Gold/Mg-Al mixed oxides catalysts for oxidative esterification of methacrolein: Effects of support size and composition on gold loading
Wangtao Li, Qiancheng Zheng, Huayu Zhang, Yunsheng Dai, Zhengbao Wang,
1 Key Laboratory of Biomass Chemical Engineering of Ministry of Education, ERC of Membrane and Water Treatment (MOE), Zhejiang Provincial Key Laboratory of Advanced Chemical Engineering Manufacture Technology, College of Chemical and Biological Engineering, Zhejiang University, Hangzhou 310027, China
2 Yunnan Precious Metals Laboratory Co., Ltd, Kunming 650106, China
Keywords:Gold catalysts Oxidative esterification Support particle size effect Mass transfer Distributions Optimization
ABSTRACT Gold catalysts supported on Mg-Al mixed oxides for oxidative esterification of methacrolein are prepared by impregnation.Effects of the support particle size,concentration of HAuCl4 solution and Mg/Al ratio on gold loading and catalytic properties are investigated.The catalysts are characterized by CO2-TPD, EDS,XPS,STEM and XRD techniques.Catalysts with smaller support particle size show more uniform gold distribution and higher gold dispersion, resulting in a higher catalytic performance, and the uniformity of gold and the activity of the catalysts with larger support particle size can be improved by decreasing the concentration of HAuCl4 solution.The Mg/Al molar ratio has significant effect on the uniformity of gold and the activity of the catalyst,and the optimum Mg/Al molar ratio is 0.1-0.2.This study underlines the importance of engineering support particle size, concentration of HAuCl4 solution and density of adsorption sites for efficient gold loading on support by impregnation.
1.Introduction
Methyl methacrylate (MMA) is a significant organic material with various downstream products such as plexiglass, polyvinyl chloride (PVC) processing aids, high-performance coatings, and adhesives [1].Currently, the main route for industrial production of MMA is acetone cyanohydrin technique (ACH route) [2].And this process requires toxic chemicals and is lengthy, which is harmful to environment and has low economic benefits.To change this situation, one-step oxidative esterification of methacrolein(MAL) to product MMA has been widely researched, because it not only shortens the reaction route, but also eliminates the need of toxic materials, making it a green and sustainable alternative[3-7].Development of effective catalysts is the key to improving the industrial feasibility of this technology.
At early stage, iodine compounds [8] and heteropoly acid [9]were used in catalytic oxidative esterification.These catalysts have various issues such as use of additives, requirement of abundant methanol, low selectivity and long reaction time.With the development of nanotechnology [10], Pd [11-13] and Au [14-24] supported catalysts showed higher catalytic capability of oxidative esterification, among which gold supported catalysts have the highest activity and selectivity.However,the activity and stability of gold supported catalysts still need to be improved.These limitations leave enough room for the investigation of new effective catalysts in this system.
From liquid-phase studies it has been found that either the addition of a base or the use of a basic support is necessary for high yield of the corresponding methyl ester in oxidative esterification.Non-reducible materials are often used as supports of oxidative esterification catalysts, such as MgO [14], Al2O3[22], MgO-Al2O3-modified silica [23], hydroxyapatite [17] and molecular sieves(SBA-15)[24],due to their acid-base properties and specific surface area,which boost catalytic performance.Mg-Al mixed oxides supported catalysts have been widely used in organic reactions such as glycerol oxidation [25,26], methane oxidation [27], CO oxidation[28], selective hydrogenation of acetylene [29], and alcohol elimination [30] without addition of a base, because of its high specific surface area and tunable basic sites.However,there are no reports on oxidative esterification of MAL using Mg-Al mixed oxides as supports.
Supported gold catalysts are often prepared by deposition-preci pitation method[31-39],especially using urea or NaOH as precipitant [32-39].The results showed that the pH of HAuCl4solution[35,36], the temperature during preparation [36], the surface area of the support [37], the acid-base properties of the support (TiO2-TiOxNy-TiN)[38]and the electronic density of the support[39]had obvious effects on gold uptake and dispersion.The main support was TiO2in those studies [35-39].However, when gold is loaded on the alkaline support, the gold catalyst with high dispersion can be obtained by a simple contact between the HAuCl4solution and the support at a certain temperature,and the method is called as the impregnation method here.For example, Pitchon et al.[40]prepared Au/Al2O3catalysts by this method, although they also called the method as direct anionic exchange (DAE).AuNiOx/SiO2-Al2O3-MgO catalysts, which have a core-shell structure of Au@NiOx and have been commercialized in oxidative esterification of MAL by Asahi-Kasei Chemical Corporation, were also prepared by this method, although it was called as co-precipitation method[23].All these indicate that impregnation is also an effective and simple method to prepare gold supported catalysts.However,there are few studies on this method, hence it is worth investigating.
In this work, gold supported catalysts for oxidativeesterification of MAL are prepared on Mg-Al mixed oxides by the impregnation method, and effects of the support particle size, the concentration of HAuCl4and the Mg/Al molar ratio on properties of catalysts are investigated in detail.The Mg-Al mixed oxides will be prepared from Mg-Al hydrotalcite precursors.The catalysts are characterized by EDS, XPS, STEM, XRD, and CO oxidation reaction.It is demonstrated that the support particle size,the concentration of HAuCl4and the Mg/Al molar ratio have significant effects on gold loading and the catalytic activity for oxidative esterification of MAL.
2.Experimental
2.1.Support synthesis
According to the procedure reported in the literature [25,41],Mg-Al mixed oxides were prepared using a co-precipitation method with a little modification.An aqueous solution (A,100 ml) containing a total 100 mmol of Mg(NO3)2·6H2O (Sinopharm Chemical Reagent Co., Ltd, Shanghai, China) and Al(NO3)3-·9H2O (Sinopharm Chemical Reagent Co., Ltd) was agitated for 20 min at 65 °C with various molar Mg/Al ratios (X = 0, 0.1, 0.2 &0.3).An equal volume solution(B,100 ml)with NaOH(Sinopharm Chemical Reagent Co., Ltd; 200 mmol) and Na2CO3(Sinopharm Chemical Reagent Co., Ltd; 40 mmol) was agitated for 10 min at room temperature.Solution A was added into solution B by a peristaltic pump(rotating speed=3 r·min-1,BT300-2 J,Baoding Longer Precision Pump Co.,Ltd,China).The formed suspensions were agitated for 6 h at 65 °C, then filtered and washed extensively with deionized water.The obtained solid was dried over night at 110 °C.And the solid was calcined in air for 5 h at 600 °C with a rate of 2.5 °C·min-1to get Mg-Al mixed oxides.The produced Mg-Al mixed oxide was labeled as MgXAl, in which X denoted Mg/Al molar ratios.The calcined MgXAl mixed oxides were ground and sieved into different sizes (150-300 μm, 74-150 μm, 50-74 μm, <50 μm) labeled as MgXAl(Y) (Y = S1, S2, S3, S4),respectively).
