Effects of morroniside on oxidative stress and cardiomyocyte apoptosis in rats with acute myocardial infarction
, , , , ,
(1.Key Laboratory of Basic Pharmacology of Ministry of Education and Joint International Research Laboratory of Ethnomedicine of Ministry of Education,Zunyi Medical University,Zunyi Guizhou 563099,China; 2.Experimental Animal Center,Xuanwu Hospital of Capital Medical University,Beijing 100053,China)
Cardiovascular disease is the leading cause of morbidity and mortality worldwide,and myocardial infarction (MI) remains the major cause of death in the developed world and a major pathology worldwide[1-2]. Acute myocardial ischemia is mainly caused by myocardial necrosis due to severe and persistent acute hypoxia and ischemia[3].In the early stage of myocardial infarction,myocardial tissues is imbalanced between oxidation and antioxidation due to hypoxia and ischemia,leading to cardiomyocyte apoptosis and oxidative stress.Myocardial apoptosis and oxidative stress can accelerate ventricular remodeling and increase the risk of death in the chronic phase of myocardial infarction[3-5].Current treatment methods for acute myocardial infarction (AMI) include thrombolytic and interventional therapies.Interventional therapies for AMI include percutaneous coronary intervention and coronary artery bypass grafting.However,these methods cannot prevent severe left ventricular dysfunction and transition to heart failure in patients with MI[6].
Myocardial tissue pathology after AMI is characterized by the imbalance of apoptosis and the expression of oxidative stress products[3].Hypoxia is a pessimal stimulus to myocardial tissue,and the imbalance between reactive oxygen species (ROS) production and antioxidant defenses further leads to oxidative stress.MDA is the product of oxidative stress,and indirectly reflects the degree of injury inflicted by oxygen free radicals.SOD,which acts as an enzymatic antioxidant,can scavenge superoxide anion radicals and indirectly reflects the ability of an organism to scavenge free radicals.Oxidative stress affects various pathological processes,as well as destroys macromolecules involved in energy production.Its finally induces apoptosis of myocardial cells and depresses cardiac function[4-5].Myocardial apoptosis after AMI not only affects the infarct size,but also contributes to ventricular remodeling[7].Therefore,the inhibition of oxidative stress and protection of myocardial cells against apoptosis are crucial for improving cardiac function.
Morroniside is one of the most abundant iridoid glycosides extracted fromCornusofficinalis,a member of the Cornaceae family.C.officinalissarcocarp is referred to as Shangzhuyu in Chinese traditional medicine.Our previous studies have showed that morroniside exerts protective effects such as reducing infarct size,inhibiting oxidative stress and apoptosis on ischemic cerebrovascular diseases[8-10].In the present study,we tested the potential protective effect of morroniside against AMI in rats and determined whether its protective effect is associated with antioxidation and antiapoptosis mechanisms.
1 Materials and methods
1.1 Drug preparation Morroniside was extracted from the sarcocarp ofC.officinalisand purified as previously described[10].C.officinaliswas purchased from Tong Ren Tang Company,Beijing,China.The final purity of the extracted morroniside was 98.5% as determined through high performance liquid chromatography (HPLC) analysis.
Metoprolol was used as the positive drug and was procured from AstraZeneca ,Shanghai,China.
1.2 Animals Male Sprague-Dawley (SD) rats weighing 260-280 g were purchased from Beijing Vital River Experimental Animal Co.(Beijing,China) and placed in a controlled environment under specific-pathogen-free (SPF) conditions.The animals had free access to standard chow and water for the entire duration of the study.
1.3 Animal model All animals were subjected to acute myocardial ischemia by permanent left anterior descending (LAD) coronary artery ligation as previously described[11-12].Before AMI model construction,the rats were subjected to 12h of fasting without water deprivation.Animals were anesthetized with enflurane,and a ventilator was connected to assist respiration.The LAD coronary artery was permanently ligated with 6-0 silk suture through the third intercostal space incision.Lungs were reinflated using positive end-expiratory pressure,and the chest wall was sutured layer by layer.Regional ischemia was confirmed by the whitening of the region under the suture and ST segment elevation on the electrocardiograph (ECG).The sham-operated model was constructed similarly as the AMI model but without ligation.
