Hydrangea serrata extract exerts tumor inhibitory activity against hepatocellular carcinoma HepG2 cells via inducing p27/CDK2-mediated cell cycle arrest and apoptosis
Ye-eun Kim ,Jeonghye Hwang ,Ki-Young Kim
1Graduate School of Biotechnology, Kyung Hee University, Seocheon, Giheung, Yongin, Gyeonggi-do 446-701, Republic of Korea
2Department of Genetics and Biotechnology, College of Life Sciences, Kyung Hee University, Seocheon, Giheung, Yongin, Gyeonggi-do 446-701,Republic of Korea
ABSTRACT Objective:To examine the inhibitory effect of Hydrangea serrata extract against hepatocellular carcinoma HepG2 cells and its underlying mechanisms.Methods:The effects of Hydrangea serrata extract on growth inhibition of tumor cells and spheroids were assessed using MTT and 3D culture assays.Quantitative real-time PCR and Western blot analyses were employed to investigate the changes in mRNA and protein expression levels of molecules related to cell cycle and apoptosis.Results:Hydrangea serrata extract effectively inhibited the growth of both tumor cells and spheroids.The extract also significantly upregulated p27 mRNA expression and downregulated CDK2 mRNA expression,leading to cell cycle arrest.Moreover,increased BAX/Bcl-2 ratio as well as caspase-9 and -3 were observed after treatment with Hydrangea serrata extract,indicating the induction of tumor cell apoptosis.Conclusions:Hydrangea serrata extract has the potential to alleviate tumors by effectively modulating cell-cycle-related gene expressions and inducing apoptosis,thereby inhibiting tumor growth.
KEYWORDS: Hydrangea serrata;Hepatocellular carcinoma;Liver cancer;Anticancer;Cell cycle arrest;Apoptosis
1.Introduction
Liver cancer remains a significant threat worldwide since it is the third leading cause of cancer-related fatalities globally[1].Among liver cancers,hepatocellular carcinoma (HCC),the most common form of primary liver cancer,accounts for up to 90% of the cases[2].Remarkably,the majority of HCCs can be attributed to chronic liver inflammation and viral infection from hepatitis B virus (HBV)and hepatitis C virus (HCV),setting it apart from other forms of cancer[3,4].Besides,due to the pivotal involvement of the liver in glucose metabolism,metabolic syndrome,and diabetes mellitus have emerged as increasingly common risk factors in especially western countries[5].
Hydrangea serrata(H.serrata),also called “San-su-guk” which means “mountain hydrangea”,is native to the mountains of Korea and Japan,and has been traditionally consumed as an herbal medicine in East Asian countries including Korea,China,and Japan[6,7].Because of its naturally sweet taste when brewed as tea,it also has been called “tea of heaven”[8].Interestingly,previous literature reported that the extract of hydrangea exhibits antiinflammatory,anti-diabetic,hepatoprotective,and anticancer effects against gastric cancer[9-12].Considering the strong association between inflammation,diabetes,and hydrangea,we sought to further investigate whetherH.serrataextract could also demonstrate potential anticancer properties against hepatocellular carcinoma,a type of liver cancer commonly associated with inflammation and diabetes,using HepG2 cell line[13].

2.Materials and methods
2.1.Cell line and cell culture
Hepatocellular carcinoma cell line,HepG2,was purchased from Korean Cell Line Bank and routinely cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) containing 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin.The cells were incubated in a humidified incubator at 37 ℃ with 5% CO2.
2.2.Preparation of plant extract
The stem and leaves ofH.serrataobtained from Jeju Island,Republic of Korea[8] were dried with a freeze dryer (IlShin,Dongducheon,Korea),extracted with distilled water at 98 ℃ for 5 h,and spray-dried to give a dried extract residue[8].The resulting dried extract was dissolved in dimethyl sulfoxide (DMSO) to the original concentration of 10 mg/mL.High performance liquid chromatography (HPLC) analysis was carried out to prepareH.serrataextract with varying concentrations (0%,7%,and 13%) of hydrangenol (Supplementary Figure 1)[8].