2.2.Catalyst preparation
A typical wet impregnation process [23], also known as direct anionic exchange (DAE) [40], was used to prepare Au/MgXAl(Y)-Z with a theoretical gold loading of 1.1% (mass).0.377 g of 6.0%(mass) HAuCl4solution was typically introduced to Z g of water with a mechanical stirring for 15 min at 90 °C.And 1 g of MgXAl(Y) was added to the diluted HAuCl4solution after being predried for 30 min at 110 °C.The formed suspension was stirred for 30 min at 90°C,followed by extensive washing with deionized water.The obtained solid was dried for 4 h at 110 °C before being calcined in air at 300 °C with a rate of 2.5 °C·min-1.After 5 h of calcination, the catalyst was labeled as Au/MgXAl(Y)-Z, where Z also refers to the liquid-solid ratio (Z:1).
2.3.Catalyst characterization
Gold contents in catalysts were measured on an Optima ICPOES spectrometer by Agilent 5110.Cl contents in catalysts were determined by ion chromatography (IC) on a Thermo Scientific AQUIONRFIC.X-ray powder diffraction (XRD) analysis was performed on a D2 PHASER (Bruker), Cu-source powder diffractometer.Cu Kα radiation was used with a power setting of 40 kV and 40 mA.A scan rate of 5(°)·min-1with a 0.04° step size was used.N2physisorption experiments were performed on a Micromeritics TriStar II 3020.The total surface area of each catalyst was obtained using BET equation, and pore size was determined by BJH desorption analysis.X-ray photoelectron spectroscopy (XPS) analysis was carried out with a Thermo Scientific K-Alpha instrument using Al Kα as X-ray source.The binding energy of elements was calibrated with reference to the C 1s peak for adventitious carbon at 284.8 eV.The high-angle annular dark filed scanning transmission electron microscopy (HAADF-STEM) was obtained on a FEI Tecnai G2 F20 S-TWIN.And at least 100 particles from high-magnification STEM pictures were counted for the estimation of average gold nanoparticle size.
Temperature-programmed desorption (TPD) experiments were performed on a PX200 Multi-sorption equipment (Tianjin Golden Eagle Technology Co., Ltd, China).The sample (70 mg) was pretreated by purging with flowing dry He (20 ml·min-1) at 650 °C for 1 h and then cooled to 100 °C for adsorption of CO2for 30 min.After purging with flowing He to remove any physically adsorbed CO2, the CO2-TPD curve was subsequently determined by passing He (20 ml·min-1) through the preheated sample whilst the temperature was increased from 100 to 700 °C at a rate of 10 °-C·min-1.A thermal conductivity detector (TCD) was used to determine the amount of CO2desorption by this process.The element of catalyst was determined by energy dispersive X-ray spectroscopy(EDS) on an Oxford X-max 80.The support particle size was measured by laser diffraction on a Malvern Mastersizer 2000.
The CO oxidative reaction was performed at atmospheric pressure using a fixed bed quartz micro-reactor packed with 100 mg of catalyst.A mixture containing 0.5% CO and 10% O2(He as eluant gas) with a flow rate of 30 ml·min-1was used over the temperature range RT - 300 °C with a ramp rate of 2 °C·min-1.A TCD detector was used to perform the analysis for CO.The catalytic performance was assessed in terms of the t50temperature, defined as the temperature at which a 50% conversion was obtained.
2.4.Catalytic performance
The catalytic performance was evaluated by the one-step oxidative esterification of methacrolein (MAL) to methyl methacrylate(MMA).The main reaction was as follows:
The main by-product is methacrylic acid (MAA) and propene,and the side reactions were as follows:
And standard reaction conditions were as follows: A combination of Au/MgXAl(Y)-Z (720 mg, Au: 0.036 mmol), MAL (2.21 g,99%(mass),31 mmol,containing 800 mg·kg-1polymerization inhibitor,hydroquinone),and methanol(MeOH,33.62 g,99.5%(mass),1045 mmol) was placed into a stainless-steel autoclave (100 ml)fitted with a mechanical stirrer.The autoclave was pressurized to 1 MPa with nitrogen and stirred (600 r·min-1) after purging six times with nitrogen.When the temperature reached 80 °C, the pressure in the autoclave was increased to 2.9 MPa with nitrogen,and then the oxygen gas was incorporated to make the system pressure reach 3.2 MPa.During reaction, the oxygen valve remained open to maintain a constant oxygen pressure.After reaction, the autoclave was cooled to ambient temperature and gently depressurized.The catalyst Au/MgXAl(Y)-Z was separated from the reaction mixture by filtration.The reaction mixture was analyzed by gas chromatography with an FID detector.Toluene was used as an internal standard to evaluate the concentrations of MAL,MMA,methacrylate acid(MAA),and other byproducts.The conversion of MAL (XMAL), yield of MMA (YMMA) and selectivity of MMA(SMMA) were calculated by following equations.
3.Results and Discussion
3.1.Influence of support particle size
Basically, the particle size of catalysts and adsorbents is an important factor in heterogeneous chemical reactions [42,43] and adsorption processes [44,45], respectively.The size-dependence of metal nanoparticles (NPs, especially for gold) has been confirmed in various catalytic reactions because nanoscale effects are stronger when the NP size decreases, in particular to below 5 nm [46-49].In supported catalyst systems, dispersed metal NPs are loaded on the supports that are usually several orders of magnitude larger than the metal NPs to ensure high dispersion.However, as far as we know, the effect of support particle size on properties of metal particles in supported catalysts remains largely unexplored,especially for gold supported catalysts.Therefore,it is useful to examine what will happen if the support size is tailored.Herein, we systematically investigated the influences of the support particle size on properties of catalysts of Au/Mg-Al mixed oxides prepared by impregnation.
3.1.1.Catalytic performance
The Mg-Al mixed oxides were synthesized imitating the preparation method of layered double hydroxides (LDHs) [25,41].The XRD patterns of MgXAl(S3)-AP before calcination (as prepared,AP) are shown in Fig.S1 in Supplementary Material.The XRD pattern of Mg0Al(S3)-AP shows the diffraction peaks of Al2O3(PDF#49-0134), while those of Mg0.1-0.3Al(S3)-AP show weak peaks at 11.7°, 23.6°, 35.6° & 39.7°, which belong to (003), (006),(009) & (015) lattice planes of Mg4Al2(OH)14(PDF#35-0964), and peaks at 28.2°, 38.2°, 48.9° attributed to (120), (031), (051) lattice planes of boehmite (AlO(OH), PDF#21-1307), respectively.This indicates that, with co-precipitation method [25,50,51], LDHs structure can be partially formed even under non-stoichiometric ratio(e.g.,Mg/Al molar ratio=0.2)and the excess Al is in the form of boehmite.
The Mg0.2Al mixed oxide was chosen as the preferred support for investigation of the support particle size effect since it has the maximum specific surface area and adequate basic sites among Mg-Al mixed oxides with different Mg/Al molar ratios[25],both of which are crucial in the oxidative esterification of aldehydes [16-18,24].The texture properties of Ma0.2Al mixed oxide were characterized by N2adsorption-desorption (Fig.S2) technique.Based on calculation results of N2adsorption-desorption isotherms,Mg0.2Al mixed oxide had BET specific surface area of 188 m2·g-1and average pore size of 8.5 nm.Calcined Mg0.2Al mixed oxide particles were ground and sieved into four grades (S1,S2,S3,S4), as shown in Table 1.The average support particle sizes of relevant grades(S1-S4) estimating by laser diffraction were 252.5, 147.1, 71.6 and 27.4 μm, respectively, which match to the aperture diameter of the corresponding mesh (Table S1, in Supplementary Material).Support particle size distributions from the laser diffraction are shown in Fig.S3.