The rats were randomly divided into six groups:Sham group,AMI model group,AMI+45 mg/(kg·d) morroniside group,AMI+90 mg/(kg·d) morroniside group,AMI+180 mg/(kg·d) morroniside group,and AMI +10 mg/(kg·d) metoprolol group.
Morroniside and metoprolol were dissolved in distilled water and administered intragastrically once a day after AMI.The AMI model and sham groups received an equal volume of distilled water.
1.4 2,3,5-triphenyltetrazolium chloride staining For the description of MI,hearts were stained with 2,3,5-triphenyltetrazolium chloride (TTC) (Sigma-Aldrich,St.Louis,MO,USA) through a previously described method[13].Three days after AMI,rats were euthanized by heart injection with 1 ml of 0.2 mol/L potassium chloride (KCl) after anesthetization.Hearts were cut freehand into transverse slices and incubated in a 2% solution of TTC in phosphate buffer for 15 minutes at 37 ℃ and pH 7.4 in the dark.The percentage of TTC-stained tissue isolated from whole heart tissue was analyzed by using Image J software (NIH,USA).
1.5 Myocardial tissue preparation Three days after AMI,rats were anesthetized with 10% chloral hydrate (0.35 ml/100 g).The myocardium was isolated from the MI border zone homogenized in RIPA Lysis Buffer (phenylmethyl sulphonyl fluoride 20 mmol/L),and centrifuged at 12 000×gfor 30 min at 4 ℃.Supernatants were then collected.
1.6 Myocardial oxidation level detection MDA and SOD levels were detected using a suite of commercial kits in accordance with the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute,Nanjing,China).
1.7 Western blot analysis Protein supernatants were assayed with a bicinchoninic acid (BCA) protein assay kit (Beyotime Biotechnology,Shanghai,China).Total proteins were separated via sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE).After electrophoresis,samples were transferred onto pure nitrocellulose blotting membranes.The membranes were blocked in 5% nonfat dry milk in TBS-Tween 20 (0.1%) prior to overnight incubation at 4 ℃ with the following primary antibodies:rabbit anti-GAPDH (1∶1 000),rabbit anti-Bax (1∶1 000),rabbit anti-Bcl-2 (1∶1 000),rabbit anti-Caspase-3 (1∶500),or rabbit anti-Caspase-8 (1∶500).Rabbit anti-GAPDH antibody,rabbit anti-Bax antibody,rabbit anti-Bcl-2 antibody,and rabbit anti-Caspase-8 antibody were procured from Cell Signaling Technology,Beverly,MA,USA.Rabbit anti-Caspase-3 antibodies were procured from Abcam,Cambridge,MA,USA.The membranes were washed,and the appropriate secondary peroxidase-conjugated antibody (ZSGB-BIO,Beijing,China) was applied for 2h at room temperature.The bound antibodies were subjected to chemiluminescence visualization (Super Signal,Pierce).
1.8 Statistical analysis Data were expressed as the mean±standard error of mean (SEM); n was the number of animals in each experimental group.The statistical significance was calculated by one-way ANOVA and LSD multiple comparison test.A value ofP<0.05 was considered statistically significant.
2 Results
2.1 Effect of morroniside on infarct size 3 days after AMI To determine the protective effects of morroniside against AMI in rats,the infarct size of myocardial tissue was examined through TTC staining.As shown in Figure 1,infarct sizes in rats treated with morroniside at the doses of 90 mg/(kg·d) and 180 mg/(kg·d) significantly decreased from 18.72% to 13.02% and 10.42% ,respectively,3 days after LAD ligation relative to those in AMI rats (P<0.05,P<0.01).Infarct size in rats treated with metoprolol at the dose of 10 mg/(kg·d) significantly decreased from 18.72% to 10.32% relative to that in AMI rats (P<0.01).These results indicated that morroniside exerts protective effects against AMI in rats.

