Hyperbaric oxygen preconditioning improves postoperative cognitive dysfunction by reducing oxidant stress and inf l ammation
Zhi-xin Gao, Jin Rao, Yuan-hai Li
Department of Anesthesiology, First Af filiated Hospital of Anhui Medical University, Hefei, Anhui Province, China
Hyperbaric oxygen preconditioning improves postoperative cognitive dysfunction by reducing oxidant stress and inf l ammation
Zhi-xin Gao, Jin Rao, Yuan-hai Li*
Department of Anesthesiology, First Af filiated Hospital of Anhui Medical University, Hefei, Anhui Province, China
Postoperative cognitive dysfunction is a crucial public health issue that has been increasingly studied in ef f orts to reduce symptoms or prevent its occurrence. However, ef f ective advances remain lacking. Hyperbaric oxygen preconditioning has proved to protect vital organs, such as the heart, liver, and brain. Recently, it has been introduced and widely studied in the prevention of postoperative cognitive dysfunction, with promising results. However, the neuroprotective mechanisms underlying this phenomenon remain controversial. This review summarizes and highlights the definition and application of hyperbaric oxygen preconditioning, the perniciousness and pathogenetic mechanism underlying postoperative cognitive dysfunction, and the ef f ects that hyperbaric oxygen preconditioning has on postoperative cognitive dysfunction. Finally, we conclude that hyperbaric oxygen preconditioning is an ef f ective and feasible method to prevent, alleviate, and improve postoperative cognitive dysfunction, and that its mechanism of action is very complex, involving the stimulation of endogenous antioxidant and anti-inf l ammation defense systems.
nerve regeneration; brain injury; hyperbaric oxygenation; preconditioning; antioxidants; antiinf l ammation; reactive oxygen species; oxidant stress; inf l ammation; protection; post-operation; cognitive dysfunction; neural regeneration
Introduction
Postoperative cognitive dysfunction (POCD) is a complication of surgery that is widely considered an important clinical problem, particularly in elderly patients (Shoair et al., 2015). However, the pathophysiology underlying POCD is fairly complex, involving numerous mechanisms including oxidant stress, inflammation, and apoptosis (Eckenhoff et al., 2004; Dong et al., 2009; Thom, 2009; Cao et al., 2012; Wilson et al., 2013). Over the past several decades, researchers have explored a wide array of methods for improving POCD, including hyperbaric oxygen preconditioning (HBOPC). HBOPC is one of the most economical, simple, safe, and ef f ective strategies among all the possible choices (Zhu et al. 2016). Indeed, studies have successfully utilized HBOPC to improve cognitive dysfunction (Alex et al., 2005; Peng et al., 2010; Sun et al., 2014). The purpose of this narrative review is to summarize and discuss the literature concerning HBOPC and POCD, with an emphasis on the evidence for a role of HBOPC in treating patients undergoing POCD. The review is organized into the following sections: introduction of HBOPC, mechanisms underlying POCD, and the effect of HBOPC on POCD (Figure 1).
HBOPC
Def i nition of HBOPC
During HBO treatment, patients usually inhale pure oxygen (100%) at pressures greater than the atmospheric pressure in a steel vessel (Löndahl, 2012), which increases both the dissolved oxygen and the partial pressure of oxygen in blood plasma (Tibbles and Edelsberg, 1996). Consequently, a large amount of oxygen-dependent reactions and signaling pathways are enhanced (Babchin et al., 2011).
Application of HBOPC
Normobaric oxygen and various levels of HBO have been widely utilized therapeutic agents, and Valenzuela pioneered the application of pure oxygen (as high as 2 MPa) in clinical research (Edwards, 2010). The use of HBO as an adjuvant treatment for a number of medical conditions has been widely supported by the experience of experts in hyperbaric medicine and the scientif i c literature in areas such as traumatic brain injury (Hu et al., 2016; Zhou et al., 2016a, b) complex refractory wounds (Morykwas and Argenta, 1996), cerebral infarction (Tian, 2015), and radiation-tissue injury (Kindwall and Hunt, 1995; Kindwall and Wheland, 1999). Along with the development of medicines, disease prevention has increasingly become recognized as important. Pre-conditioning is a type of primary prevention that activates endogenous protective mechanisms, which can reduce the risk of morphologic and functional sequelae. The preconditioning state is typically def i ned by the response to a sublethal stimulus that extends beyond its presence in the system. This response significantly lessens the level of signal cascades for stress-activated and stress-reactive proteins, which subsequently shows a protective effect for cells. Recently, HBO has become recognized as an ef f ective preconditioning method for reducing mental and cellular stresses, especially in regimented sessions of moderate HBO (Nie et al., 2006; Li et al., 2008). In the clinic, however, HBOPC has had only minimal impact before surgery, and no role in the surgery or post-surgical care of patients (Allen et al., 2014). HBO protocols are performed at 2.0—2.5 atmosphere absolute (ATA) oxygen partial pressures, and usually only applied for one or a few days. The physical adaptations in response to alterations in atmospheric oxygen appear to extend not only to survival, but also a preconditioned state. Similar to ischemic and stress preconditioning, many dif f erent paradigms have been used to demonstrate that either rapid or delayed tolerance is affected by the HBO (Stetler et al., 2014). To achieve the best outcome using HBOPC, it requires a certain O2concentration and high pressure. When air (20% oxygen) rafter than 100% O2was infused into the hyperbaric chamber, the tolerance was negated, demonstrating the need for high O2concentration in the hyperbaric preconditioned state (Wada et al., 2001). Additionally, Kocaoğullar et al. (2004) compared normobaric oxygen with HBO treatment of rats with cerebral vasospasm after subarachnoid hemorrhage and found that normobaric oxygen was less ef f ective in ameliorating neurological deficits associated with the central nervous system. Many experiments have shown that HBOPC can protect against subsequent multi-organ injury to the brain, heart, or liver (Alex et al., 2005; Yu et al., 2005; Qin et al., 2008). Previous studies suggest that preconditioning with pressures of 2 ATA 3–5 sessions/every other day was effective in inducing tolerance against global ischemia in gerbils (Wad et al., 1996; Wada et al., 2001). Cheng et al. (2011) reported that 2.5 ATA preconditioning, 1 hour daily for 5 days, protected against subsequent global ischemic injury in rats (Cheng et al., 2011). Similarly, pretreatment with HBO has been found both to improve the degree and accelerate the rate of neurologic recovery. Additionally, long-term HBOPC paradigms have been shown to be more ef f ective at establishing tolerance than are acute paradigms (Xiong et al., 2000; Dong et al., 2002; Nie et al., 2006; Liu et al., 2012). Animal studies of ischemia/reperfusion (I/R) injury in the myocardium have indicated that HBO preconditioning can lead to ischemia tolerance, resulting in protection against myocardial ischemia (Kim et al., 2001). In addition to these experimental studies, clinical studies have demonstrated that preconditioning patients who have coronary artery disease with HBO before on-pump cardiopulmonary bypass or coronary artery graTh bypass were in a position improved myocardial function, and reduced myocardial injury, the duration of staying in the intensive care unit, blood loss, postoperative complications, and cost (Yogaratnam et al., 2010; Li et al., 2011). Karu et al. (2010) indicated that exposure to hyperoxia for a limited time before ischemia induced a mild oxidative stress and resulted in an (ischemic) preconditioning-like effect in the myocardium, which protected the heart from subsequent injury. Yu et al. (2005) performed an experimental study in rats and reported preconditioning with single-dose HBO (90 minutes) protects the rat liver against subsequent I/R injury. Ren et al. (2008) similarly reported that HBO preconditioning increased the number of new cells and the density of microcirculation in the regenerating liver. Therefore, HBO preconditioning is an encouraging and feasible therapeutic strategy for protecting organs from the subsequent lethal stimulus. The ef f ect andmechanism of HBOPC on POCD will be described below.
