The construction of aggregation-induced charge transfer emission systems in aqueous solution directed by supramolecular strategy
Weirui Qin,Minzn Zuo,Pengo Niu,Xio-Yu Hu,*,Leyong Wng,c,*
a Key Laboratory of Mesoscopic Chemistry of MOE,Jiangsu Key Laboratory of Advanced Organic Materials,School of Chemistry and Chemical Engineering,Nanjing University,Nanjing 210023,China
b College of Materials Science and Technology,Nanjing University of Aeronautics and Astronautics, Nanjing 211106, China
c Department of Chemistry, Xihua University, Chengdu 610039, China
ABSTRACT Novel aggregation-induced charge transfer(CT)emission systems with long luminescence lifetime directed by supramolecular strategy have been successfully developed in water.The dimethylacridine-based electron donor(BrAc)with excellent aggregation ability can co-aggregate with a triazine-based electron acceptor(TRZ)to form nanorods in water,which exhibit CT emission with long lifetime(τ = 0.92 μs).As for a similar electron donor(QaAc)with poor aggregation ability,water-soluble pillar[5]arene(WP5)can be introduced to promote the aggregation process,leading to the obvious CT emission with long lifetime(τ = 0.61 μs).In addition,structural modification of the acceptor with substituent groups possessing stronger electron-accepting capabilities will cause red-shift(about 50 nm)of the emission,which allows conveniently constructing long lifetime organic luminescent materials with different emission colors.
Keywords:Aggregation-induced charge transfer emission Supramolecular strategy Long luminescence lifetime Self-assemble Co-aggregate
Organic luminescent materials directed by supramolecular strategy in aqueous solution have been widely investigated in recent years[1-6].Based on this design,different types of molecules can be noncovalently incorporated into the formed nanoaggregates through weak intermolecular interactions,which can avoid multiple steps of synthesis and purification during the structure fabrication.In addition,these highly ordered nanoaggregates can effectively shorten the distance between the donor and acceptor to ensure efficient energy transfer(ET)or charge transfer(CT),which provide a convenient way for fabricating functional nanosystems[7-11].For example,in our previous work,a highly efficient energy transfer(ET)system based on the self-assembly of a watersoluble pillar[5]arene(WP5),a bis(4-phenyl)acrylonitrile derivative(BPT),and two types of fluorescence dye was successfully fabricated,and it showed potential application in artificial lightharvesting[7].However,these ET systems are usually emissive with very short luminescence lifetime(τ <10 ns),as the same level as the autofluorescence from organisms,which greatly limits their applications in the field such as time-resolved luminescence imaging[12-16].
Comparing with ET materials,charge transfer(CT)materials with long luminescence lifetime at the microsecond level have always been a research hotspot for scientists,owing to their wide applications in many fields such as organic light-emitting diodes,time-resolved luminescence imaging,and photocatalytic synthesis[17-20].In the construction of CT molecules,electron donor(D)and acceptor(A)are often linked through conjugated bonds to ensure efficient charge transfer[21-24].However,the resulted through-bond charge transfer(TBCT)effect tends to induce a large red-shift of the emission,which is undesired for the construction of short-wavelength CT materials[25].Recently,a new construction strategy for CT materials with long lifetime has aroused great interests,in which D and A units are non-conjugated but spatially proximate.For example,Wanget al.developed a kind of CT polymers based on non-conjugated polyethylene backbones with through-space charge transfer(TSCT)effect between pendant D and A units[26].Such TSCT effect cannot only ensure efficient charge transfer,but also can avoid the large red-shift of emission for the conjugated architectures[27-29].However,among those reported CT materials with TSCT effect,D and A units are still covalently connected based on complicated synthesis procedures to ensure efficient charge transfer,which greatly limits the design of various novel CT materials.From this perspective,delicate design of CT materials with long lifetime directed by supramolecular strategy featuring non-covalent interactions and structural diversity is highly appealing.