Table 1 Support sizes, Au contents, specific activity and basicity of Au/Mg0.2Al(Y)-5 catalysts
As shown in Fig.1 and Table 1,the support particle size showed a stronginfluence on the catalyticactivity in oxidative esterification of MAL over Au/Mg0.2Al(Y)-5 catalysts.With the decrease of the support particle size,the conversion of MAL increased while the selectivity remained almost unchanged (SMMA≈ 93%).When the average support particle size is larger than 140 μm (Mg0.2Al(S1)and Mg0.2Al(S2)),the conversion of MAL is much lower(e.g.,<40%)and the reaction rate (rMAL) is lower than 28.2 mmol·min-1·g-1,while the conversion of MAL is higher than 87% when the average support particle size is smaller than 70 μm(Mg0.2Al(S3)and Mg0.2-Al(S4)),and the rMALincreases to 69.7 mmol·min-1·g-1.This shows that the activity of the catalyst can be doubled when the size of the support particle is small enough.Moreover,when the support particle size is further reduced from 70 μm to 27 μm,the activity of the catalyst is not significantly increased.It is worth mentioning that Au/Mg0.2Al(S3)-5 and Au/Mg0.2Al(S4)-5 reached a very high level of MAL conversion.It only takes 1 h to achieve ~88%of XMAL,whereas other reported catalysts (e.g., Au/MgO, Au/SBA-15, etc.) need 2 h under a similar reaction condition[14,16,19-21].
It is known that basicity and gold dispersion of catalyst are both crucial in oxidative esterification [14,17,22-24].Hence, CO2-TPD was carried out on Au/Mg0.2Al(Y)-5 catalysts (Fig.S4), and basic sites of catalysts were calculated based on TPD profiles (Table 1).Results showed that four catalysts have the similar number of basic sites (average value: 58.7 μmol·g-1).Therefore, we turned our attention on the gold dispersion.The CO oxidation reaction as a model reaction can be used to evaluate the activity of gold active sites in catalysts.Compared to the MAL oxidative esterification, basicity of supports and internal diffusion are not primary in the CO oxidation, and the gold active site is more important[52,53].The CO oxidation reaction is structure-sensitive and therefore, a difference in activity reflects a change of the particle size.Therefore, to determine whether the difference in the catalytic oxidative esterification activity is caused by the difference in gold active sites between Au/Mg0.2Al(Y)-5 catalysts, the temperatureprogrammed CO oxidation reaction was conducted over these catalysts.As shown in Fig.2, the CO conversion increased with the reaction temperature over all catalysts.With the reduction of support particle size, the reactivity curves are from right to left, and the t50are 251, 174, 63 and 47 °C for the catalysts Au/Mg0.2Al(S1,S2, S3, S4)-5, respectively.This indicates that the activity of Au/Mg0.2Al(Y)-5 catalysts increased with the decrease of the support particle size.This is consistent with the oxidative esterification reaction of MAL, indicating that the difference in the catalytic oxidative esterification activity between Au/Mg0.2Al(Y)-5 catalysts is caused by the difference in gold active sites.Therefore,the characterization of gold active sites in Au/Mg0.2Al(Y)-5 catalysts was carried out in detail.
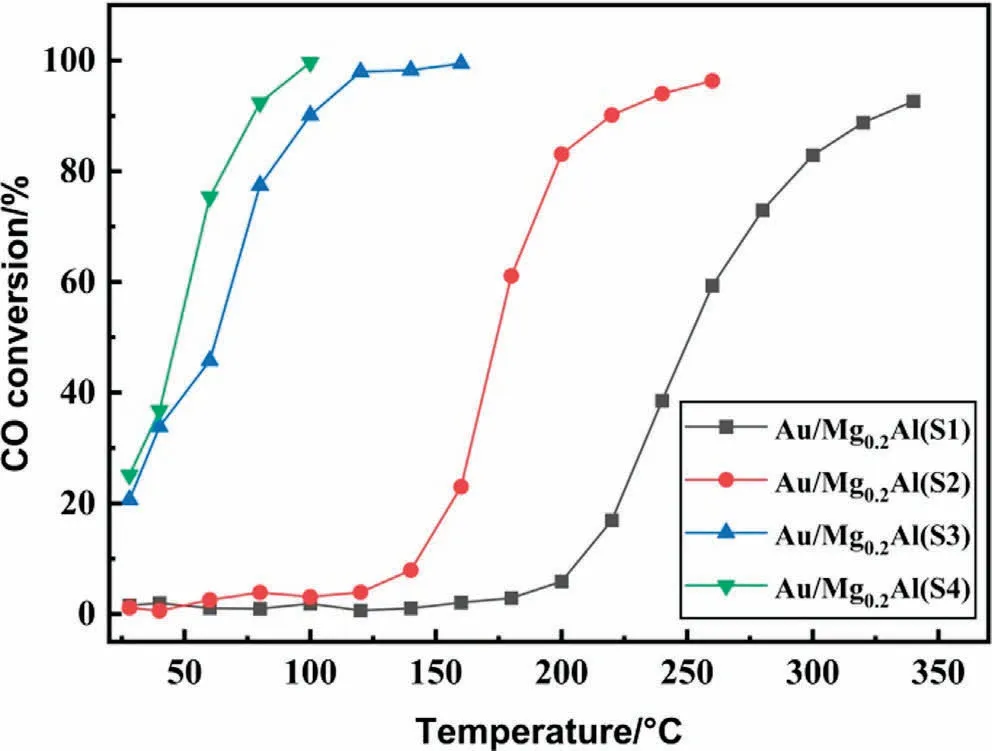
Fig.2.Catalytic activity of Au/Mg0.2Al(Y)-5 catalysts for CO oxidation.
3.1.2.Characterization of gold catalysts
To observe the appearance of catalysts, optical pictures were taken, which is a simple and typical way to identify the gold state[54].As shown in Fig.S5,the color of catalysts became lighter with the decrease of the support particle size.That is, Au/Mg0.2Al(S1)-5 appears bluish violet (Fig.S5(a)), indicating the presence of large gold particles.Au/Mg0.2Al(S2)-5 appears purplish red (Fig.S5(b)),indicating the presence of relatively large gold particles.The similar light color of Au/Mg0.2Al(S3)-5 and Au/Mg0.2Al(S4)-5 proves that their gold dispersion is good and close to each other.Variations in catalyst color and catalytic activity are comparable, the high catalytic activity of Au/Mg0.2Al(S3)-5 and Au/Mg0.2Al(S4)-5 with smaller support particle size than ~70 μm is due to the high gold dispersion.
