Crystal lattice free volume in a study of initiation reactivity of nitramines:Friction sensitivity
Svtopluk Zemn,Ning Liu,Mrel Jungová,Ahmed K.Hussein,Qi-long Yn
aInstitute of Energetic Materials,Faculty of Chemical Technology,University of Pardubice,CZ-532 10,Pardubice,Czech Republic
bXi'an Modern Chemistry Research Institute,Xi'an,Shaanxi,710065,China
cSchool of Astronautics,Northwestern Polytechnical University,Xi'an 710072,Shaanxi,China
1.Introduction
Over the last 30 years,the main interest in initiation of high energy materials has been focused on studies of shock and impact sensitivities[1,2].In comparison with these,no similar attention has been paid to the friction sensitivity(FS)of such materials.We have dealt with FS intensely over the last five years[3-8].Our experience shows that the results of the FS determination can be heavily in fluenced by “human variability”.Nevertheless,careful measurements[3]by a single researcher provide results showing relationships which correlate with the output of other sensitivity parameters[4-8],with15N NMR chemical shifts of the key nitrogen atoms in the reaction center of the molecule[7]or with the DFT calculation outputs[8].Therefore,it should not be without interest to use in the FS outputs analysis recent knowledge about free spaces in crystal lattice of energetic materials(EMs),ΔV,and about their in fluence on the EMs'impact sensitivity[9-13].Recently we have analyzed a relationship between theΔV values and impact sensitivity of eighteen nitramines[13].In the present paper,which can be taken as a continuation of those studies[4-8],we use the same approach in analyzing similar relationships for friction sensitivity of thirteen nitramines.
2.Data sources
2.1.Nitramines under study
Chemical names,code designations and impact sensitivity,expressed as friction energy of the nitramines studied are summarized inTable 1.For a better illustrative view,structural formulas of these nitramines are presented in Scheme 1.
2.2.Friction sensitivity
The friction sensitivity of all the nitramines studied were determined[4,7]by means of the BAM friction test apparatus operated under standard test conditions[15]with evaluation of the output by Probit analysis[16](only the normal force at which 50%of initiations occur is reported in Table 1).

Table 1A review of the nitramines studied and their friction sensitivity(FS)from papers[4,7].
2.3.Results of calculation for crystal lattice free volume of nitramine explosives
The object molecules were optimized at computational level of B3LYP/6-311+g(d,p)by using the Gaussian 09 package[17].All of the optimized structures were characterized to be true local energy minima on the potential energy surfaces without imaginary frequencies.The crystal volume[V(0.003)]was calculated by using the Multiwfn[17-19]software.The effective volume per molecule(Veff)is calculated as
where M is molecular mass,and d is crystal density.The intrinsic gas phase molecular volume(Vint)is calculated by the 0.003 au surface according to Ref.[13],Vint=V(0.003).Therefore,the free space per molecule(ΔV)is
Results of these calculations are summarized inTable 2,together with crystal densities of the nitramines studied.
3.Results and discussion
In papers[4,7]we found a semi-logarithmic relationship between impact and friction sensitivity of the nitramines which isdivided into a number of partial relationships in close correlation with the molecular structure characteristics of these compounds[7].If we also take into account our findings from paper(impact sensitivity versus theΔV values)[13],it should be clear that friction sensitivity is in a semi-logarithmic relationship with the crystal lattice free volume per molecule,ΔV,of the nitramines studied,as shown in Fig.1.