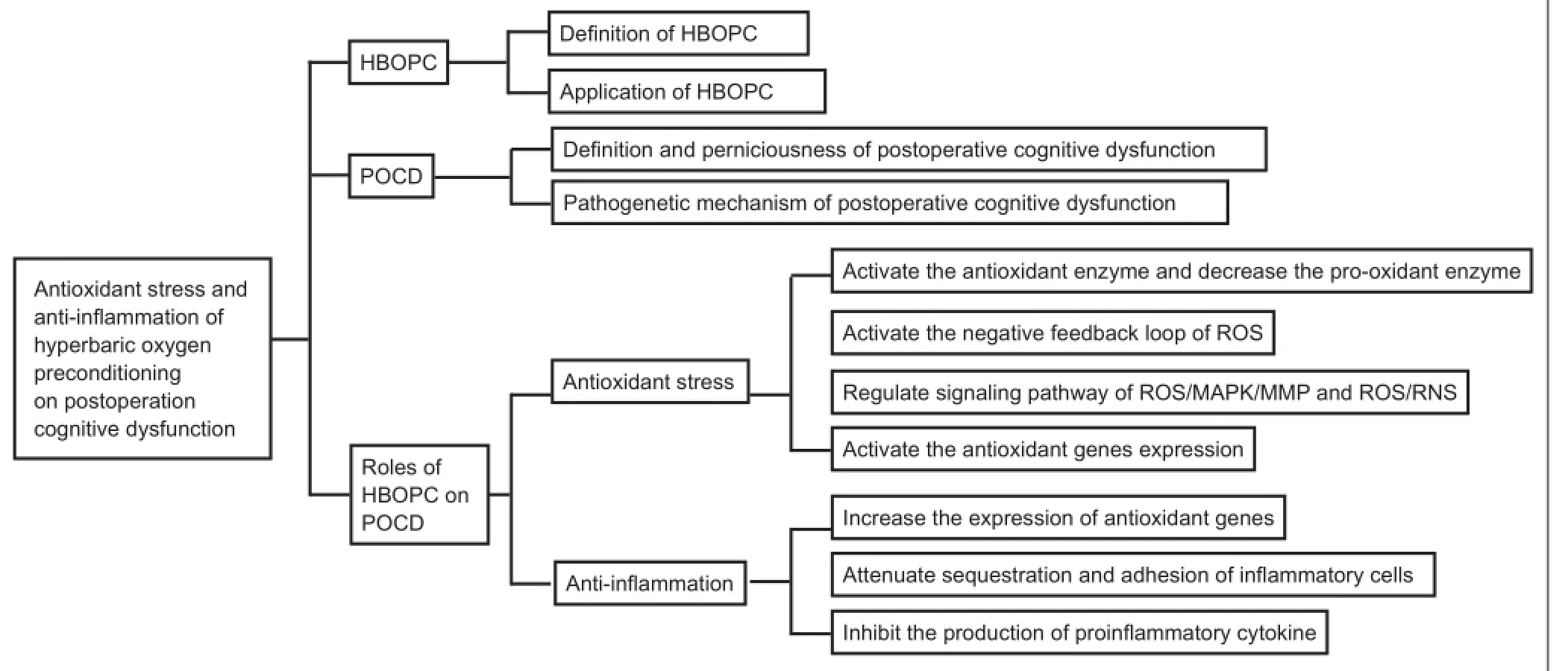
Figure 1 Summary of article structure.
POCD
Def i nition and perniciousness of POCD
Every year, numerous people undergo surgery hoping that the operation will lighten symptoms, heal diseases, and improve quality of life (Berger et al., 2015). Although there is much interest in, and controversy about, the mechanism and treatment of POCD, there is little doubt that cognitive decline after surgery (especially in the elderly population) is a critical clinical issue that shows a high morbidity. POCD is def i ned as an impairment in mental processes of perception, memory, and information processing that occurs in the postoperative period (Hanning, 2005), and which is diagnosed by specif i c tests after exclusion of other neurological complications. Both cardiac surgery and non-cardiac surgery are associated with the cognitive dysfunction after hospital discharge from 30% to 50% of patients (Newman et al., 2001; McDonagh et al., 2010; Selnes et al., 2012). One study reported the incidence of cognitive decline to be 53% at discharge, 36% at 6 weeks after discharge, 24% at 6 months after discharge, and 42% at 5 years after coronary-artery bypass graThing (Newman et al., 2001). Shoair et al. (2015) showed that 15.9% of older adult patients developed POCD within 3 months after elective major non-cardiac surgery. Other studies have found that POCD is associated with poor shortterm and long-term outcomes, including an increased risk of disability, increased expenditure on hospitalization, inability to cope independently, reduced quality of life, and possible permanent dementia (Hovens et al., 2014; Shoair et al., 2015). Patients with POCD are at an increased risk of death in the fi rst year after surgery and the elderly (aged 60 years or older) are at a signif i cant risk for long-term cognitive problems (Moller et al., 1998; Monk et al., 2008; Avidan et al., 2009; Steinmetz et al., 2009).
Pathogenic mechanism of POCD
There is strong standpoint that cognitive decline experienced by elderly patients is directly mediated by neuro-inflammation and the enhancement of amyloid-beta oligomerization after surgery and general anesthesia (Bedford, 1955; Eckenhof f et al., 2004; Müller et al., 2004; Newman et al., 2007; Dong et al., 2009; Cao et al., 2012). At the same time, some results suggest that surgery results in neuro-inflammation and cognitive impairment, and that anesthesia might not be an essential influential factor for these effects (Zhang et al., 2015; Zhou et al., 2015). Despite these controversies, existing evidence has conf i rmed that neuro-inf l ammatory response to operative stress is an independent risk factor associated with the development of POCD (Wan et al., 2007; Barrientos et al., 2012; Hovens et al., 2014; Lu et al., 2015; Ma et al., 2015; Zheng et al., 2015). Amyloid beta, the peptide associated with Alzheimer’s disease, was also detected in the serum of POCD patients. Another important player is acetylcholine, which has signif i cant roles in memory, learning, and attention (Hshieh et al., 2008). The most likely mechanism underlying POCD is a central cholinergic def i ciency caused by the deregulation of cholinergic anti-inflammatory pathways, which results in increased inflammation (Inouye, 2006; Androsova et al., 2015). Many scholars have highlighted the importance of the cholinergic ref l ex in resolving the inf l ammatory pathogenesis of several diseases, including sepsis (Borovikova et al., 2000), rheumatoid arthritis (van Maanen et al., 2009), and colitis (Ghia et al., 2007). Researchers have reported that pro-inf l ammatory cytokines, including interleukin (IL)-1 and tumor necrosis factor-alpha (TNF-α), play key roles in mediating surgery-induced neuro-inf l ammation and subsequent cognitive decline (Cibelli et al., 2010; Terrando et al., 2010). Results reveal that surgery, not propofol-based anesthesia, induces neuro-inflammation and the impairment of learning and memory. Pyrrolidine dithiocarbamate attenuates these ef f ects by inhibiting nuclear factor-kappa B activation and downstream matrix metallopeptidase 9 activity (Zhao et al., 2013; Zhang et al., 2014). Other studies have demonstrated that peripheral surgery af f ects the blood-brain barrier through the release of TNF-α. This promotes macrophage migrating into the hippocampus (Rudolph et al., 2008; Terrando et al., 2011; Vacas et al., 2013). Activation of α7 nicotinic acetylcholine receptors trigger an endogenous inflammation-resolving pathway that has been proven to be useful in blocking TNF-α-induced nuclear factor-kappa B activation and cognitive decline after surgery (Terrando et al., 2011). Jiang et al. (2015) suggested that IL-6 has a crucial role in POCD, and that IL-6R antagonists may serve as novel agents for its prevention or treatment. Chen et al. (2015) also demonstrated that dexmedetomidine reduces the incidence of POCD by suppressing inf l ammation in aged patients.
