Influences of process parameters on solvent-free toluene oxidation over Au/γ-MnO2 catalyst
Jiang Feng Xiao Guomin
(School of Chemistry and Chemical Engineering, Southeast University, Nanjing 211189, China)
Influences of process parameters on solvent-free toluene oxidation over Au/γ-MnO2catalyst
Jiang Feng Xiao Guomin
(School of Chemistry and Chemical Engineering, Southeast University, Nanjing 211189, China)
γ-MnO2nanorobs and Au/γ-MnO2catalysts were synthesized and characterized by theX-ray powder diffraction (XRD), the scanning electron microscope (SEM), and transmission electron microscope (TEM). The characterizations show that Au particles are well dispersed on the surface of γ-MnO2nanorobs with a particle size of about 10 nm. The catalytic performance is evaluated in solvent-free toluene oxidation with oxygen. The influences of several process parameters such as reaction time, reaction temperature, initial oxygen pressure and catalyst amounts on the catalytic performance are studied. Catalytic results reveal that Au/γ-MnO2catalyst has a unique selectivity to benzaldehyde and all these factors greatly influence the conversion of toluene and selectivity of bezaldehyde, benzoic acid and benzyl benzoate. However, these factors have slight influence on the selectivity of benzyl alcohol.
nano-gold; manganese dioxide; toluene oxidation; solvent-free
Toluene oxidation has received more and more attention, since all the oxidation products, such as benzyl alcohol, benzaldehyde, benzoic acid, and benzyl benzoate, are commercially significant versatile intermediates in the manufacture of pharmaceuticals, dyes, solvents, perfumes, plasticizers and preservatives[1]. Industrially, a homogeneous catalyst and a halogenic or acidic solvent are applied to toluene oxidation for high conversion, but the use of such solvents and homogeneous catalyst cause pollution and waste[2]. Thus, a solvent-free method for toluene oxidation catalyzed by a heterogeneous catalyst has attracted attention and been studied in recent years. Mn3O4, MnO2and Au-Pd/C catalysts have shown to be effective in the solvent-free oxidation of toluene to benzoic acid, benzaldehyde and benzyl benzoate reaction[3]. These findings suggest that noble metals and manganese oxides are good candidates for solvent-free toluene oxidation.
Manganese oxides are well known as materials for clean energy storage due to their low material cost, acceptable environmental characteristics, and chemical stability. Many studies have focused on the modification of their crystal forms, morphology and particle size. In particular, three crystal forms, i.e., α (2×2 tunnel structure), γ (1×1 and 1×2 tunnel structure), and δ (layer structure), that are called manganese oxide octahedral molecular sieves (OMS)[4]. The basic unit of OMS is MnO6octahedra which can share their edges and vertices to generate a wide range of periodic structures.
The Au nanoparticle has been widely used as catalyst in three kinds of reactions: CO oxidation, alkene epoxidation, and selective oxidation of alkanes. Many factors are known to affect Au activity, such as particle size and geometric structure, the type of support, methods of preparation, calcination, and the presence of trace compounds. Supported gold catalyst demonstrates excellent catalytic performance, and the supports reported include zirconia[5], cerium oxide[6], iron oxide[7], magnesium oxide[8], and titanium oxide[9]. Therefore, it is a good choice to synthesize a catalyst using nanosized Au particles as active centers and manganese (IV) oxide as support.
In our previous study[10], we synthesized three kinds of MnO2nanostructures (α-, γ-, δ-MnO2) by the redox method. Among them, γ-MnO2has a suitable morphology and surface area for the use of support, which also shows high activity in toluene oxidation. To continue our study, in this work, Au/γ-MnO2catalyst is prepared by a technique of in situ reduction. Then, process parameters such as reaction time, temperature, initial oxygen pressure and catalyst amount are studied in solvent-free toluene oxidation over Au/γ-MnO2catalyst.
1 Experimental
The γ-MnO2nanostructure was prepared according to the procedure of our previous work by the redox reaction of Mn2+and Mn7+[10]. Au/γ-MnO2catalyst was prepared by an in situ reduction method. Chloroauric acid and ascorbic acid were used as precursors and reductants, respectively. At 80℃, the required amount of aqueous chloroauric acid was added to the PVP aqueous solution (0.7‰ g/mL, 300 mL) by dripping. After stirring, 0.9 g of dispersed MnO2powder was added into the solution. Six hours later, 30 mL aqueous ascorbic acid (0.01 mol/L) was dropped into the mixture and stirred for another 1 h. Finally, the product was obtained by filtration and washed several times with deionized water until the filtrate did not react with AgNO3solution. Then absolute alcohol was used to remove any residual reactants. Finally, the product was dried at 60℃ for 16 h. The mole percentage of Au loading was 1.0%.