Herein,different aggregation-induced charge transfer(CT)emission systems with long lifetime directed by supramolecular strategy have been successfully developed in aqueous phase(Scheme 1).A dimethylacridine-based derivative(BrAc)with excellent aggregation ability was designed as the electron donor,which could co-aggregate with the triazine-based electron acceptor(TRZ)to form nanorods in water,leading to the observation of greenish CT emission with long lifetime(τ= 0.92 μs).As for another electron donor(QaAc)with similar structure but enhanced watersolubility and poor aggregation ability,water-soluble pillar[5]arene(WP5)could be introduced to promote the aggregation process,thus resulting in the CT emission with long lifetime(τ= 0.61 μs).The formation of nanoaggregates not only shortens the distance between the donor and acceptor to promote charge transfer process,but also reduces the adverse effects of external oxygen on the CT emission.In addition,structural modification of the acceptor with substituent groups bearing stronger electron-accepting abilities would cause obvious red-shift of the emission,which allows the convenient construction of long lifetime organic luminescent materials with different emission colors.
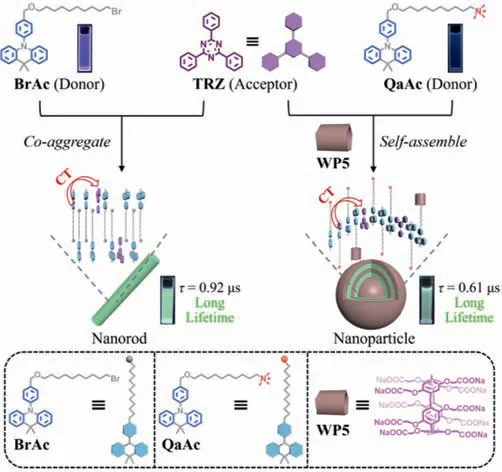
Scheme 1.Schematic illustration of the aggregation-induced charge transfer emission systems in water directed by supramolecular strategy.
BrAc,a typical dimethylacridine-based electron donor,was first synthesized by using 9,10-dihydro-9,9-dimethylacridine and 4-bromobenzaldehyde as the starting materials(Scheme S1 and Figs.S1-S6 in Supporting information).The long alkyl chain of BrAc was designed to increase the stability of nanoaggregates in aqueous solution.It has been widely reported that CT materials showing long lifetime can be successfully constructed when dimethylacridine-based electron donors and triazine-based electron acceptors are covalently modified on the side chains of polymers[24-26].Hence,a triazine-based derivative(TRZ)was selected as acceptor and it was inferred that the system would exhibit CT emission with long lifetime when BrAc and TRZ coaggregated to form abundant nanoaggregates throughπ-πinteraction and hydrophobic interaction in aqueous phase.
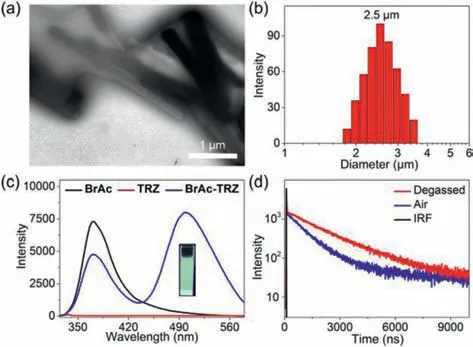
Fig.1.TEM image(a)and DLS data(b)of BrAc-TRZ nanoaggregates.(c)PL spectra of BrAc,TRZ,and BrAc-TRZ in water(λex = 300 nm).Inset:PL images of BrAc-TRZ in water.(d)Transient PL decay spectra of BrAc-TRZ nanoaggregates at 499 nm in air and after degassed(λex = 300 nm).IRF:Instrument response function.[BrAc]= 250 μmol/L,and[TRZ]= 20 μmol/L.