Roles of HBOPC on POCD
Cognitive decline after surgery includes deterioration in cognition, disturbance in attention, and reduced awareness of the environment. In light of recent clinical developments, HBO preconditioning has been shown to protect against focal and global cerebral ischemia as well as traumatic brain injury (Cheng et al., 2011; Yan et al., 2011; Lin et al., 2012). Furthermore, HBO preconditioning can promote both cerebral-protective and cardiac-protective effects, as determined by biochemical markers of neuronal and myocardial injury and clinical consequences in patients experiencing on-pump coronary artery bypass-graTh surgery (Yogaratnam et al., 2010; Li et al., 2011). Additionally, Alex et al. (2005) indicated that while pretreatment with 2.4-ATA HBO can reduce neuropsychometric dysfunction and modulate the inflammatory response that occurs after cardiopulmonary bypass. In basic studies, Sun et al. (2014) indicated that HBO preconditioning can significantly lessen cognitive impairment, and that it can be considered responsible for decreases in pro-inflammatory (either systemic or central) cytokines and caspase-3 activity. Similarly, Peng et al. (2010) indicated that continuous HBOPC could lead to an apparent improvement in impairments of associative learning and spatial memory. The study also showed that HBOPC has an ef f ective anxiolytic effect and provided experimental evidence that supports the idea that HBOPC is useful for treating some affective disorders, including post-traumatic stress disorder. Allthese results showed that HBO preconditioning is a safe and feasible procedure that can attenuate cognitive impairments after surgery. Additionally, they show that it is associated with anti-oxidants stress, anti-inflammation, and anti-apoptosis, as well as increased regional cerebral blood fl ow distribution and improvement of blood-brain barrier integrity (Li et al., 2007; Micarelli et al., 2013; Tian et al., 2013; Sun et al., 2014). Among these phenomena, the anti-oxidative stress and anti-inf l ammatory action of HBOPC are considered two crucial mechanisms with respect to easing POCD.
Antioxidant stress
(1) Anti-oxidative stress is achieved through activation of antioxidant enzymes and the decrease of pro-oxidant enzymes. HBO can elevate the partial pressure of oxygen and enhance the cellular tolerance against harmful stimuli by inducing the expression of cell protective proteins (Thom, 2009). Several studies have shown that the endogenous antioxidant-defense system becomes active in parallel with the development of HBOPC-induced neuroprotection (Nie et al., 2006; Thom, 2009; Huang et al., 2014). Numerous studies have shown that repeated preconditioning with HBO, but not normal conditions, can protect the spinal cord against I/R damage (Nie et al., 2006; Lu et al., 2012; Huang et al., 2014). These results have been attributed to the protective effect of upregulated HO-1, and the activity of catalase and superoxide dismutase (SOD), which are triggered by HBO preconditioning (Li et al., 2007). Further investigations have shown that when dimethylthiourea, a potent free radical scavenger, was administered before each session of HBO treatment, the HBO-induced catalase and SOD activities were abolished. Similarly, when the catalase inhibitor 3-amino-1,2,4-triazole or dimethylthiourea was administered before spinal cord ischemia, the ischemic tolerance induced by HBOPC was attenuated (Nie et al., 2006; Huang et al., 2014). HBOPC was shown to decrease mortality rate, improve neurological recovery, lessen neuronal injury, reduce the level of malondialdehyde, and increase antioxidant activity of catalase and SOD (Li et al., 2008). Repeated HBO exposure supplies protection against oxygen toxicity in the central nervous system and this may be attributed to the decreased enzymatic activity of the antioxidant system and reduced levels of peroxynitrite, primarily in the hippocampus (Arieli et al., 2014). In related work, Peng et al. (2010) suggested that HBOPC is beneficial for the improvement of anxiety-like behavior and cognitive impairments arising from a single prolonged exposure to stress, and that this effect might be associated with inhibition of neuronal apoptosis via upregulation of thioredoxin reductase in stressed rats. These results confi rmed that HBO preconditioning can induce upregulation of antioxidant-enzyme activity, leading to the generation of tolerance against I/R injury in the brain (Li et al., 2008). Expression of antioxidant enzymes, including Cu/Zn-superoxide dismutase, catalase, and glutathione peroxidase, have been shown to be enhanced by HBOPC (Kim et al., 2001; Li et al., 2008). Additionally, levels of pro-oxidant enzymes such as inducible nitric oxide synthase and gp91-phox have been shown to significantly decrease after HBOPC (Zhang and Gould, 2014). However, few animal experiments reported that in the hippocampus of preconditioned rats, the activities of glutathione reductase and glucose-6-phosphate dehydrogenase were substantially decreased, while the activity of glutathione peroxidase was greatly increased (Arieli et al., 2014).
(2) Anti-oxidative stress is also achieved through the reactive oxygen species negative feedback loop. Transiently increased reactive oxygen species (ROS) levels activate a negative feedback loop, which leads to downregulation of oxidant enzymes and upregulation of antioxidant enzymes, thereby limiting subsequent higher levels of reactive species of oxygen and nitrogen production (Zhang and Gould, 2014). Furthermore, these results also indicate that ROS-related enzymes, including inducible nitric oxide synthase and nicotinamide adenine dinucleotide phosphate oxidase, rafter than the ROS itself, can be crucial therapeutic targets for inhibiting oxidative stress.
(3) Anti-oxidative stress can also result from regulation of the ROS/mitogen-activated protein kinase (MAPK)/matrix metalloproteinase (MMP) and ROS/reactive nitrogen species (RNS) signaling pathways. HBO repairs ischemic wounds by decreasing the phosphorylation of extracellular signal-regulated kinases 1/2, c-Jun N-terminal kinase, and c-Jun, which suggests that mitogen-activated protein kinase is downregulated. All these results demonstrate that HBO acts via the ROS/MAPK/MMP signaling pathway to decrease neurodegeneration and ameliorate healing of ischemic wounds (Zhang and Gould, 2014). For example, the level of oxidative stress in ischemic wound tissue will be highly enhanced when the effect of HBO is completely blocked (Zhang and Gould, 2014). The oxidized N-linoleoyl tyrosine marker is sufficiently sensitive to detect oxidative stress imposed on cells and cell-free systems and to react selectively with the various ROS/RNS that are induced as a result. Thus, it is very useful for characterizing oxidative stress in general, and possibly also in oxidative stress-associated diseases (Szuchman et al., 2006). In one ingenious and delicate experiment, the oxidized N-linoleoyl tyrosine marker and the protein products of advanced oxidation were analyzed to demonstrate that preconditioning with multiple short HBO exposures followed by a long exposure will lead to a decrease in oxidative adducts, reaching even lower levels than that which initially existed in the control group. Endogenous antioxidant defense mechanisms induced by HBOPC play an important role in the formation of tolerance against long HBO exposure (Palzur et al., 2011).
