Gastrointestinal complications after kidney transplantation
Rossella Gioco, Daniela Corona, Burcin Ekser, Lidia Puzzo, Gaetano Inserra, Flavia Pinto, Chiara Schipa,Francesca Privitera, Pierfrancesco Veroux, Massimiliano Veroux
Abstract Gastrointestinal complications are common after renal transplantation, and they have a wide clinical spectrum, varying from diarrhoea to post-transplant inflammatory bowel disease (IBD). Chronic immunosuppression may increase the risk of post-transplant infection and medication-related injury and may also be responsible for IBD in kidney transplant re-cipients despite immunosuppression. Differentiating the various forms of post-transplant colitis is challenging, since most have similar clinical and histological features. Drug-related colitis are the most frequently encountered colitis after kidney transplantation, particularly those related to the chronic use of mycophenolate mofetil, while de novo IBDs are quite rare. This review will explore colitis after kidney transplantation, with a particular focus on different clinical and histological features, attempting to clearly identify the right treatment, thereby improving the final outcome of patients.
Key Words: Inflammatory bowel disease; Kidney transplantation; Solid organ transplantation; Crohn disease; Ulcerative colitis; Mycophenolate mofetil colitis; Mycophenolate mofetil; Colitis; Cytomegalovirus
INTRODUCTION
Kidney transplantation is considered the gold standard treatment in patients affected by end-stage renal disease since it significantly improves the quality of life and patient survival compared to dialysis[1]. Although much progress has been made over the years, the management of patients during post-transplant follow-up still presents numerous difficult clinical problems. The success of a kidney transplant is related to the prevention of acute rejection, and newer immunosuppressive therapy provides a significant improvement in transplant outcomes, reducing the incidence of acute rejection[1].
However, chronic immunosuppression may increase the risk of various complications, including chronic allograft nephropathy and post-transplant infections and cancers[1-4]. In addition, kidney transplant recipients are at increased risk of gastrointestinal complications, which represent a major cause of morbidity and mortality after transplantation[5,6].
Gastrointestinal complications in kidney transplant reci-pients may be a consequence of typical infections occurring in transplant recipients, such as cytomegalovirus (CMV) infection[7-10], and of immunosuppression-mediated injury to the gastrointestinal mucosa. Post-transplant inflammatory bowel disease (IBD) may arise from an inappropriate immune response to intestinal antigens resulting in continuous intestinal inflammation[5]. Immunosuppressive therapy, which could theoretically combat this inflammatory process, may paradoxically allow for dysregulation of the intestinal immune system, finally resulting in the development of post-transplant IBD[5,11,12]. Therefore, the use of mycophenolate mofetil (MMF), which is a part of the standard immunosuppressive protocol in kidney transplantation, may increase the risk of gastrointestinal complications, including diarrhoea, gastritis and specific MMF-related colitis[6,13,14]. Furthermore, kidney transplant recipients may develop a form ofde novoIBD despite being immunosuppressed[13,15-19].
Gastrointestinal inflammatory diseases in transplanted patients are mostly colitis and are characterized by similar symptoms but different physiopathological features[20]. A variety of clinical conditions have been described, including the following (Table 1): (1) Graft-versus-host disease (GVHD); (2) Infection-related gastrointestinal colitis (mainly CMV-derived colitis); (3) Drug-induced colitis (mainly MMF-related colitis); and (4)De novoIBD: Crohn’s disease (CD) and ulcerative colitis (UC).
Although some studies report that the incidence of IBD in solid organ transplantation is approximately 10 times higher than that observed among the general population, particularly in liver transplant recipients[21], the occurrence of IBD in kidney transplantation is rarely reported[5].
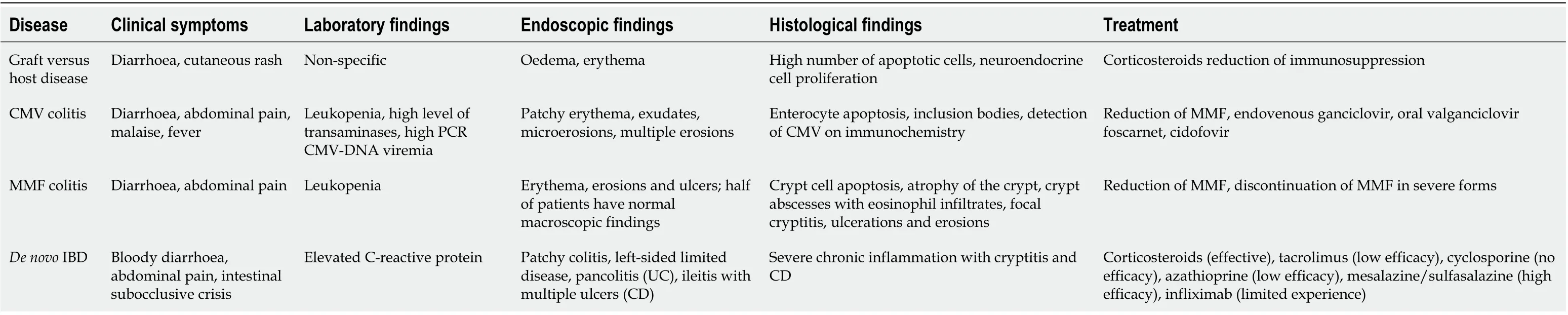
Table 1 Clinical and histological characteristics of inflammatory bowel diseases in kidney transplantation
The aim of this review is to evaluate the natural history of gastrointestinal inflammatory disease in renal transplant recipients, with particular emphasis on the incidence, clinical characteristics, and potential for effective therapy. Moreover, a brief overview of the outcomes of kidney transplantation in patients with previous inflammatory bowel disease is also reported.