The XRD characterization of MnO2and Au/MnO2samples was carried out on a Bruker D8 FocusX-ray diffractometer using CuKαradiation and a nickel filter (λ=0.15406 nm) with a voltage and current of 40 kV and 40 mA, respectively.
Selective oxidation of toluene was performed in an autoclave (50 mL) with mechanical stirring. After the vessel was charged with 30 mL of toluene and the required amount of catalyst, it was filled with pure oxygen to a desired initial pressure. Then the reaction was time was measured as soon as the temperature reached the set value. When the reaction was finished, we analyzed the liquid phase by GC and the results were compared to known commercially pure standard samples. Finally, conversion and product selectivity were calculated based on the carbon balance with the calibration curves.
2 Results and Discussion
2.1 Characterization results
The XRD patterns of the prepared samples are shown in Fig.1. Some broad peaks and some sharp peaks are observed, as well as shifting in the positions of the peaks. Broadening of diffraction peaks occurs when certain types of random intergrowth of pyrolusite (1×2 tunnel structure) and ramsdellite (1×2 tunnel structure) manganese dioxide are present according to the de Wolff model[11]. After comparison with the reference data for the IBA-11 XRD patterns[12], the sample was confirmed to be a crystal form of γ-MnO2. The other sample shows a similar XRD pattern with γ-MnO2except that it has the peaks at 2θof 38.5° (111), 44.8° (200) due to the Au particles (JCPDS Card No.1-1172). This indicates that the loading of Au particles do not change the crystal structure of MnO2and the size of Au particles is larger than 4 nm. In