(4) Antioxidant gene expression is another factor that increases anti-oxidative stress. Ferrer et al. (2007) showed that HBO can also act to activate antioxidant genes in human tissue. Endothelial cells are sensitive to high pressure oxygen exposure, which easily triggers the expression of many Nrf2-regulated antioxidant genes and molecular chaperones (Godman et al., 2010a, b). Additionally, the expression of antioxidant genes also occurs in other cells and tissues activated by HBO (Padgaonkar et al., 1997; Dennog et al., 1999; Rothfuss et al.,2001; Verma et al., 2015).
All these observations serve to illustrate central role that anti-oxidative stress has as a mechanism underlying HBO treatment. The fi ndings strongly suggest that HBO preconditioning is a potentially promising treatment for preventing the development of cognitive impairment after surgery.
Anti-inf l ammation
Despite advances in surgical techniques, the incidence of neuropsychometric dysfunction after surgery is high. Previous studies have demonstrated that the systemic and central inflammatory response plays a critical role in the development of postoperative cognitive impairment (Cibelli et al., 2010; Fidalgo et al., 2011; Barrientos et al., 2012; He et al., 2012; Hovens et al., 2014; Sun et al., 2014), and HBO treatment can improve POCD by attenuating inf l ammatory responses (Alex et al., 2005; Daniel et al., 2011; Lin et al., 2012a, b).
(1) Inf l ammatory responses can be reduced by increased expression of antioxidant genes. ROS plays a signif i cant role in transduction cascades and pathways (Allen and Balin, 1989; Maulik, 2002; Ushio-Fukai and Alexander, 2004; Calabrese et al., 2007). HBO-related anti-inf l ammatory action can be partially induced through increased expression of antioxidant genes and other cellular defense genes via non-cytotoxic oxidative stimuli (Godman et al., 2010a, b; Matsunami et al., 2010, 2011; He et al., 2011; Simsek et al., 2011).
(2) Attenuation of inflammatory cells sequestration and adhesion can also reduce inflammatory responses. Tissue inf l ammation can occur when circulating neutrophils adhere to vascular endothelium through interactions with B2-integrins. However, neutrophil B2-integrin function is inhibited by exposure to HBO (Thom et al., 2008; Thom, 2009). In some cases, when animals or humans are exposed to HBO (2.8—3.0 ATA), the ability of circulating neutrophils to adhere to target tissues is temporarily inhibited, and inf l ammation is subsequently reduced (Thom, 1993; Zamboni et al., 1993; Thom et al., 1997; Labrouche et al., 1999; Kalns et al., 2002). In ameliorating I/R injuries, HBO is notably superior to B2-integrin monoclonal antibodies because it does not compromise the immune system (Mileski et al., 1990; Buras et al., 2006). At the same time, HBO exposure also leads to the impaired synthesis of cyclic guanosine monophosphate (Chen et al., 1996), which consequently reduces the activity of the neutrophil specific adhesion molecule CD18 (Malik and Lo, 1996). In the meantime, intercellular adhesion molecule 1, which is a marker of acute and chronic inf l ammation, acts as the receptor of leukocyte function associated antigen-1 (CD11a/CDx18). This antigen is expressed on various inf l ammatory cells, including neutrophils, monocytes, and lymphocytes. For example, some studies have indicated that levels of intercellular adhesion molecule 1 are downregulated by HBO (Buras et al., 2000). By downregulating the accumulation of these cellular adhesion molecules, neutrophil sequestration and adhesion is attenuated, which reduced inflammation (Zamboni et al., 1993).
(3) Inflammation is also reduced through the inhibition of pro-inflammatory cytokine production. The production of pro-inflammatory cytokines by monocyte-macrophages is inhibited after exposure to HBO. Pro-inflammatory cytokine-regulating adhesion molecules and enhancement of heme oxygenase-1 and heat shock proteins (e.g., heat shock protein 70) (Rothfuss et al., 2001) are all mechanisms considered to play important roles in the anti-inf l ammatory ef f ects of HBO. Compared with cells isolated from HBO-exposed rats (Lahat et al., 1995), those isolated from rats that were not previously exposed to HBO released more TNF-α. Additionally, in endotoxic rats, HBO treatment inhibits the endotoxin lipopolysaccharide-induced pro-inflammatory cytokines in monocytes and macrophages (Benson et al., 2003). Niu et al. (2007) reported that pyrogenic fever is prevented and suppressed by HBO via decreased overproduction of circulating TNF-α and hypothalamic prostaglandin E2. Similarly, several studies have demonstrated that the rise of TNF-α (Huang et al., 2006) and IL-6 (Niu et al., 2009) induced by lipopolysaccharide administration also can be signif i cantly decreased by HBO pretreatment. Further, HBO decreases the release of IL-1B and TNF-a in monocytes and macrophages derived from human blood (Benson et al., 2003). HBO exposure is also indicated to lessen cytokine induction (Yamashita and Yamashita, 2000; Kang et al., 2014). Additionally, HBO pretreatment inhibits activated inf l ammation and gliosis, and stimulates angiogenesis, neurogenesis, and production of IL-10. This consequently improves outcomes of traumatic brain injury (Lin et al., 2012). Pretreatment with HBO is benef i cial for recovery after brain surgery, and can enhance expression of osteopontin, which reduces the expression of IL-1B/nuclear factor-κ-gene binding and expansive protein kinase B (Akt) (Hu et al., 2015).
Discussion
As presented here, there is substantial evidence for a central involvement of oxidant stress and inflammatory response in POCD. In addition, numerous basic and clinical studies have demonstrated that HBOPC has a protective ef f ect on POCD by reducing the detrimental inf l ammation and balancing the oxygen free radicals. The mechanism underlying preconditioning is not yet fully understood. Many researchers have suggested that HBOPC can alleviate cognitive impairment after surgery (Sun et al., 2014) and subsequently decrease the density of apoptotic cells and further recovery of nerve function (Wang et al., 2009; Lu et al., 2013). The mechanism underlying the protection might involve the reduction of systemic and hippocampal pro-inf l ammatory cytokines (Cheng et al., 2011) and upregulation of heat-shock protein 32 (Nie et al., 2006). Based on the experimental evidence, the prospect for using HBOPC to reduce cognitive impairment after surgery is bright. However, the number of relevant clinical studies remains low at present. Therefore, further studies are critical for understanding the fundamental mechanisms of this phenomenon and to explore the optimal parameters for pretreatment.
Author contributions:ZXG prepared the paper. JR prepared the reference format. YHL presented the idea of the review. All authors approved the fi nal version of the paper.
Conf l icts of interest:None declared.
Plagiarism check:This paper was screened twice using CrossCheck to verify originality before publication.
Peer review:This paper was double-blinded and stringently reviewed by international expert reviewers.
