A stable and reliable animal model for hepatocellular carcinoma with portal vein tumor thrombus
Zong-To Chi , Zhen-Hu Chen , Xiu-Ping Zhng , , Jin-Ki Feng , Zong-Hn Liu ,Shu-Qun Cheng ,*
a Department of Hepatic Surgery VI, Eastern Hepatobiliary Surgery Hospital, Second Military Medical University, 225 Changhai Road, Shanghai 200433,China
b Faculty of Hepato-Biliary-Pancreatic Surgery, the First Medical Center of Chinese People’s Liberation Army (PLA) General Hospital, Beijing 10 0 039, China
TotheEditor:
Hepatocellular carcinoma (HCC) is one of the most common cancers worldwide [ 1 , 2 ]. Portal vein tumor thrombus (PVTT) has been demonstrated to be a poor prognostic indicator for HCC [3-5] .However, effective treatment for the condition is still limited. Understanding the insight into the molecular mechanisms behind PVTT development may help to establish a new therapeutic strategy. Animal models which mimic the development of PVTT in humans are necessary to figure out the molecular mechanisms behind PVTT development. Currently, a wide range of animal models have been established for HCC from different angles, including chemically-induced models like diethylnitrosamine, carbon tetrachloride, thioacetamide, and phenobarbital, geneticallyengineered mouse (GEM) models like Wnt/β-catenin signaling pathway (CTNNB1), telomerase reverse transcriptase (TERT) activation, aflatoxin B1 and HBV infection, and engrafted models [6-8] .However, all the above animal models cannot be used in the study of PVTT, since these models were not able to present all the histological, physiological, and clinical features of human PVTT.
Our team has established a novel cell line (CSQT-2) from PVTT [9] . However, the previous study focused on the establishment of the cell line and the generation of PVTT was not stable and clinically applicable [10] . Hence, a stable and reliable animal model for PVTT is needed for understanding the pathogenesis of PVTT, identification of therapeutic targets, as well as screening for effective drugs. Thus, we designed a study to verify whether the intrasplenic injection of tumor cells is a stable and reliable way to build animal model for the development of PVTT and aimed to provide a useful tool for the basic research of PVTT.
To verify whether animal model induced by intrasplenic injection of CSQT-2 cells could produce a stable PVTT animal model,a total of five mice were used. After exploring spleen via the skin and peritoneal incisions, CSQT-2 cells (1 × 106/mL) were injected into the spleen of those mice. Twenty-four days later, all the 5 mice were sacrificed by cervical dislocation and the liver and lung tissues were collected to assess whether there were micrometastases by using Hematoxylin-Eosin (HE) staining. The results showed that the micrometastases were found in the vessels of liver, but not in the lung tissues ( Fig. 1 A, B, C). Next, we established intrasplenic injection model and orthotopic implantation model by using CSQT-2 to compare which was better in generating PVTT. There were 5 mice in each group and the results showed that more vessel metastases were detected in the mice of the intrasplenic injection group than in the orthotopic xenograft model group ( Fig. 1 D, E,P= 0.001 and Table S1). Just like the patient derived xenografts, we next wanted to verify whether intrasplenic injection model can be established by using PVTT tissue of HCC patient, and we collected the primary HCC and PVTT cells from the tissues of 15 HCC patients to establish this model and only one case succussed ( Table 1 ). We found that the PVTT cells from the patients can generate more liver metastases than the primary tumor cells ( Fig. 2 A, B, C,P<0.001 and Table S2). Moreover, a survival experiment was conducted (five mice in each group) and the Kaplan-Meier curves showed that the mice injected with PVTT cells had worse overall survival (OS) than those injected with primary tumor cells ( Fig. 2 D,P= 0.002). This result indicated that primary tumor thrombus cells was different from the primary tumor cells and that intrasplenic injections can be used to establish patient derived PVTT model in mice.
Overall, our study revealed that the PVTT animal model induced by the intrasplenic injection approach was superior to that induced by orthotopic implantation in the aspects of time span,PVTT formation rate, formation stability, and practical applicability.And the fact that CSQT-2 cells can generate PVTT greatly facilitates our research of PVTT progression. In addition, the primary tumor thrombus cells possessed a highly metastatic potential and PVTT formation ability, which indicated that the primary tumor thrombus cells were more suitable for the establishment of the PVTT animal model. Nevertheless, we demonstrated that this model partially mimics the whole progression of PVTT formation in primary HCC, and fully elucidates various phenotypes of PVTT.
In conclusion, our study may contribute to the identification of treatment targets, modeling of disease progression, and development of novel therapeutic strategies against PVTT.