A:Representative image of TTC staining for infarct size in the left ventricle; B:Quantitative analysis of infarct size.Values are expressed as mean ± SEM,n=8.*P<0.05,***P<0.01 vs.AMI model group.Infarct sizes were determined through TTC staining.Fig 1 Effects of morroniside on infarct size in the AMI rat model
2.2 Effect of morroniside on oxidative stress 3 days after AMI To investigate whether the protective mechanisms of morroniside on AMI are associated with oxidative stress,MDA and SOD levels in heart tissues were examined.As shown in Figure 2A,compared with that in the sham group,the amount of MDA,which is an index of lipid peroxidation,significantly increased (P<0.01) in the AMI model group.Compared with that in AMI rats,MDA levels in rats treated with morroniside at the dose of 180 mg/(kg·d) or metoprolol at the dose of 10 mg/(kg·d) significantly decreased from 13.32 nmol/mgprot to 10.34 nmol/mgprot and 10.98 nmol/mgprot (P<0.01,P<0.01).As shown in Figure 2B,compared with that in the sham group,the activity of the antioxidant enzyme SOD decreased significantly (P<0.01) in the AMI model group.Compared with that in the AMI model group,SOD activity in rats treated with morroniside at the doses of 90 mg/(kg·d) and 180 mg/kg·d or metoprolol at the dose of 10 mg/(kg·d) increased from 119.63 U/mgprot to 132.77 U/mgprot,140.14 U/mgprot and 137.21 U/mgprot,respectively (P<0.05,P<0.01,P<0.01).

A:MDA level in each experimental group at 3 days after AMI; B:SOD activity in each experimental group at 3 days after AMI.Values are expressed as mean ± SEM,n=6.##P<0.01 vs.sham group; and *P<0.05,**P<0.01 vs.AMI model group.Fig 2 Effects of morroniside on oxidative stress indexes (MDA and SOD) in heart tissues after AMI
2.3 Effect of morroniside on apoptosis-related protein expression 3 days after AMI To establish whether the mechanisms underlying the protective effects of morroniside against AMI are associated with apoptosis,the expression levels of the apoptosis-related proteins Bax,Bcl-2,cleaved-Caspase-3 and Caspase-8 were detected through Western blotting analysis.As shown in Figure 3,compared with those in the sham group,Bax protein expression significantly increased and Bcl-2 protein expression significantly decreased in the AMI model group (P<0.01,P<0.01).Treatment with morroniside at the dose of 45 mg/(kg·d) and metoprolol at the dose of 10 mg/(kg·d) suppressed Bax expression in AMI rats (P<0.01,P<0.01).Bcl-2 protein expression significantly increased in AMI rats compared with that in rats treated with morroniside at the doses of 45 mg/(kg·d) and 90 mg/(kg·d) or metoprolol at the dose of 10 mg/(kg·d) (P<0.05,P<0.01,P<0.01).The ratio of Bax/Bcl-2 in the morroniside-treated group and metoprolol-treated group also decreased significantly (P<0.01).As shown in Figure 4,compared with those in the sham group,cleaved-Caspase-3 and Caspase-8 protein expression levels significantly increased in the AMI model group (P<0.01,P<0.01).Treatment with morroniside at the dose of 90 mg/(kg·d) and metoprolol at the dose of 10 mg/(kg·d) decreased cleaved-Caspase-3 protein expression in AMI rats (P<0.01,P<0.01).Treatment with morroniside at the dose of 180 mg/(kg·d) and metoprolol at the dose of 10 mg/(kg·d) decreased cleaved-Caspase-8 protein expression in AMI rats (P<0.05,P<0.01).

