Aquaporin 5 in Alzheimer’s disease:a link between oral and brain pathology?
Cristina Municio ,Eva Carro
The involvement of aquaporins (AQPs) in the development of diseases has been widely described (Azad et al.,2021).AQP5 has been described in astrocytes changing after traumatic brain injuries (Chai et al.,2013),but the precise role of AQP5 in Alzheimer’s disease (AD) pathology is yet to be understood.We have recently reported that AQP5 expression changes during the development of AD (Antequera et al.,2022).The AQP5 expression in salivary glands is decreased in 6-month-old APP/PS1 mice and AD patients.This decrease in AQP5 expression could be involved in the mechanism of salivary gland dysfunction described in a previous study (Antequera et al.,2021).Now,we propose a new indirect role of AQP5 in the connection between infectioninduced oral dysbiosis and AD (Sureda et al.,2020).Here,we suggest that the proinflammatory response induced by oral pathogen infection results in the downregulation of AQP5 contributing to the salivary gland secretory dysfunction.All these alterations destabilize the peripheral immune-inflammatory balance and exacerbate neuroinflammation and neurodegeneration leading to AD pathology.
AQPs overview:AQPs are a group of small transmembrane proteins that facilitate water transport.They are present in all kingdoms,from viruses to vertebrates.To date,13 different members have been described in mammals.In humans,they are widely distributed among various tissues and organs,as well as different cell types.They have a critical role in physiological functions and pathological processes as mediators of fluids transport.Traditionally,they have been defined as water channels across cell membranes.The functions of osmoregulation and fluid homeostasis are carried out mainly in epithelium and endothelium,for example,in renal tubules for urine concentration,and glandular epithelium for the formation of saliva,tears,sweat,and choroid plexus.
Currently,other functions in AQPs are also involved and are known as channels for the transport of small molecules such as glycerol,gases,anions,or some sugars.They participate in processes of cell signaling,migration,angiogenesis,or wound healing.Although many functions of AQPs have been described,some remain to be known,such as their roles in tumor metastases and neurological diseases (Wagner et al.,2022).
AQ Ps are monomers composed of six transmembrane alpha-helical segments and two short helical segments joined by connecting loops.Two consensus motifs of asparagineproline-alanine are conserved in the short helical segment and constitute a central pore.Although each monomer constitutes by itself an independent pore,the functional unit present in the membranes is a homotetramer (Verkman and Mitra,2000).
AQPs function and expression levels are regulated by post-translational modifications such as phosphorylations,glycosylations,or ubiquitinations.The processes of exocytosis and endocytosis regulate the abundance of AQPs in biological membranes.
AQPs are classified into four subfamilies according to their permeability capacity of glycerol in addition to water,the homology presented by the conserved sequence asparagine-prolinealanine,and the molecular phylogeny.The first group is classical or water-selective AQPs (AQP0,AQP1,AQP2,AQP4,AQP5,and AQP6).The second group refers to aquaglyceroporins (AQP3,AQP7,AQP9,and AQP10),which are permeable to glycerol.The group of superaquaporins or unorthodoxaquaporins (AQP11 and AQP12) is characterized by a unique asparagine-prolinealanine box with little homology concerning the previous groups.The AQP8 type or aquaammoniaporins are water channels with an unusual structure with long N-terminus and short C-terminus.
Thanks to its role in various physiological and pathological processes,its expression and regulation are considered potential therapeutic targets.Although common ion transport inhibitors such as diuretics and antiepileptics have been studied with little success,more positive results come from studies with biological drugs including anti bodies like AQP4-IgG,used for the treatment of inflammatory demyelinating diseases such as optic neuromyelitis.
AQPs in neurological diseases:In the central nervous system,the most abundant AQPs are AQP1 and AQP4,which are involved in cerebrospinal fluid homeostasis.AQP1 is mainly expressed in the epithelial cells of the choroid plexus,meanwhile,AQP4 is localized in the astrocytic endfeet that form the glial limiting membrane in the cortex.AQP5 is also expressed in astrocytes and its levels change during traumatic brain injuries (Chai et al.,2013).
The role of AQP4 is widely described in brain diseases.For example,alterations in the expression or localization of AQP4 have been associated with brain inflammation since it was reported to be a target anti gen in the autoimmune disease optic neuromyelitis,where patients express antibodies against AQP4.AQP4 is also involved in hydrocephalus,although its role is not yet well defined.Synaptic plasticity and memory are also affected by AQP4 deficiency,through its role as a regulator of glutamate transporter 1.Downregulation of glutamate transporter 1 is observed in central nervous system disorders,including neurodegenerative diseases.AQP4 also has an important role in AD,the most common cause of dementia in adults,due to its plausible effects on amyloid-beta (Aβ) and tau clearance.The glymphatic system facilitates brain fluid clearance and waste removal,including Aβ and tau,by the flow of cerebrospinal fluid into the brain.AQP4 is an integral part of the glymphatic system.AQP4 is in contact with the perivascular space adjacent to the blood vessels,facilitating cerebrospinal fluid influx into the brain parenchyma and its efflux back to the perivascular space.In AD,glymphatic dysfunction,related to perturbed AQP4 expression,has been reported.Indeed,AQP4 has been proposed as an interesting therapeutic target for AD,due to its plausible effects in Aβ and tau clearance.
