喷昔洛韦-铜Ⅱ配合物的合成、晶体结构及DNA键合作用
刘瑞雪 赖德霖 邓前军 程风杰 范兰琼 刘延成*,
(1广西师范大学化学与药学学院,省部共建药用资源化学与药物分子工程国家重点实验室,桂林 541004)
(2佛山科学技术学院材料科学与能源工程学院,佛山 528000)
0 Introduction
Nucleoside drugs are a series of therapeutic agents for viral diseases.In particular,the successful development of the antiviral drug,acyclovir,marked the opening of a new era for anti-herpes virus drugs[1].Nucleoside drugs are classified into the following categories:antiretroviral drugs,anti-herpes virus drugs,anti-cytomegalovirus drugs and so on[2-3].As the acyclovir derivative,penciclovir (PCV)is one of the most successful nucleoside drugs[4-6],which isdeveloped by SmithKline Beecham of U.S.It is the metabolic product of the precursor drug of acyclovir actually,and it can inhibit the proliferation of the herpes simplex virus,herpeszoster and EBviruseffectively[5,7].Its action mechanism is similar to acyclovir,but with the increased cell accumulation and survival rate,which can maintain the antiviral activity effectively.Meanwhile,the side effects are reduced obviously.PCV can also block the normal band-lymphocyte lesions caused by EB virus.Therefore,it can be assumed that PCV not only exhibits a significant effect on the herpes virus[8-10],but also shows certain resistance to the lymphatic cancer.
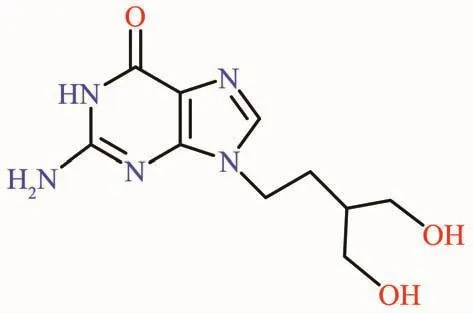
Scheme 1 Chemical structure of PCV
Cancer has endangered the health of human beings seriously since the 20th century.It is generally accepted that cancer is caused by the disorder of cell growth and proliferation mechanisms[11].Most of the antitumor drugs were primarily exert their antitumor mechanisms by preventing the synthesis of DNA,RNA and protein,or by acting on these large molecules directly[12].Therefore,nucleotide drugs can be considered potential anticancer drugs,although they are not yet fully applied to the cancer treatment.
On the other hand,the discovery of cisplatin as new metal-based anticancer drug and its clinical application have explored a new and broad field of anticancer drugs development[13-15].More and more metal complexes have been synthesized and designed in recent years.As an indispensable element of human body,copperⅡhas attracted an increasing number of researchers to explore its potential biological applications from its metal complexes,especially for its potential anticancer activity,since quite some copperⅡcomplexes were found to be significantly higher cytotoxic against some human tumor cell lines[16-18]. Herein we selected PCV as a typical nucleotide drug targeting DNA,and combined it with copperⅡby coordination chemistry method,to obtain the first metal complex of PCV,[Cu(PCV)2(H2O)3]SO4(1).This new complex 1 was structurally characterized and was screened on its in vitro antitumor activity.Its DNA binding property with CT-DNA was also discussed by means of typical experimental methods.
1 Experimental
All the starting materials and the reaction solvents were commercial available with reagent grade and were used without further purification.PCV was purchased from Yuancheng Co.Ltd.of Hubei Province.CuSO4·5H2O and the reaction solvents were all purchased from Xilong Co.Ltd.of Guangdong Province.Calf thymus DNA (CT-DNA),MTT,Tris and PBSwere all purchased from Sigma-Aldrich.