Literature search
The PubMed database was searched for articles by using the following terms: “chronic kidney disease”, “chronic renal insufficiency”, “Crohn’s disease”, “kidney transplantation”, “ulcerative colitis”, “inflammatory bowel disease”, “graft-versushost”, “colitis”, “mycophenolate-mofetil colitis”, and “mycophenolic acid”. Titles and abstracts were screened by two authors (Rossella Gioco and Massimiliano Veroux) to identify potentially relevant studies, and all potentially eligible studies were subsequently evaluated in detail by three authors (Massimiliano Veroux, Daniela Corona and Rossella Gioco) through consideration of the full text. The reference lists of retrieved articles were also searched for relevant publications. Experimental studies, clinical trials, meta-analyses, narrative reviews, and systematic reviews published in the last 20 years were included. Bibliographies of relevant articles and reviews were manually screened to identify additional studies. Studies were excluded if they were not in the English language, if they did not fit the research question, or if they had insufficient data. Initial database searches yielded 247 studies from PubMed in the last 20 years. After the evaluation of the bibliographies of the relevant articles, we evaluated 32 eligible full-text articles.
EPIDEMIOLOGY AND PATHOGENESIS OF GASTROINTESTINAL COMPLICATIONS IN KIDNEY TRANSPLANT RECIPIENTS
Very few studies have investigated the incidence of gastrointestinal inflammatory complications after kidney transplantation. A true incidence is difficult to assess due to the heterogeneity of classification and clinical manifestations. Clinical manifestations vary from diarrhoea, which is the most common symptom, to true IBD, which is largely less common. Moreover, most of the studies focused on a small proportion of patients undergoing diagnostic colonoscopy for symptomatic diarrhoea, and in most cases, the final diagnosis was a non-specific colitis, which could underestimate the true prevalence of the disease.De novoIBD after solid organ transplantation (SOT) is extremely rare (206 cases/100000): The majority of cases occur in liver transplant recipients, while only a few cases have been reported in renal transplant recipients[22,23].
In a review, Wörnset al[24]reported 44 cases ofde novoIBD, but only 2 were detected in kidney transplant re-cipients. A more recent review[16]identified a total of 27de novoIBD patients (15 patients with UC and 12 patients with CD) after renal transplantation. In a descriptive study on histological features of IBD in kidney transplant recipients, Pittmanet al[5], among 700 kidney transplant recipients, identified 51 patients (7.2%) with gastrointestinal symptoms. Most of them (33%) were ultimately considered to have medication-related colonic injury, mainly MMF colitis, while 11 (22%) had infectious colitis, mainly fromClostridium difficileand CMV infections. Four (8%) patients had clinical and histopathological colitis suggesting ade novoIBD. In a cohort of 940 kidney transplant recipients, Dobieset al[25]found an IBD in 7 patients (0.7%). An additional case ofde novoCD was recently reported by Mottéet al[19], making for a total of 46de novohistologically proven IBD cases (23 UC cases and 21 CD cases, plus 2 cases not otherwise specified) reported to date in kidney transplant recipients[5,15,19,20,22-34], including three paediatric patients[18,32]. In contrast, MMF-related colitis has a higher incidence, since it is present in up to 47% of patients undergoing colonoscopy for chronic diarrhoea[5,13,25,35].
Post-transplantde novoIBDs present more frequently in males, with a mean age of 35 years[5,15,18,21,22,26-35]. The main presenting symptoms are diarrhoea, abdominal pain and bright red haematochezia[5,15,18,21,22,26-34], and the mean time after transplantation to IBD presentation is 4.6 years[5,15,19,20,22-31]. Only in one patient did IBD present early, within one year after transplantation[18].