2.3.MTT assay
HepG2 cells were seeded at a density of 5×103cells per well of 96-well plates and further incubated for 24 h at 37 ℃ with 5% CO2.Subsequently,the medium was suctioned,and cells were treated with serum-free DMEM medium containing 0,1,5,10,50,and 100 μg/mL ofH.serrataextract for 24 h.Dilution of theH.serrataextract proceeded while maintaining the same concentration of DMSO in order to prevent it from affecting the results[14].After 24 hours of treatment,100 μL of 0.5 mg/mL MTT solution in the serum-free DMEM medium was added to each well after suction[15].After 3 hours of incubation at 37 ℃,the wells were replaced with 100 μL of DMSO to dissolve formazan crystals.Then,the plate was covered with aluminum foil and shaken for 15 min.Absorption values at 540 nm were measured using a microplate reader (Bio Tek Instruments,Korea)[16].
2.4.Generation of HCC spheroids and 3D culture assay
HepG2 cells were seeded at a density of 5×103cells per well of 96-well SPL3D™Cell Floater (SPL,#34896) and incubated for 24 h.Thereafter,100 μg/mL ofH.serrataextract was added to the medium to determine the effect of the extract on the spheroid-forming ability[17,18].After 3,4,and 6 d,the pictures of HCC spheroids were taken under a camera-attached microscope (EVOS®FL Cell Imaging System,Thermo Fisher Scientific,Waltham,MA,USA),and the area was calculated using ImageJ software[19].
2.5.Quantitative real-time PCR
HepG2 cells were seeded at a density of 5×104cells per well of 6-well plates in 2 mL of DMEM medium and cultured for 24 h.Then,cells were treated withH.serrataextract at concentrations of 0,5,10,and 50 μg/mL in the serum-free DMEM medium and further incubated for 24 h.After 24-hour treatment,total RNA was isolatedusing Trizol reagent (Life Technology,Thermo Fisher Scientific,USA) according to the manufacturer’s protocol,and cDNA was synthesized using 1 μg of total RNA[20].Quantitative real-time PCR was performed using QGreen 2X SybrGreen qPCR Master Mix(CellSafe,Korea)[21].Sequences of the primers used in this study are listed in Table 1.GAPDHwas used as an internal control[22].The amount of gene expression was calculated as the fold change value using 2-ΔΔCq.
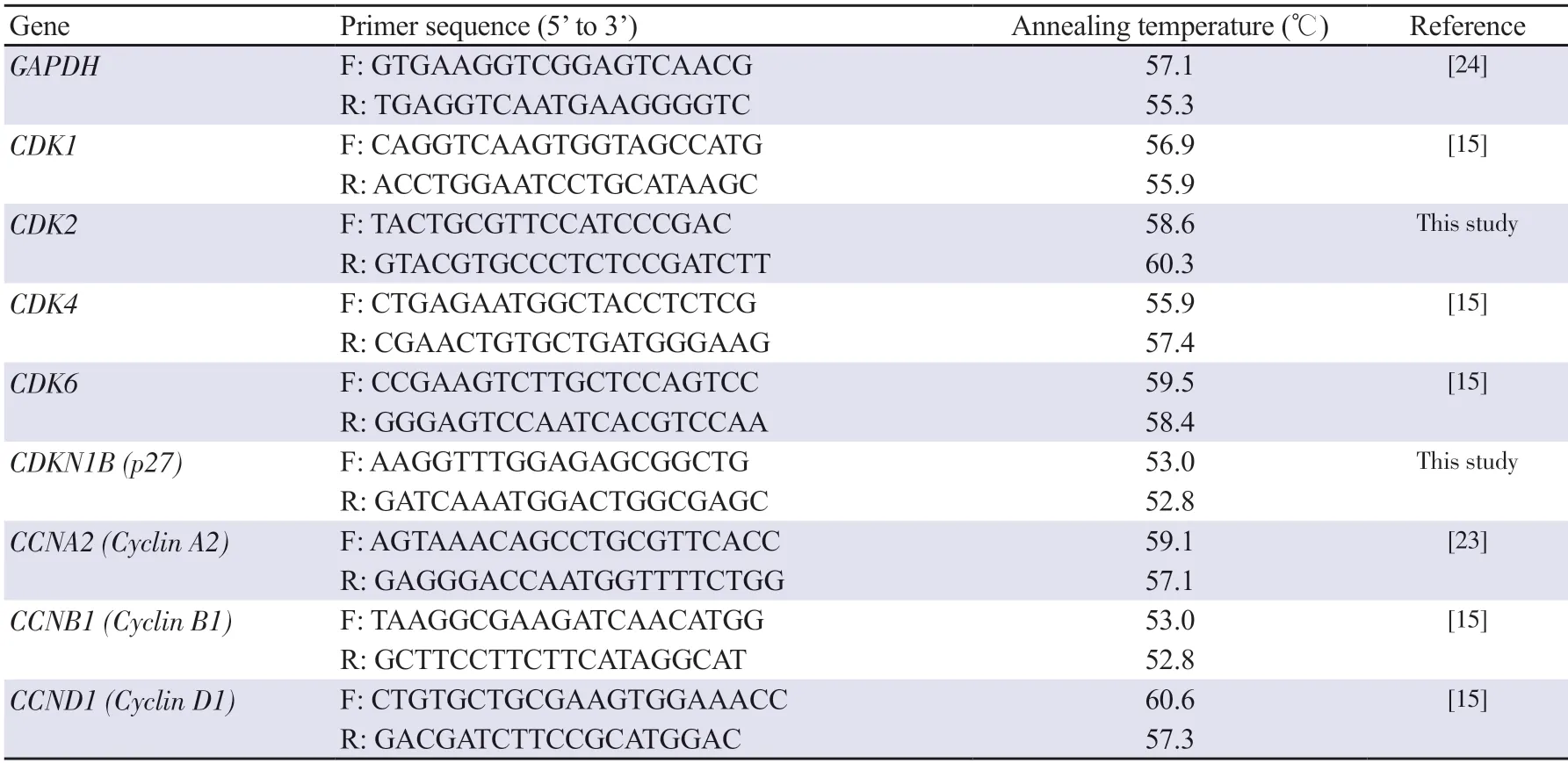
Table 1.List of primers used in qRT-PCR analysis.
2.6.Western blot