Alex J, Laden G, Cale AR, Bennett S, Flowers K, Madden L, Gardiner E, McCollum PT, Grif fin SC (2005) Pretreatment with hyperbaric oxygen and its ef f ect on neuropsychometric dysfunction and systemic inf l ammatory response after cardiopulmonary bypass: a prospective randomized double-blind trial. J Thorac Cardiovasc Surg 130:1623-1630.
Allen M, Golembe E, Gorenstein S, Butler G (2014) Protective ef f ects of hyperbaric oxygen therapy (HBO2) in cardiac care-A proposal to conduct a study into the effects of hyperbaric pre-conditioning in elective coronary artery bypass graTh surgery (CABG). Undersea Hyperb Med 42:107-114.
Allen R, Balin AK (1989) Oxidative influence on development and dif f erentiation: an overview of a free radical theory of development. Free Radic Biol Med 6:631-661.
Androsova G, Krause R, Winterer G, Schneider R (2015) Biomarkers of postoperative delirium and cognitive dysfunction. Front Aging Neurosci 7:112.
Arieli Y, Kotler D, Eynan M, Hochman A (2014) Hyperbaric oxygen preconditioning protects rats against CNS oxygen toxicity. Respir Physiol Neurobiol 197:29-35.
Avidan MS, Searleman AC, Storandt M, Barnett K, Vannucci A, Saager L, Xiong C, Grant EA, Kaiser D, Morris JC (2009) Long-term cognitive decline in older subjects was not attributable to non-cardiac surgery or major illness. Anesthesiology 111:964.
Babchin A, Levich E, Sivashinsky G (2011) Osmotic phenomena in application for hyperbaric oxygen treatment. Colloids Surf B Biointerfaces 83:128-132.
Barrientos RM, Hein AM, Frank MG, Watkins LR, Maier SF (2012) Intracisternal interleukin-1 receptor antagonist prevents postoperative cognitive decline and neuroinflammatory response in aged rats. J Neurosci 32:14641-14648.
Bedford P (1955) Adverse cerebral ef f ects of anaesthesia on old people. Lancet 266:259-264.
Benson R, Minter L, Osborne B, Granowitz E (2003) Hyperbaric oxygen inhibits stimulus-induced proinflammatory cytokine synthesis by human blood-derived monocyte-macrophages. Clin Exp Immunol 134:57-62.
Berger M, Nadler JW, Browndyke J, Terrando N, Ponnusamy V, Cohen HJ, Whitson HE, Mathew JP (2015) Postoperative cognitive dysfunction: minding the gaps in our knowledge of a common postoperative complication in the elderly. Anesthesiol Clin 33:517-550.
Borovikova LV, Ivanova S, Zhang M, Yang H, Botchkina GI, Watkins LR, Wang H, Abumrad N, Eaton JW, Tracey KJ (2000) Vagus nerve stimulation attenuates the systemic inf l ammatory response to endotoxin. Nature 405:458-462.
Buras JA, Stahl GL, Svoboda KK, Reenstra WR (2000) Hyperbaric oxygen downregulates ICAM-1 expression induced by hypoxia and hypoglycemia: the role of NOS. Am J Physiol Cell Physiol 278:C292-C302.
Buras JA, Holt D, Orlow D, Belikof f B, Pavlides S, Reenstra WR (2006) Hyperbaric oxygen protects from sepsis mortality via an interleukin-10—dependent mechanism. Crit Care Med 34:2624-2629.
Calabrese V, Mancuso C, Calvani M, Rizzarelli E, Butterf i eld DA, Stella AMG (2007) Nitric oxide in the central nervous system: neuroprotection versus neurotoxicity. Nat Neurosci 8:766-775.
Cao L, Li L, Lin D, Zuo Z (2012) Isoflurane induces learning impairment that is mediated by interleukin 1B in rodents. PLoS One 7:e51431.
Chen Q, Banick PD, Thom SR (1996) Functional inhibition of rat polymorphonuclear leukocyte B2 integrins by hyperbaric oxygen is associated with impaired cGMP synthesis. J Pharmacol Exp Ther 276:929-933.
Cheng O, Ostrowski RP, Wu B, Liu W, Chen C, Zhang JH (2011) Cyclooxygenase-2 mediates hyperbaric oxygen preconditioning in the rat model of transient global cerebral ischemia. Stroke 42:484-490.
Cibelli M, Fidalgo AR, Terrando N, Ma D, Monaco C, Feldmann M, Takata M, Lever IJ, Nanchahal J, Fanselow MS (2010) Role of interleukin-1B in postoperative cognitive dysfunction. Ann Neurol 68:360-368.
Daniel RAF, Cardoso VK, Góis Jr E, Parra RS, Garcia SB, Rocha JJRd, Féres O (2011) Ef f ect of hyperbaric oxygen therapy on the intestinal ischemia reperfusion injury. Acta Cir Bras 26:463-469.
Dennog C, Radermacher P, Barnett YA, Speit G (1999) Antioxidant status in humans after exposure to hyperbaric oxygen. Mutat Res 428:83-89.
Dong H, Xiong L, Zhu Z, Chen S, Hou L, Sakabe T (2002) Preconditioning with hyperbaric oxygen and hyperoxia induces tolerance against spinal cord ischemia in rabbits. Anesthesiology 96:907-912.
Dong Y, Zhang G, Zhang B, Moir RD, Xia W, Marcantonio ER, Culley DJ, Crosby G, Tanzi RE, Xie Z (2009) The common inhalational anesthetic sevoflurane induces apoptosis and increases B-amyloid protein levels. Arch Neurol 66:620-631.
Eckenhof f RG, Johansson JS, Wei H, Carnini A, Kang B, Wei W, Pidikiti R, Keller JM, Eckenhof f MF (2004) Inhaled anesthetic enhancement of amyloid-beta oligomerization and cytotoxicity. Anesthesiology 101:703-709.
Edwards ML (2010) Hyperbaric oxygen therapy. Part 1: history and principles. J Vet Emerg Crit Care 20:284-288.
Fidalgo AR, Cibelli M, White JP, Nagy I, Maze M, Ma D (2011) Systemic inf l ammation enhances surgery-induced cognitive dysfunction in mice. Neurosci Lett 498:63-66.
Ghia JE, Blennerhassett P, El-Sharkawy RT, Collins SM (2007) The protective ef f ect of the vagus nerve in a murine model of chronic relapsing colitis. Am J Physiol Gastrointest Liver Physiol 293:G711-718.
Godman CA, Joshi R, Giardina C, Perdrizet G, Hightower LE (2010a) Hyperbaric oxygen treatment induces antioxidant gene expression. Ann N Y Acad Sci 1197:178-183.
Godman CA, Chheda KP, Hightower LE, Perdrizet G, Shin D-G, Giardina C (2010b) Hyperbaric oxygen induces a cytoprotective and angiogenic response in human microvascular endothelial cells. Cell Stress Chaperones 15:431-442.
Hanning C (2005) Postoperative cognitive dysfunction. Br J Anaesth 95:82-87.
He HJ, Wang Y, Le Y, Duan KM, Yan XB, Liao Q, Liao Y, Tong JB, Terrando N, Ouyang W (2012) Surgery upregulates high mobility group box-1 and disrupts the blood-brain barrier causing cognitive dysfunction in aged rats. CNS Neurosci Ther 18:994-1002.