Several hypotheses have been suggested to explain the paradoxical development ofde novoIBD in kidney transplant recipients. In the nontransplant population, IBD is generally caused by the activation of the immune system towards intestinal antigens, which may include normal intestinal microbiota[36-38]. In a kidney transplant setting, immunosuppressive therapy may provoke dysregulation of the intestinal immune environment, making it more susceptible to various insults that may damage the epithelial barrier of the intestinal mucosa, allowing prolonged exposure to luminal antigens. This exposure could lead to chronic immune stimulation and IBD, similar to what happens in non-immunosuppressed individuals who develop CD[39,40]. Moreover, a gut microbiota dysbiosis may be responsible of an increased risk of post-transplant diarrhoea and gastrointestinal complications[41,42].
Immunosuppression may increase the patient’s susceptibility to opportunistic infections, such as CMV,Escherichia coli, Campylobacter, orSalmonellainfections, which may trigger IBD[22,23,39,40,43,44], as demonstrated by the likely simultaneous occurrence of IBD and gastrointestinal infections[9,10].
Moreover, immunosuppressive drugs themselves may be responsible for the increased susceptibility of the gastrointestinal mucosa. Experimental studies in mice have shown that interleukin-2 (IL-2) has important inhibitory effects on T cells, and a reduction in IL-2 may provoke autoimmune colitis similar to UC[45]. The extensive use of IL-2 inhibitors such as basiliximab and tacrolimus in the induction and maintenance of post-transplant immunosuppressive therapy may therefore predispose patients to an increased risk of IBD[7,12,46], while the use of azathioprine might exhibit a protective role[11]. However, in the nontransplant population, the use of tacrolimus showed a clinical benefit in the management of IBD in the short term[47,48], suggesting that in the transplant population, the pathogenesis ofde novoIBD is multifactorial and requires a multihit process.
Moreover, MMF and mycophenolic acid (MPA), which are two of the most effective immunosuppressants in renal transplant recipients[1], are a frequent cause of posttransplant colitis with diarrhoea[5,6,13]. MPA and MMF exposure seems to directly cause local gastrointestinal toxicity[13], which may ultimately determine apoptotic cell death and crypt damage through cytotoxic or immune-mediated mechanisms[13].
Finally, it has been suggested that steroids may have a protective role due to the likely occurrence of IBDs late in posttransplant follow-up, when the dosage of steroids is at its minimum[23,26]. Indeed, the few patients who presented IBD in the first posttransplant semester had early steroid withdrawal[22,23].
DIAGNOSIS
IBD may appear as an exacerbation of a pre-existing disease or, more rarely, asde novoIBD occurring in patients without any previous symptoms, and post-transplantde novodiseases may have a more aggressive clinical course[22,23,26]. A combination of clinical, endoscopic, and histologic features is useful to distinguish between causes of gastrointestinal symptoms affecting renal transplant recipients. The clinical manifestations are extremely varied, and patients are usually diagnosed with a form of IBD after excluding other aetiologies. In most cases, patients present symptoms such as bloody diarrhoea, abdominal cramping and bright red haematochezia.
Detailed descriptions of the clinical and endoscopic features of post-transplant IBD are limited[35]. The main feature is the presence of chronic inflammation of the mucosa, involving any tract of the digestive system in the case of CD or only the colon in the case of UC. They are both chronic intermittent diseases that can evolve into severe forms. The endoscopic pictures are patchy colitis, left-sided limited disease, pancolitis or ileal disease suggestive of CD or UC[22-31], while the presence of erythema and erosion/ulcers may be significantly associated with MMF-related colitis[35]. However, up to half of patients may have normal endoscopic findings, particularly in MMFrelated colitis[35,49]or nonspecific colitis[50]. Interestingly, to date, no studies have evaluated the utility of faecal and blood markers for the detection of endoscopically active post-transplant IBD[51].
CLINICAL AND HISTOLOGICAL FEATURES
GVHD
The term GVHD refers to a clinical syndrome that occurs in transplanted patients with injury to target organs such as skin, liver, gastrointestinal tract and, more rarely, other organs[52]. It is more common in bone marrow transplantation and rarely occurs in SOT recipients[52]. Its incidence is higher in small bowel transplant recipients (5%), while in liver transplant recipients, it occurs 1 to 11 wk after transplantation, with a frequency ranging between 0.1% and 1%[53]and a mortality exceeding 75%[54]. GVHD in kidney transplant recipients is extremely rare, with only six cases reported in a recent review by Guoet al[55]. Clinical manifestations include diarrhoea (in the case of gastrointestinal tract involvement) and cutaneous rashes (in the case of skin involvement), with some degree of kidney function impairment[56]. The prognosis of GVHD after kidney transplantation is usually better than that GVHD following other SOTs, probably as a consequence of the fewer donor-derived lymphocytes in kidney grafts than in other solid organ grafts, and only 2 of 6 patients have died of GVHD in the kidney transplant setting[55].