HAADF-STEM can be used to characterize the gold particle size and their dispersion.As shown in Fig.3, the four catalysts Au/Mg0.2Al(Y)-5 with different support particle sizes have large and small gold particles.It is worth noting that average gold particle sizes (Fig.3(i)-(l)) acquired from high-magnification STEM pictures (Fig.3(e)-(h)) did not include large gold particles.Although the difference of small gold particle size (0.9-1.0 nm) between the four catalysts is small, it is obvious that the size of large gold particles decreases with the decrease of support size.Among them,the Au/Mg0.2Al(S1)-5 catalyst with the largest support particle size has large gold particles up to ~40 nm(Fig.3(a)).The occurrence of such enormous gold particles will result in a drop in gold dispersion as well as the number of gold active sites,which is the primary reason for lower catalytic activity.The size of large gold particles in Au/Mg0.2Al(S2)-5 is ~10 nm.Such oversized gold particles are difficult to see in STEM images of Au/Mg0.2Al(S3)-5 and Au/Mg0.2Al(S4)-5 under the same magnification (Fig.3(c) and (d)).And the greatest gold particle size is similarly 5 nm in these two catalysts.In addition, the average size of small gold particles of these two catalysts is also smaller than that of the Au/Mg0.2Al(S1,S2)-5 catalysts(Fig.3(k)and(l)).Therefore,these two catalysts with smaller support particle size than ~70 μm have high gold dispersion and high catalytic activity.
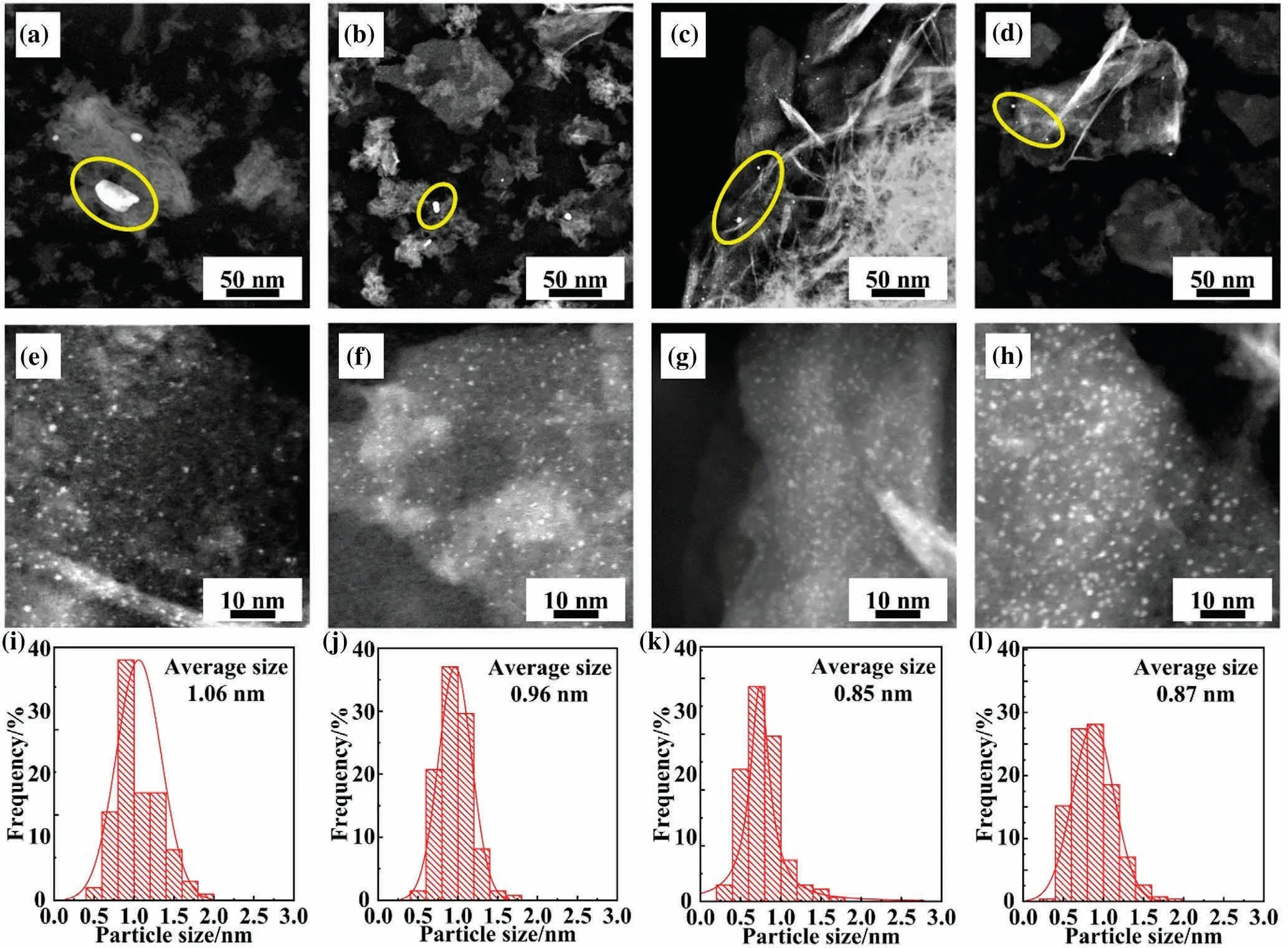
Fig.3.STEM images and gold particle size distributions of catalysts:(a,e, i) Au/Mg0.2Al(S1)-5, (b, f,j)Au/Mg0.2Al(S2)-5, (c,g,k) Au/Mg0.2Al(S3)-5,(d, h,l) Au/Mg0.2Al(S4)-5.
XRD patterns in Fig.4 show a noticeable variation in the degree of gold aggregation of the four catalysts with different support particle sizes.The diffraction peak of Au(111)at 38.2°(PDF#04-0784)can be clearly observed in the pattern of Au/Mg0.2Al(S1)-5,which is provided by oversized gold particles, indicating that there is a significant amount of gold aggregation.When the support particle size decreases, the characteristic peak of Au(111) in Au/Mg0.2Al(S2)-5 pattern is clearly diminished, and it is no longer visible in Au/Mg0.2Al(S3)-5 and Au/Mg0.2Al(S4)-5.This also demonstrates that when the support particle size decreases, the degree of gold aggregation reduces and dispersion increases.This is also consistent with the tendency of catalytic activity change.
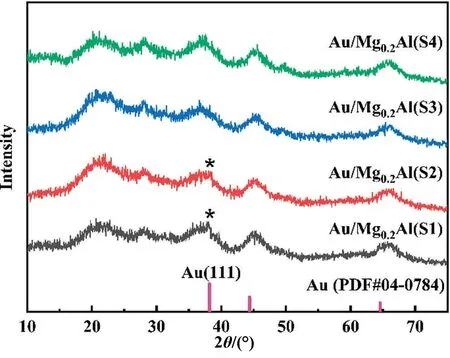
Fig.4.XRD patterns of Au/Mg0.2Al(Y)-5 catalysts.
A number of compositional analytical characterizations were employed to describe the content and the chemical state of gold in catalysts.According to ICP-OES data in Table 1,the real Au content in Au/Mg0.2Al(Y)-5 is ~1%(mass),close to the theoretical loading (1.14% (mass)), which infers that gold is affectively loaded on the surface of Mg0.2Al(Y) by impregnation method.And as previously reported[23],this is a typical method to locally load AuNiOxon the support surface, resulting in an egg-shell type catalyst.