He X, Xu X, Fan M, Chen X, Sun X, Luo G, Chen L, Mu Q, Feng Y, Mao Q (2011) Preconditioning with hyperbaric oxygen induces tolerance against renal ischemia-reperfusion injury via increased expression of heme oxygenase-1. J Surg Res 170:e271-277.
Hovens IB, Schoemaker RG, van der Zee EA, Absalom AR, Heineman E, van Leeuwen BL (2014) Postoperative cognitive dysfunction: Involvement of neuroinflammation and neuronal functioning. Brain Behav Immun 38:202-210.
Hshieh TT, Fong TG, Marcantonio ER, Inouye SK (2008) Cholinergic def i ciency hypothesis in delirium: a synthesis of current evidence. J Gerontol A Biol Sci Med Sci 63:764-772.
Hu Q, Manaenko A, Xu T, Guo Z, Tang J, Zhang JH (2016) Hyperbaric oxygen therapy for traumatic brain injury: bench-to-bedside. Med Gas Res 6:102-110.
Hu SL, Huang YX, Hu R, Li F, Feng H (2015) Osteopontin mediates hyperbaric oxygen preconditioning-induced neuroprotection against ischemic stroke. Mol Neurobiol 52:236-243.
Huang G, Xu J, Xu L, Wang S, Li R, Liu K, Zheng J, Cai Z, Zhang K, Luo Y (2014) Hyperbaric oxygen preconditioning induces tolerance against oxidative injury and oxygen-glucose deprivation by up-regulating heat shock protein 32 in rat spinal neurons. PLoS One 9:e85967.
Huang WT, Lin MT, Chang CP (2006) An NMDA receptor-dependent hydroxyl radical pathway in the rabbit hypothalamus may mediate lipopolysaccharide fever. Neuropharmacology 50:504-511.
Inouye SK (2006) Delirium in older persons. N Engl J Med 354:1157-1165.
Kalns J, Lane J, Delgado A, Scruggs J, Ayala E, Gutierrez E, Warren D, Niemeyer D, Wolf EG, Bowden RA (2002) Hyperbaric oxygen exposure temporarily reduces Mac-1 mediated functions of human neutrophils. Immunol Lett 83:125-131.
Kang N, Hai Y, Liang F, Gao CJ, Liu XH (2014) Preconditioned hyperbaric oxygenation protects skin fl ap graThs in rats against ischemia/ reperfusion injury. Mol Med Report 9:2124-2130.
Kim CH, Choi H, Chun YS, Kim GT, Park JW, Kim MS (2001) Hyperbaric oxygenation pretreatment induces catalase and reduces infarct size in ischemic rat myocardium. Pf l ügers Archiv 442:519-525.
Kindwall EP, Hunt TK (1995) Hyperbaric medicine practice. Plast Reconstr Surg 96:985.
Kindwall EP, Wheland HT (1999) Hyperbaric medicine practice: Best Publishing Company.
Kocaoğullar Y, Üstün ME, Avci E, Karabacakoglu A, Fossett D (2004)The role of hyperbaric oxygen in the management of subarachnoid hemorrhage. Intensive Care Med 30:141-146.
Löndahl M (2012) Hyperbaric oxygen therapy as treatment of diabetic foot ulcers. Diabetes Metab Res Rev 28:78-84.
Labrouche S, Javorschi S, Leroy D, Gbikpi-Benissan G, Freyburger G (1999) Influence of hyperbaric oxygen on leukocyte functions and haemostasis in normal volunteer divers. Thromb Res 96:309-315.
Lahat N, Bitterman H, Yaniv N, Kinarty A, Bitterman N (1995) Exposure to hyperbaric oxygen induces tumour necrosis factoralpha (TNF-α) secretion from rat macrophages. Clin Exp Immunol 102:655-659.
Li J, Liu W, Ding S, Xu W, Guan Y, Zhang JH, Sun X (2008) Hyperbaric oxygen preconditioning induces tolerance against brain ischemia—reperfusion injury by upregulation of antioxidant enzymes in rats. Brain Res 1210:223-229.
Li Q, Li J, Zhang L, Wang B, Xiong L (2007) Preconditioning with hyperbaric oxygen induces tolerance against oxidative injury via increased expression of heme oxygenase-1 in primary cultured spinal cord neurons. Life Sci 80:1087-1093.
Li Y, Dong H, Chen M, Liu J, Yang L, Chen S, Xiong L (2011) Preconditioning with repeated hyperbaric oxygen induces myocardial and cerebral protection in patients undergoing coronary artery bypass graTh surgery: a prospective, randomized, controlled clinical trial. J Cardiothorac Vasc Anesth 25:908-916.
Lin H, Chang CP, Lin HJ, Lin MT, Tsai CC (2012a) Attenuating brain edema, hippocampal oxidative stress, and cognitive dysfunction in rats using hyperbaric oxygen preconditioning during simulated high-altitude exposure. J Trauma Acute Care Surg 72:1220-1227.
Lin KC, Niu KC, Tsai KJ, Kuo JR, Wang LC, Chio CC, Chang CP (2012b) Attenuating inflammation but stimulating both angiogenesis and neurogenesis using hyperbaric oxygen in rats with traumatic brain injury. J Trauma Acute Care Surg 72:650-659.
Liu W, Liu K, Tao H, Chen C, Zhang JH, Sun X (2012) Hyperoxia preconditioning: the next frontier in neurology? Neurol Res 34:415-421.
Lu P, Feng H, Yuan S, Zhang R, Li M, Hu R, Liu Z, Yin J (2013) Ef f ect of preconditioning with hyperbaric oxygen on neural cell apoptosis after spinal cord injury in rats. J Neurosurg Sci 57:253-258.
Lu PG, Hu SL, Hu R, Wu N, Chen Z, Meng H, Lin JK, Feng H (2012) Functional recovery in rat spinal cord injury induced by hyperbaric oxygen preconditioning. Neurol Res 34:944-951.
Lu SM, Gui B, Dong HQ, Zhang X, Zhang SS, Hu LQ, Liu HL, Sun J, Qian YN (2015) Prophylactic lithium alleviates splenectomy-induced cognitive dysfunction possibly by inhibiting hippocampal TLR4 activation in aged rats. Brain Res Bull 114:31-41.
Müller S, Krause N, Schmidt M, Münte T, Münte S (2004) Cognitive dysfunction after abdominal surgery in elderly patients. Z Gerontol Geriatr 37:475-485.
Ma Y, Cheng Q, Wang E, Li L, Zhang X (2015) Inhibiting tumor necrosis factor-α signaling attenuates postoperative cognitive dysfunction in aged rats. Mol Med Report 12:3095-3100.
Malik AB, Lo SK (1996) Vascular endothelial adhesion molecules and tissue inf l ammation. Pharmacol Rev 48:213-229.
Matsunami T, Sato Y, Sato T, Ariga S, Shimomura T, Yukawa M (2010) Oxidative stress and gene expression of antioxidant enzymes in the streptozotocin-induced diabetic rats under hyperbaric oxygen exposure. Int J Clin Exp Pathol 3:177.
Matsunami T, Sato Y, Hasegawa Y, Ariga S, Kashimura H, Sato T, Yukawa M (2011) Enhancement of reactive oxygen species and induction of apoptosis in streptozotocin-induced diabetic rats under hyperbaric oxygen exposure. Int J Clin Exp Pathol 4:255.
Maulik N (2002) Redox signaling of angiogenesis. Antioxid Redox Signal 4:805-815.