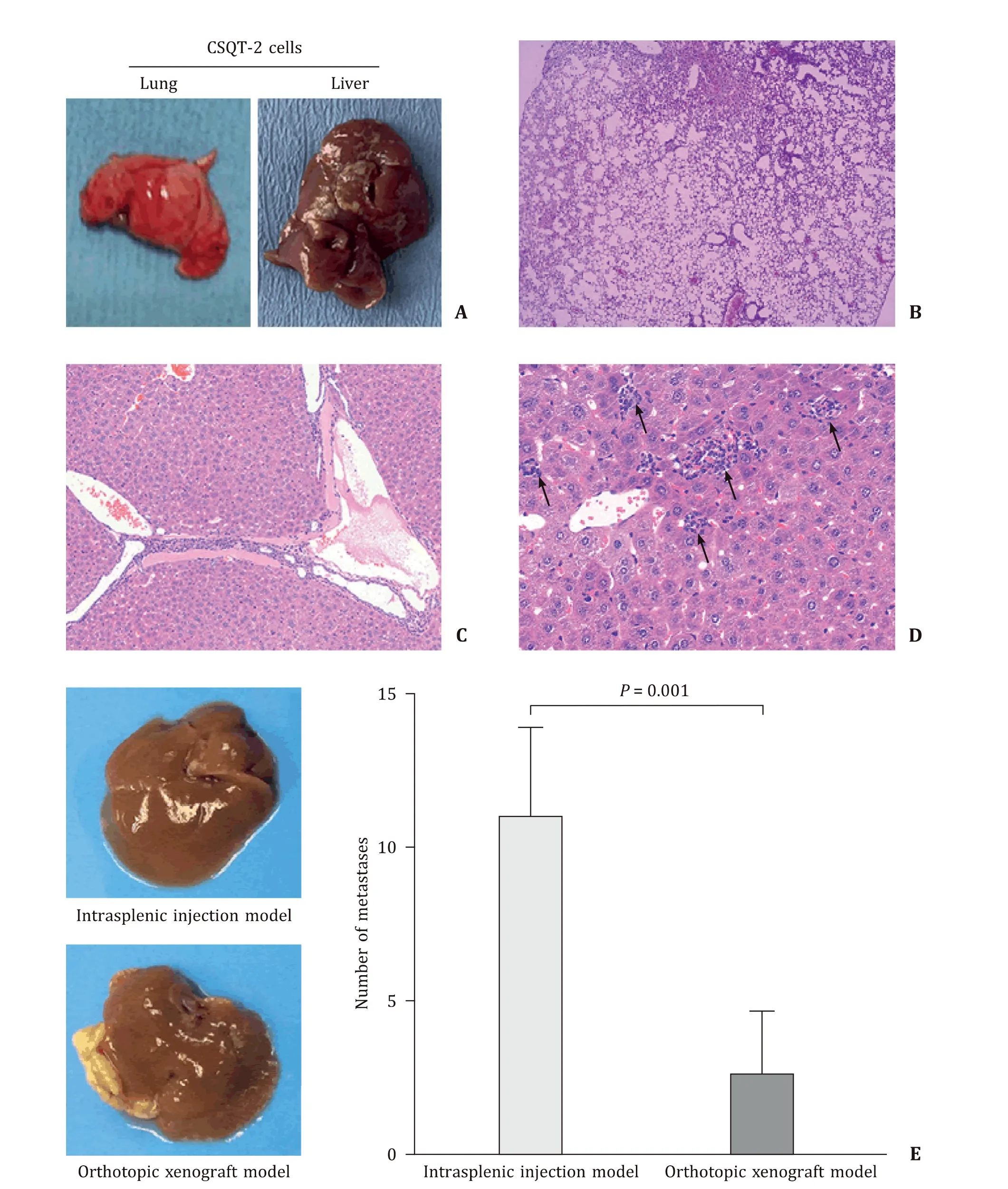
Fig. 1. PVTT animal model induced by CSQT-2 cells. A : The lungs and livers of mice injected with CSQT-2; B : the representative images of lung in the mice injected with CSQT-2 (HE staining, original magnification × 50); C : the representative images of vessel metastasis of liver in the mice injected with CSQT-2 (HE staining, original magnification × 200); D : the representative images of live metastasis induced by orthotopic xenograft models (original magnification × 400); E : intrasplenic injections model generated more PVTT sites than orthotopic xenograft mode ( P = 0.001). PVTT: portal vein tumor thrombus; HE: hematoxylin-eosin.