To verify the above assumption,the self-aggregate behavior of BrAc-TRZ complex in water was explored.BrAc-TRZ solution showed obvious Tyndall effect,suggesting the existence of abundant nanoaggregates.The morphology and size of the nanoaggregates formed by BrAc-TRZ complex were determined by transmission electron microscopy(TEM)and dynamic light scattering(DLS)measurements,respectively.The corresponding results revealed the formation of large-sized nanorods with an average diameter of 2.5 μm(Figs.1a and b).Subsequently,photoluminescence(PL)performance of BrAc-TRZ aggregates in water was studied.As shown in Fig.1c,TRZ was basically not emissive,but the BrAc-TRZ coaggregates showed strong greenish fluorescence with a maximum emission peak at 499 nm,which displayed red-shifted emission relative to BrAc(λem,max= 371 nm).Upon gradually increasing the concentration of TRZ,the fluorescence intensity of BrAc at 371 nm decreased,while the emission peak at 499 nm increased(Fig.S12 in Supporting information).The above phenomena suggest distinct intermolecular charge transfer between BrAc and TRZ has taken place,which is similar to the phenomena in the reported literature[24].Then,transient PL decay spectra in water were measured under different conditions.With respect to free BrAc,the average lifetime(τ)was below 5 ns(Fig.S13 in Supporting information).TRZ is not emissive,hence the spectrum was absent.However,for the BrAc-TRZ aggregates in air and after degassed,the average lifetime(τ)was 0.92 μs and 1.78 μs,respectively(Fig.1d),which showed obvious long lifetime emission compared with free BrAc.
For in-depth understanding the PL properties of BrAc-TRZ aggregates,the absorption and excitation spectra were measured(Fig.S14 in Supporting information).The absorption spectrum of BrAc-TRZ aggregates was a simple overlay of BrAc and TRZ.No new absorption peak appeared,which confirmed that weak intermolecular interaction occurred between BrAc and TRZ in the ground state.In addition,the excitation spectrum of BrAc-TRZ aggregates matched well with that of free BrAc,which indicated the above two systems have the same excitation pathway[30].Based on the above experiments,we inferred that the emission at 499 nm might be attributed to the charge transfer in the excited state[31,32].Moreover,the photoluminescence quantum yield(PLQY)of BrAc-TRZ aggregates was measured to be 19.70% by integrating sphere(Fig.S18 in Supporting information).
To further understand the mechanism of CT emission,PL behavior of BrAc-TRZ in good solvent(THF)was further studied.Compared with free BrAc and TRZ,the appearance of new emission peak could not be observed in the PL spectra of BrAc-TRZ,implying that no charge transfer occurred(Fig.2a).In addition,transient PL decay spectra revealed that the average lifetime(τ)of BrAc-TRZ in THF was below 5 ns no matter in air or after degassed,indicating no long lifetime emission in the above system(Fig.2b).Therefore,it could be concluded that the CT emission of BrAc-TRZ with long lifetime in water was attributed to the formation of nanoaggregates,which can shorten the distance between the donor and acceptor to induce efficient charge transfer.This is also consistent with our assumption.In addition,considering that CT materials showing long lifetimes are related to the long-lived triplet excited(T1)state,which can be quenched by oxygen,it is also proposed that the aggregation process can reduce the adverse effect of oxygen on CT emission[14,24].
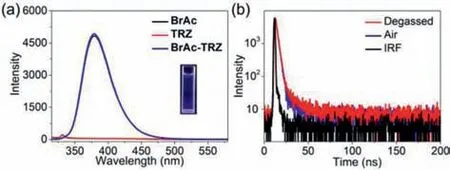
Fig.2.(a)PL spectra of BrAc,TRZ,and BrAc-TRZ in THF(λex = 300 nm).Inset:PL images of BrAc-TRZ in THF.(b)Transient PL decay spectra of BrAc-TRZ at 380 nm in air and after degassed in THF(λex = 300 nm).IRF:Instrument response function.[BrAc]= 250 μmol/L,and[TRZ]= 20 μmol/L.