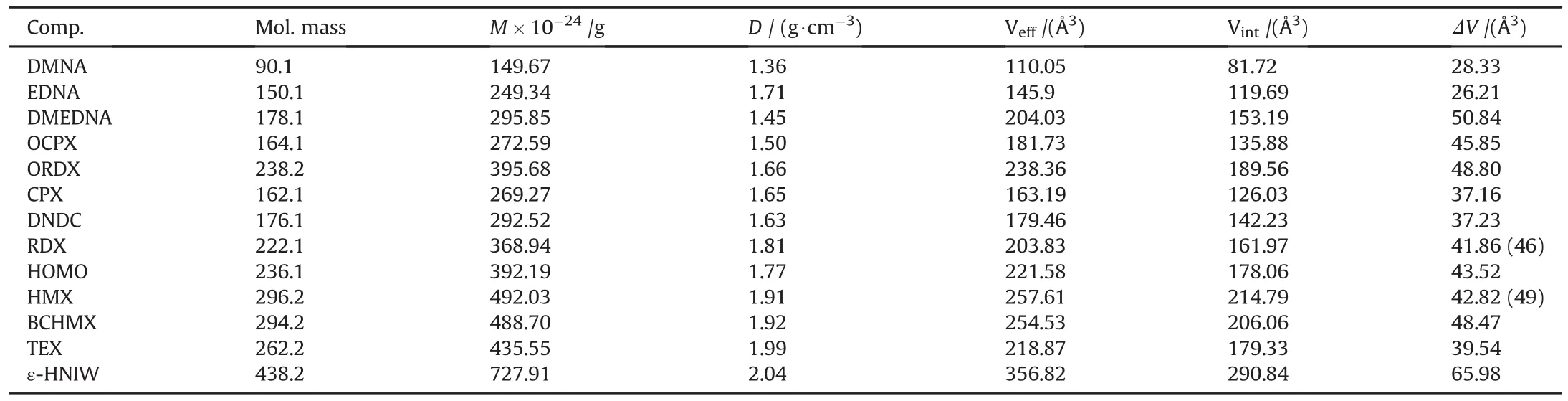
Table 2A survey of the molecular mass,M,crystal densities,d,effective volume per molecule,Veff,intrinsic gas phase volume,Vint,and free space per molecule,ΔV.
This relationship is broken into the partial straight lines which are strongly limited by the molecular structure similarity.Only straight lines D and F correspond to the expected trend of increase in friction sensitivity with the increase in theΔV values;in all the nitramines,the data for which are associated with these lines,it is possible to find the molecular skeleton of DMEDNA(or part of it).
Data for compounds which can theoretically be generated from the DMNA molecule are associated with the straight lines B and C.The OCPX molecule,whose data lie at the straight line A,is as if an“opened”molecule of 1,3-dinitroimidazolidine(CPX)in whose skeleton the ethylenedinitramine grouping can be found.
The position of ε-HNIW in Fig.1 is interesting.Its data correlate well with those of DMEDNA and EDNA using the straight line E.Comparison with the nitramines associated with the straight line D clearly shows that the difference between ε-HNIW and its RS-analogue(product with reduced sensitivity[14])rests in the difference in the intensity and uniformity ofintermolecular interactions in their crystals.From this comparison it seems as if in the ε-HNIW crystals only the nitro groups in positions 2,4,6,and 8 had a major part in the intermolecular force in its crystals-in so doing,the most reactive nitramino grouping is in position 2 of this particular nitramine[1,7,20-22].
The above-mentioned disintegration of the nitramine groups in Fig.1,namely differences between compositions of groups with positive and negative slopes of the corresponding straight lines,on the first look is connected with kind and character of intermolecular interactions in crystals of these compounds.Interpretation of these facts needs another study by means the quantum chemical approaches in combination with X-ray spectroscopy.
For all the nitramines studied a direct proportionality exists between their intrinsic gas phase volume,Vint(V(0.003)),and corresponding friction sensitivity,as shown in Fig.2.This is understandable because the friction force during shearing is proportional to the contact area.Also here it is possible to observe a limitation by molecular structure similarity.In this case,the difference in both kinds of HNIW is good visible by comparison of the straight lines I and J,with the same possible explanation as in the case of Fig.1.In each case this comparison shows that ε-HNIW(technical or“common”quality)gives the impression of disorder in the distribution of the forces in its crystal lattice in comparisonwith its RS or chemically pure analogue[13].