1.1 Synthesis of complex 1
Penciclovir (PCV) (0.2 mmol,0.050 6 g)and CuSO4·5H2O (0.15 mmol,0.037 5 g)were put into a Pyrex glass tube,adding to 1 mL of mixed solvent of ethanol and water (3∶1,V/V).Thereaction mixturewas frozen by liquid N2,and then the tube was vacuumed and sealed by a torch.Then it was placed in 80℃in the dark for 48 h,before it was gradually cooled down to roomtemperature.The green crystalswere harvested,which were determined to be the title complex 1(Yield:66%).The green stick crystals suitable for X-ray single crystal diffraction analysis was selected to determine the structure.IR (KBr,cm-1):3 400,2 931,2 375,1 666,1 633,1 487,1 413,1 384,1 120,1 033,859,776,689,619,510.Anal.Calcd.for C20H42CuN10O16S(%):C,31.00;H,5.42;N,18.08.Found(%):C 29.77,H 5.68,N 17.93.
1.2 X-ray structure analysis
The data collection of single crystal was carried out on a Rigaku Mercury CCDequipped with a graphite monochromated Mo Kα radiation (λ=0.071 073 nm).The structures were solved with direct methods and refined using SHELX-97[19]programs.Thenon-hydrogen atoms were located in successive difference Fourier synthesis.The final refinement was performed by fullmatrix least-squares methods with anisotropic thermal parametersfor non-hydrogen atomson F2.Thehydrogen atoms were added theoretically and riding on the concerned atoms.No H atoms associated with guest water molecules were located from the difference Fourier map.The crystallographic data and refinement details of the structure analyses were summarized in Table 1 and Table 2.