A:Bax,Bcl-2 protein expression levels as detected through immunoblotting; B:Quantitative analysis of Bax/Bcl-2 expression; C:Quantitative analysis of Bax expressed as a fraction of the respective levels of GAPDH; D:Quantitative analysis of Bcl-2 expressed as a fraction of the respective levels of GAPDH.Values are expressed as mean ± SEM,n=4.##P<0.01 vs.sham group; and *P<0.05,**P<0.01 vs.AMI model group.Fig 3 Effects of morroniside on Bax and Bcl-2 protein expression in heart tissues 3 days after AMI
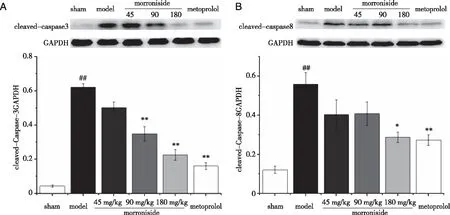
A:Cleaved-Caspase-3 protein expression levels as detected through immunoblotting.Quantitative analysis of cleaved-Caspase-3 expressed as a fraction of the respective levels of GAPDH; B:Cleaved-Caspase-8 protein expression levels as detected through immunoblotting.Quantitative analysis of cleaved-Caspase-8 expressed as a fraction of the respective levels of GAPDH.Values are expressed as mean ± SEM,n=4.##P<0.01 vs.sham group; and *P<0.05,**P<0.01 vs.AMI model group. Fig 4 Effects of morroniside on cleaved-Caspase-3 and Caspase-8 protein expression in heart tissues 3 days after AMI
3 Discussions
Cardiovascular disease has become prevalent with the aging of the population and the acceleration of urbanization.AMI,a major ischemic heart disease,is a grave threat to human health.Its pathological characteristics are myocardial vascular stenosis and blockage,which further lead to myocardial infarction.At present,the main therapies for AMI are thrombolytic therapy and percutaneous coronary intervention (PCI)[6].However,the recovery of ischemic myocardial blood flow may lead to myocardial reperfusion injury,which could reduce treatment efficacy[14].Drugs that protect the myocardium from ischemic injury are potential treatments for ischemic heart disease.
In previous studies,we found that morroniside,a primary active ingredient extracted from the Chinese herbC.officinalis,acts as a protective agent of nerve function in an oxidative stress-induced neurotoxic cell model and a focal cerebral ischemia rat model[8,9,15].Cerebrovascular disease and cardivascular disease have similar pathological basis and treatment methods.Patients with cerebrovascular disease often have cardiovascular disease.However,the effects of morroniside on the regulation of oxidative stress and Caspase families related factors of apoptosis signaling pathway in AMI have not been reported.Here,we first identified the antioxidative stress effect of morroniside on AMI in rats,and first demonstrated its antiapoptosis effect of inhibiting the activation of Caspase families.The result of TTC staining showed that morroniside treatment could decrease infarct size 3 days after AMI.This result indicated that morroniside has a protective effect against myocardial ischemia.
Oxidative stress is involved in ventricular remodeling and subsequent heart failure after AMI[16-17].A large amount of oxygen free radicals produced by the ischemic myocardium damage the structure and disrupt the function of myocardial cells,as well as destroy macromolecules,such as lipids,chondriosome,proteins,DNA,and enzymes,involved in energy production.Eventually these effects accelerate myocardial cell death through apoptosis and necrosis[18].MDA is an end-product of oxidative stress,and high MDA levels can cause irreversible damage to the heart[19].The extent of myocardial injury induced by the increase in oxygen free radicals can be assessed on the basis of the activity of the endogenous antioxidant enzyme SOD in myocardial tissues.SOD acts as the most efficient enzymatic antioxidant by scavenging superoxide anions,preventing cellular damage,and maintaining the balance of oxidation and antioxidation[20].In the present study,we quantified MDA levels and SOD activity to study the probable antioxidative mechanism underlying the protective effects of morroniside against AMI.