Already mentioned usage of the 15N NMR chemical shifts in specification of the key nitrogen atoms in the reaction center of nitramine molecules[7]led to Fig.3 like to the one from corresponding outputs.Here a skeleton of DMEDNA(better of ethylene-1,2-diimine)seems to be a dominating-directional structure in these relationships.
It is interestingthat the composition and sequence of nitramines associated with straight line I in Fig.2 are the same as in the corresponding partial relationship between FS and the15N NMR chemical shifts of nitrogen atoms in the most reactive nitro groups of these nitramines(see straight line A in Fig.3)[7].The oxygen atoms of nitro groups,by their dipole-dipole interactions,contact the oxygen and nitrogen atoms of nitro groups in neighboring nitramine molecules in the crystal[24-26],which is the decisive factor governing the crystal structure of nitramines.The type of interaction mentioned will act against the shear slide during friction.Aza atoms(i.e.bearers of these nitro groups)in fluence the mutual orientations of nitro groups in neighboring molecules by means of conformation of molecular skeletons.This conformation has a determining in fluence on the crystal lattice free volume.As aza atoms are “inner”atoms of the nitramines'molecular skeleton,their effect on the intermolecular potential will be lower than that of the nitro groups attached to them[7].Therefore,the crystal lattice free volumes should have a similar in fluence on this potential.This has already been shown in the case of the relationship between impact sensitivityand crystal lattice free volumes inpaper[13].
4.Conclusion
The relationship between friction sensitivity(FS,shear slide with fixed volume)and the crystal lattice free space per molecule,ΔV,of the nitramines studied is described by a linear equation which is divided into a number of partial relationships with a strong limitation created by the molecular structure of such compounds.It is not possible to say clearly that increasingΔV values leads to increasing FS and vice versa.It is possible to see some similarity with the in fluence of aza atoms on FS[7].These atoms,as part of the molecular skeleton of nitramines,in fluence the mutual orientations of nitro groups in neighboring molecules by means of conformation of the nitramine molecules and thus determine simultaneously the crystal lattice free space per molecule.Also,in friction sensitivity,the dominating in fluence is thus the dipoledipole interaction on the base of mutual contacts of the oxygen and nitrogen atoms of nitro groups in neighboring nitramine molecules.In concordance with this interaction,a directly proportional relationship was found between FS and the intrinsic gas phase molecular volume,Vint,of the nitramines mentioned,which is divided into several straight lines according to relatively tight molecular structure similarity.Both types of friction sensitivity relationships found again con firm unbalance in distribution of the forcesin the crystallattice ofthe “common” quality of ε-2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane, in comparison with its RS or chemically pure analogue.
Acknowledgement
The work described in this paper partially received financial support from the Students Grant Projects No.SGSFCHT_2016002 of the Faculty of Chemical Technology at the University of Pardubice,partially it was created in the framework of the six month traineeship of Dr.LIU Ning in Institute of Energetic Materials at University of Pardubice in 2016 under financial support of the Chinese State Administration of Foreign Experts Affairs.
Appendix A.Supplementary data
Supplementary data related to this article can be found at https://doi.org/10.1016/j.dt.2018.01.001.
[1]Zeman S.Sensitivities of high energy compounds.In:Klapoetke T,editor.High energy density compounds,structure&bonding,vol.125.Heidelberg,Germany:Springer;2007.p.195-271.
[2]Zeman S,Jungova M.Sensitivity and performance of energetic materials.Propellants,Explos Pyrotech 2016;41(3):426-51.
[4]Jungová M,Zeman S,Husarová A.Friction sensitivity of nitramines.Part I:comparison with impact sensitivity and heat of fusion.Chin J Energetic Mater 2011;19(6):603-6.
[5]Jungová M,Zeman S,Husarová A.Friction sensitivity of nitramines.Part II:comparison with thermal reactivity.Chin J Energetic Mater 2011;19(6):607-9.
[6]Zeman S,Jungová M,Husarová A.Friction sensitivity of nitramines.Part III:comparison with detonation performance.Chin J Energetic Mater 2011;19(6):610-2.