The pathogenesis of GVHD is still incompletely understood, but it is probably triggered by the destruction of host tissues through several different mechanisms involving donor cytotoxic T cells, natural killer cells, cross reactivity between antigens on intestinal bacteria and the epithelium and the release of cytotoxic agents following the interaction between the host and donor cells and tissue injury[56-58]. Koyamaet al[59]suggested that GVHD is initiated by the interaction between recipient antigenpresenting cells and donor T cells. After transplantation, these antigen-presenting cells are modified by cellular pattern signals derived from the intestinal microbiota. Donor dendritic cells in the gastrointestinal tract are activated in the colonic mucosa, resulting in the development of GVHD[59].
GVHD involving the gastrointestinal tract is characterized by crypt cell apoptosis, which is a histologic feature also described in other disorders associated with immune dysregulation and IBD, such as MMF colitis[35,60]. However, the absolute number of apoptotic cells may be significantly higher in GVHD than in MMF colitis[61]. Moreover, among the other features characterizing the histology of GVHD, there is neuroendocrine cell proliferation, which is probably a compensatory response to cell loss, periglandular inflammatory infiltrates, crypt cell cytologic atypia and histologic features of chronic inflammation similar to those described in inflammatory bowel disease and MMF colitis[35,60-62].
Treatment of GVHD includes methylprednisolone and decreased immunosuppression with the aim of destroying activated donor-derived lymphocytes as well as deleting donor-derived lymphocytes with the host's native immune system[55].
CMV colitis
Infectious complications are the most important cause of morbidity and mortality after transplantation[1,3]. The incidence varies according to many factors, including the type of transplant, the patient's immune system and the immunosuppressive therapy. It is estimated that approximately 80% of transplant recipients develop an infection after transplantation[63], and the progressive reduction in the incidence of acute rejection has led to a significant increase in infectious diseases, particularly those associated with latent viruses such as CMV. Although the introduction of CMV prophylaxis has significantly reduced the incidence of clinically evident CMV disease in the early period after transplantation, CMV is the virus that most commonly infects patients undergoing SOT, with significant consequences on graft and patient survival. In the first year after transplantation, 50%-70% of patients experience primary infection, reactivation or reinfection[63]. When CMV infection causes significant viral replication and symptomatic illness, it may cause tissue-invasive disease with end-organ damage from the virus[64,65]. The gastrointestinal tract is most commonly affected during CMV tissue-invasive disease, resulting in oesophagitis, gastritis, enteritis, or colitis[66,67]. Diagnosis requires a biopsy obtained during oesophagogastroduodenoscopy and/or colonoscopy with histologic or culture-based evidence of CMV[66,68]. Gastrointestinal involvement during CMV infection is described in approximately 5% of patients undergoing SOT and may involve any part of the digestive tract[68], but its incidence may rise up to 25% in patients with clinical symptoms suggestive of CMV infection[68].
Indeed, CMV infects epithelial and mesenchymal cells and destroys them, causing ulcerations on the epithelial layer in different organs, including the small intestine and colon[69]. Moreover, several studies have highlighted an association between IBD and CMV, which is attributed to the virus’s role in terms of both disease onset and severity[70,71]. Clinical symptoms of CMV colitis include fever, malaise and abdominal pain with diarrhoea, while laboratory findings include leukopenia, thrombocytopenia and high levels of transaminases[10,72,73].
Endoscopic lesions range from patchy erythema, exudates, and microerosions to oedematous mucosa with multiple erosions[10,72,73]. The main histologic feature of CMV colitis is increased enterocyte apoptosis, which is caused by viral infection, so it is difficult to distinguish CMV colitis from GVHD on gastrointestinal biopsy[72]. Definitive diagnosis in kidney transplant patients requires histologic findings of characteristic inclusion bodies on haematoxylin and eosin staining (Figure 1), in addition to macroscopic lesions on endoscopy[72]. Moreover, the detection of CMV in formalin-fixed tissue with immunochemistry, eventually integrated with polymerase chain reaction (PCR) of the paraffin-embedded tissue, is a highly valuable method for confirming the diagnosis of CMV colitis[74].