After AMI,cardiomyocyte apoptosis or necrosis,occurs and ultimately decreases the number of cardiac myocytes.Infarct size is mainly determined by apoptosis.Myocardial cells are not renewable,and the recovery of cardiac function after AMI is closely related to the number of healthy myocardial cells.Therefore,reducing cardiomyocyte apoptosis is crucial for improving cardiac function after AMI[21-23].The endogenous pathway,also known as mitochondrial pathway,mediates apoptosis in cardiomyocytes.Bcl-2 family proteins are important modulators of the mitochondrial pathway,and are classified as antiapoptotic or proapoptotic.Bcl-2,which acts as antiapoptosis gene,can promote cell survival and inhibit apoptosis.Bax is a member of the proapoptotic in the Bcl-2 family.When apoptosis is induced,Bax migrates from the cytoplasm to the mitochondria and the nuclear membrane.When Bcl-2 is highly expressed,two heterogenous polymers of Bax/Bcl-2 are formed to inhibit apoptosis[23].Caspase families play a key regulatory role in cell apoptosis,and the activation of Caspase-8 is the first step.Cleaved-Caspase-8 is involved in apoptosis and induces Caspase-3 activation.Cleaved-Caspase-3 is a protease involved in enzymatic hydrolysis,which is downstream of the apoptotic pathway,and acts as the final effector molecule of apoptosis[24].We found that morroniside can reduce apoptosis 3 days after AMI by regulating the expression of Bax,Bcl-2,cleaved-Caspase-3 and Caspase-8 proteins.
Our study demonstrated that the ischemia and hypoxia of myocardial tissue increase oxidative stress and decreases antioxidant activity after AMI,as indicated by MDA levels and SOD activity.However,morroniside decreased MDA production and increased SOD activity to levels observed in sham-operated rats,suggesting that morrroniside could scavenge endogenous oxygen free radicals to inhibit oxidative stress and consequently prevent myocardial tissue from oxidative damage.We also found that compared with those in the sham group,the protein expression levels of Bax,cleaved-Caspase-3 and Caspase-8 in ischemic myocardial tissue increased,and Bcl-2 protein expression decreased in the AMI model group.However,Bax,cleaved-Caspase-3 and Caspase-8 protein expression levels decreased,and Bcl-2 protein expression level increased after morroniside treatment.These results indicated that morroniside can reduce apoptosis by regulating the protein expression levels of Bax,Bcl-2,Bax/Bcl-2,cleaved-Caspase-3 and Caspase-8.
In conclusion,this study showed that morroniside has a protective effect on myocardial cells three days after AMI.Its protective mechanism is related to the inhibition of oxidative stress and the reduction of apoptosis.Further studies and long-time observations to define the protective effects of morroniside through antioxidants and antiapoptosis mechanisms are warranted.
[References]
[1] Adams M R.Prevention of myocardial infarction[J].Int Med J,2002,32(12):595-600.
[2] Sivakumar R,Babu P V,Shyamaladevi C S.Aspartate and glutamate prevents isoproterenol-induced cardiac toxicity by alleviating oxidative stress in rats[J].Exp Toxicol Pathol,2011,63(1-2):137-142.
[3] Chen H G,Xu Y F,Wang J Z,et al.Baicalin ameliorates isoproterenol-induced acute myocardial infarction through iNOS,inflammation and oxidative stress in rat[J].Int J Clin Exp Pathol,2015,8(9):10139-10147.
[4] Sun L,Hao Y,Nie X,et al.Construction of PR39 recombinant AAV under control of the HRE promoter and the effect of recombinant AAV on gene therapy of ischemic heart disease[J].Exp Ther Med,2012,4(5):811-814.
[5] Song Y H,Li B S,Chen X M,et al.Ethanol extract from Epimedium brevicornum attenuates left ventricular dysfunction and cardiac remodeling through down-regulating matrix metalloproteinase-2 and -9 activity and myocardial apoptosis in rats with congestive heart failure[J].Int J Mol Med,2008,21(1):117-124.