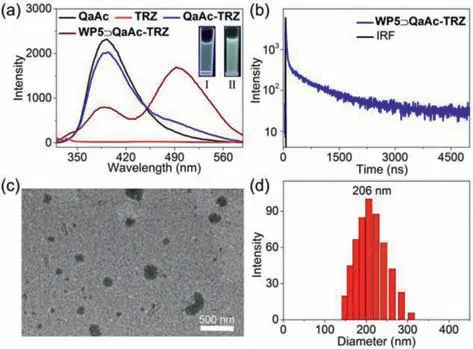
Fig.3.(a)PL spectra of QaAc,TRZ,QaAc-TRZ,and WP5⊃QaAc-TRZ in water(λex = 300 nm).Inset:PL images of QaAc-TRZ(I)and WP5⊃QaAc-TRZ(II)in water.(b)Transient PL decay spectra of WP5⊃QaAc-TRZ at 492 nm in water(λex = 300 nm).IRF:Instrument response function.TEM image(c)and DLS data(d)of WP5⊃QaAc-TRZ nanoassemblies.[WP5]= 31.25 μmol/L,[QaAc]= 250 μmol/L,and[TRZ]= 20 μmol/L.
Considering that the excellent aggregation ability of BrAc-TRZ complex in water was the key point to induce CT emission,it was questioned whether the complex could still exhibit CT emission when the water-solubility of electron donor was increased.To answer this question,another electron donor(QaAc)with better water-solubility while poor aggregation ability was synthesized by quaternization reaction of BrAc(Scheme S1 and Figs.S7-S9 in Supporting information).Then,the aggregate behavior of QaAc-TRZ complex in water was explored.QaAc-TRZ complex showed weak opalescence in water and the optical transmittance at 600 nm was nearly to 98%(Fig.S16 in Supporting information),indicating its very limited aggregation behavior.PL spectra revealed that QaAc-TRZ showed no obvious new emission peak compared with free QaAc(Fig.3a),implying the charge transfer efficiency was low,and the CT emission was quite weak under this condition.
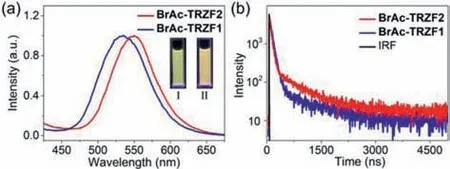
Fig.4.(a)PL spectra of BrAc-TRZF1,BrAc-TRZF2 in water(λex = 300 nm).Inset:PL images of BrAc-TRZF1(Ⅰ)and BrAc-TRZF2(Ⅱ).(b)Transient PL decay spectra of BrAc-TRZF1 at 536 nm and BrAc-TRZF2 at 550 nm in water(λex = 300 nm).IRF:Instrument response function.[BrAc]= 250 μmol/L,[TRZF1]= 20 μmol/L,and[TRZF2]= 20 μmol/L.
It has been widely reported that water-soluble pillar[5]arene(WP5)can bind with the quaternary ammonium group of different guest molecules and induce the formation of various nanostructures[33-35].Hence,it was inferred that a large number of nanoaggregates could form upon the strong host–guest interaction between WP5 and QaAc,resulting in the strong CT emission with long lifetime.To verify this assumption,the binding behavior between WP5 and QaAc was firstly investigated.Since the strong absorption of QaAc below 320 nm would disturb the UVvis titration measurement,amyltrimethylammonium bromide(GM)with the same binding site of QaAc was taken as a model guest molecule.Job’s plot indicated that WP5 and GMcould form host–guest complex with a 1:1 stoichiometry(Fig.S15 in Supporting information).The self-aggregate behavior of WP5⊃QaAc-TRZ assemblies was further explored.Compared with free QaAc-TRZ solution,WP5⊃QaAc-TRZ solution showed notable opalescence as well as obvious Tyndall effect.Moreover,the transmittance of WP5⊃QaAc-TRZ at 600 nm was much lower than that of QaAc-TRZ solution(Fig.S16 in Supporting information).The above phenomena were mainly attributed to the macrocyclic host WP5-promoted aggregation of guest molecule.DLS result and TEM image revealed that WP5⊃QaAc-TRZ complex could form nanoparticles with an average diameter of 206 nm(Figs.3c and d).As presented in Fig.3a,when WP5 was added into the QaAc-TRZ solution,the fluorescence emission intensity at 394 nm decreased significantly,while the emission intensity at 492 nm increased obviously,suggesting the efficient charge transfer between QaAc and TRZ has taken place.As expected,transient PL decay spectra revealed that the WP5⊃QaAc-TRZ assemblies displayed a long lifetime emission(τ= 608.2 ns)(Fig.3b).The above phenomena further proved that the aggregation behavior is a critical factor for the fabrication of CT materials with long lifetime based on supramolecular strategy.