However,increasing evidence suggests that AD pathology is not restricted to the central nervous system but affects other peripheral tissues.Changes in function and morphology have been described in the salivary glands of AD patients,as well as alterations in saliva composition.A reduction in muscarinic M3 receptors and acetylcholine levels was observed in AD,resulting in salivary gland dysfunction,including salivary secretion (Antequera et al.,2021).Lactoferrin,one of the main antimicrobial proteins in saliva,was decreased in the saliva of AD patients and transgenic mice models (Carro et al.,2017;González-Sánchez et al.,2020;Antequera et al.,2021).A deficiency of lactoferrin might facilitate pathogen proliferation and expansion to the central nervous system.
In the salivary gland,AQP1,AQP3,AQP4,AQP5,and AQP8 are expressed.But only in AQP5 significant changes have been described(Antequera et al.,2022).APQ5 represents the main water channel for normal physiology of the salivary gland and other exocrine glands,as shown in studies with animal models after the decrease or elimination of AQP5.Saliva secretion is affected during the development of certain pathologies,including Sjögren’s syndrome,where an abnormal distribution of AQP5 in acinar cells has been described.
Because the salivary gland presents morphological and functional changes in AD,our group proposed to study AQP5 expression in AD experimental models.In Antequera et al.,we have described how the expression of AQP5 changes in pathological situations,specifically during the development of AD (Antequera et al.,2022).In both human and mice experimental models,AQP5 is abundantly expressed in the acinar cells of the submandibular glands,confirming its role in maintaining salivary gland homeostasis.In salivary glands,we found that AQP5 expression is decreased in 6-month-old APP/PS1 mice and AD patients (Antequera et al.,2022).
We propose that the decrease in AQP5 expression in the salivary gland of APP/PS1 mice could be explained by systemic inflammatory signaling.In mice,tumor necrosis factor-α overexpression induces AQP5 reduction in the salivary glands.There is evidence of dysfunction in the salivary gland of AD patients associated with chronic inflammation since the plasma of AD patients has high levels of pro-inflammatory proteins such as interleukin 1β,interleukin 6,tumor necrosis factor α,or macrophage inflammatory protein 2.Tumor necrosis factor α is an important mediator of inflammation in response to infection,and increased tumor necrosis factor α expression has been often associated with inflammatory conditions that cause salivary gland hypofunction.Previous studies on infecti ous processes describe that nuclear factor-κB and mitogen-activated protein kinase pathways,induced by bacterial stimuli such as lipopolysaccharide,regulate the protein levels of some AQPs.Moreover,lipopolysaccharide treatment increases the expression of AQPs in different brain regions.In experimental animal models,lipopolysaccharide induces the secretion of the pro-inflammatory cytokine interleukin 1β through the salivary glands.A reduction in saliva production along with a decrease in the genes that regulate the expression of AQP5 is also present.This downregulation of AQP5 is reversed using inhibitors of the mitogenactivated protein kinase and/or nuclear factor κB signaling pathways,particularly by blocking the transcription factors p65,c-Jun,and c-Fos (Hosoi et al.,2020).These results support the implication of AQPs in neuroinflammatory processes.
AD as an autoimmune disorder:the responsibility of oral dysbiosis: In addition to genetic and environmental factors,an important correlation between oral health and AD has been described,with a link between infections resulting from oral dysbiosis (Figure 1).Chronic bacterial and viral infection may cause and initi ate the degenerative process in AD,sustaining chronic inflammation,and leading to progressive neuronal damage and Aβ deposition.The oral microbiome may increase the infection of opportunistic pathogens in the brain of AD patients and thus contributes to the development of AD (Sureda et al.,2020).Oral microbiota dysbiosis can directly cause cavities and periodontal disease and,in some cases,can affect the brain directly by Aβ production,or indirectly by producing neuroinflammation(Olsen and Singhrao,2021).The flow and composition of saliva are key elements for the maintenance of healthy oral microbiota.Saliva contains peptides and proteins secreted by the salivary glands involved in the defense of the oral cavity.Indeed,the innate immune system uses antimicrobial peptides and proteins to attack invading pathogens.Salivary alterations may result not only in a rapid deterioration in oral health but also in brain health,leading to a negative impact on a patient’s quality of life.In patients with AD,salivary levels of lactoferrin,one of the main antimicrobial peptides,are decreased probably,at least in part,due to a consequence of salivary gland dysfunction (Antequera et al.,2021).Lactoferrin represents one of the main first lines of defense against pathogens,plays an important role in regulating the oral microbiota and the inflammatory state of the oral mucosa,and contributes to the maintenance of symbiosis in the host-microbiome relationship.Additionally,the dysregulated microbiota activates inflammatory mediators such as interleukin 1β,interleukin 6,or tumor necrosis factor α that can produce chronic inflammation,leading to detrimental effects on submandibular AQP5 levels and salivary gland function.We propose the hypothesis by which systemic inflammatory signaling may induce AQP5 downregulation in submandibular glands in AD.Decreased expression of AQP5 led to decreased salivary secretion,resulting in lower levels of anti microbial proteins in saliva,including lactoferrin.