HepG2 cells were seeded at a density of 5×104cells per well of 6-well plates in 2 mL of the DMEM medium and cultured for 24 h.Then,cells were treated withH.serrataextract at concentrations of 0,5,10,and 50 μg/mL in the serum-free DMEM medium and further incubated for 24 h.Protein concentration was determined by Bradford assay.A total of 30 μg of total protein from cell lysate was loaded per well,separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE),transferred to a polyvinylidene difluoride (PVDF) membrane,and blocked with 5% bovine serum albumin (BSA) in TBST buffer (20 mM Tris-HCl,150 mM NaCl,and 0.1% Tween 20,pH 7.6)[24].Membranes were probed with primary antibodies at the following dilutions: p-p53 (sc-51690,1∶1 000),BAX (sc-493,1∶500),Bcl-2 (sc-7382,1∶500),caspase-9 p35 (sc-8355,1∶1 000),GAPDH(sc -25778,1∶1 000),p38 (sc-535,1∶1 000),p-p38 (sc-17852-R,1∶1 000),JNK (sc-7345,1∶500),p-JNK (sc-6254,1∶500) (Santa Cruz Biotechnology,CA,USA),p53 (9282s,1∶1 000),caspase-3 (9662S,1∶1 000),p44/42 MAPK (ERK1/2) (9102S,1∶1 000),and p-p44/42 MAPK (p-ERK1/2) (9101S,1∶1 000) (Cell Signaling Technology,MA,USA).After 1-hour incubation with secondary antibodies,protein bands were developed with ECL reagent (Bio-Rad,USA)and detected with UVITEC imaging system equipment (UVITEC Cambridge,UK)[25].Quantitation of Western blot bands was conducted using ImageJ software.
2.7.Statistical analysis
All the experiments were performed in duplicate and repeated more than three times.Results are presented as mean ± standard deviation(SD).Statistically significant differences were analyzed using oneway ANOVA with Dunnett’spost hoctest[26].GraphPad Prism software version 5.0 was used to perform all statistical analyses.P<0.05 was considered significantly different.
3.Results
3.1.H.serrata extract shows anticancer effects against hepatocellular carcinoma HepG2 cell line regardless of a key indicator component, hydrangenol
To evaluate whetherH.serrataextract possesses anti-proliferative properties against liver cancer,MTT assay was conducted.As shown in Figure 1A,the growth of HepG2 cells was dose-dependently inhibited by treatment ofH.serrataextract,with a maximum inhibition of 40% observed at the highest concentration tested(100 μg/mL).However,theH.serrataextract did not demonstrate a substantial anticancer impact on PLC/PRF5 cells,implying its efficacy may be selective to specific forms of liver cancer(Supplementary Figure 2).To further investigate if hydrangenol,a key indicator component of hydrangea extract with known antiproliferative properties against multiple types of cancer,affects the efficacy exerted byH.serrataextract,we conducted a follow-up experiment usingH.serrataextract with varying concentrations (0%,7%,and 13%) of hydrangenol.AllH.serrataextracts showed similar inhibition activities at the highest concentration tested (100 μg/mL),suggesting the anti-proliferative property ofH.serrataextract is likely independent of hydrangenol (Figure 1B-D).
