Synthesis and Characterization of Brush Copolymer Poly(propylene oxide)—graft—Poly(N,N—dimethylaminoethyl methacrylate)
ZHANG+Wen-qian
【Abstract】In this paper, a amphiphilic brush copolymer poly(propylene oxide)-graft -poly(N,N-dimethylaminoethyl methacrylate)(PPO-g-PDMAEMA) was successfully prepared via the combine of anionic ring opening polymerization and atom transfer radical polymerization(ATRP). The target products were confirmed by GPC and 1H NMR. This well-defined copolymer can supply a promising material as drug and gene carriers and protective materials.
【Key words】Characterization of Copolymer; Poly propylene oxide; methacrylate
0 Introduction
Brush copolymers[1] are a class of copolymers that grafting with side chains covalently linked to a polymer backbone. Their unique molecular architectures gives potential applications in biomedicine and nanotechnologies, and the synthesis of well-defined brush copolymers had well developed over the past decades. With the arising of the controlled radical polymerization (CRP) techniques such as atom transfer radical polymerization(ATRP)[2], stable free radial polymerization[3], and reversible addition fragmentation chain transfer(RAFT)[4]polymerization many brush copolymers were achieved. Generally, well-defined brush copolymers could be synthesized through two strategies: “grafting from” and “grafting onto” methods, the “grafting onto” method had the virtue that both backbone and side chains could be presynthesized individually and could also produce reactive junction point with functionalities between the backbone and side chain for postmodification. However, due to the steric congestion between the side chain and polymer backbone and low reactivity between two polymers, the grafting density of the resulting brush copolymers via “grafting onto” method was usually lower than that from“grafting from” methods. Poly(N,N-dimethylaminoethyl methacrylate) is a temperature, pH sensitive polymer, in the past decades various architectures PDMAEMA copolymer was synthesized for preparing smart materials and be widely applied in modification of nanoparticle, smart hydrogel, porous films, oil/water separation, drug, gene release, or flow behavior under control condition.
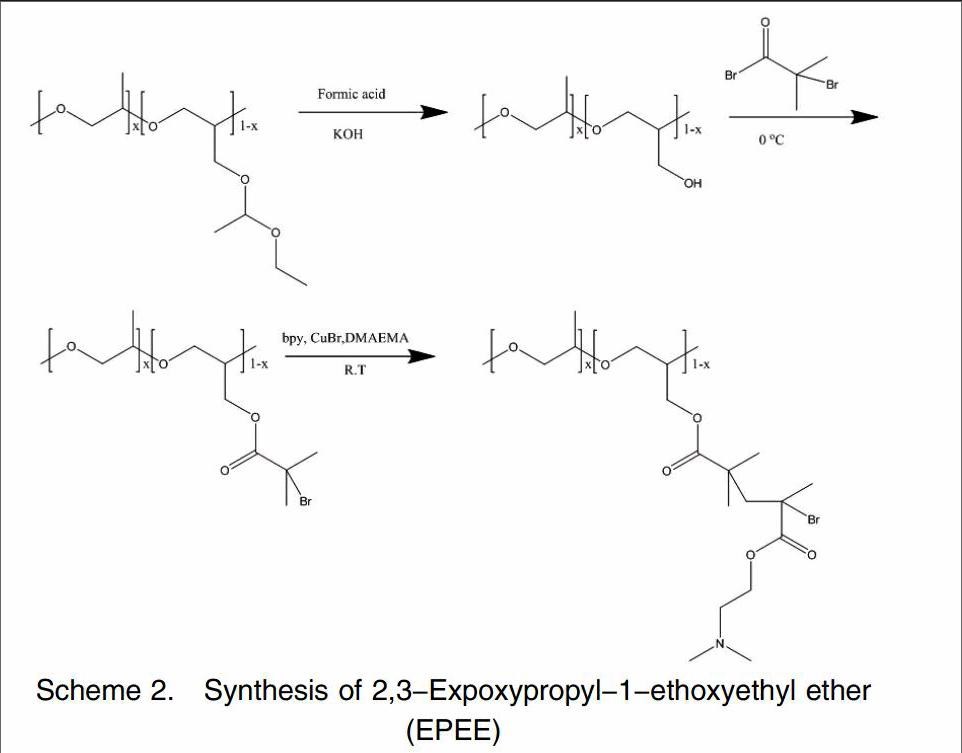
Here, a “grafting from” strategy were employed and the combine of anionic ring opening polymerization and atom transfer radical polymerization (ATRP)method was applied to synthesis amphiphilic brush copolymer PPO-g-PDMAEMA. Firstly the backbone was achieved by ring opening polymerization in the presence of PO and EPEE, after deprotection of EPEE units of copolymer, the hydroxy was modified to ATRP site, secondly the side chain was achieved by ATRP. The target products were characterized by GPC and 1H NMR.