To identify the Au content on catalyst surface (Ausurcontent),EDS was employed.Ausurcontents were determined by measuring all potential elements in catalysts.As shown in Table 1, the Ausurcontent of catalyst has a positive correlation with the support particle size while the real Au content is similar to each other.Au/Mg0.2Al(S1)-5 with the largest support particle size among four catalysts has the highest Ausurcontent of 1.83%, while Au/Mg0.2Al(S4)-5 with the smallest particle size has the lowest Ausurcontent of 1.00%.For Au/Mg0.2Al(S1)-5, its Ausurcontent is about two-fold of the average Au content by ICP, in which a significant quantity of gold is concentrated on the surface, making it an egg-shell type catalyst (non-uniform distribution).The Ausurcontent decreases with the decrease of support particle size, suggesting that nonuniform distribution of gold is lessened.The result that the size of metal particles is related to the size of support particles has been reported for supported Ru[55,56]and Ni[57]catalysts.It is important to note that for Au/Mg0.2Al(S3)-5 and Au/Mg0.2Al(S4)-5, their Ausurcontents are quite similar to the average Au content and there is no significant difference between two catalysts.This demonstrates that non-uniform distribution of gold essentially vanishes when the size of support particle is smaller than 70 μm.
XPS analysis was also carried out to measure the distribution of gold particles in catalysts.As shown in Fig.5,the surface composition and oxidation state of Au/Mg0.2Al(Y)-5 catalysts were obtained.Au 4f7/2peaks at (84.6 ± 0.1) eV (green pattern) and(86.8±0.1)eV(pink pattern),corresponding to the typical binding energy of Au0and Auδ+,respectively[58-61],are found in the area of Au 4f signal.The XPS pattern also contains Mg 2s peak (yellow pattern).As the support particle size decreases, the relative intensity of Au 4f peaks decreases, suggesting that a decrease of Ausurcontent.To further verify this phenomenon, surface atomic Au/Mg ratios (Fig.5(b)) were calculated by using the intensity of Au 4f and Mg 1s(Fig.S6)signals and the respective atomic sensitivity factors.As previously predicted, the Au/Mg ratio reduces with the decrease of the support particle size.This further demonstrates that gold is more uniformly dispersed on the small particle size support.And gold in the large particle size support (e.g., Mg0.2Al(S1)) is more likely a non-uniform distribution.
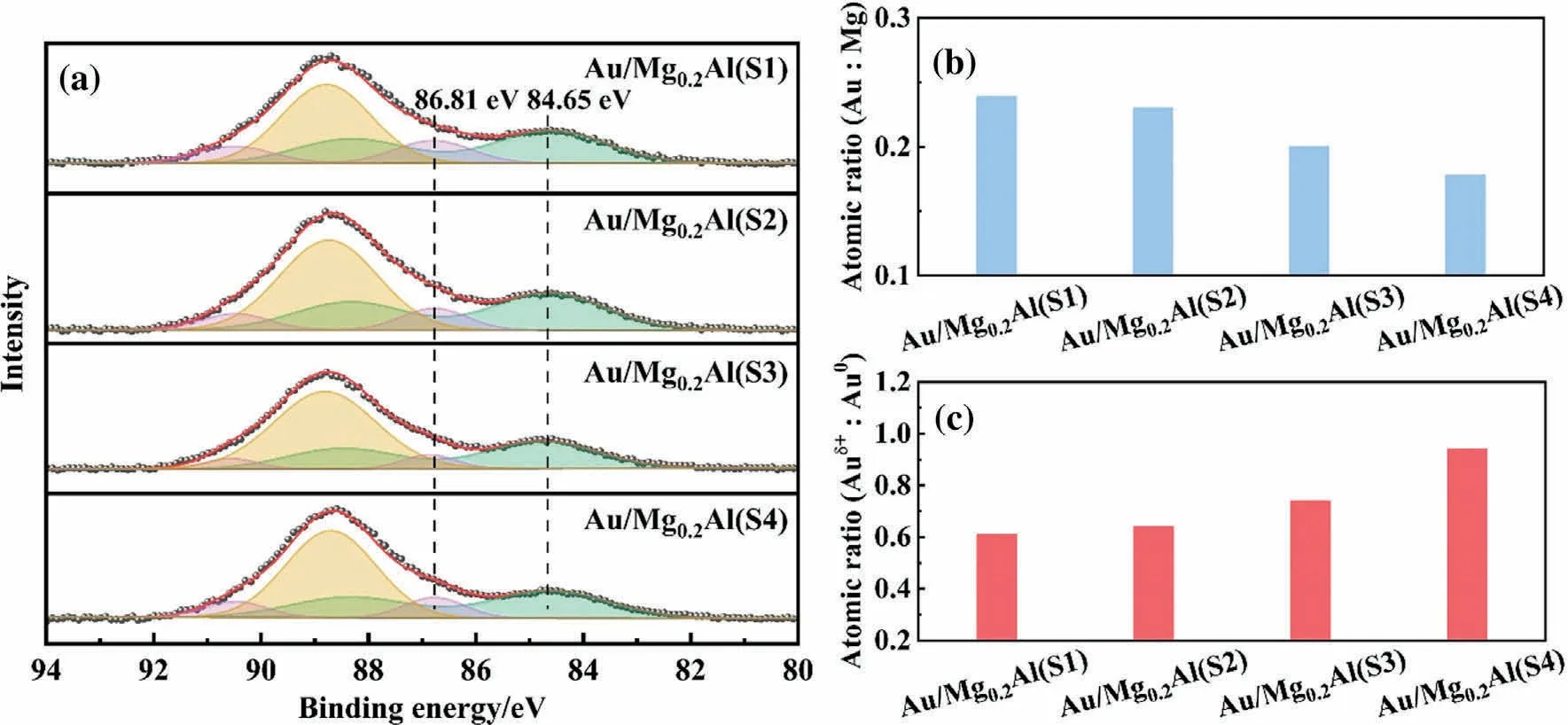
Fig.5.(a)XPS patterns of Au 4f and Mg 2s regions of Au/Mg0.2Al(Y)-5 catalysts,yellow pattern refers to Mg2+,pink pattern refers to Auδ+,green pattern refers to Au0,red line refers to peak sum,black scatter refers to raw intensity; (b) Surface atomic Au/Mg ratios and (c) atomic Auδ+/Au0 ratios of catalysts as derived from Au 4f and Mg 1s peaks.
As previously reported [62], the degree of gold dispersion can also be evaluated by studying its electronic state.Auδ+species mostly appears at the interface between gold particles and support,whereas Au0species appears between gold atoms.This indicates that the higher the Auδ+/Au0ratio,the more the gold species in contact with support and the higher the gold dispersion.In this work,the Auδ+/Au0ratios were determined by Au 4f signal in XPS patterns and are shown in Fig.5(c).The ratio increases as the support particle size reduces,suggesting that the average gold particle size lowers and gold dispersion increases.