3.2.H.serrata extract inhibits the growth of liver cancer spheroids
To further investigate the inhibitory effect ofH.serrataextract on the growth of tumor spheroids as well as the growth of cells,3D culture assay was performed using HCC spheroids.HepG2 cells were treated with 100 μg/mLH.serrataextract and the volume of spheroids formed was measured on days 3,4,and 6,respectively.The volume of spheroids treated withH.serrataextract was significantly smaller than that of spheroids treated with DMSO control (Figure 2),with only 56% remaining on day 6.Moreover,H.serrataextract also seemed to inhibit the tumor cells from spreading outward,indicating its potential role in the suppression of invasion at least in part.
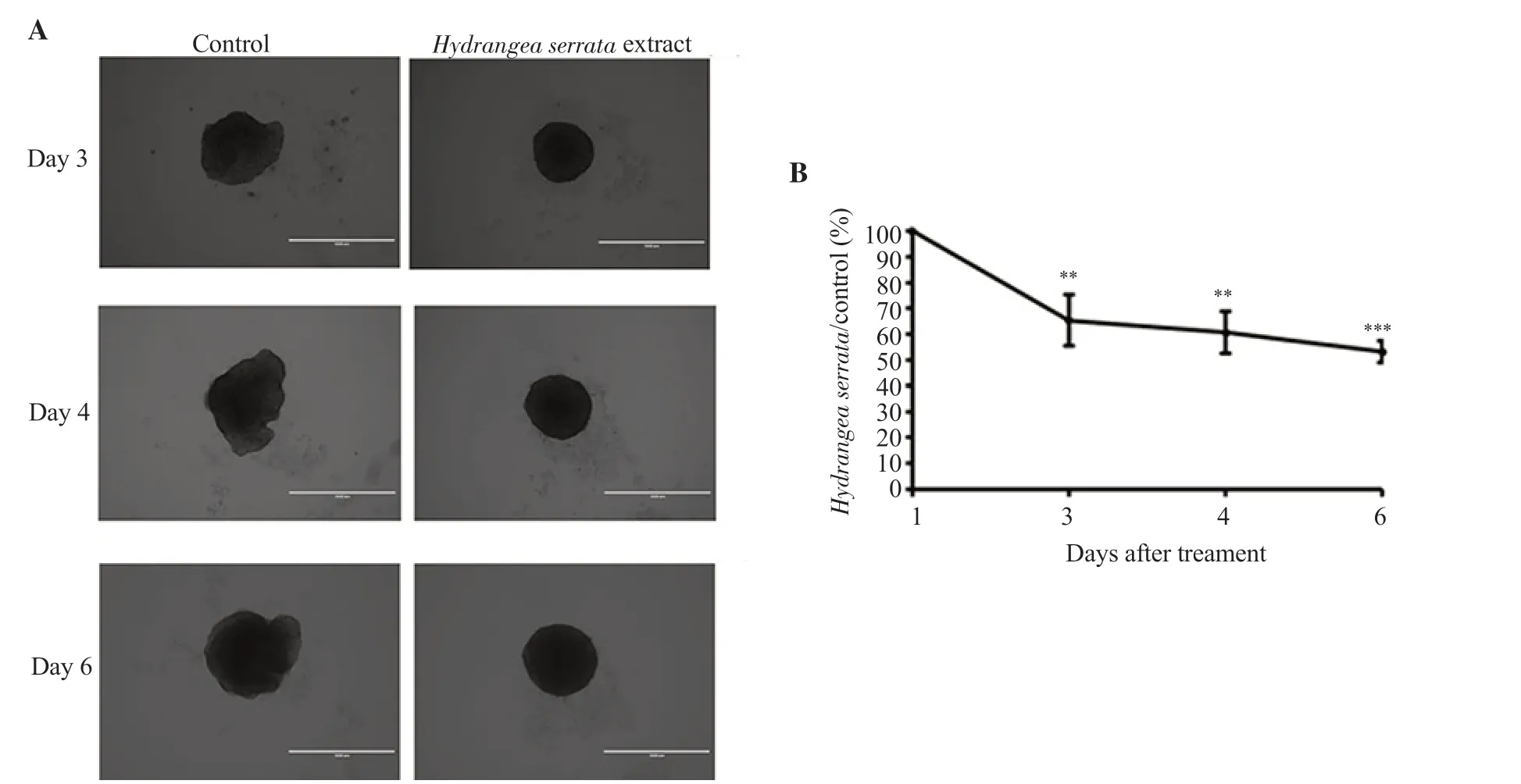
Figure 2.H.serrata extract inhibits the growth of liver cancer spheroids.(A) Microscopic images at 40× magnification depicting tumor spheroids 3,4,and 6 days after treatment with 100 μg/mL of H.serrata extract.(B) Quantification of spheroid area showing a time-dependent decrease.Relative spheroid volume of DMSO control was set as 100%.The area of the spheroid was quantified using ImageJ software.Data are presented as mean ± SD and analyzed by ANOVA.Each experiment was performed in duplicate and repeated at least three times.**P < 0.01,***P < 0.001 compared with the control.
