Nogo-A receptor internalization by cyclic adenosine monophosphate in overcoming axonal growth inhibitors after stroke
Rayudu Gopalakrishna, Charlotte Lin, Mark S. Kindy, William J. Mack
Currently, there are no clinically proven drugs for recovery from stroke and other neuronal injuries such as traumatic brain injury and spinal cord injury. Recovery therapy requires axonal regeneration, which is inhibited by diverse axonal growth inhibitors, such as Nogo-A, myelin-associated glycoprotein (MAG),oligodendrocyte myelin glycoprotein (OMgp),and chondroitin sulfate proteoglycans (CSPGs)(Chaudhry and Filbin, 2007; Carmichael,2010; Schwab and Strittmatter, 2014). A cellsurface receptor for Nogo-A, NgR1, mediates the inhibitory action of not only Nogo-A but also other axonal inhibitors (MAG, OMgp, and CSPGs) as well. Intracellular cyclic adenosine monophosphate (cAMP) overcomes neuronal growth inhibition caused by myelin and improves functional recovery from neuronal injuries (Chaudhry and Filbin, 2007). It may block the actions of axonal growth inhibitors by inducing a transcriptional activation of specific genes. In some cases, cAMP rapidly prevents axonal growth inhibitors from acting through an unknown mechanism (Murray and Shewan,2008).
Recently, we have found that various intracellular cAMP-elevating agents induce a rapid internalization of cell-surface associated NgR1 (Gopalakrishna et al., 2020)Internalization of NgR1 is dependent on the exchange protein directly activated by cAMP(Epac) and is independent of protein kinase A(PKA) (Figure 1). The internalization of NgR1 desensitized neuronal cells to Nogo-66-induced neurite growth cone collapse. We postulate that not only do extracellular cues, such as axonal growth inhibitors, influence neurite outgrowth but neurons also self-regulate their sensitivity to axonal growth inhibitors. Several physiological agents and natural products have neuroprotective and neuritogenic abilities that elevate intracellular cAMP. We speculate that these agents may induce internalization of NgR1 to overcome axonal growth inhibitors and enhance functional recovery after stroke and other neurological injuries.
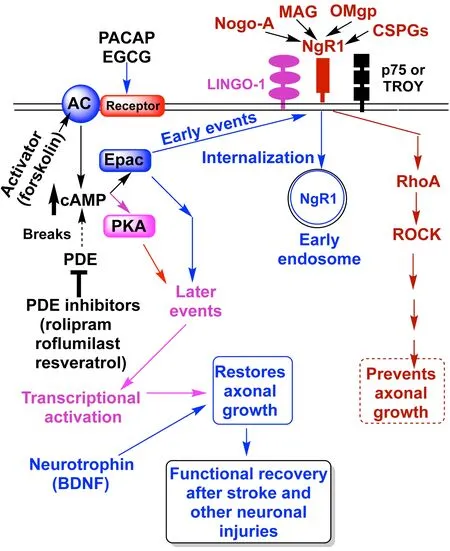
Figure 1|Schematic presentation of the possible role of cAMP in the internalization of NgR1 and in overcoming axonal growth inhibitors for improving functional recovery after neuronal injuries.
Current pharmacological approaches in overcoming axonal growth inhibitors and their limitations:Previous studies have confirmed the functional role of Nogo-A and other axonal growth inhibitors by utilizing blocking-antibodies and knockout studies in experimental animals (Chaudhry and Filbin,2007; Schwab and Strittmatter, 2014). A triple knockout of Nogo-A, MAG, and OMgp promotes higher axonal growth and functional recovery than does Nogo-A knockout alone (Schwab and Strittmatter, 2014). NgR1 elicits cell signaling by associating with transmembrane coreceptor proteins, LINGO-1, and p75 or TROY (Figure 1). This leads to the activation of RhoA and downstream activation of its effector, ROCK,which eventually causes a perturbation in the actin cytoskeletal organization. This can result in the collapse of growth cones and inhibition of neurite outgrowth. Presently, various pharmacological agents are being evaluated in the preclinical and clinical settings for their ability to inhibit NgR1 signaling in order to enhance functional recovery after stroke and spinal cord injury (Carmichael, 2010). These agents include NEP1-40, which is an NgR1 antagonistic peptide, antibodies to block Nogo-A and MAG, inhibitors to prevent RhoA and ROCK from activating, inosine to activate MST3b protein kinase, and daidzein to induce arginase-1 (Carmichael, 2010; Ma et al., 2010).Recently, a monotherapy clinical trial using an anti-MAG neutralizing antibody failed to achieve stroke recovery (Cramer et al., 2017).Instead of blocking one axonal growth inhibitor at a time, by inhibiting the function of NgR1 makes it is possible to block the actions of several axonal growth inhibitors to which NgR1 is a common receptor.