1 Experimental Section
1.1 Materials
Propylene oxide(PO)was supplied by Sinopharm Chemical Reagent, ethoxyethene, 2,3-Epoxy-1-propano, Tetrabutylphosphonium bromide 2-bromoisobutyryl bromide and aluminium triisobutyl were purchased from Aldrich and used without further puri?覱cation. N,N-(Dimethylamino)ethyl methacrylate (DMAEMA) was provided by Aladdin and purified by passing through basic alumina to remove inhibitors. CuBr (95%) was stirred overnight in acetic acid, ?覱ltered, washed with ethanol and diethyl ether successively, and dried in vacuum. 2,3-Expoxypropyl-1-ethoxyethyl ether(EPEE)was prepared in our lab.
1.2 Synthesis of PPO-g-PDMAEMA
The synthesis of PPO-g-PDMAEMA was according to Scheme 2. 0.4 g Tetrabutylphosphonium bromide was dissolved in 50 mL toluene and introducted to 500 mL ampoule bottle, then 3.7 mL EPEE, 10 mL PO and 150 mL toluene were added. Subsequently, 5 mL aluminium triisobutyl was injected to initiate polymerization. The reaction was carried out at 0°C for 4h. After the polymerization was terminated by methanol(5.0 mL), the solvent was evaporated, dissolved in dichloromethane, and extracted twice with a mixed solution of sodium chloride and potassium hydroxide, and then extracted once with sodium chloride solution Finally, the organic layer was evaporated, pale yellow viscous product poly (EPEE-co-PO)was obtained.
5g of the copolymer poly(EPEE-co-PO)placed in 100mL flask, 24mL methanol and 4mL concentrated hydrochloric acid was added, after stirring for 2 hours the solvent was spin, precipitated five times with n-hexane, the precipitate was dried in vacuo at 40 °C for 12 hours to give a pale yellow objects. After deprotection of EPEE units, the copolymers was dissolved in anhydrous toluene 1 mL 2-Bromoisobutyryl bromide was added and stirring 24 h after separation salt poly(PO-co-Gly)-Br was obtained.
Scheme 2. Synthesis of 2,3-Expoxypropyl-1-ethoxyethyl ether(EPEE)
The poly(PO-co-Gly)-Br initiator 54 mg 2,2-bipyridine 50 mg CuBr and 4.0 mL DMAEMA were dissolved in THF. The solution was deoxygenated by freeze-pump-thaw cycles under argon. Polymerization was performed at room temperature for 24 h terminated by methanol dilution and exposure to air prior to copper removal on alumina columns. The crude copolymer was collected by freeze drying from aqueous solution and extracted with n-hexane at room temperature three times to remove unreacted DMAEMA monomer.