3.3.H.serrata extract induces p27 upregulation and subsequent CDK2 downregulation in HepG2 cells
To evaluate whetherH.serrataextract induces cell cycle arrest,we performed quantitative real-time PCR and Western blot analyses in HepG2 cells treated with various concentrations ofH.serrataextract (0,5,10,and 50 μg/mL) for 24 h.As depicted in Figure 3A,the relative mRNA expression ofCDKN1B(p27) was markedly increased while that ofCDK2was significantly decreased.The mRNA expression ofCDK1,CDK4,CCNA2(Cyclin A2),CCNB1(Cyclin B1),andCCND1(Cyclin D1) showed no significant change while that ofCDK6was significantly increased (Figure 3B).The protein expressions of p38,p53,p-JNK,and ERK1/2 were decreased dose-dependently (Figure 3C).However,the expression levels of p-p38,p-p53,JNK,and p-ERK1/2 did not show significant changes at various concentrations,with a noticeable decrease only observed at the highest concentration of 50 μg/mL (Figure 3C).Collectively,these findings suggest that treatment withH.serrataextract upregulatesp27expression in HepG2 cells,leading to subsequent downregulation ofCDK2expression (Figure 3D).
3.4.H.serrata extract promotes apoptosis in HepG2 cells
To examine whetherH.serrataextract further enhances tumor cell apoptosis,Western blot analysis was conducted to detect apoptosisrelated proteins.The expression of BAX was increased while the expression of Bcl-2 showed a slight decrease,increasing the BAX/Bcl-2 ratio (Figure 4A and B).Furthermore,the expression of caspase-9,an initiator caspase that plays a critical role in initiating the apoptotic cascade,significantly increased while that of caspase-3,a downstream effector caspase that is activated by caspase-9,showed a modest increase,suggestingH.serrataextract promoted apoptosis in HepG2 cells (Figure 4A and C).
4.Discussion
Given the known anti-inflammatory,anti-diabetic,and hepatoprotective effects of hydrangea,we further investigated whetherH.serratahas the potential to ameliorate liver cancer,a cancer type particularly related to inflammation and diabetes.As a result,H.serrataextract effectively inhibited the growth of both tumor cells and spheroid by inducing cell cycle arrest and apoptosis.Moreover,becauseH.serrataextract also prevented the cells from spreading outward in 3D culture assay,we observed a possibility thatH.serrataextract likely suppresses tumor invasion.However,to verify this hypothesis,it needs to be further identified in a follow-up study using cell migration assay and cell invasion assay.