Fig. 2. PVTT animal model induced by either patients’ primary tumor cells or tumor thrombus cells. A : The livers of mice injected with patients’ primary tumor cells and tumor thrombus cells; B : the representative images of liver metastasis of the mice injected with patients’ primary tumor thrombus cells and primary tumor cells (HE staining, left column, original magnification × 200; right column, original magnification × 400); C : intrasplenic injections of primary tumor thrombus cells in the nude mice generated more PVTT sites in the portal vein systems of the host than the primary tumor cells ( P < 0.001); D : the mice in the PVTT cells group had poorer OS than those in the tumor cells group ( P = 0.002). PVTT: portal vein tumor thrombus; HE: hematoxylin-eosin; OS: overall survival.
Acknowledgments
We thank and appreciate the Institute of Nutritional Sciences,Shanghai Institutes for Biological Sciences, Chinese Academy of Science for providing us with experimental platforms and materials as well as technical support.
CRediT authorship contribution statement
Zong-Tao Chai : Conceptualization, Formal analysis, Investigation, Methodology, Writing - original draft. Zhen-Hua Chen : Conceptualization, Formal analysis, Investigation, Writing - original draft. Xiu-Ping Zhang : Conceptualization, Formal analysis, Investigation, Writing - original draft. Jin-Kai Feng : Data curtion, Resources, Validation. Zong-Han Liu : Data curtion, Resources, Validation. Shu-Qun Cheng : Conceptualization, Funding acquisition,Project administration, Supervision, Writing - review & editing.
Funding
This study was supported by a grant from the National Natural Science Foundation of China ( 81702335 ).
Ethical approval
This study was approved by the Shanghai Medical Experimental Animal Care Commission and the Institutional Review Board of Eastern Hepatobiliary Surgery Hospital. Written informed consent was obtained from all the participants for their data to be used for research purposes.
Competing interest
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
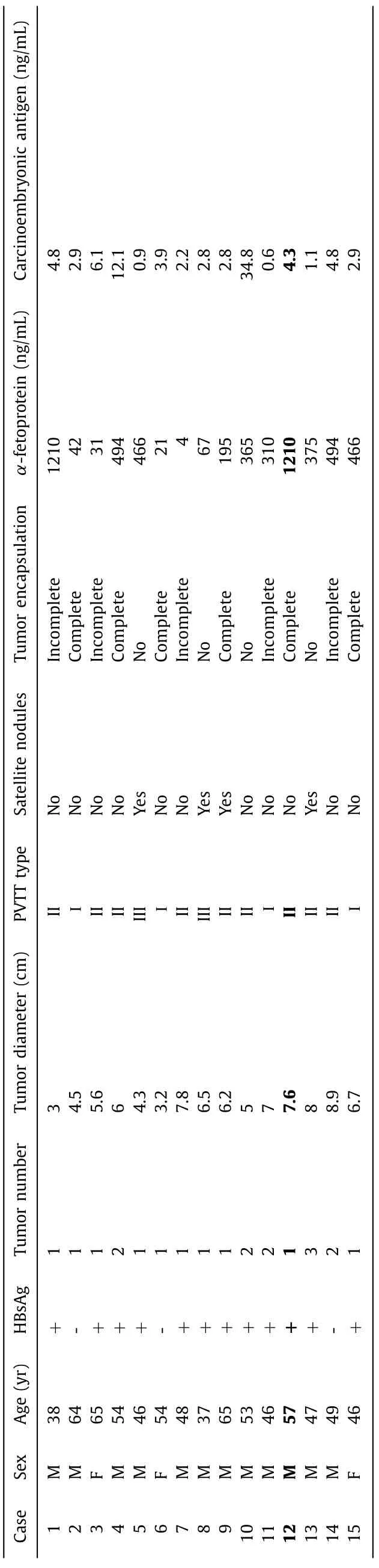
Table 1 Clinicopathological features of the 15 patients.
Supplementary materials
Supplementary material associated with this article can be found, in the online version, at 10.1016/j.hbpd.2021.03.003 .
 Hepatobiliary & Pancreatic Diseases International2022年1期
Hepatobiliary & Pancreatic Diseases International2022年1期
- Hepatobiliary & Pancreatic Diseases International的其它文章
- Primary pancreatic lymphoma diagnosed by endoscopic ultrasound-guided fine needle biopsy
- Expert consensus on the application of the magnetic anchoring and traction technique in thoracoscopic and laparoscopic surgery
- Diverse and precision therapies open new horizons for patients with advanced pancreatic ductal adenocarcinoma
- SNHG16 promotes hepatocellular carcinoma development via activating ECM receptor interaction pathway
- Predictors of recurrent bile duct stone after clearance by endoscopic retrograde cholangiopancreatography: A case-control study
- Call for action: Increased healthcare utilization with growing use of percutaneous cholecystectomy tube over initial cholecystectomy in cirrhotics