cAMP in overcoming myelin-derived axonal growth inhibitors:Elevation of intracellular cAMP overcomes the axonal growth inhibitors,MAG and myelin, in dorsal root ganglion,cerebellar, cortical, and hippocampal neurons(Chaudhry and Filbin, 2007). Compared to adult neurons, embryonic neurons have higher levels of cAMP, which suppresses axonal growth inhibitors and allows them to promote growth, whereas lower cAMP levels in adult neurons allow these inhibitors to prevent neuronal growth (Murray and Shewan, 2008).Others have shown that cAMP mediates phosphorylation of cAMP-response elementbinding protein via the PKA pathway, leading to transcriptional activation of arginase-1 and other specific genes (Chaudhry and Filbin, 2007). Elevation of arginase activity ultimately leads to an increase in the synthesis of polyamines which play a role in overcoming axonal growth inhibitors. Some effects of cAMP in decreasing the action of axonal growth inhibitors are also PKA-independent. Recent studies have shown that another effector of cAMP, Epac, especially Epac2, plays a crucial role in cAMP-mediated axonal regeneration(Guijarro-Belmar et al., 2021). Some studies showed rapid effects of cAMP in overcoming axonal growth inhibitors by mechanisms that are not clear (Murray and Shewan, 2008).
Cell-surface expression of NgR1 influenced by cAMP and Epac:Our recent studies have shown that treatment of neuronal cells(Neuroscreen-1) with dibutyryl-cAMP, a cellpermeable analog of cAMP, induced a rapid internalization of cell-surface associated NgR1(Gopalakrishna et al., 2020). Within 1 hour after the removal of dibutyryl-cAMP, NgR1 returned to the cell surface, suggesting that this is a reversible process. Furthermore, intracellular cAMP-elevating agents, such as forskolin,which directly activates adenylyl cyclase, and rolipram, which inhibits cyclic nucleotide phosphodiesterase (PDE), also induced this reversible decrease in cell-surface associated NgR1 (Figure 1). Internalization of NgR1 is dependent on Epac and independent of PKA.This rapid cAMP-regulated mechanism may complement the subsequent late mechanism involving cAMP-induced transcriptional activation of specific genes (Figure 1).
Internalization of NgR1 desensitizes neuronal cells to Nogo-A and other axonal growth inhibitors:Our studies showed a correlation between the cAMP-influenced cell-surface expression of NgR1 and the ability of Nogo-66 to induce rapid neurite growth cone collapse (Gopalakrishna et al., 2020). Upon internalization of NgR1, the neuronal cells failed to respond to Nogo-66 to induce growth cone collapse. However, after relocalization of NgR1 to the cell surface by removal of cAMP, Nogo-66’s ability to induce growth cone collapse was restored. Since NgR1 is also a receptor for other axonal growth inhibitors such as MAG, OMgp, and CSPGs, the internalization of NgR1 by cAMP may desensitize neurons to these axonal growth inhibitors as well. These axonal growth inhibitors also bind to other receptors such as NgR2 and NgR3, which are also glycosylphosphatidylinositol-linked cellsurface proteins similar to NgR1 (Schwab and Strittmatter, 2014). These receptors have sequence homology to NgR1 and may be localized to lipid rafts similar to NgR1. If these receptors are also internalized by cAMP, it will cause more effective desensitization of neurons to a variety of axonal growth inhibitors.
Complementary actions of brain-derived neurotrophic factor (BDNF):Since NgR1 is localized to lipid rafts, its coreceptors, p75NTR and LINGO-1, may also be localized to lipid rafts and may be internalized by the cAMP-Epac regulated system. The signals that desensitize neurons to axonal growth inhibitors may be coupled to signals that potentiate the action of neurotrophins andvice versa. For example,the internalization and degradation of p75NTRcould inhibit NgR1, to which p75NTRis an activating coreceptor of, while simultaneously enhancing BDNF-TrkB signaling to which p75NTRis an inhibitor of. Previous studies have shown that simply blocking the axonal growth inhibitors has not resulted in substantial axonal sprouting, particularly in long axonal projection (Carmichael, 2010). Instead, it also requires neurotrophic factors and growthassociated proteins. There is ample evidence that BDNF plays an important role in recovery from stroke and spinal cord injury (Chaudhry and Filbin, 2007). Priming neurons with BDNF decreases MAG and myelin neurite growth inhibition (Chaudhry and Filbin, 2007). BDNF treatment elevates cAMP which may contribute to its efficacy against axonal growth inhibitors.It is not known whether BDNF can induce internalization of NgR1 via elevating cAMP.