CT materials with long-wavelength emission could be conveniently constructed by modifying the acceptor with substituent groups possessing stronger electron-accepting abilities.TRZF1 and TRZF2 were designed as acceptors due to the high electron affinity of trifluoromethyl(Schemes S2 and S3,Figs.S10 and S11 in Supporting information).Free TRZF1 and TRZF2 were both basically not emissive in water(Fig.S17 in Supporting information).However,when the donor and acceptor co-aggregated into nanoaggregates,strong CT emission could be induced,and the emission colors could range from original green(499 nm)to orange(550 nm)(Fig.4a).The CT emission of BrAc-TRZF1 and BrAc-TRZF2 also exhibited long lifetime,which was 264.2 ns and 413.6 ns,respectively(Fig.4b).In addition,PLQY was measured to be 17.22% and 13.58%,respectively(Fig.S18 in Supporting information).
In summary,novel aggregation-induced charge transfer(CT)emission systems with long lifetime directed by supramolecular strategy have been successfully developed in aqueous phase.For the electron donor(BrAc)with excellent aggregation ability,it can co-aggregate with the electron acceptor(TRZ)to form nanorods in water,leading to the obvious CT emission with long lifetime(τ= 0.92 μs).As for the electron donor(QaAc)with poor aggregation ability,WP5 can be added to promote the aggregation process,resulting in the CT emission with long lifetime(τ= 0.61 μs).The formation of nanoaggregates can not only promote the charge transfer between the electron donor and acceptor,but also can reduce the adverse effects of external oxygen on the CT emission.In addition,CT materials with different emission colors can be conveniently constructed by replacing the acceptor with different electron-accepting capabilities.The present work provides a new strategy for the construction of long lifetime organic luminescent materials,which might have potential applications in the fields of time-resolved fluorescence imaging and photocatalysis.
Declaration of competing interest
The authors report no declarations of interest.
Acknowledgments
We thank Prof.Youxuan Zheng and Dr.Guangping Sun for assistance with data analyses.This work was supported by the National Natural Science Foundation of China(No.21871136),the Natural Science Foundation of Jiangsu Province(No.BK20211179),and the Fundamental Research Funds for the Central Universities(No.NE2019002).
Supplementary materials
Supplementary material associated with this article can be found,in the online version,at doi:10.1016/j.cclet.2021.09.070.
 Chinese Chemical Letters2022年4期
Chinese Chemical Letters2022年4期
- Chinese Chemical Letters的其它文章
- Key progresses of MOE key laboratory of macromolecular synthesis and functionalization in 2020
- Small nanoparticles bring big prospect:The synthesis,modification,photoluminescence and sensing applications of carbon dots
- Cell membrane-coated nanoparticles for immunotherapy
- Diketopyrrolopyrrole-derived organic small molecular dyes for tumor phototheranostics
- Exosome based miRNA delivery strategy for disease treatment
- Recent advances in targeted stimuli-responsive nano-based drug delivery systems combating atherosclerosis