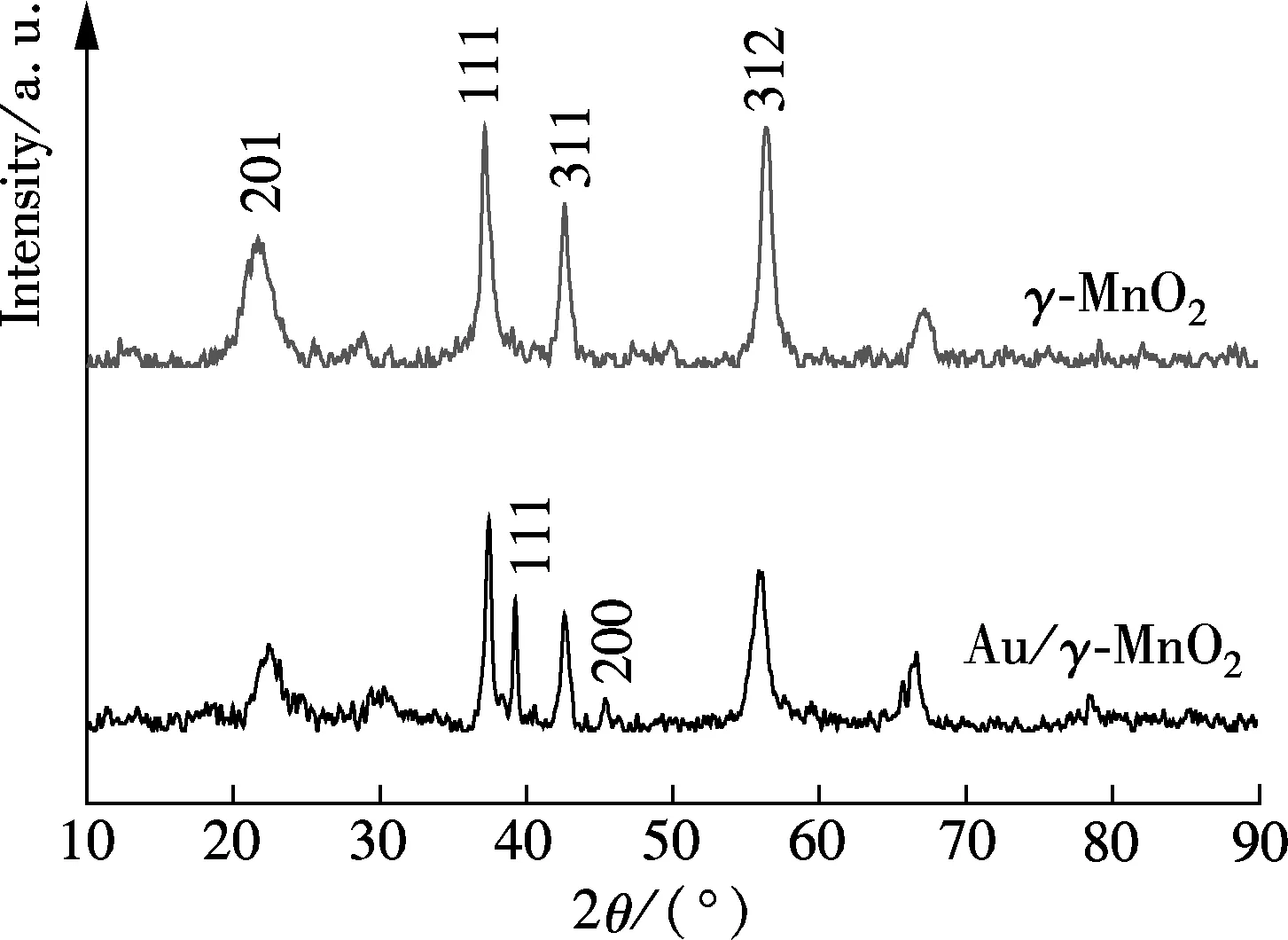
Fig.1 XRD patterns of prepared samples
fact, TEM and SEM images show that Au particles are well dispersed on γ-MnO2nanorods and the size of which was about 10 nm, the same as in our previous research[10].
2.2 Catalytic results
The effect of initial oxygen pressure on toluene oxidation reaction is shown in Fig.2. Higher oxygen pressure obviously improves the conversion of toluene, and a drastic improvement appears between 2.5 and 3.0 MPa. Meanwhile, benzoic acid selectivity presents the same variation trend, even though there is no big change between the pressure of 1.5 and 2.0 MPa. By contrast, the selectivity of benzaldehyde reduces significantly. At the pressure of 3.0 MPa, the main products are benzaldehyde and benzoic acid, which share almost the same selectivity with each other. In this single-factor reaction, the selectivities of benzyl alcohol and benzyl benzoate have not changed much, and only show a slight decreasing trend between 0.5 and 3.0 MPa. In general, the increasing initial pressure of oxygen improves the conversion of toluene precisely. This may be caused by the increasing oxygen content in the unit volume, which enhances the reaction. As a result, benzyl alcohol and benzaldehyde are further oxidized to benzoic acid and benzyl benzoate. Therefore, even though benzaldehyde has the highest selectivity, it presents a decreasing trend, while the selectivity of benzoic acid increases. Moreover, because of the limited benzyl alcohol, benzyl benzoate selectivity is not changed significantly.
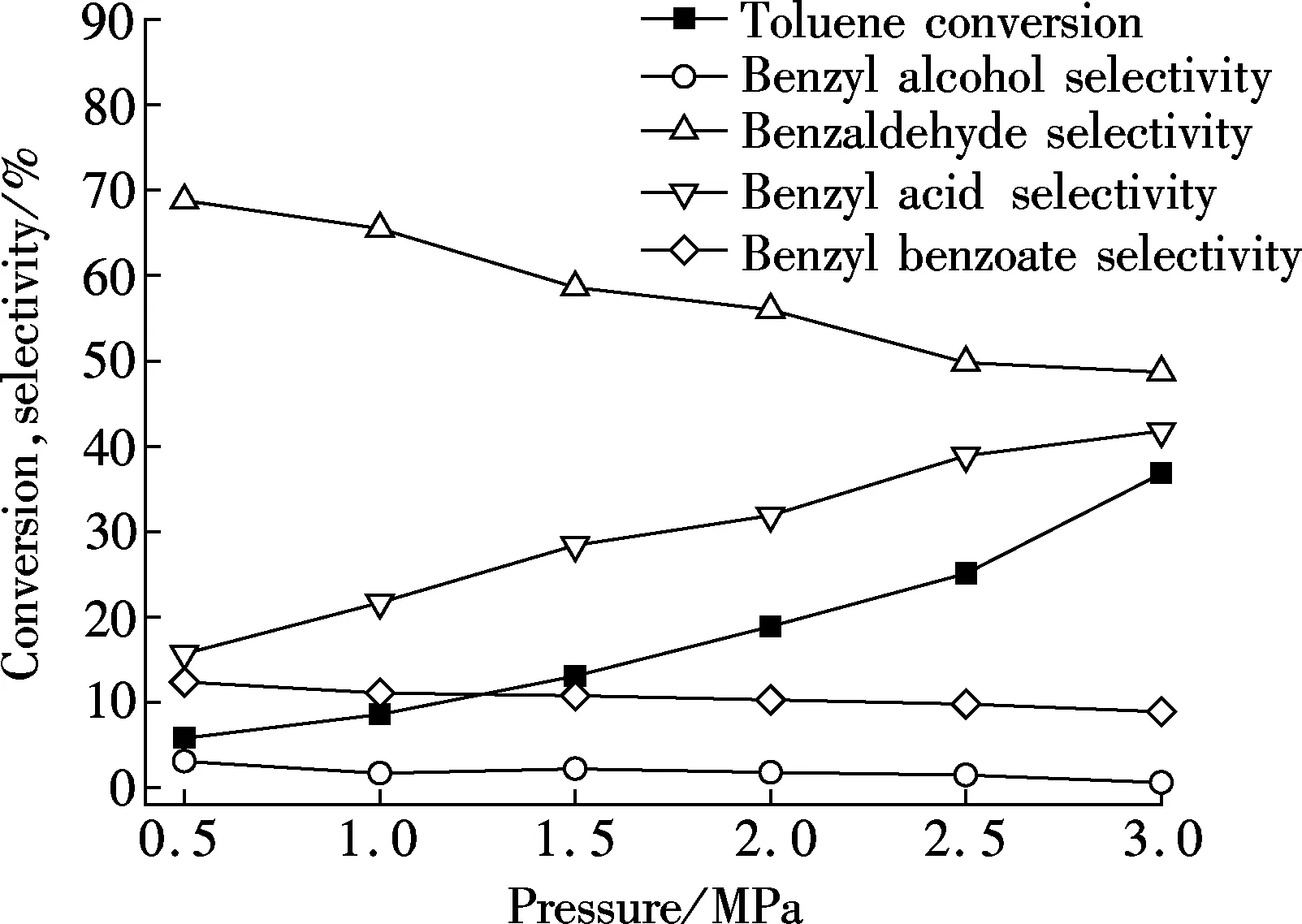