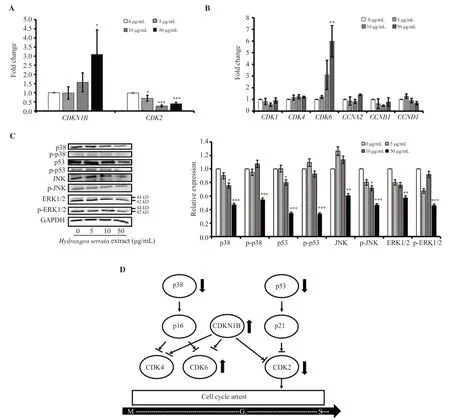
Figure 3.H.serrata extract induces p27/CDK2-mediated cell cycle arrest in HepG2 cells.(A) Relative mRNA expression of CDKN1B (p27) and CDK2.(B)Relative mRNA expression of CDK1,CDK4,CDK6,CCNA2 (Cyclin A2),CCNB1 (Cyclin B1),and CCND1 (Cyclin D1) related to cell cycle was measured by qRT-PCR analysis after HepG2 cells were treated with H.serrata extract for 24 h.(C) Western blot image depicting the expression of cell cycle-related proteins.Western blot data was quantified using ImageJ software.Relative expression of 0 μg/mL control was set as 1.Data are presented as mean ± SD and analyzed by ANOVA.Each experiment was performed in duplicate and repeated at least three times.*P < 0.05,**P < 0.01,***P < 0.001 compared with the control.(D)Schematic representation of the mechanism of cell cycle arrest induced by H.serrata extract.
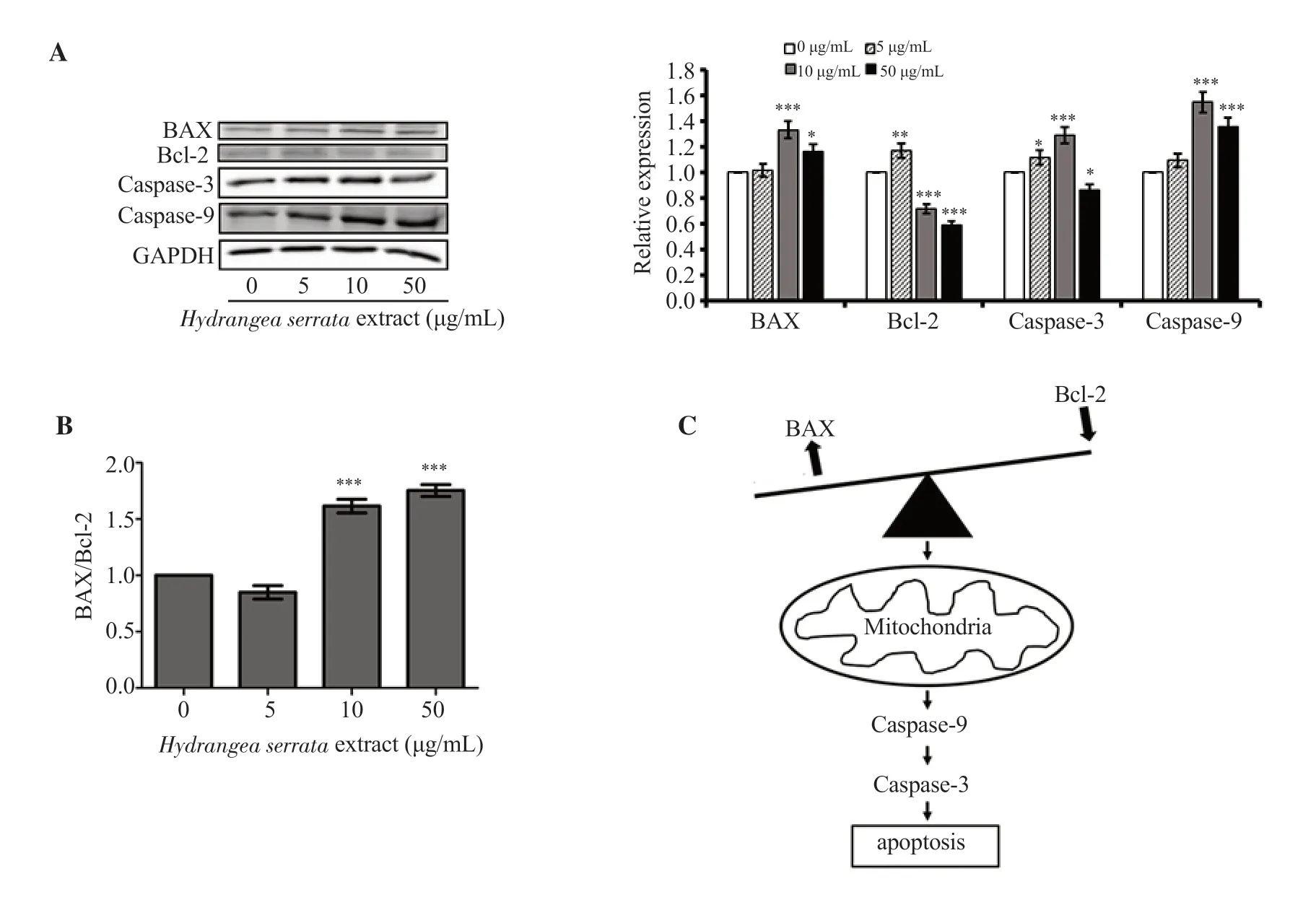
Figure 4.H.serrata extract induces apoptosis in HepG2 cells.(A) Western blot images of BAX,caspase-3,caspase-9 and Bcl-2.(B) BAX/Bcl-2 ratio.Western blot data was quantified using ImageJ software.Relative expression of 0 μg/mL control was set as 1.Data are presented as mean ± SD and analyzed by ANOVA.Each experiment was performed in duplicate and repeated at least three times.*P < 0.05,**P < 0.01,***P < 0.001 compared with the control.(C)Schematic representation of apoptosis induced by H.serrata extract.