CCDC:1825089.
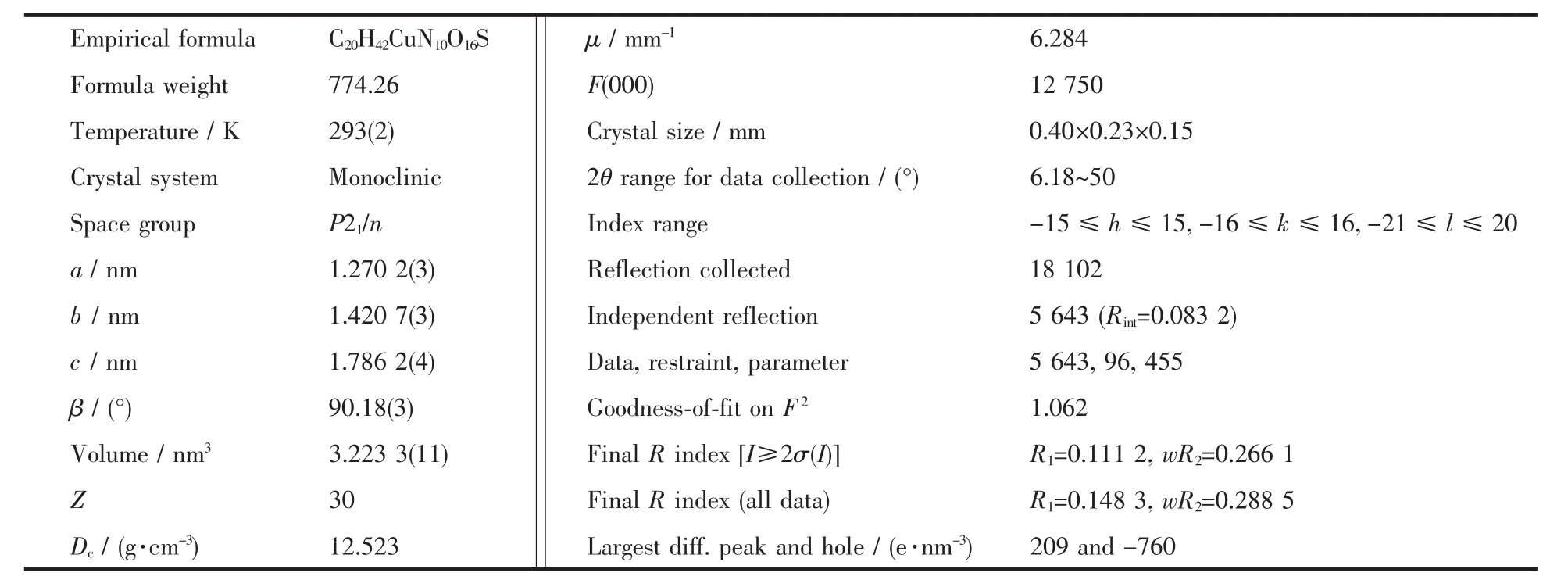
Table 1 Crystal data and structural refinement parameters for complex 1
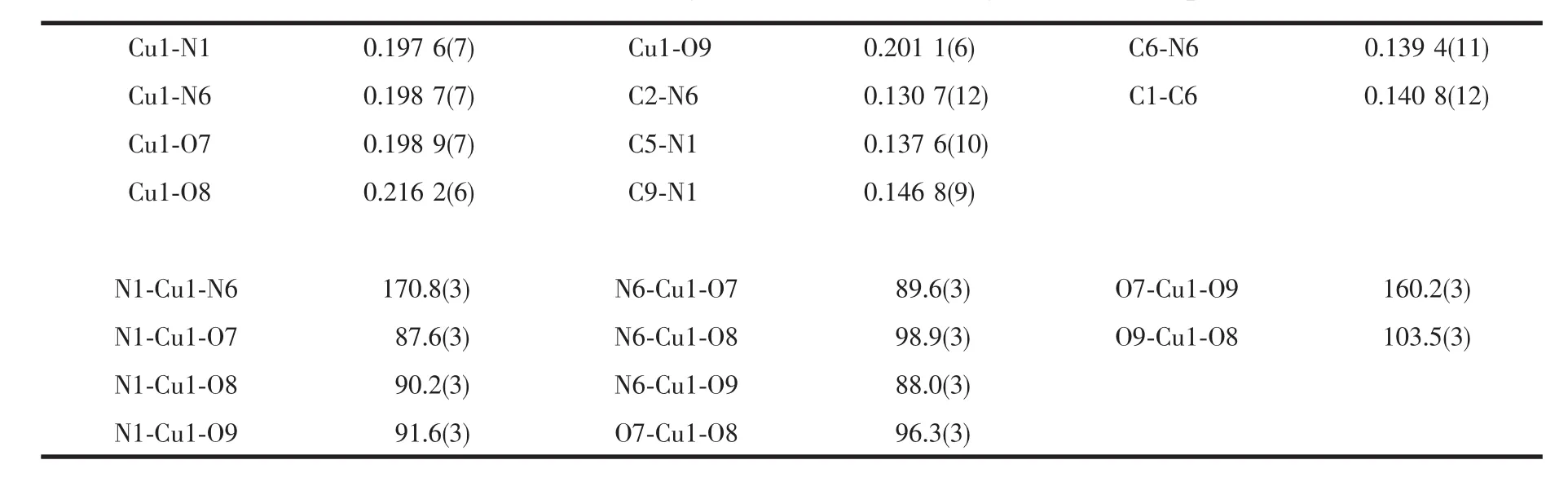
Table 2 Selected bond lengths(nm)and bond angles(°)for complex 1
1.3 In vitro cytotoxicity
The human tumor cell lines,including BEL-7404,SGC-7901,HeLa,MCF-7 and A549,were obtained from the Shanghai Cell Bank of Chinese Academy of Science.
The title complex,complex 1,compared with PCV as positive control,were dissolved in DMSO to prepare the 5.00 mmol·L-1DMSO stock solutions.Each cell was maintained with the medium supplemented containing the fetal bovine and other essential medium in order to guarantee a good state of survival before it was tested.Then they were transferred to 96-well plants as far as possible to ensure the number of cells in each well plant was similar.After 24 h of continuous culturing in the 37℃incubator,cells growth reached 70%in the well.Then each compound was added to the wells,which has been diluted with the phosphate buffer saline(PBS)solution already,and then proceeded to culture for 48 h in 5% (V/V)CO2humidified atmosphere.After 48 h of cultivation,0.1 mg of MTT (dissolved in 20 μL PBS)was added to each well.Finally,the formed blue formazan crystals were dissolved in 100μL of DMSO,and the absorbance was measured using the enzyme-labeled reading instrument with 490 nm/630 nm double wavelength.The growth inhibition ratio for each cell line under the concentration of 20 μmol·L-1of complex 1 or PCV was evaluated according to the absorbance,ultimately.