3.1.3.Discussion
According to the above characterization of gold dispersion,average Au content, and gold distribution (Ausur), the following results are clear.(1) The gold distribution is not uniform for the Au/Mg0.2Al(S1,S2)-5 catalysts with the support particle size larger than 140 μm; the Ausurcontent is higher than 1.8% (mass) for Au/Mg0.2Al(S1)-5 with the support particle size larger than 250 μm,which is almost twice the average Au content (1.0% (mass)).(2)The gold distribution is uniform for the Au/Mg0.2Al(S3, S4)-5 catalysts with the support particle size smaller than 70 μm;their Ausurcontent is 1.1%-1.0% (mass),which is very close to the average Au content (1.0% (mass)).Therefore, the schematic formation of gold particles on the Au/Mg0.2Al support is shown in Fig.6.For various porous supports (Mg0.2Al(S1-S4)) with the same mass but different particle sizes, the small particle support has a larger surface area and more open pores on the surface,which improves the uniform loading of AuCl4-on highly dispersed spinel sites(Fig.S7)during impregnation (Fig.6(a2)).After drying and calcination, a more uniform gold distribution and higher gold dispersion can be obtained on the smaller particle support(Fig.6(b2)),and the Ausurcontent is close to the average Au content in the whole catalyst particle.However, the large particle support has a lower surface area and less open pores on the surface,which is not advantageous for the uniform loading of AuCl4-species during impregnation.More AuClspecies are adsorbed on the outer surface of the support particle, resulting in a non-uniform gold distribution and egg-shell structure (Fig.6(a1)).After drying and calcination, some large gold particles and lower gold dispersion are obtained due to aggregation of high gold loading on the coating area (Fig.6(b1)).Highly active gold catalysts might be prepared on large particle supports(e.g.,Mg0.2Al(S2))as long as the gold loading area is raised by modulating mass transfer process during impregnation process,such as slowing down the adsorption rate or speeding up the diffusion rate.In the following, influences of the liquid-solid ratio (the concentration of HAuCl4) and the Mg/Al molar ratio will be investigated.
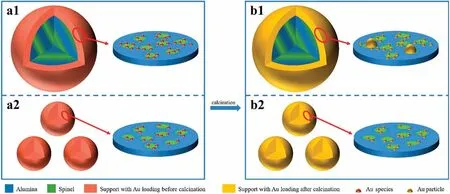
Fig.6.Schematics of Au/Mg-Al mixed oxide catalysts with different support particle sizes (1: large, 2: small): (a) as-prepared after impregnation, and (b) after calcination.
In order to confirm the schematic formation of gold species in Fig.6, the pH evolution of impregnation solution was studied.As shown in Fig.7, although the final pHs at 30 min are close for the four catalysts with different support particle sizes, the pH increased very quickly within 10 min of impregnation and the increase rates are different for the four catalysts.For Au/Mg0.2Al(S4)-5 with the smallest particle size,the pH quickly increased during impregnation process and is close to 7 at 5 min, at which gold species are free of Cl-[40].However,for Au/Mg0.2Al(S1)-5 with the largest particle size,the pH at 5 min is lower than 5.0,at which the gold species are present as [AuCl2(OH)] [40].These results prove that the interaction (diffusion and adsorption) speed of HAuCl4impregnation solution with different size supports is different,that is, the interaction speed of small size supports with impregnation solution is fast,resulting in the quick increase of pH.This is consistent with the speculation in Fig.6.
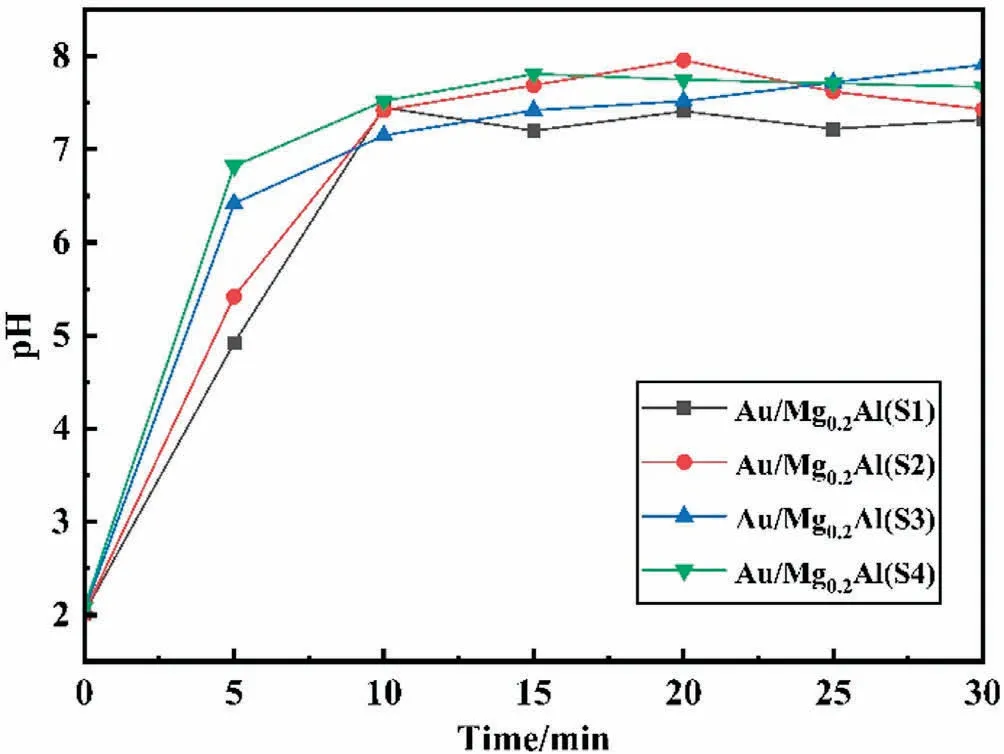
Fig.7.Evolutions of the impregnation solution pH with time after addition of Mg0.2Al(Y).
As reported before [40], the lower pH of gold species solution,the higher Cl-content in gold species.That is, gold is present as[AuCl] at pH <2 and gold is present as [Au(OH)] at pH >7.It is known that the higher the Cl-content,the easier the gold aggregation [40,63].Therefore, it is considered that the evolution of pH during impregnation reflects the residual amount of Cl-in catalysts.That is, the chlorine content in Au/Mg0.2Al(S4) might be lower than that in Au/Mg0.2Al(S1) due to the quick increase of pH to ~7.0 at 5 min for the former.The difference in element analysis of chlorine is shown in Table S2, and the Cl-content of Au/Mg0.2Al(S1)-5 is two-fold that of Au/Mg0.2Al(S4)-5.The high Clresidual leads to gold aggregation during calcination, resulting in large gold particles in the catalyst.This might be another reason for the low gold dispersion of Au/Mg0.2Al with larger support particle size than 140 μm.
3.2.Influence of HAuCl4 concentration
In order to find out whether a highly active catalyst can be obtained from the large particle support (e.g., Mg0.2Al(S2)) in the low concentration HAuCl4solution, the influence of HAuCl4concentration was investigated by adding different amounts of water for dilution while maintaining a certain amount of total HAuCl4in solution.As shown in Table 2, when liquid-solid ratios are 5,10, 20, 40 & 60, the HAuCl4concentration are 1.2 × 10-2mol·L-1,5.9 × 10-3mol·L-1, 2.9 × 10-3mol·L-1, 1.5 × 10-3mol·L-1and 9.8 × 10-4mol·L-1, respectively.Fig.8 depicts the MAL oxidative esterification reaction activity and selectivity over Au/Mg0.2Al(S2)-Z.The activity of catalysts rises as the HAuCl4concentration decreases down to 1.5 × 10-3mol·L-1.That is, when the liquidsolid ratio = 40, the activity reaches the highest level (XMAL= 92%,rMAL= 82.9 mmol·min-1·g-1, Table 2), which is even higher than the one of Au/Mg0.2Al(S4)-5(rMAL=69.7 mmol·min-1·g-1,Table 1).When the HAuCl4concentration decreases to 9.8 × 10-4mol·L-1(Au/Mg0.2Al(S2)-60), the conversion of MAL starts to decrease(rMAL=81.0 mmol·min-1·g-1,Table 2).It is worth noting that SMMA(~92%)of catalysts hardly changes with the HAuCl4concentration,which is consistent with the literature’s exposition that SMMAin MAL oxidative esterification reaction is primarily controlled by basic sites on supports [16,21,64].