Fig.2 Influence of initial oxygen pressure on catalytic performance over Au/γ-MnO2 catalyst with the reaction temperatures of 160℃, the reaction time of 8 h, the catalyst amount of 1 % in mass fraction, and the stirring rate of 600 r/min
In Fig.3, toluene conversion increases with the increase of temperature. At the temperature of 140℃ or lower, the conversion of toluene remains below 5%. However, when the reaction temperature increases, the reaction becomes more severe. Toluene conversion is improved by a clear increase rate progressively, and at the temperature of 220℃, it reaches the value of 34.1%. Another improvement appears on the selectivity of benzyl benzoate. When the temperature is low, little benzyl benzoate can be obtained. However, a reaction at 220℃ provides the greatest production of benzyl benzoate and the value of selectivity changes to 47.1%. On the contrary, the other three products show decreasing trends in Fig.3. Benzyl benzoate selectivity shows a sharp increase between the temperature of 180 and 220℃, while the selectivity of benzaldehyde decreases by 37.4%. Meanwhile, benzoic acid and benzyl alcohol do not change much. This single-factor experiment shows a result that the increasing temperature provides more energy for greater reactions. Some toluene may be directly oxidized to benzyl benzoate. This is proved to be the most effective method to improve toluene conversion in this reaction system. This enhancement in catalytic activity may be caused by the increase in the intrinsic activity of the existing active sites and also by the creation of more active sites with the increase in reaction temperature[13].