1.4 UV-Vis absorption and fluorescence spectral analysis
Tris-NaCl buffer solution(which included 5 mmol Tris,50 mmol NaCl,and was adjusted to pH=7.3 by hydrochloric acid)was prepared using double distilled water.The 5.00 mmol·L-1DMSO stock solution of complex 1 was used for DNA binding studies.It was diluted by Tris buffer to prepare the corresponding working solutions with the concentration of 20 μmol·L-1,in which the final DMSO concentration was no more than 1%.CT-DNA was also dissolved in Tris buffer solution to give a final concentration of 2.00 mmol·L-1,and it was kept at 4 ℃ for no more than 5 days before using.
The UV-Vis absorption spectroscopy was a kind of classic technique to study the interaction between small molecules and DNA.The absorbance of each sample was recorded on a PerkinElmer Lambda45 UV-Visible Spectrophotometer,in the wavelength range from 200 to 700 nm.The fluorescence spectra were recorded on a Shimadzu RF-5301/PC spectrofluorophotometer,in the wavelength range from 350 to 700 nm,with slit width of 15 nm.The optimized excited wavelength (λEx)of the fluorescence emission was determined to be 340 nm.
1.5 DNA viscosity experiment
DNA viscosity experiment was recorded on a Brookfield DV-Ⅱpro digital viscometer equipped with TC-502D thermostatic bath.A 20 mL of CT-DNA solution (1.80 mmol·L-1)was prepared in Tris buffer as a working solution for DNA viscosity experiment.The stock solution of complex 1 was added according to the gradual increasing ratios for c1/cDNAfrom 0.01 to 0.09.The circulating bath temperature was maintained at(35.0±0.5)℃.The viscosity value,η (mPa·s),was obtained by running the spindle in working samples at 30 r·min-1via the equipped ULA adapter.The data were presented directly as plot ofηversus c1.The effect of DMSO on the viscosity was eliminated based on its 100%viscosity value of 2.03 mPa·s measured at 35.0℃.

Scheme 2 Synthetic route of complex 1
2 Results and discussion
2.1 Synthesis and structural characterization of complex 1
As shown in Scheme 2,the synthetic reaction for the title complex,complex 1,has been stated in the experimental section.The product yielded green crystals.The complex was characterized by IR,elemental analysis,and its coordination geometry was determined by X-ray singlecrystal diffraction analysis.
As shown in Fig.1,the crystal structure of complex 1 shows a distorted trigonal bipyramid coordination mode,in which two PCV ligands coordinate to the central CuⅡvia the N atom of imidazole,and occupied the axial positions of the bipyramid,respectively.Three H2O molecules occupy the equatorial plane of the trigonal bipyramid via the coordinated O atoms.The Cu-N bond lengths are 0.197 6(7)nm (Cu1-N1)and 0.198 7(7)nm (Cu1-N6),respectively,while the angle of N1-Cu1-N6 is 170.8(3)°.The Cu-O bond lengths are 0.198 9(7)nm(Cu1-O7),0.216 2(6)nm (Cu1-O8)and 0.201 1(6)nm(Cu1-O9),respectively.Each coordinating unit showed+2 positive charged,and one sulfate anion (SO42-)existed in each unit as the counter-ion.