[6] Ng R,Yeghiazarians Y.Post myocardial infarction cardiogenic shock:a review of current therapies[J].J Intensive Care Med,2013,28(3):151-165.
[7] Liang T,Zhang Y,Yin S,et al.Cardio-protecteffect of qiliqiangxin capsule on left ventricular remodeling,dysfunction and apoptosis in heart failure rats after chronic myocardial infarction[J].Am J Transl Res,2016,8(5):2047-2058.
[8] Wang W,Huang W,Li L,et al.Morroniside prevents peroxide-induced apoptosis by induction of endogenous glutathione in human neuroblastoma cells[J].Cell Mol Neurobiol,2008,28(2):293-305.
[9] Liu T T,Xiang B X,Guo D Y,et al.Morroniside promotes angiogenesis and further improves microvascular circulation after focal cerebral ischemia/reperfusion[J].Brain Research Bulletin,2016,127:111-118.
[10] Wang W,Sun F L,An Y,et al.Morroniside protects human neuroblastoma SH-SY5Y cells against hydrogen peroxide-induced cytotoxicity[J].European Journal of Pharmacology,2009,613(1-3):19-23.
[11]Gao E,Lei Y H,Shang X,et al.A novel and efficient model of coronary artery ligation and myocardial infarction in the mouse[J].Circ Res,2010,107(12):1445-1453.
[12]Zhu F,Li Y,Zhang J,et al.Senescent cardiac fibroblast is critical for cardiac fibrosis after myocardial infarction[J].Plos One,2013,8(9):e74535.
[13]Vivaldi M T,Kloner R A,Schoen F J.Triphenyltetrazolium staining of irreversible ischemic injury following coronary artery occlusion in rats[J].Am J Pathol,1985,121(3):522-530.
[14]Yellon D M,Hausenloy D J.Myocardial reperfusion injury[J].N Engl J Med,2007,357(11):1121-1135.
[15]Sun F L,Wang W,Cheng H,et al.Morroniside improves microvascular functional integrity of the neurovascular unit after cerebral ischemia[J].Plos One,2014,9(6):e101194.
[16]de Castro A L,Tavares A V,Campos C,et al.Cardioprotective effects of thyroid hormones in a rat model of myocardial infarction are associated with oxidative stress reduction[J].Mol Cell Endocrinol,2014,391(1-2):22-29.
[17]Schenkel P C.Time profile of hydrogen peroxide/thioredoxin balance and its influence in the intracellular signaling post-myocardial infarction[J].Exp Physiol,2012,97(6):741-749.
[18]Bagatini M D,Martins C C,Battisti V,et al.Oxidative stress versus antioxidant defenses in patients with acute myocardial infarction[J].Heart Vessels,2011,26(1):55-63.
[19]Zhou R,He L F,Li Y J,et al.Cardioprotective effect of water and ethanol extract of Salvia miltiorrhiza in an experimental model of myocardial infarction[J].Journal of Ethnopharmacology,2012,139(2):440-446.
[20]Mo X,Zhao N,Du X,et al.The protective effect of peony extract on acute myocardial infarction in rats[J].Phytomedicine,2011,18(6):451-457.
[21]Fragasso G,Spoladore R,Cuko A.Modulation of fatty acids oxidation in heart failure by selective pharmacological inhibition of 3-ketoacyl coenzyme-A thiolase[J].Curr Clin Pharmacol,2007,2(3):190-196.
[22]Abbate A,Bussani R,Amin M S.Acute myocardial infarction and heart failure:role of apoptosis[J].Int J Biochem Cell Biol,2006,38(11):1834-1840.
[23]Tan L,Pan N,Yu L,et al.The effects of fasudil at different doses on acute myocardial infarction in rats[J].Acta Cardiol Sin,2013,29(6):524-530.
[24]Liu Q.Lentivirus mediated interference of Caspase-3 expression ameliorates the heart function on rats with acute myocardial infarction[J].Eur Rev Med Pharmacol Sci,2014,18(13):1852-1858.