2 Results and Discussion
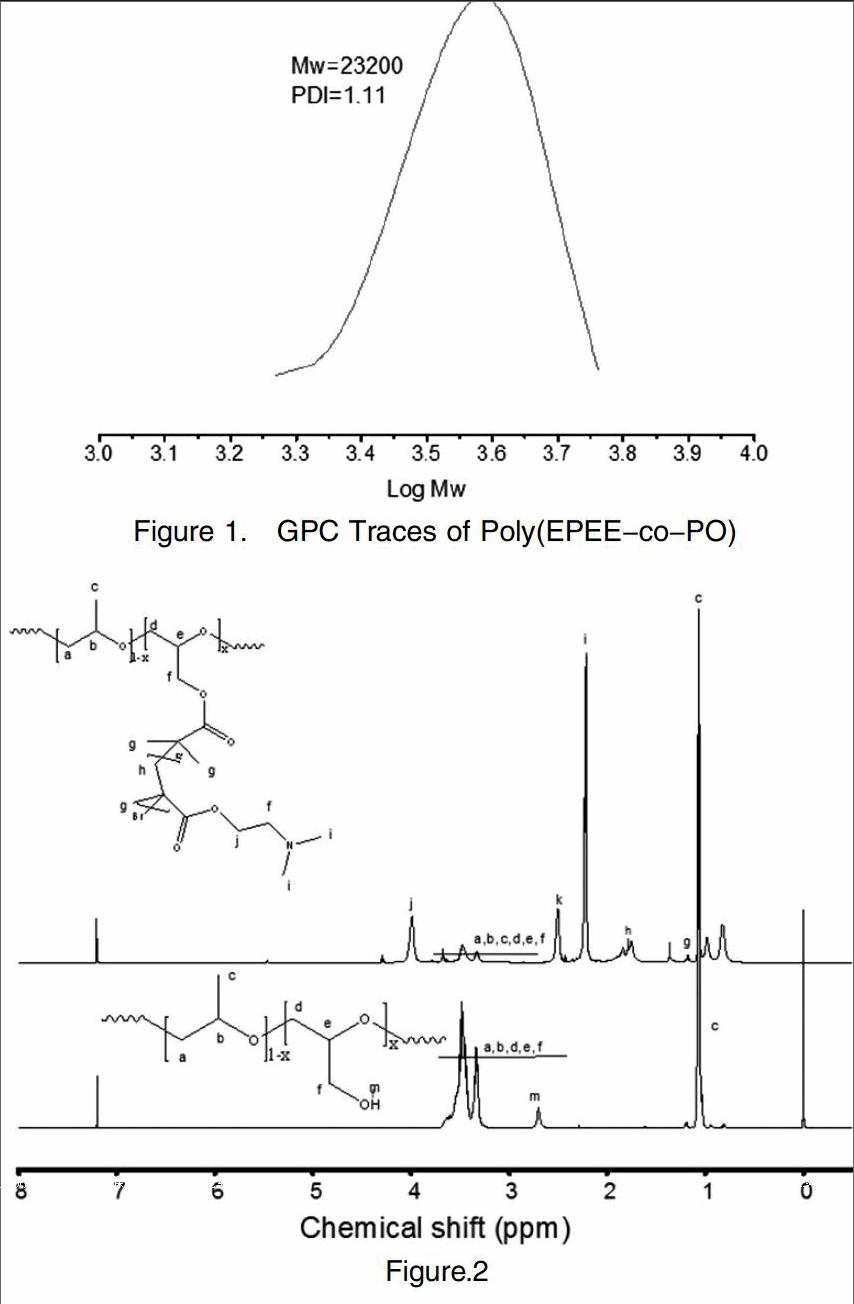
Figure 1 shows the GPC trace of the poly(PO-co-EPEE). The single peak in the GPC trace indicated formation of desired copolymers. The number average molecular weights(Mw)of the copolymers is 23200, and relatively narrow molecular weight distributions were observed (PDI=1.11).
In the 1H NMR spectrum of poly(PO-co-Gly) and PPO-g-PDMAEMA (Figure.2), compared to poly(PO-co-Gly) the characteristic proton signals of PDMAEMA in PPO-g-PDMAEMA are clearly detected. The methyl proton signals of of the PDMAEMA are located at chemical shifts of 0.9~1.2 ppm and 2.3 ppm, which are mainly attributable to the methyl protons C-CH3 and N-CH3, respectively. The methylene protons peaks located at chemical shifts of 1.8 ppm, 2.6 ppm and 4.1 ppm are corresponds to N-CH2, C-CH2, CH2-O-C=O, respectively. 1H NMR analysis results confirm PPO-g-PDMAEMA has been successfully prepared.
3 Conclusion
In summary, brush copolymer PPO-g-PDMAEMA was successfully prepared via the combine of anionic ring opening polymerization and atom transfer radical polymerization(ATRP). The target graft polymer and the intermediates were well characterized by the measurements of GPC and 1H NMR.
【Reference】
[1]Giannelis E P, Krishnamoorti R, Manias E. Polymer-silicate nanocomposites: model systems for confined polymers and polymer brushes[C]//Polymers in confined environments. Springer Berlin Heidelberg, 1999: 107-147.
[2]Wang J S, Matyjaszewski K. Controlled/“living” radical polymerization. Atom transfer radical polymerization in the presence of transition-metal complexes[J]. Journal of the American Chemical Society, 1995, 117(20): 5614-5615.
[3]Georges M K, Veregin R P N, Kazmaier P M, et al. Narrow molecular weight resins by a free-radical polymerization process[J]. Macromolecules, 1993, 26(11): 2987-2988.
[4]Chiefari J, Chong Y K, Ercole F, et al. Living free-radical polymerization by reversible addition-fragmentation chain transfer: the RAFT process[J]. Macromolecules, 1998, 31(16): 5559-5562.
[责任编辑:杨玉洁]