McDonagh DL, Mathew JP, White WD, Phillips-Bute B, Laskowitz DT, Podgoreanu MV, Newman MF (2010) Cognitive function after major noncardiac surgery, apolipoprotein E4 genotype, and biomarkers of brain injury. Anesthesiology 112:852.
Micarelli A, Jacobsson H, Larsson S, Jonsson C, Pagani M (2013) Neurobiological insight into hyperbaric hyperoxia. Acta Physiologica 209:69-76.
Mileski W, Winn R, Vedder N, Pohlman T, Harlan J, Rice C (1990) Inhibition of CD18-dependent neutrophil adherence reduces organ injury after hemorrhagic shock in primates. Surgery 108:206-212.
Moller J, Cluitmans P, Rasmussen L, Houx P, Rasmussen H, Canet J, Rabbitt P, Jolles J, Larsen K, Hanning C (1998) Long-term postoperative cognitive dysfunction in the elderly: ISPOCD1 study. Lancet 351:857-861.
Monk TG, Weldon BC, Garvan CW, Dede DE, Van Der Aa MT, Heilman KM, Gravenstein JS (2008) Predictors of cognitive dysfunction after major noncardiac surgery. Anesthesiololy 108:18-30.
Morykwas MJ, Argenta L (1996) Nonsurgical modalities to enhance healing and care of soTh tissue wounds. J South Orthop Assoc 6:279-288.
Newman MF, Kirchner JL, Phillips-Bute B, Gaver V, Grocott H, Jones RH, Mark DB, Reves JG, Blumenthal JA (2001) Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. N Engl J Med 344:395-402.
Newman S, Stygall J, Hirani S, Shaef i S, Maze M (2007) Postoperative cognitive dysfunction after noncardiac surgery: a systematic review. Anesthesiology 106:572-590.
Nie H, Xiong L, Lao N, Chen S, Xu N, Zhu Z (2006) Hyperbaric oxygen preconditioning induces tolerance against spinal cord ischemia by upregulation of antioxidant enzymes in rabbits. J Cereb Blood Flow Metab 26:666-674.
Niu KC, Huang WT, Lin MT, Huang KF (2009) Hyperbaric oxygen causes both antiinf l ammation and antipyresis in rabbits. Eur J Pharmacol 606:240-245.
Padgaonkar VA, Giblin FJ, Fowler K, Leverenz VR, Reddan JR, Dziedzic DC (1997) Heme oxygenase synthesis is induced in cultured lens epithelium by hyperbaric oxygen or puromycin. Exp Eye Res 65:435-443.
Palzur E, Soliman K, Vaya J, Chezar I, Michelis R, Sela S (2011) Ef f ects of hyperbaric oxygen (HBO) preconditioning on plasma constituents. Free Radic Biol Med 51:S97-S98.
Peng Y, Feng SF, Wang Q, Wang HN, Hou WG, Xiong L, Luo ZJ, Tan QR (2010) Hyperbaric oxygen preconditioning ameliorates anxiety-like behavior and cognitive impairments via upregulation of thioredoxin reductases in stressed rats. Prog Neuropsychopharmacol Biol Psychiatry 34:1018-1025.
Rothfuss A, Radermacher P, Speit G (2001) Involvement of heme oxygenase-1 (HO-1) in the adaptive protection of human lymphocytes after hyperbaric oxygen (HBO) treatment. Carcinogenesis 22:1979-1985.
Rudolph JL, Ramlawi B, Kuchel GA, McElhaney JE, Xie D, Sellke FW, Khabbaz K, Levkoff SE, Marcantonio ER (2008) Chemokines are associated with delirium after cardiac surgery. J Gerontol A Biol Sci Med Sci 63:184-189.
Selnes OA, Gottesman RF, Grega MA, Baumgartner WA, Zeger SL, McKhann GM (2012) Cognitive and neurologic outcomes after coronary-artery bypass surgery. N Engl J Med 366:250-257.
Shoair OA, Grasso II MP, Lahaye LA, Daniel R, Biddle CJ, Slattum PW (2015) Incidence and risk factors for postoperative cognitive dysfunction in older adults undergoing major noncardiac surgery: A prospective study. J Anaesthesiol Clin Pharmacol 31:30.
Simsek K, Ay H, Topal T, Ozler M, Uysal B, Ucar E, Acikel CH, Yesilyurt O, Korkmaz A, Oter S (2011) Long-term exposure to repetitive hyperbaric oxygen results in cumulative oxidative stress in rat lung tissue. Inhal Toxicol 23:166-172.
Steinmetz J, Christensen KB, Lund T, Lohse N, Rasmussen LS (2009) Long-term consequences of postoperative cognitive dysfunction. Anesthesiology 110:548-555.
Stetler RA, Leak RK, Gan Y, Li P, Zhang F, Hu X, Jing Z, Chen J, Zigmond MJ, Gao Y (2014) Preconditioning provides neuroprotection in models of CNS disease: paradigms and clinical signif i cance. Prog Neurobiol 114:58-83.
Sun L, Xie K, Zhang C, Song R, Zhang H (2014) Hyperbaric oxygen preconditioning attenuates postoperative cognitive impairment in aged rats. Neuroreport 25:718-724.
Szuchman A, Aviram M, Soliman K, Tamir S, Vaya J (2006) Exogenous N-linoleoyl tyrosine marker as a tool for the characterization of cellular oxidative stress in macrophages. Free Radic Res 40:41-52.
Terrando N, Monaco C, Ma D, Foxwell BM, Feldmann M, Maze M (2010) Tumor necrosis factor-α triggers a cytokine cascade yielding postoperative cognitive decline. Proc Natl Acad Sci U S A 107:20518-20522.
Terrando N, Eriksson LI, Kyu Ryu J, Yang T, Monaco C, Feldmann M, Jonsson Fagerlund M, Charo IF, Akassoglou K, Maze M (2011) Resolving postoperative neuroinf l ammation and cognitive decline. Ann Neurol 70:986-995.
Thom S, Mendiguren I, Hardy K, Bolotin T, Fisher D, Nebolon M, Kilpatrick L (1997) Inhibition of human neutrophil beta2-integrin-dependent adherence by hyperbaric O2. Am J Physiol Cell Physiol 272:C770-777.
Thom SR (1993) Functional inhibition of leukocyte B 2 integrins by hyperbaric oxygen in carbon monoxide-mediated brain injury in rats. Toxicol Appl Pharmacol 123:248-256.
Thom SR (2009) Oxidative stress is fundamental to hyperbaric oxygen therapy. J Appl Physiol 106:988-995.
Thom SR, Bhopale VM, Mancini DJ, Milovanova TN (2008) Actin S-nitrosylation inhibits neutrophil B2 integrin function. J Biol Chem 283:10822-10834.
Tian X (2015) Variations of brain edema and neurological function of rat models of cerebral infarction after hyperbaric oxygen therapy. Zhongguo Zuzhi Gongcheng Yanjiu 19:6423-6428.
Tian X, Zhang L, Wang J, Dai J, Shen S, Yang L, Huang P (2013) The possible protective mechanism of hyperbaric oxygen (HBO) in memory impairments induced by AB25—35in rats. Open Med 8:468-475.
Tibbles PM, Edelsberg JS (1996) Hyperbaric-oxygen therapy. N Engl J Med 334:1642-1648.