Serologic testing (for CMVIgG) is ineffective for diagnosing CMV disease because most cases present in a state of viral reactivation of latent virus[72]. The late CMV antigen (pp65) and quantitative CMV PCR assays have a high sensitivity for CMV viremia, but they are not good predictors for CMV tissue-invasive disease[72,73]and have a poor sensitivity (range, 48%–73%) in the detection of GI tract disease[72,73,75]. In a recent study, Durandet al[68]evaluated the sensitivity of quantitative PCR (qPCR) for plasma CMV deoxyribonucleic acid for the diagnosis of gastrointestinal CMV infections. Among 81 solid organ transplant recipients (liver and kidney), 20 endoscopic biopsy-proven cases of CMV of the gastrointestinal tract were identified. Overall, the sensitivity of qPCR for diagnosing CMV gastrointestinal tract disease was 85%, and the specificity was 95%. Interestingly, the sensitivity of qPCR in CMVseronegative recipients with CMV-seropositive donors (D+/R-) was 100%, while in CMV D+/R+ recipients, it was 72.3%, probably as a consequence of the difference in immune response[68]. Indeed, in CMV R-patients, gastrointestinal CMV disease is a consequence of primary infection with high-grade viremia. In contrast, in CMV R+ patients, gastrointestinal disease follows a reactivation of CMV that could be limited by pre-existing immunity[68].
Treatment of CMV colitis includes reduction of immunosuppression and the introduction of specific endovenous antiviral drugs, such as I.V. ganciclovir (5 mg/kg BID) for a period of 10-14 d until resolution of symptoms, followed by oral valganciclovir (900 mg once a day) for 3-6 mo. High-dose valganciclovir (up to 1800 mg twice a day) and/or foscarnet and cidofovir along with immunosuppression reduction may be a treatment option for CMV colitis with ganciclovir resistance[76].
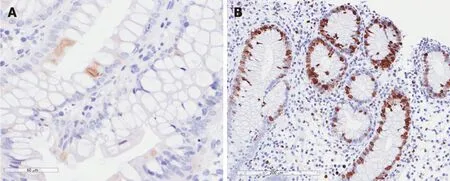
Figure 1 Cytomegalovirus colitis. A: Positivity for early cytomegalovirus antigen on immunochemistry; and B: Positivity for Ki-67 antigen on immunochemistry.
Mycophenolate mofetil-related colitis
Diarrhoea and other gastrointestinal symptoms are frequent complications after SOT, with an incidence of 12.6%, and up to 34% of cases may be related to the use of immunosuppressive drugs[19].
The introduction of MMF in the immunosuppressive regimen has led to a significant reduction in the incidence of acute rejection[1,61], although its use is associated with an increased rate of gastrointestinal complications[1,61]. It is a derivative of MPA, an antibiotic extracted fromPenicillium stoloniferum. After oral administration, it is hydrolysed into its active metabolite, MMF. MPA inhibits the type II isoform of inosine monophosphate dehydrogenase, a key enzyme in thede novosynthesis of purines, causing the depletion of guanine and deoxyguanosine nucleotides, inhibiting the proliferation of T and B lymphocytes and the formation of antibodies[77].
The most common adverse effect in kidney transplant patients is watery afebrile diarrhoea, with an incidence reaching 36% in renal transplant recipients[21,78], which may persist even after drug withdrawal[21,78].
The mechanism by which MMF induces changes in the gastrointestinal mucosa is unknown, but several hypotheses have been formulated. MMF could have a direct cytotoxic effect in reducing the presence of lymphocytes in the colon and the proliferation of enterocytes, which are partially dependent on the pathway ofde novosynthesis of purines, thus contributing to gastrointestinal toxicity, which occurs with diarrhoea. In the bowel, MMF may even cause apoptosis of lymphocytes activated following contact with luminal antigens[77-79]. In addition, MPA, having an antibacterial effect, may cause a change in the autochthonous flora of the gastrointestinal tract, which could promote the growth of anaerobic bacteria responsible for tissue damage[35]. Moreover, intestinal damage could be indirectly mediated by the immunosuppressive effect of MMF and the consequent modification of inflammatory responses[35,77-79].
Over the last few years, it has come to light that MMF can cause many gastrointestinal complications, and numerous studies have tried to determine whether MMF-related colitis shows typical histological features[5,6,13,35,51,60,61]. As MMF damage may be similar to that of IBD, it is essential to distinguish MMF colitis fromde novoIBD since the treatment and outcome are completely different, as this could avoid unnecessary reduction of immunosuppression[13,15,35,61].