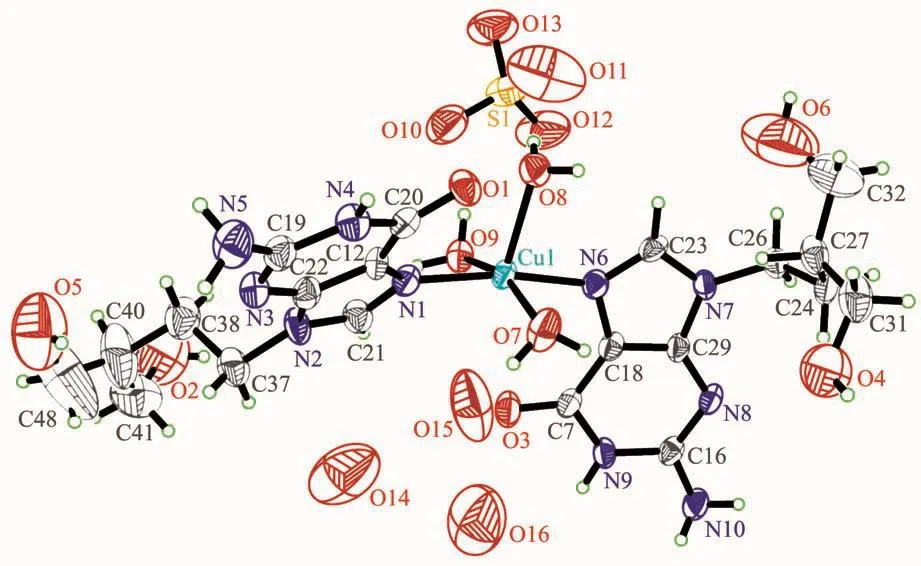
Fig.1 ORTEPpresentation of the crystal structure of complex 1 in a 50%ellipsoid
2.2 In vitro cytotoxicity screening
The in vitro cytotoxicity wastested asapreliminary screening by MTT assay,towards a series of tumor cell lines,including the A549,BEL-7404,SGC-7901,HeLa and MCF-7.The screening concentration for complex 1,PCV and CuSO4·5H2O was equal to 20 μmol·L-1.The results were listed in Table 3.
Viewed from the data of the screening test,complex 1 showed higher growth inhibition abilities than PCV alone in terms of all these tested tumor cell lines.The inhibition ratio of PCV did not exceed 50%under 20 μmol·L-1.Comparatively,the inhibition ratios for complex 1 toward all the tumor cell lines were higher than 50%.The growth inhibition ratios of complex 1 were higher than PCV in the range from ca.20%to 220%.Especially,the growth inhibition ratios for complex 1 against BEL-7404 and A549 were about 2 and 3 times of those for PCV,respectively.Furthermore,CuSO4·5H2O as the raw metal salt was found to cytotoxic selectively on the BEL-7404 and SGC-7901 tumor cell lines.However,it is much less cytotoxic against the other three tumor cell lines.It should be noticed that CuSO4alone can′t function as an effective antitumor agent.So PCV might be also regarded as an appropriate carrier to bring CuⅡinto the cells to exert its bioactivity.In all,it suggested that there is the positive synergistic effect when combining the PCV and CuⅡtowards some typical tumor cell lines,such as HeLa,MCF-7 and A549.The enhancement on the antitumor activity of complex 1 might be ascribed to the combination of PCV and CuⅡ,where the central CuⅡ plays a key role[16-18].This is of positive significance for exploring new applications on PCV as new type of drugs.
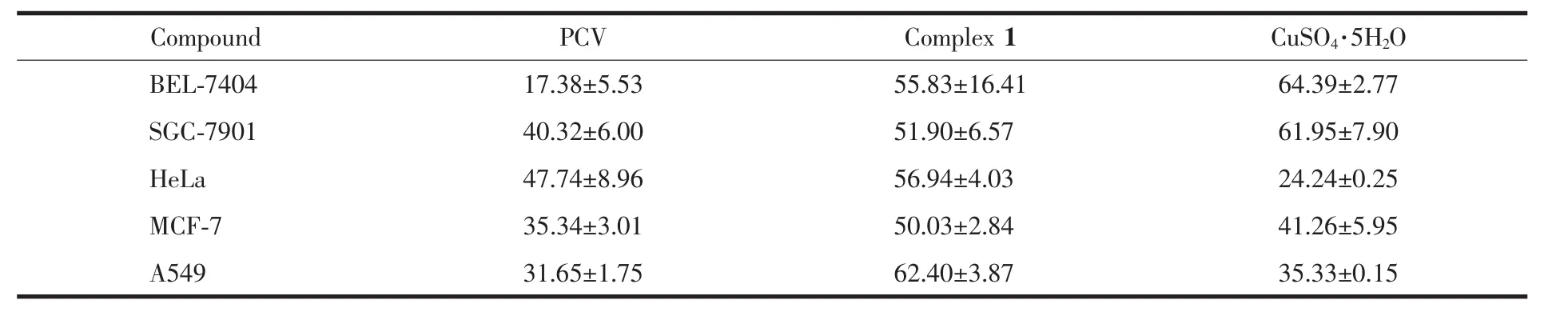
Table 3 Cell growth inhibition ratios induced by PCV,complex 1 and CuSO4·5H 2O towards five typical human tumor cell lines%
2.3 DNA binding analysis
DNA is widely recognized as a target for many antitumor drugs.The mechanism of interaction between small molecules and DNA has been studied for several decades,so not only it is necessary to study the DNA binding mechanism of new compounds,but also the study has important significance for designing DNA-targeting drugs.Therefore,the DNA binding property of complex 1 was studied by UV-Vis,fluorescence spectroscopy,and DNA viscosity experiment,which are the most classic methods for the DNA binding study.