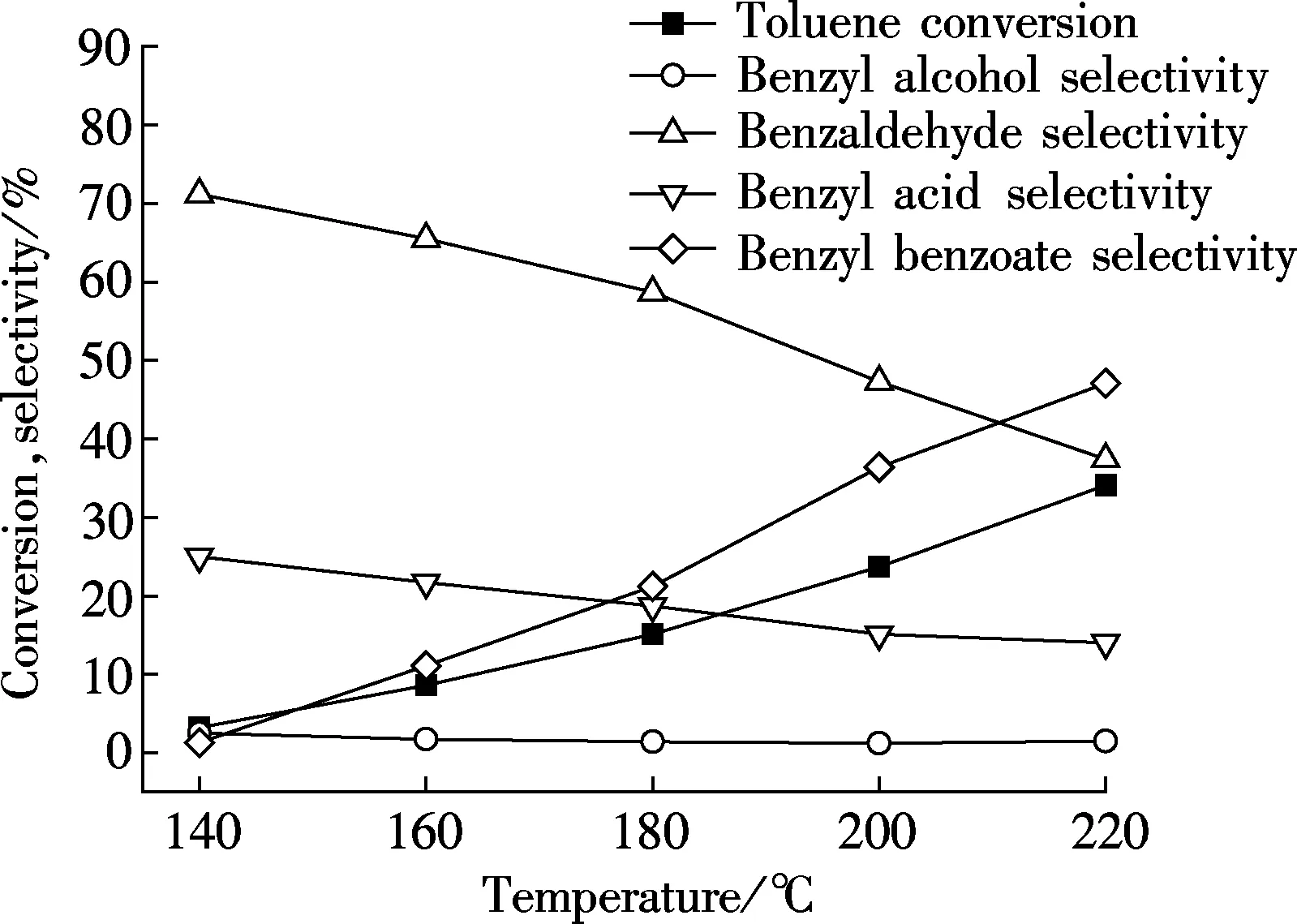
Fig.3 Influence of reaction temperature on catalytic performance over Au/γ-MnO2 catalyst with the initial pressure of 1 MPa, the catalyst amount of 1% in mass friction, the reaction time of 8 h, and the stirring rate of 600 r/min
It is clear to see from Fig.4 that a long reaction time can clearly improve toluene conversion. However, in the first 12 h, oxidation reaction proceeds slowly. On the other hand, both benzyl alcohol and benzaldehyde decrease as time goes by. The change in benzyl alcohol selectivity is not obvious, while a sharp decrease appears
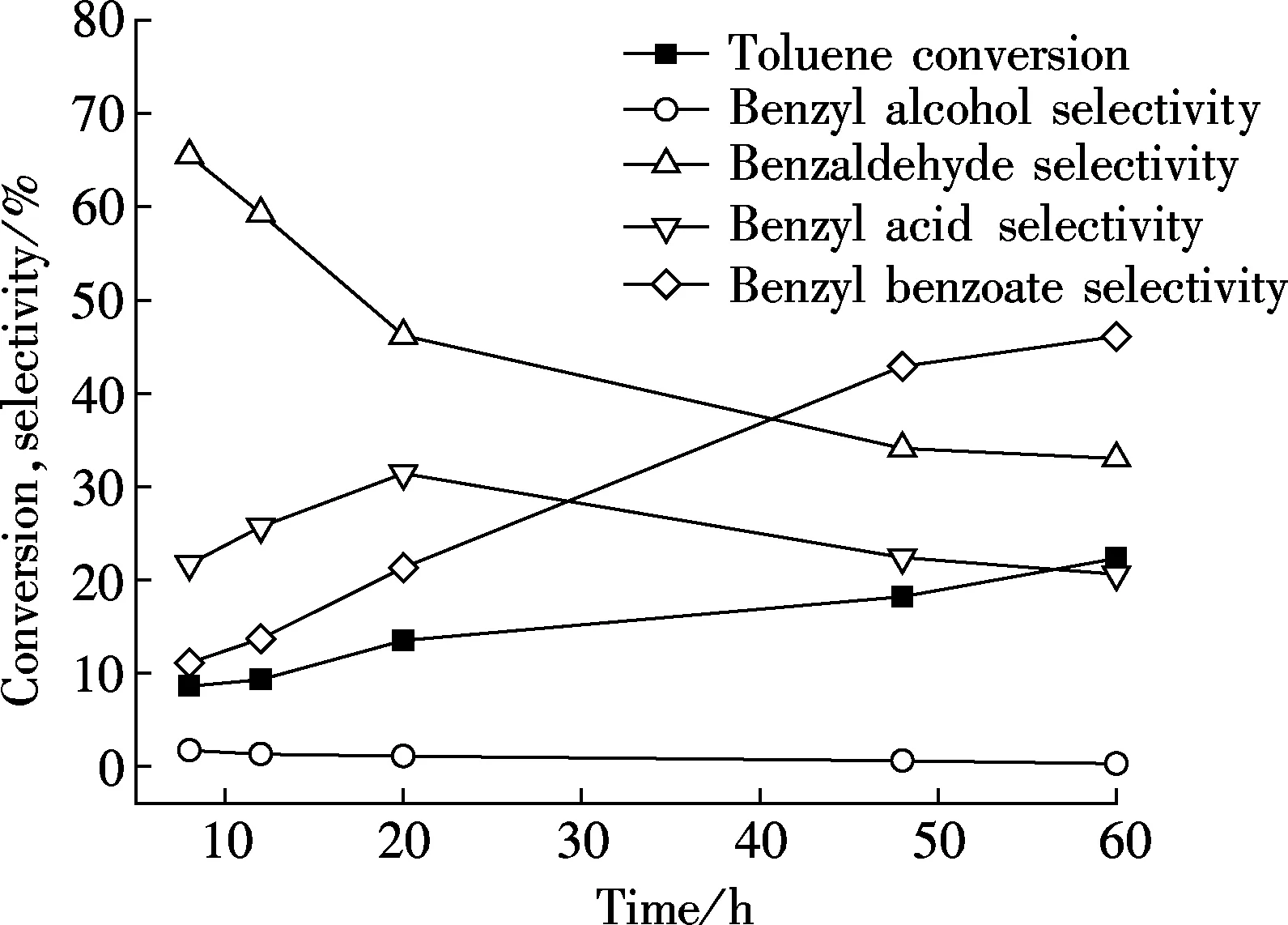
Fig.4 Influence of reaction time on catalytic performance over Au/γ-MnO2 catalyst with the reaction temperature of 160℃, the initial pressure of 1 MPa, the catalyst amount of 1% in mass friction, and the stirring rate of 600 r/min
in the first 20 h of benzaldehyde selectivity. On the contrary, benzyl benzoate production increases more over time and exceeds benzaldehyde after a reaction for 42 h. Unlike the three products, benzoic acid selectivity first increases to 31.4% at 20 h and then decreases to 20.6% at 60 h. This phenomenon is attributed to more benzaldehyde turning into benzyl acid with oxygen at the beginning of the reaction. As time passes, the benzyl acid and benzyl alcohol begin further reacting to form benzyl benzoate. This observation can also be confirmed by the increasing trend of benzyl benzoate selectivity. A similar result was also discovered by Fu et al[14]. In this way, we can obtain the most benzoic acid product by controlling reaction time.