Ushio-Fukai M, Alexander RW (2004) Reactive oxygen species as mediators of angiogenesis signaling. Role of NAD (P) H oxidase. Mol Cell Biochem 264:85-97.
Vacas S, Degos V, Feng X, Maze M (2013) The neuroinf l ammatory response of postoperative cognitive decline. Br Med Bull 106:161-178.
van Maanen MA, Lebre MC, van der Poll T, LaRosa GJ, Elbaum D, Vervoordeldonk MJ, Tak PP (2009) Stimulation of nicotinic acetylcholine receptors attenuates collagen-induced arthritis in mice. Arthritis Rheum 60:114-122.
Verma R, Chopra A, Giardina C, Sabbisetti V, Smyth JA, Hightower LE, Perdrizet GA (2015) Hyperbaric oxygen therapy (HBOT) suppresses biomarkers of cell stress and kidney injury in diabetic mice. Cell Stress Chaperones 20:495-505.
Wad K, Ito M, Miyazawa T, Katoh H, Nawashiro H, Shima K, Chigasaki H (1996) Repeated hyperbaric oxygen induces ischemic tolerance in gerbil hippocampus. Brain Res 740:15-20.
Wada K, Miyazawa T, Nomura N, Tsuzuki N, Nawashiro H, Shima K (2001) Preferential conditions for and possible mechanisms of induction of ischemic tolerance by repeated hyperbaric oxygenation in gerbil hippocampus. Neurosurgery 49:160-167.
Wan Y, Xu J, Ma D, Zeng Y, Cibelli M, Maze M (2007) Postoperative impairment of cognitive function in rats: a possible role for cytokine-mediated inflammation in the hippocampus. Anesthesiology 106:436-443.
Wang L, Li W, Kang Z, Liu Y, Deng X, Tao H, Xu W, Li R, Sun X, Zhang JH (2009) Hyperbaric oxygen preconditioning attenuates early apoptosis after spinal cord ischemia in rats. J Neurotrauma 26:55-66.
Wilson B, McLaughlin L, Nair AR, Dange R, Francis J (2013) Inf l ammation, oxidative stress, and neuroprotective factors in the pathophysiology of PTSD in an animal model. FASEB J 27:691.695.
Xiong L, Zhu Z, Dong H, Hu W, Hou L, Chen S (2000) Hyperbaric oxygen preconditioning induces neuroprotection against ischemia in transient not permanent middle cerebral artery occlusion rat model. Chin Med J (Engl) 113:836-839.
Yamashita M, Yamashita M (2000) Hyperbaric oxygen treatment attenuates cytokine induction after massive hemorrhage. Am J Physiol Endocrinol Metab 278:E811-816.
Yan W, Zhang H, Bai X, Lu Y, Dong H, Xiong L (2011) Autophagy activation is involved in neuroprotection induced by hyperbaric oxygen preconditioning against focal cerebral ischemia in rats. Brain Res 1402:109-121.
Yogaratnam JZ, Laden G, Guvendik L, Cowen M, Cale A, Griffin S (2010) Hyperbaric oxygen preconditioning improves myocardial function, reduces length of intensive care stay, and limits complications post coronary artery bypass graTh surgery. Cardiovasc Revasc Med 11:8-19.
Zamboni WA, Roth AC, Russell RC, Graham B, Suchy H, Kucan JO (1993) Morphologic analysis of the microcirculation during reperfusion of ischemic skeletal muscle and the ef f ect of hyperbaric oxygen. Plast Reconstr Surg 91:1110-1123.
Zhang J, Jiang W, Zuo Z (2014) Pyrrolidine dithiocarbamate attenuates surgery-induced neuroinf l ammation and cognitive dysfunction possibly via inhibition of nuclear factor κB. Neuroscience 261:1-10.
Zhang J, Tan H, Jiang W, Zuo Z (2015) The choice of general anesthetics may not affect neuroinflammation and impairment of learning and memory after surgery in elderly rats. J Neuroimmune Pharmacol 10:179-189.
Zhang Q, Gould LJ (2014) Hyperbaric oxygen reduces matrix metalloproteinases in ischemic wounds through a redox-dependent mechanism. J Invest Dermatol 134:237-246.
Zhao Z, Huang G, Wang B, Zhong Y (2013) Inhibition of NF-kappaB activation by Pyrrolidine dithiocarbamate partially attenuates hippocampal MMP-9 activation and improves cognitive def i cits in streptozotocin-induced diabetic rats. Behav Brain Res 238:44-47.
Zheng X, Ma Z, Gu X (2015) Plasma levels of tumor necrosis factor-α in adolescent idiopathic scoliosis patients serve as a predictor for the incidence of early postoperative cognitive dysfunction following orthopedic surgery. Exp Ther Med 9:1443-1447.
Zhou BC, Liu LJ, Liu B (2016a) Neuroprotection of hyperbaric oxygen therapy in sub-acute traumatic brain injury: not by immediately improving cerebral oxygen saturation and oxygen partial pressure. Neural Regen Res 11:1445-1449.
Zhou HX, Liu ZG, Liu XJ, Chen QX (2016b) Umbilical cord-derived mesenchymal stem cell transplantation combined with hyperbaric oxygen treatment for repair of traumatic brain injury. Neural Regen Res 11:107-113.
Zhou L, Wang Z, Zhou H, Liu T, Lu F, Wang S, Li J, Peng S, Zuo Z (2015) Neonatal exposure to sevof l urane may not cause learning and memory def i cits and behavioral abnormality in the childhood of Cynomolgus monkeys. Sci Rep 5:11145.
Zhu JY, Yin GQ, Pang JJ, Chen ZX, Pan XY, Lu SD, Wei QX, Xie ZD (2016) Ef f ect of hyperbaric oxygen pretreatment on the expressions of vascular endothelial growth factor and transforming growth factor beta in over-length dorsal random skin flaps. Zhongguo Zuzhi Gongcheng Yanjiu 20:1525-1531.
Copyedited by Phillips A, Robens J, Wang J, Li CH, Qiu Y, Song LP, Zhao M
How to cite this article: Gao ZX, Rao J, Li YH (2017) Hyperbaric oxygen preconditioning improves postoperative cognitive dysfunction by reducing oxidant stress and inf l ammation. Neural Regen Res 12(2):329-336.
Open access statement: This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Funding: This study was supported by the Special Research Foundation of Doctoral Course in Colleges and Universities of China in 2013, No. 20133420110009.
*Correspondence to:
Yuan-hai Li, M.D., Ph.D., liyuanhai-1@163.com.
orcid:
0000-0001-6763-9624
(Yuan-hai Li)
10.4103/1673-5374.200816
Accepted: 2016-12-22
- 中国神经再生研究(英文版)的其它文章
- Nonhuman primate models of focal cerebral ischemia
- The cortical activation pattern during bilateral arm raising movements
- Neuroprotective ef f ect of the Chinese medicine Tiantai No. 1 and its molecular mechanism in the senescence-accelerated mouse prone 8
- Adenyl cyclase activator forskolin protects against Huntington’s disease-like neurodegenerative disorders
- Edaravone protects against oxygen-glucose-serum deprivation/restoration-induced apoptosis in spinal cord astrocytes by inhibiting integrated stress response
- Ef f ect of electroacupuncture on the mRNA and protein expression of Rho-A and Rho-associated kinase II in spinal cord injury rats