MMF colitis is defined by the presence of gastrointestinal symptoms not otherwise related to any other aetiology, with endoscopic and histological features suggesting MMF colitis, and by marked improvement or resolution of symptoms with no treatment other than the discontinuation of MMF or a 50% reduction in the initial dose of MMF[13,61]. Diarrhoea is a common symptom in MMF colitis, and up to 76.5% of patients undergoing a colonoscopy for diarrhoea have histological features of MMF colitis[11,13,35]. MMF colitis commonly presents at a later stage after transplantation, usually after 4 years[11,35], although some authors reported that MMF colitis could present two years after transplantation[13]. Renal transplant recipients present a higher incidence of MMF colitis than other organ transplant recipients[35]. The reason for that is unclear, although it may be related to the heavier immunosuppression and higher doses of MMF required after renal transplantation than after other SOTs. Moreover, MMF toxicity is more severe when it is introduced later after transplantation, and it is more frequent in patients with higher serum creatinine concentrations[80]. Moreover, tacrolimus coadministered with MMF may significantly alter enterohepatic circulation, thereby increasing contact with intestinal cells and ultimately causing colitis[6,81]. In contrast, cyclosporin coadministered with MMF reduces the excretion of MPA metabolites, and could therefore reduce the incidence of gastrointestinal injury[82]. Finally, a significant improvement in MMF-related gastrointestinal symptoms was observed after replacement of MMF with mizoribine[83].
Common colonoscopic findings include erythema, erosions and ulcers, but half of patients have normal macroscopic findings[13,35,61].
The histological appearance of MMF colitis is characterized by the presence of architectural distortion, which resembles the appearance of chronic colitis. Therefore, the diagnosis of MMF colitis is based on specific histologic features and mainly on the exclusion of an alternative aetiology for these histological findings, such as acute colitis, IBD, and GVHD[13,35,59,60]. In their study, Selbstet al[61]found that MMF-induced changes were similar to those in IBDs (28%), GVHD (19%), acute colitis (16%), and ischaemia (3%). Similar findings were reported by de Andradeet al[6], who analysed 36 patients undergoing a colonoscopy for MMF-related diarrhoea: The most frequent histologic patterns were nonspecific colitis (31.3%), IBD-like colitis (25%), normal/near normal colitis (18.8%), GVHD-like colitis (18.8%), and ischaemia-like colitis (12.5%).
Liapiset al[13]evaluated colonic biopsies obtained from 43 renal transplant recipients with a clinical history of MMF administration and persistent afebrile diarrhoea. The main histological features were as follows: (1) Atrophy of the crypts assessed as absent, mild, moderate or severe; (2) Distortion of the crypts, cryptic abscesses, inflammatory infiltrates and changed numbers of eosinophils classified according to severity and extent; (3) Changed numbers of eosinophils defined as low if < 40 eosinophils per high pass filter (HPF) and high if > 40 eosinophils per HPF; and (4) Oedema, ulceration and crypts with flattened epithelium classified as absent or present.
The severity of colitis is estimated as absent, mild, moderate or severe based on the presence of eosinophils, cryptic abscesses and ulceration.
In summary, MMF colitis usually presents (1) irregular or extensive atrophy of the crypt; (2) alterations of the crypts in terms of cryptic abscesses, neutrophils, eosinophils and mucin inside the lumen of the crypt; (3) mild, moderate or severe inflammatory infiltrates, mainly plasma cells and in some cases eosinophils ( > 40 per HPF); and (4) focal cryptitis, ulcerations and erosions[5,6,13,60,61]. MMF colitis presents in a more severe form in the right colon than in the left colon, probably as a consequence of longer MPA exposure in the right colon and of the diminishing compound concentration in peripheral colonic segments[13,35]. Moreover, the degree of colitis is inversely correlated with the duration of MMF administration, such that a longer therapy is associated with moderate and severe degrees of colitis[13].
As reported by other studies[5,6,59-61], the histological characteristics of MMF colitis resemble those of IBD-like conditions and GVHD. However, compared with MMF colitis and IBD-like conditions, GVHD is characterized by a higher number of apoptotic bodies[6,60]and mild to moderate inflammation with mild or absent crypt distortion[13,60]. The IBD-like pattern is mainly characterized by moderate or severe crypt abnormalities with erosion and ulcerations[13].
Increased apoptosis of crypt epithelial cells plays an important role in the pathogenesis of MMF colitis. Increased cell apoptosis in association with crypt distortion irrespective of disease activity speaks in favour of a long-acting toxic effect of MMF attributed to its pharmacodynamics[5,13,60]. Increased cell apoptosis has been correlated with MMF colitis[5,13,60,61], although the absolute numbers of apoptotic cells were significantly higher in GVHD than in MMF colitis (P< 0.0001)[60]. The treatment of MMF colitis includes a reduction or, in more severe forms, complete discontinuation of MMF. A 50% dosage reduction[5,61]or switch to another immunosuppressant[6]usually results in a complete resolution or in a significant improvement of symptoms in most patients, while only a minority of patients require complete discontinuation of MMF[6,60].