The influence of catalyst amount in this system is described in Fig.5. Similar to the influence of the above discussed parameters of reaction temperatures and initial oxygen pressure, the toluene conversion increases dramatically with the increase in catalyst amount, which can be raised from the initial 8.6% to above 30%. However, the conversion maintains constant when the catalyst amount is above 7%. This phenomenon may be ascribed to two reasons. One is the limitation of the oxygen amount in the liquid phase of this reaction system. The other possible reason is the findings that metal complexes of transition metals, particularly in the media of low polarity such as neat hydrocarbons, often act as catalysts at low loadings but inhibitors at high loadings[15]. The selectivity of benzoic acid and benzyl benzoate first increases then decreases, while the selectivity of benzyl alcohol does not change too much. The increase of catalyst amount may drive the reaction into a further stage and results in the increase of selectivity of the more reactive oxidation products. However, due to the limitations of reaction conditions, this selectivity declines when the amount of catalyst reaches a certain value. Another product is benzaldehyde, the selectivity of which reaches a maxmum value when the amount is 7%. It is known that benzoic acid and benzyl benzoate are the further oxidation products
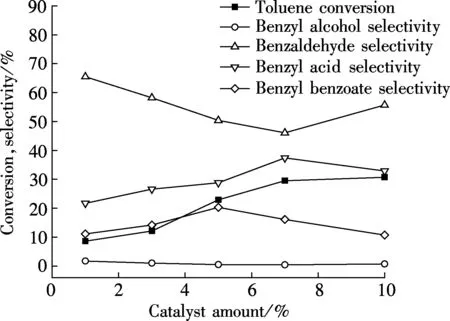
Fig.5 Influence of catalyst amount on catalytic performance over Au/γ-MnO2 catalyst with the reaction temperature of 160℃, the initial pressure of 1 MPa, the reaction time of 8 h and the stirring rate of 600 r/min
of benzaldehyde, so it is easy to understand that the behavior of benzaldehyde selectivity can be contrasted with that of benzoic acid and benzyl benzoate. Moreover, the selectivity of benzaldehyde is always the highest during the increase of catalyst amount from 1% to 10%, indicating that the Au/γ-MnO2catalyst has a unique selectivity to benzaldehyde in this reaction condition.
3 Conclusion
In this paper, the well dispersed Au/γ-MnO2catalyst is synthesized. The sample shows effective catalytic performance in solvent-free toluene oxidation. Several process parameters can enhance the conversion of toluene, which are reaction temperature, initial oxygen pressure, catalyst amount, and reaction time. All these factors influence the selectivity of benzaldehyde, benzoic acid and benzyl benzoate significantly, but affect that of benzyl alcohol only slightly. Under the conditions of a reaction time within 20 h, a reaction temperature below 200℃ and the initial oxygen pressure within 3 MPa, this Au/γ-MnO2catalyst has a unique selectivity to benzaldehyde.
[1]Suresh A K, Sharma M M, Sridhar T. Engineering aspects of industrial liquid-phase air oxidation of hydrocarbons [J].Industrial&EngineeringChemistryResearch, 2000, 39(11): 3958-3997.
[2]Kantam M L, Sreekanth P, Rao K K, et al. An improved process for selective liquid-phase air oxidation of toluene [J].CatalysisLetters, 2002, 81(3/4): 223-232.
[3]Kesavan L, Tiruvalam R, Ab Rahim M H, et al. Solvent-free oxidation of primary carbon-hydrogen bonds in toluene using Au-Pd alloy nanoparticles [J].Science, 2011, 331(6014): 195-199.
[4]Qiu G H, Huang H, Dharmarathna S, et al. Hydrothermal synthesis of manganese oxide nanomaterials and their catalytic and electrochemical properties [J].ChemistryofMaterials, 2011, 23(17): 3892-3901.
[5]Zhang Y B, Shen Y N, Yang X Z, et al. Gold catalysts supported on the mesoporous nanoparticles composited of zirconia and silicate for oxidation of formaldehyde [J].JournalofMolecularCatalysisA:General, 2010, 316(1/2): 100-105.