2.3.1 UV-Vis absorption analysis
The UV-Vis absorption spectral analysis was firstly applied to study the DNA binding property of the complex.As shown in Fig.2,the UV-Vis spectrum of complex 1 in the absence of CT-DNA (dashed line)showed the maximum absorption peak at 252 nm,which corresponds to theπ→π*electronic transition of 1.With the increasing amounts of CT-DNA,it was found that the maximum absorbance at 252 nm decreased by gradient (solid lines).When the DNA concentration reached 1.6×10-4mol·L-1,the maximum absorbance (Amax)of complex 1 decreased from 0.422 to 0.369,with a decreasing ratio of 12.6%.It suggested an intercalative binding mode for complex 1 to DNA,since the small molecules intercalated between the neighboring base pairs of DNA might weaken theπ→π*electronic transition and lead to obvious hypochromic effect.However,comparing with the binding constant of EtBr,which is the classic DNA intercalator,the intercalative binding ability of complex 1 to DNA might be not strong.
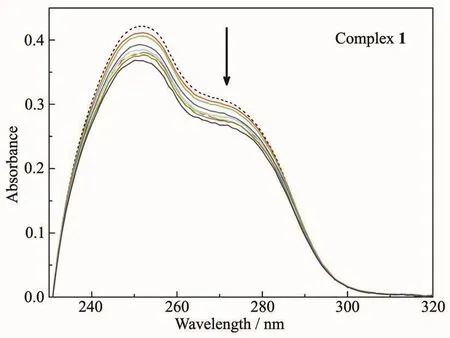
Fig.2 UV-Vis absorption spectra of complex 1 in the absence (dashed line)and presence (solid lines)of CT-DNA with increasing concentration ratios(c DNA/c1)in the range from 1∶1 to 8∶1
2.3.2 Fluorescence spectra analysis
Fluorescence quenching is a phenomenon of the reduction on the fluorescence intensity caused by the interaction between the fluorescent molecules and the quenching molecules. In water solution, the fluorescent molecules may be quenched by H2O molecules.But when they intercalated between the neighboring base pairs of DNA,they might be protected by the hydrophobic circumstance of DNA,so that their fluorescence would not be quenched.
As shown in Fig.3,in the absence of CT-DNA,the fluorescence emitted of complex 1 at 20 μmol·L-1was very weak.The maximum fluorescent emission peak was at 470 nm,with fluorescence intensity of I=45.4,asindicated by the dashed line.After the addition of increasing amounts of CT-DNA,it was found that the fluorescence intensity of complex 1 enhanced obviously.The fluorescence intensity enhanced to I=136.52 when the cDNA/c1=1∶1,which was ca.3 times of complex 1 alone.When the concentration of DNA reached 1.4×10-4mol·L-1,with cDNA/c1=7∶1,the fluorescent intensity of complex 1 enhanced to 487.3,which was more than 10 times of complex 1 alone.And then the fluorescent intensity of this complex tended to be saturated with further addition of DNA.It further suggested that complex 1 might bind DNA by an intercalative mode,so that its fluorescent emission was effectively prevented from quenching by H2O.Herein,we assumed that the central CuⅡplayed a key role in facilitating the DNA binding ability of PCV.