De novo IBD
Although IBDs are characterized by a likely autoimmune etiopathogenesis, and transplanted patients are already under immunosuppression, the incidence of IBD after SOT (up to 550 cases/100000 individuals) is approximately 10 times higher than that observed among the general population (approximately 7-10 cases/100000 individuals)[5,21-23,71]. However,de novoIBDs after kidney transplantation are not common, with only 46 cases reported in the literature[15,19,20,22-24,26-34]. An additional 7 cases were reported by a French multicentric study[84]. Moreover, mostde novoIBDs occur in liver transplant recipients[16], with only 5% of SOT-related IBDs occurring in renal recipients[16]. Interestingly, a recent multicentre study of IBDs and kidney transplantation found no correlation between pre-existing autoimmune disease or immunosuppressive treatment and IBDs before or after transplantation[84].
De novoIBDs after transplantation usually present late in the follow-up, with a mean delay time to presentation up to 91 mo[5,15,19,20,22-31,84]. Clinical manifestations ofde novoIBDs resemble those occurring in the general population and include bloody diarrhoea and abdominal pain[5,15,19,20,22-32]. Endoscopic findings included patchy colitis, left-sided limited disease, pancolitis, and ileal disease suggestive of CD or UC[5,15,19,20,22-32]. The histological features ofde novoCD are expansion of the lamina propria by a dense lymphoplasmacytic infiltrate with basal plasmacytosis, crypt architectural distortion, and cryptitis[5,19], while UC usually presents with chronic active colitis limited to the rectum[5].
The course of post-transplant IBD appears much more aggressive than that of IBD in the general population, and it is associated with increased mortality and difficult therapeutic management, especially due to the possible interaction between immunosuppressive drugs and IBD-specific therapy[5,15,19,20,22-32,34]. Corticosteroids may induce clinical remission of the IBD, but they are unable to maintain it as monotherapy, probably because of their failure in causing apoptosis of mature T lymphocytes, which allows chronic and acute episodes of IBD exacerbation[85]. Studies on MMF have achieved contradictory results, as in some works, MMF was unable to maintain remission in patients with IBD[86], while others showed that its administration led to an improvement of symptoms[87]. Tacrolimus and cyclosporine, although highly effective in the prevention of acute rejection after transplantation, have been proven to be ineffective in the treatment of IBD[88], although recent observations in a nontransplant population suggest that tacrolimus could have a short-term clinical benefit in the management of IBD[47,48]. Conflicting results have been obtained even regarding the use of azathioprine in maintenance therapy. Timmeret al[89]demonstrated that azathioprine is less effective than sulfasalazine or mesalazine due to its likely side effects, including bone marrow suppression and consequent increased susceptibility to infections in already immunocompromised renal recipients. Azathioprine should be administered as maintenance therapy only in the case of failure of a mesalazine-based therapeutic regimen or in the case of a patient who requires repeated courses of steroids[89].
Among all reported cases ofde novoIBD, 16 occurred in kidney transplant recipients, who were successfully treated with conventional medical IBD therapy (mesalazine, cortico-steroids, and azathioprine) to achieve clinical remission. Approximately half of patients are resistant to conventional IBD therapy combined with immunosuppression[5,15,19,20,22-32].
Infliximab is a chimeric monoclonal IgG1 against tumour necrosis factor α that is used for steroid-resistant IBD. In the SOT setting, infliximab has been used in heart and liver transplantation to treat refractory IBD[31,90]. Temmeet al[31]reported a case of steroid-refractory UC successfully treated with infliximab. Infliximab was used at a dose of 5 mg/kg body weight at week 0, 2, and 4, followed by infusions every 8 wk. After completing the infliximab regimen, the stool frequency decreased, with endoscopic resolution of the colitis. Interestingly, the authors did not observe any deterioration of graft function[31]. More recently, Garrousteet al[34]reported the use of anti-tumour necrosis factor α antibodies in seven kidney transplant recipients withde novoIBD (5 patients with Crohn’s disease and 2 patients with UC). Three patients had complete remission, while in the other four patients, the disease recurred or progressed. There was no significant increase in the infection rate, and only one graft was lost. However, compared with the IBD group, in the non-IBD group, the use of infliximab resulted in a higher risk of death from infection. Although this approach has the potential to be a safe therapeutic option in patients refractory to standard therapy, the clinical experience is very limited, and other supportive data are required for this approach to be used safely in the kidney transplant setting. Approximately 20% of patients are refractory to therapy and ultimately need surgical treatment with colectomy[60,61].