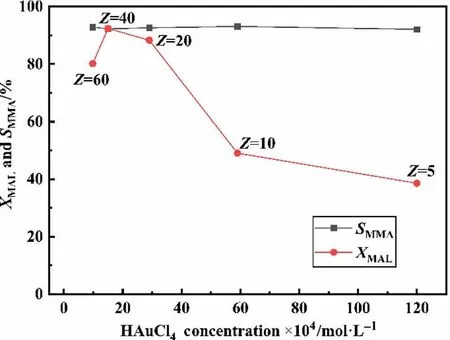
Fig.8.Catalytic performances of Au/Mg0.2Al(S2)-Z for oxidative esterification of MAL with methanol to MMA.
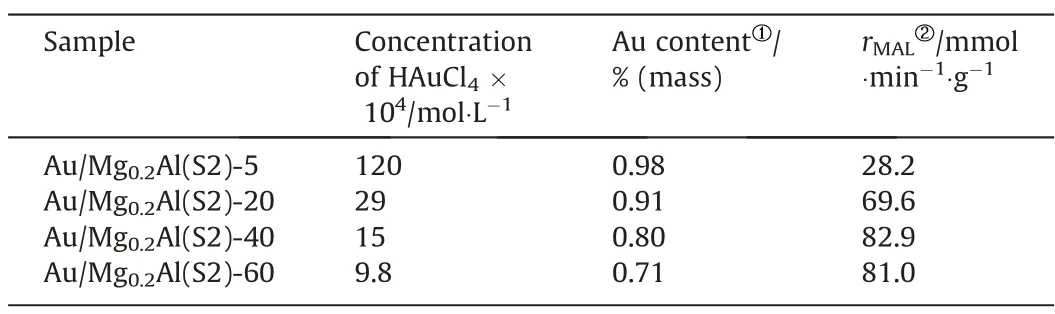
Table 2 Au contents and specific activity of Au/Mg0.2Al(S2)-Z catalysts
In order to find out what happened to physical properties of Au/Mg0.2Al(S2) when the HAuCl4concentration was decreased, STEM was carried out.As shown in Fig.9, the size of oversized gold particles in the catalyst decreased with the increase of liquid-solid ratio (the decrease of the HAuCl4concentration) to 40 (Fig.9(a)and (b)).Compared with Au/Mg0.2Al(S2)-5 (Fig.3(b) and (j)) and Au/Mg0.2Al(S2)-20 (Fig.9(a) and (g)), the oversized gold particles(size = ~5 nm, Fig.9(b)) and the gold particles under higher magnification (average size = 0.69 nm, Fig.9(h)) of Au/Mg0.2Al(S2)-40 are both smaller,which means that decreasing the HAuCl4concentration in a suitable range can improve the gold dispersion and increase active sites, leading to higher catalytic activity.However,when the liquid-solid ratio was further increased to 60,the size of oversized gold particles increased.
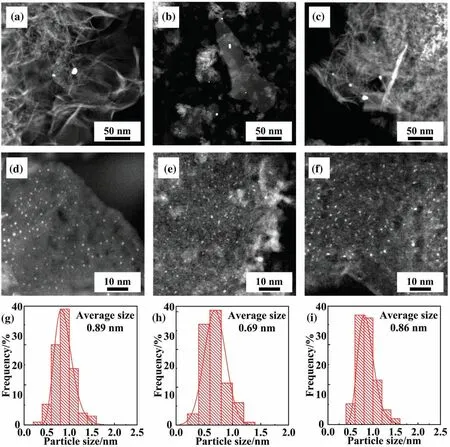
Fig.9.STEM images and gold particle size distributions of Au/Mg0.2Al(S2)-20 (a, d, g), Au/Mg0.2Al(S2)-40 (b, e, h), Au/Mg0.2Al(S2)-60 (c, f, i) catalysts.
The HAuCl4concentration was decreased as the liquid-solid ratio increases, which leads to the decrease of the adsorption rate of AuCl4-species on support particles.Nevertheless,the decrease of adsorption rate will lead to deeper gold loading depth and larger loading area of large particle support(e.g.,Mg0.2Al(S2)).Therefore,the Ausurcontent reduced with the decrease of the HAuCl4concentration, as well as gold aggregation (oversized gold particles) during calcination, resulting in higher gold dispersion (Fig.9(h)) and higher catalytic activity of the catalyst.The decrease of HAuCl4concentration further reduces not only the adsorption rate but also the diffusion rate during impregnation.Loading area of gold is lessened and inhibition of aggregation is decreased when the liquidsolid ratio = 60 (HAuCl4concentration = 9.8 × 10-4mol·L-1).As shown in Fig.9(c), for Au/Mg0.2Al(S2)-60, oversized gold particles with size of ~10 nm appeared again,and the average size of small gold particle rise to 0.86 nm, demonstrating a reduction of gold dispersion.
It can be seen from Table 2 that when the liquid-solid ratio increases,the Au content of the catalyst decreases.Especially when the liquid-solid ratio is greater than 40, the Au content of the catalyst decreases faster.This is due to the limitation of adsorption equilibrium, because the lower HAuCl4concentration of solution,the lower gold loading ratio [65].Even though the Au content of Au/Mg0.2Al-40 catalyst decreased significantly, the dispersion of gold increased a lot, so its catalytic activity was still higher than that of Au/Mg0.2Al-20.For Au/Mg0.2Al-60 catalyst, not only the Au content decreased significantly, but also the dispersion of gold was lower than that of Au/Mg0.2Al-40, so its catalytic activity decreased instead.
In summary,although the particle size of Mg0.2Al(S2)is greater than 140 μm, the gold loading behavior on the support can be greatly improved by reducing the concentration of HAuCl4impregnation solution.When the concentration of HAuCl4impregnation solution decreased to 1.5 × 10-3mol·L-1, the MAL conversion of the catalyst obtained is up to 92%.