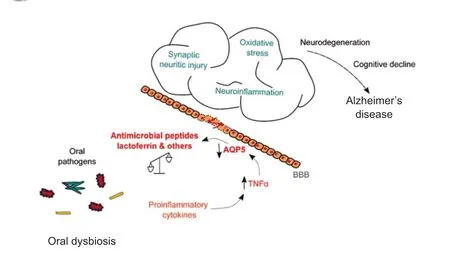
Figure 1 |Oral dysbiosis may lead to Alzheimer’s disease pathology.
The repercussion of this dysfunction on the immune response decreases the secretion of anti microbial proteins in saliva like lactoferrin.As consequence lower levels of these proteins with immunity and host protection functions increases the risk of developing AD.
Potenti al role of AQP5 in oral dysbiosis-induced AD:The involvement of AQPs in the development of neurodegenerative diseases is widely described(Azad et al.,2021),but the role of AQP5 in AD pathology is still unclear.Based on altogether previous findings,alterations in the location and expression of AQP5 in the salivary glands can produce deficiencies in saliva secretion,a dysregulation of the immune response,oral dysbiosis,and,consequently,a higher risk of both oral and cerebral infections.
Several experimental and clinical data confirm the key role of oral dysbiosis in neurodegeneration(Olsen et al.,2018).The merging of oral-derived inflammatory response together with aging in the elderly may contribute to the pathogenesis of AD.We speculate that salivary lactoferrin deficiency may act as an unknown trigger of oral microbial dysbiosis.Once the lactoferrin level begins to decrease,this becomes a vicious cycle for sustained dysbiosis,based on pathogens,such asPorphyromonas gingivalis,and may degrade lactoferrin.It was also reported that the presence of oral bacteria may induce the production of anti -AQP5 autoantibodies,associated with the low resting salivary flow.The anti -AQP5 autoanti bodies could have different effects,such as inhibiting the function of AQP5.In summary,we propose that the proinflammatory response induced by oral pathogen infection results in the downregulation of AQP5 contributing to the salivary gland secretory dysfunction.All these alterations destabilize the peripheral immune-inflammatory balance and exacerbate neuroinflammation and neurodegeneration leading to AD pathology(Figure 1).
These results obtained from our last studies also support the view that,in AD,several protective mechanisms against pathogen-targeting agents generating infections,and inflammatory conditions have been established.
Cristina Municio*,Eva Carro*
Group of Neurodegenerative Diseases,Hospital Universitario 12 de Octubre Research Institute(imas12),Madrid,Spain (Municio C)
Network Center for Biomedical Research in Neurodegenerative Diseases (CIBERNED),Instituto de Salud Carlos III,Madrid,Spain (Municio C,Carro E)
Neurobiology of Alzheimer’s Disease Unit,Functional Unit for Research into Chronic Diseases,Instituto de Salud Carlos III,Madrid,Spain (Carro E)
*Correspondence to:Eva Carro,PhD,eva.carro@isciii.es;Cristina Municio,PhD,cmunicio.imas12@h12o.es.
https://orcid.org/0000-0002-6504-4579(Eva Carro)
https://orcid.org/0000-0002-8552-8183(Cristina Municio)
Date of submission:September 23,2022
Date of decision:October 13,2022
Date of acceptance:October 20,2022
Date of web publication:November 25,2022
https://doi.org/10.4103/1673-5374.361545
How to cite this article:Municio C,Carro E (2023)Aquaporin 5 in Alzheimer’s disease: a link between oral and brain pathology? Neural Regen Res 18(7):1491-1492.
Open access statement:This is an open access journal,and articles are distributed under the terms of the Creative Commons AttributionNonCommercial-ShareAlike 4.0 License,which allows others to remix,tweak,and build upon the work non-commercially,as long as appropriate credit is given and the new creations are licensed under the identical terms.
- 中国神经再生研究(英文版)的其它文章
- Bystanders or not? Microglia and lymphocytes in aging and stroke
- Alzheimer’s disease risk after COVID-19: a view from the perspective of the infecti ous hypothesis of neurodegeneration
- Serine and arginine rich splicing factor 1: a potenti al target for neuroprotection and other diseases
- Can glial cells save neurons in epilepsy?
- Lights for epilepsy: can photobiomodulation reduce seizures and offer neuroprotection?
- The landscape of cognitive impairment in superoxide dismutase 1-amyotrophic lateral sclerosis