[7]Jungová M,Zeman S,Husarová A.New aspect of friction sensitivity of nitramines.J Energetic Mater 2012;VII(1(14)):13-8[in Persian].
[8]Friedl Z,Jungová M,Zeman S,Husarová A.Friction sensitivity of nitramines.Part IV:links to surface electrostatic potentials.Chin J Energetic Mater 2011;19(6):613-5.
[11]Politzer P,Murray JS.Impact sensitivity and crystal lattice compressibility/free space.J Mol Model 2014;20:2223.https://doi.org/10.1007/s00894-014-2223-7.
[12]Politzer P,Murray JS.Some molecular-crystalline factors that affect the sensitivities of energetic materials:molecular surface electrostatic potentials,lattice free space and maximum heat of detonation per unit volume.J Mol Model 2015;21:1-11.
[13]Zeman S,Liu N,Jungová M,Hussein AK,Yan Q-L.Crystal lattice free volume in a study of initiation reactivity of nitramines:impact sensitivity,Defence Technology-MS No.DT_2017_147.
[14]ElbeihA,HusarováA,ZemanS.Methodofpreparationofepsilon-2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane with reduced impact sensitivity.U.S.Pat.9,227,981 B2.Univ.of Pardubice;Jan.5,2016.
[15]Su′ceska M.Test methods for explosives.Heidelberg,Germany:Springer;1995.
[17]Frisch MJ,Trucks GW,Schlegel HB,Scuseria GE,Robb MA,Cheeseman JR,et al.Gaussian 09,Revision B.01.Wallingford,CT,USA:Gaussian,Inc.;2009.
[18]Lu T,Chen F.Multiwfn:a multifunctional wave function analyzer.J Comput Chem 2012;33(5):580-92.
[19]Lu T,Chen F.Quantitative analysis of molecular surface based on improved Marching Tetrahedra algorithm.J Mol Graph Model 2012;38:314-23.
[20]Zeman S.Analysis and prediction of the arrhenius parameters of lowtemperature thermolysis of nitramines by means of the15N NMR spectroscopy.Thermochim Acta 1999;333:121-9.
[21]Zeman S.New aspect of initiation reactivity of energetic materials demonstrated on nitramines.J Hazard Mater 2006;132:155-64.
[22]Zeman S.Study of the initiation reactivity of energetic materials.Chapter 8.In:Armstrong RW,Short JM,Kavetsky RA,Anand DK,editors.Energetics science and Technology in Central Europe.College Park,Maryland:CECDS,University of Maryland;2012.p.131-67.
[23]Ou Y,Chen Y,Chi Y,Ma Q,Wang J,Huang J,et al.Synthesis of 1-amino2,4-dinitroimidazole optimized by online infrared spectroscopy and its energetic properties.ChemPhysChem 2017;82:287-94.
[24]Atovmyan LO,Golovina NI,Zolotoy AB,Zhitomirskaya NG,Fedorov BS,Eremenko LT.Structure and packing of primary and secondary nitro amines.Z Organicheskoy Khimii 1998;24(9):1848-56.
[25]Krebs B,Mandt J,Cobbledick RE,Small RWH.The structure of N,N-dimethylnitramine.Acta Crystallogr B 1979;35:402-4.
[26]Filhol A,Bravic G,Rey-Lafon M,Thomas M.X-Ray and neutron studies of a displacive phase transition in N,N-dimethylnitramine(DMN).Acta Crystallogr B 198(36):575-586.
- Defence Technology的其它文章
- Evaluating location specific strain rates,temperatures,and accumulated strains in friction welds through microstructure modeling
- Crystal lattice free volume in a study of initiation reactivity of nitramines:Impact sensitivity
- The effects of compressibility and strength on penetration of long rod and jet
- A DFT study on TNGU isomers and aluminized cis-TNGU composites
- Numerical simulation and optimized design of cased telescoped ammunition interior ballistic
- The kinetic of mass loss of grades A and B of melted TNT by isothermal and non-isothermal gravimetric methods