3.3.Influence of Mg/Al ratio
It was reported that a preferential deposition of gold could occur on certain sites or atoms for gold supported on mixed oxides(CeO2/Al2O3) prepared by deposition-precipitation method [39],which means that the modulation of support composition might change gold loading behavior during preparation.In order to find out whether the density of adsorption sites (spinel species) of smaller particle support affects the gold loading behavior, MgXAl(S3) with different Mg/Al molar ratios (X = 0, 0.1, 0.2 & 0.3) were prepared by the same method.After calcination, LDHs will transform to the Mg-Al spinel [50,51], while excessive Al exists in the form of oxides or hydroxides, and plays a role in dispersing Mg-Al spinel.The XRD patterns of supports in Fig.S7 show that, with increase of Mg/Al molar ratio of MgXAl(S3), spinel phase begins to appear.When Mg/Al molar ratio = 0, the XRD pattern of Mg0Al(S3) only shows the characteristic peak of Al2O3(PDF#49-0134)at 46.2°and 67.3°.And the XRD patterns of Mg0.2Al(S3)and Mg0.3-Al(S3)show the characteristic peak of Mg-Al spinel(PDF#21-1152)at 19.0°, 36.9°, 44.8° and 65.2°.CO2-TPD profiles of supports with Mg/Al molar ratios of 0, 0.1, 0.2 and 0.3 are shown in Fig.S8.Two desorption peaks at 100-270°C and 270-550°C are observed in CO2-TPD profiles of four samples.It is worth noting that only one peak at 100-270 °C was observed for the similar supports, which were recorded by quadrupole mass spectrometer [25].Therefore,we infer that the peak at 270-550 °C might due to the release of water from the sample and only the peak at 100-270 °C is contributed to desorption of CO2adsorbed on basic sites of Mg-Al mixed oxides in Fig.S8.When the Mg/Al molar ratio was increased,the CO2desorption peak area at low temperature increased, indicating that there are more basic sites in the support.It was reported that from Mg0Al to Mg0.3Al, the number of basic sites increased from 11.1 to 68.0 μmol·g-1[25].
The MAL oxidative esterification activity of catalysts with different Mg/Al ratios is shown in Fig.10.The activity of catalysts with the Mg/Al ratios of 0.1-0.2 is the highest(XMAL=~88%),while the activity of catalyst with the Mg/Al ratio of 0.3 is lower.Optical images of Au/MgXAl(S3)-5(X =0.1,0.2, 0.3)in Fig.S9 show lighter color of Au/MgXAl(S3)-5(X=0.1,0.2)and violet of Au/Mg0.3Al(S3)-5,illustrating the difference in gold dispersion.That is,Au/Mg0.3Al(S3)-5 has lower gold dispersion, resulting in lower activity in the reaction of oxidative esterification.It is speculated that when the number of basic sites is higher (e.g., Mg0.3Al), the adsorption of AuClis faster, resulting in the non-uniform distribution of gold species.That is,the Ausuris higher than that inside.It is worth noting that SMMA(~92%)of the three catalysts hardly changes with the Mg/Al molar ratio.
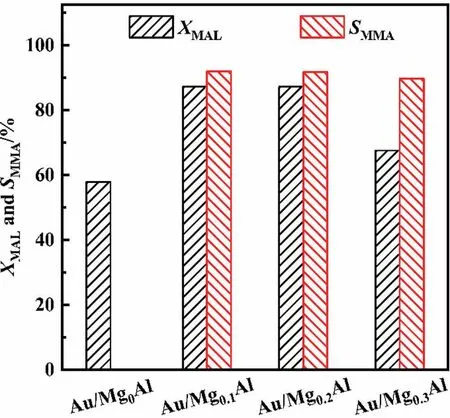
Fig.10.Catalytic performance of Au/MgXAl(S3)-5 catalysts for oxidative esterification of MAL with methanol to MMA.
Au/Mg0Al(S3)-5 is totally different from others.The MAL conversion is 58%,while the MMA selectivity is 0%and the methacrylic acid (MAA) selectivity is close to 100% (data not shown in the figure).The lack of Mg in Mg0Al leads to lower basic sites,resulting in the presence of less hydrolyzed gold complexes during the preparation and especially during the calcination.This leads to the formation of Au-Cl-Au bridges and particle agglomeration [40,63].From the color of optical image of Au/Mg0Al(S3)-5 (Fig.S9(a)),non-uniform gold distribution and gold aggregation can be seen directly.The gold color can even be seen in the reaction product,indicating the loss of gold particles from the catalyst.Therefore,the low activity of Au/Mg0Al(S3)-5 is due to the large gold particles and low gold dispersion.As for the very low MMA selectivity,possible reason is that the reaction stopped in the oxidation stage due to the lack of enough basic sites in Mg0Al and the lack of enough interfaces between gold particles and support surface.This also illustrates the mechanism of the oxidative esterification process,which needs the simultaneous activity of gold and the basic sites[66].There are two steps for the oxidative esterification process on the gold supported catalyst.In the first step, methanol is adsorbed by the acidic and basic sites on the support surface around the gold nanoparticles to form methoxyl groups, which then react with MAL to form the intermediate product hemiacetal[17,66].In the second step, the active sites in the gold nanoparticles activate the C-H bonds in the hemiacetal,while the basic sites around the gold nanoparticles activate the C-O bonds in the hemiacetal.Under the synergistic effect of these sites, the hemiacetal undergoes dehydrogenation to obtain MMA [67,68].However,when the gold is separated from the support,the two-step reaction pathways in the oxidative esterification process cannot be realized.That is to say, if MAL can only interact with free gold particles in the liquid phase, then it can only generate MAA by oxidation reaction.
4.Conclusions
Au/MgXAl(Y)-Z catalysts were prepared by the impregnation method and effects of the support particle size (Y), the HAuCl4impregnation solution concentration(Z)and the Mg/Al molar ratio(X) on properties of catalysts were investigated.Au/Mg0.2Al-5 catalysts with smaller support particle size (e.g., ≤70 μm) showed more uniform gold distribution and higher gold dispersion at the concentration of HAuCl4impregnation solution of 0.012 mol·L-1,resulting in higher catalytic performance, and the uniformity of gold and the activity of the catalysts with larger support particle size(e.g.,>140 μm)can be improved by decreasing the concentration of HAuCl4impregnation solution (e.g., 0.0015 mol·L-1).The density of the adsorption sites(e.g.,the Mg/Al molar ratio)had significant effect on the uniformity of gold and the activity of the catalyst, and the optimum Mg/Al molar ratio was 0.1-0.2 at the HAuCl4concentration of 0.012 mol·L-1.The results underline the importance of engineering the support particle size,the concentration of HAuCl4solution and the density of adsorption sites for efficient gold loading on support by impregnation.
Author Contributions
The manuscript was written through contributions of all authors.All authors have given approval to the final version of the manuscript.
Data Availability
Data will be made available on request.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work is supported by Open Project of Yunnan Precious Metals Laboratory Co., Ltd (YPML-2023050269) and the Fundamental Research Funds for the Central Universities (226-2023-00085, 226-2023-00057).
Supplementary Material
Supplementary material to this article can be found online at https://doi.org/10.1016/j.cjche.2023.05.006.
 Chinese Journal of Chemical Engineering2023年12期
Chinese Journal of Chemical Engineering2023年12期
- Chinese Journal of Chemical Engineering的其它文章
- Intrinsic kinetics of catalytic hydrogenation of 2-nitro-4-acetylamino anisole to 2-amino-4-acetylamino anisole over Raney nickel catalyst
- Experiments and model development of p-nitrochlorobenzene and naphthalene purification in a continuous tower melting crystallizer
- α-Synuclein: A fusion chaperone significantly boosting the enzymatic performance of PET hydrolase
- Influence of water vapor on the separation of volatile organic compound/nitrogen mixture by polydimethylsiloxane membrane
- Mass transfer mechanism and relationship of gas-liquid annular flow in a microfluidic cross-junction device
- Enhanced photocatalytic activity of methylene blue using heterojunction Ag@TiO2 nanocomposite: Mechanistic and optimization study