Potential therapeutic application of cAMPinduced internalization of NgR1 for recovery from stroke and other neuronal injuries as well as neurodegenerative diseases:Several natural products, such as green tea polyphenols, resveratrol, daidzein, and inosine,have the potential for treating stroke, traumatic brain injury, and spinal cord injury (Raval et al., 2006; Kim et al., 2015; Lopez et al., 2016).Basement membrane protein laminin, which supports neurite outgrowth, overrides the neurite growth inhibitory activity of myelin(David et al., 1995). All of these agents induce elevation of cAMP in the neurons. We postulate that these agents that elevate cAMP can also induce NgR1 internalization.
Recent studies show that by binding to NgR1,Nogo-A plays a crucial role in the pathogenesis of Alzheimer’s disease (AD) (Karlsson et al.,2013). Although injection of NgR(310)ecto-Fc reduces β-amyloid plaque load in the brain,neuronal overexpression of NgR1 impairs spatial cognition tasks (Karlsson et al., 2013).Therefore, it is possible that the internalization of NgR1 by cAMP may contribute to the previously reported protective role of cAMP towards AD (Nabavi et al., 2019). There is a need to address this regulation in relation to AD, as several inhibitors for PDE isoenzymes have been reported to show encouraging results for the treatment of AD (Nabavi et al.,2019).
Adenylyl cyclase and PDE have several isoenzymes with different localization within the neurons. This may lead to localized production of cAMP and altered expression of NgR1 on specific sites on the neuronal surface, which may result in a decrease or increase in axonal growth in a specific direction. Unlike this physiological regulation,continuous pharmacological global activation of cAMP pathways can cause cognitive decline,hyperexcitability, and hyperalgesia (Song et al., 2006). Nevertheless, it is possible to locally stimulate the cAMP-Epac system necessary for NgR1 internalization by selecting the appropriate natural product or isoenzymespecific PDE inhibitor. With this approach,neuroregeneration may be achieved while minimizing neurotoxicity.
In summation, neurons are not only influenced by extracellular cues such as axonal growth inhibitors, but are also able to self-regulate their sensitivity to axonal growth inhibitors,which is influenced by localized production of cAMP in the neurons. This physiological process may be pharmacologically exploited to develop drugs or utilize natural products to overcome axonal growth inhibitors and enhance functional recovery after stroke and other neurological injuries such as traumatic brain injury and spinal cord injury.
This work was supported by NINDS, NIH
NS116720 (to RG and WJM).
Rayudu Gopalakrishna*, Charlotte Lin,Mark S. Kindy, William J. Mack
Department of Integrative Anatomical Sciences,Keck School of Medicine, University of Southern California, Los Angeles, CA, USA (Gopalakrishna R,Lin C)
Department of Neurological Surgery, Keck School of Medicine, University of Southern California, Los Angeles, CA, USA (Mack WJ)Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa,FL, USA (Kindy MS)
*Correspondence to:Rayudu Gopalakrishna, PhD,rgopalak@usc.edu.
https://orcid.org/0000-0002-0398-3166(Rayudu Gopalakrishna)
Date of submission:January 11, 2021
Date of decision:January 26, 2021
Date of acceptance:March 17, 2021
Date of web publication:June 7, 2021
https://doi.org/10.4103/1673-5374.314298
How to cite this article:Gopalakrishna R, Lin C,Kindy MS, Mack WJ (2022) Nogo-A receptor internalization by cyclic adenosine monophosphate in overcoming axonal growth inhibitors after stroke. Neural Regen Res 17(1):91-92.
Copyright license agreement:The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check:Checked twice by iThenticate.
Peer review:Externally peer reviewed.
Open access statement:This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
Open peer reviewer:Ulf Strauß, Charite Universitatsmedizin Berlin, Germany.
Additional file:Open peer review report 1.
- 中国神经再生研究(英文版)的其它文章
- Genes for RNA-binding proteins involved in neuralspecific functions and diseases are downregulated in Rubinstein-Taybi iNeurons
- Research advances on how metformin improves memory impairment in “chemobrain”
- Dendritic spine density changes and homeostatic synaptic scaling: a meta-analysis of animal studies
- Optogenetic activation of intracellular signaling based on light-inducible protein-protein homo-interactions
- Presenilin mutations and their impact on neuronal differentiation in Alzheimer’s disease
- Growth differentiation factor 5: a neurotrophic factor with neuroprotective potential in Parkinson’s disease