OUTCOMES OF KIDNEY TRANSPLANTATION IN PATIENTS WITH INFLAMMATORY BOWEL DISEASE
The clinical course of IBD presents flares and remissions, with intestinal and extraintestinal manifestations, including kidney impairment, with AA amyloidosis and IgA nephropathy as the most common diagnoses[91,92]; these complications may even result in renal insufficiency requiring kidney transplantation[90,92,93], although renal failure is a rare complication, especially in patients with CD[94]. Age and duration of IBD are not risk factors for developing renal failure[95]. There is an association between oxalate nephropathy and IBD since the prevalence of calcium oxalate urolithiasis is up to five-fold higher in patients with CD than in the general population[96]. Moreover, CD seems to be a likely predisposing factor for haemolytic-uremic syndrome because of recurrent gastrointestinal tract infections[97]. Very few studies have reported the outcome of kidney transplantation in patients with IBD who develop end-stage renal disease[33,90,93]. In one study, a total of 21 patients with IBD (12 patients with CD and 6 patients with UC, as well as 3 patients with disease not otherwise defined) received kidney transplantation[33,31,90,93]. An additional 28 cases were reported in a French multicentric study[85].
Schnitzleret al[92]reported 6 patients with IBD (5 patients with CD and 1 patient with UC) who received a kidney transplant. The female/male ratio was 5/1, and the mean age was 54.1 years. Three patients received IBD treatment before transplantation (mainly 5-ASA and steroids) and underwent ileocecal resection and fistula surgery. At the time of kidney transplantation, all patients were in clinical remission. Three patients required IBD treatment after transplantation (two CD patients were treated with steroids and 6-MP and one UC patient was treated with 5-ASA and steroids). Interestingly, among the three patients requiring treatment after transplantation, two were treated before kidney transplantation. One patient developed a post-transplant lymphoproliferative disorder, and one developed kidney graft cancer. At a median follow-up of 112.5 mo, all patients were alive, and only one patient required retransplantation[92].
In the series of Grupperet al[90], 12 patients with IBD (7 patients with CD and 5 patients with UC) received kidney transplantation. Kidney transplantation was more frequent in males than in females, and the median age was 48.4 years. IgA nephropathy and autosomal dominant polycystic kidney disease were the most common causes of end-stage renal disease. When compared with that in a matched control group, the rate of late rehospitalization was significantly higher in the IBD group. Moreover, patient survival was significantly lower in patients with IBD, with an estimated 5-year patient survival of 80.8%vs96.8% for patients with and without IBD, respectively, with a hazard ratio for the risk of death with a functioning graft of 1.41. However, the death-censored graft survival of the IBD group was comparable with that of the non-IBD group[90]. The increased risks of rehospitalization and death were related to an increased incidence of infections, probably as a consequence of the worse nutritional status among IBD patients than non-IBD patients, as demonstrated by lower BMIs and haemoglobin levels, or as a consequence of the higher chronic immunosuppression status because of IBD-related treatments[90]. Interestingly, most patients remained in clinical remission or did not experience response deterioration after transplantation[90,92], probably as a consequence of the heavier immunosuppression in the kidney transplant population than in the liver transplant population, in which the rate of recurrence is higher[92]. However, in patients in whom IBD recurred after transplantation, the median time to flare-up after transplantation was 17 mo, and CMV infection increased the risk of recurrence[85].
CONCLUSION
Gastrointestinal diseases are common after kidney transplantation and may present with a variety of clinical and histological features. The diagnosis and management of IBD after transplantation are challenging since there are no definitive histological criteria to clearly diagnose post-transplant IBD. Indeed, many histological features may be common between different clinical forms, such as mycophenolate mofetil colitis with Graft-versus-host disease, and this could render the treatment controversial.
De novoIBD after renal transplantation should be part of the differential diagnosis in patients with chronic diarrhoea and abdominal pain, even without a previous history of gastrointestinal disease, along with infectious causes, drug-related side effects, or other comorbidities. Management of post-transplant IBD can be challenging due to the contemporary use of immunosuppressive therapy, which can increase the risk of infectious complications. Moreover, the clinical course of post-transplant IBD may be more severe than that of IBD in the general population. A better definition of clinical and histological features could help to standardize the treatment and to improve the outcome of IBD after transplantation. Due to the clinical complexity of IBD patients, a close multidisciplinary approach is necessary to achieve the best clinical outcomes of IBDs after kidney transplantation.
 World Journal of Gastroenterology2020年38期
World Journal of Gastroenterology2020年38期
- World Journal of Gastroenterology的其它文章
- Role of betaine in liver disease-worth revisiting or has the die been cast?
- Management of an endoscopy center during the outbreak of COVID-19: Experience from West China Hospital
- Is vitamin D receptor a druggable target for non-alcoholic steatohepatitis?
- Acetyl-11-keto-β-boswellic acid inhibits proliferation and induces apoptosis of gastric cancer cells through the phosphatase and tensin homolog /Akt/ cyclooxygenase-2 signaling pathway
- Endogenous motion of liver correlates to the severity of portal hypertension
- Longitudinal decrease in platelet counts as a surrogate marker of liver fibrosis