[6]Wang H, Zhu H Q, Qin Z F, et al. Deactivation of a Au/CeO2-Co3O4catalyst during CO preferential oxidation in H2-rich stream [J].JournalofCatalysis, 2009, 264(2): 154-162.
[7]Li C Y, Shen Y N, Jia M L, et al. Catalytic combustion of formaldehyde on gold/iron-oxide catalysts [J].CatalysisCommunications, 2008, 9(3): 355-361.
[8]Blick K, Mitrelias T D, Hargreaves J S J, et al. Methane oxidation using Au/MgO catalysts [J].CatalysisLetters, 1998, 50(3/4): 211-218.
[9]Campbell C T. The active site in nanoparticle gold catalysis [J].Science, 2004, 306(5694): 234-235.
[10]Jiang F, Zhu X W, Fu B S, et al. Au/γ-MnO2catalyst for solvent-free toluene oxidation with oxygen [J].ChineseJournalofCatalysis, 2013, 34(9): 1683-1689.
[11]de Wolff P M. Interpretation of some γ-MnO2diffraction patterns [J].ActaCryst, 1959, 12: 341-345.
[12]Malpas D G, Tye F L.Handbookofmanganesedioxidesbatterygrade[M]. Brunswick: IBA Inc & JEC Press, 1989.
[13]Kalevaru V N, Raju B D, Rao V V, et al. Preparation, characterisation and catalytic evaluation of MgF2supported V2O5catalysts for ammoxidation of 3-picoline [J].AppliedCatalysisA:General, 2009, 352(1/2): 223-233.
[14]Fu B S, Zhu X W, Xiao G M. Solvent-free selective aerobic oxidation of toluene by ultra fine nano-palladium catalyst [J].AppliedCatalysisA:Gernal, 2012, 415-416: 47-52.
[15]Sheldon R A.Metal-catalyzedoxidationoforganiccompounds[M]. New York:Academic Press, 1981.
反应条件对Au/γ-MnO2催化甲苯氧化反应的影响
姜 枫 肖国民
(东南大学化学化工学院, 南京 211189)
制备了γ-MnO2纳米棒以及Au/γ-MnO2催化剂,并用X射线衍射(XRD)、扫描电镜(SEM)和透射电镜(TEM)等手段对其进行表征.结果表明:Au颗粒均匀分散在载体γ-MnO2的表面,颗粒大小约为10 nm.在无溶剂存在下甲苯和氧气的氧化反应中测试了样品的催化活性.研究了反应时间、反应温度、初始氧气压力及催化剂加入量对催化活性的影响.活性测试结果表明:Au/γ-MnO2催化剂对苯甲醛有特殊的选择性;反应时间、温度、初始压力及催化剂加入量等条件对甲苯转化率及苯甲醛、苯甲酸、苯甲酸苄酯的选择性都有很大影响,但对苯甲醇的选择性影响不大.
纳米金;二氧化锰;甲苯氧化;无溶剂
TQ139.2
s:The National Natural Science Foundation of China (No.21276050), the Scientific Research Foundation of Graduate School of Southeast University (No.YBJJ1341).
:Jiang Feng, Xiao Guomin.Influences of process parameters on solvent-free toluene oxidation over Au/γ-MnO2catalyst[J].Journal of Southeast University (English Edition),2014,30(3):387-390.
10.3969/j.issn.1003-7985.2014.03.024
10.3969/j.issn.1003-7985.2014.03.024
Received 2014-01-20.
Biographies:Jiang Feng (1987—), femal,graduate; Xiao guomin (corresponding author), male, doctor, professor, xiaogm@seu.edu.cn.
 Journal of Southeast University(English Edition)2014年3期
Journal of Southeast University(English Edition)2014年3期
- Journal of Southeast University(English Edition)的其它文章
- P-FFT and FG-FFT with real coefficients algorithm for the EFIE
- Compressed sensing estimation of sparse underwateracoustic channels with a large time delay spread
- Improved metrics for evaluating fault detection efficiency of test suite
- Early-stage Internet traffic identification based on packet payload size
- An adaptive generation method for free curve trajectory based on NURBS
- Stability analysis of time-varying systems via parameter-dependent homogeneous Lyapunov functions