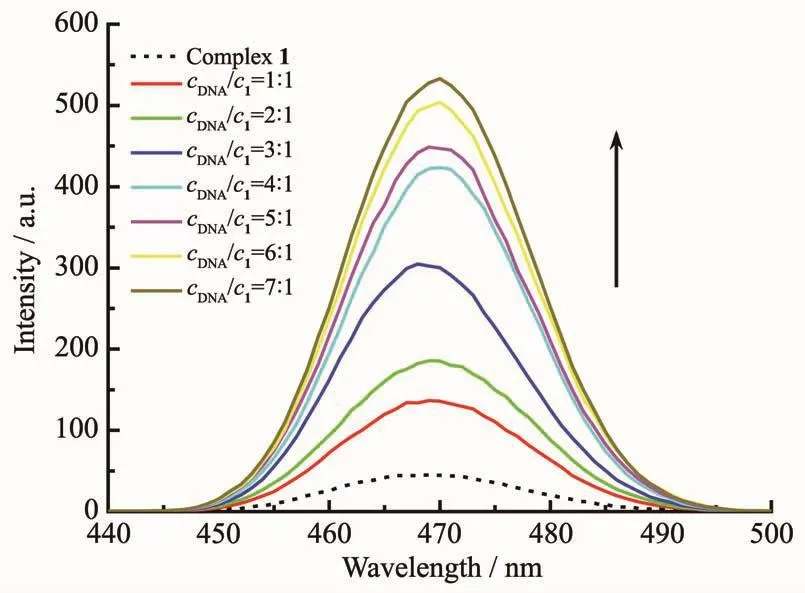
Fig.3 Fluorescent emission spectra of complex 1 in the absence and presence of CT-DNA with increasing concentration ratios (c DNA/c1)in the range from 1∶1 to 7∶1
2.3.3 DNA viscosity experiment
When the small molecules intercalated between the neighboring base pairs of DNA,the viscosity of the DNA solution will increased due to the lengthening of the DNA double helix.So the DNA viscosity experiment was generally considered to be a more direct method to determine the DNA binding mode of small molecules,which might be possible to further confirm the interaction mechanism of complex 1 with DNA.
As shown in Fig.4,the viscosity of the DNA solution alone was measured to be η0=3.495 mPa·s,when the rotational speed was 50 r·min-1and the temperature of solution was 30℃.However,with the increasing amounts of complex 1 added,the viscosity of the DNA solution increased significantly.When the ratio of c1/cDNAreached 0.09,the viscosity increased to η=3.660 mPa·s,with η/η0=1.05.So it also suggested that complex 1 should bind with CT-DNA via a classic intercalative mode,resulting in the significant enhancement on the viscosity of the DNA solution.

Fig.4 Viscosity measurement of CT-DNA solution under the addition of increasing concentrations of complex 1,with the ratios of c1/c DNA in the range from 0.02 to 0.09
3 Conclusions
In this work,a new copperⅡcomplex of the antivirus drug,penciclovir(PCV),was synthesized and structurally characterized.To our knowledge,this is the first metal complex of penciclovir reported.Furtherly,its in vitro antitumor activity was screened,which indicated that the antitumor activity towards several typical tumor cell lines was significantly improved when PCV coordinated with CuⅡ.Subsequent studies strongly suggested that the complex could bind with CT-DNA via an intercalative binding mode,even though it was not as strong as the classic DNA intercalator,EtBr.This was supported by the experimental results,including the hypochromicity from the UV-Vis spectra and the increase on the fluorescence intensity of the complex under the addition of CT-DNA,as well as the enhancement on the viscosity of the DNA solution when bound with the complex.It suggested that the CuⅡplayed an indispensable role in the complex.In addition,considering the bioactivity of CuⅡitself,it provided an approach for designing and developing new type of drugs from penciclovir.And the further studies on its antiviral activity towards HSV might help to explore new metal-based antiviral drugs.
Acknowledgements:This work was supported by the National Natural Science Foundation (Grant No.21561005),the Natural Science Foundation of Guangxi Province (Grant No.2017GXNSFDA198048),the Open Foundation of Characteristic Key Subject on Materials of Guangdong Province (Grant No.2017-13)and the Talents′Small Highland in Medicinal Industry of Guangxi (Grant No.1405/1505).