Even though hydrangenol,which is known as the key indicator component ofH.serrataextract,has been shown to inhibit the proliferation of EJ bladder cancer in previous literature[27],our observations from MTT assay indicate that hydrangenol does not affectH.serrataextract’s anticancer effect against HepG2 cells.This suggests that the anticancer effect of hydrangenol may be restricted to certain types of cancer including bladder cancer and the observed anticancer effect ofH.serrataextract may not be attributed to hydrangenol.
The results from the analysis of cell cycle arrest induced byH.serrataextract revealed that upregulation ofp27,a negative regulator of cell cycle progression,and consequent downregulation ofCDK2,a main target of p27 that regulates G1/S transition,were the possible mechanisms governingH.serrataextract-induced cell cycle arrest[28,29].It is reported that cells in the G0/G1phases can accumulate when there is a reduction of cell cycle progression through the G1/S phases,due to their inability to advance through the cell cycle[30].Since CDK6 is a cyclin-dependent kinase that releases the cells from the G0phase,the increase inCDK6mRNA expression could potentially be interpreted as an accumulation of cells in the G0phase caused by the G1/S cell cycle arrest[31].
Since Bcl-2 is known as an anti-apoptotic protein and BAX is known as a pro-apoptotic protein,an upregulation in BAX/Bcl-2 ratio likely indicates that cells are going under apoptosis[32-34].Furthermore,it is known that the expression of caspase-3,and caspase-9 is correlated to the activation of apoptotic cascade[35].The increase in the BAX/Bcl-2 ratio,caspase-3,and caspase-9 expression from our results leads us to the conclusion that theH.serrataextract possibly induces tumor apoptosis,which could serve as an effective strategy to combat cancer.
Taken together,these findings demonstrate thatH.serrataextract has the potential to alleviate tumors by effectively inhibiting tumor growth through cell cycle arrest and the induction of apoptosis.Our study acknowledges two primary limitations.First,while we have established the anticancer efficacy ofH.serrataextract against HepG2 cells,further research is needed to generalize these findings across a broader spectrum of liver cancer types.Future studies should include a variety of cell lines to comprehensively validateH.serrataextract’s anticancer effects in diverse liver cancer contexts.Second,our investigation was confined to cellular-level analysis.To advance our understanding,forthcoming research will employ xenograft models to explore thein vivoanticancer efficacy ofH.serrataextract.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Funding
This research was funded by the GRRC Program of Gyeonggi province [GRRC-KyungHee2023(B01)],Republic of Korea.
Data availability statement
The data supporting the findings of this study are available from the corresponding authors upon request.
Authors’contributions
Both YEK and JH wrote the manuscript,performed experiments,collected and analyzed data,and designed this study.KYK contributed to supervision and revision of the manuscript.All authors reviewed the final manuscript.
 Asian Pacific Journal of Tropical Biomedicine2024年2期
Asian Pacific Journal of Tropical Biomedicine2024年2期
- Asian Pacific Journal of Tropical Biomedicine的其它文章
- Catalpa bignonioides extract improves exercise performance through regulation of growth and metabolism in skeletal muscles
- Icariin plus curcumol enhances autophagy through the mTOR pathway and promotes cathepsin B-mediated pyroptosis of prostate cancer cells
- Benzydamine hydrochloride ameliorates ethanol-induced inflammation in RAW 264.7 macrophages by stabilizing redox homeostasis
- NUDT5 promotes the growth,metastasis,and Warburg effect of IDH wild-type glioblastoma multiforme cells by upregulating TRIM47
