission.Therefore,the determination of infection in the lungs relied on chest X-rays in this study.
In conclusion,this study describes the clinical characteristics of pediatric cases infected with the Omicron variant of SARS-CoV-2.Omicron variant infection in children is associated with less severe disease.A high SARS-CoV-2 viral load upon admission is associated with leukocytopenia,neutropenia,and thrombocytopenia.In addition,the time until SARS-CoV-2 viral clearance is longer in younger children or those with underlying diseases.
Supplementary InformationThe online version contains supplementary material available at https:// doi.org/ 10.1007/ s12519-022-00621-6.
AcknowledgementsWe thank all the hospital staffmembers who worked hard under this pandemic's pressure.We also thank Bin Zhang(Medical Department,Shanghai Children's Medical Center,National Children’s Medical Center) for his effort in collecting the information used in this study.In particular,we would like to thank all our patients who were willing to participate in the study.
Author contributionsSN,WYF,CYW,and FXY contributed equally to this study.CQ and ZH conceived and designed this study.WYF,CYW,ZM,WWY,TMY,SBY,PQH,and MJ performed the study.FXY analyzed the data.SN performed the study and wrote the manuscript.All authors approved the final version of the manuscript.
FundingThis work was funded by the Innovative Research Team of High-Level Local Universities in Shanghai (SHSMUZDCX20212800),Science and Technology Commission Shanghai Municipality (20JC1410200,20JC1410204),2021 Shanghai University Teacher Training Plan-Industry University Research Practice Plan Project (EYJ26.RL017),Shanghai Municipal Health Commission (2019SY049),and Shanghai Medical Leading Talents Program(2019LJ22).
Data availabilityThe datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethical approvalThe study was approved by the Institutional Review Board and the Ethics Committee of Shanghai Children’s Medical Center (SCMCIRB-K2022053-3).
Conflict of interestNo financial or non-financial benefits have been received or will be received from any party related directly or indirectly to the subject of this article.Nan Shen is a member of the Editorial Board forWorld Journal of Pediatrics.The paper was handled by the other Editor and has undergone a rigorous peer review process.Nan Shen was not involved in the journal's review of,or decisions related to,this manuscript.
 World Journal of Pediatrics2023年1期
World Journal of Pediatrics2023年1期
- World Journal of Pediatrics的其它文章
- Innovative treatments for congenital heart defects
- Perioperative extracorporeal membrane oxygenation in pediatric congenital heart disease: Chinese expert consensus
- Role of ultrasound in the treatment of pediatric infectious diseases:case series and narrative review
- Manual and alternative therapies as non-pharmacological interventions for pain and stress control in newborns: a systematic review
- Vitamin D therapy in pediatric patients with inflammatory bowel disease: a systematic review and meta-analysis
- Assessment of compatibility of rhIGF-1/rhIGFBP-3 with neonatal intravenous medications
