Clinical efficacy of intravitreal corticoid as an adjunctive therapy to anti-VEGF treatment of neovascular agerelated macular degeneration: a Meta-analysis
Bo-Hao Cui, Wei Zhou, Wen-Wen Wang, Hao Yang, Ya-Lan Dong, Yuan-Yuan Liu, Hua Yan
Abstract
· KEYWORDS: age-related macular degeneration;dexamethasone; triamcinolone; anti-vascular endothelial growth factor; Meta-analysis
INTRODUCTION
Age‐related macular degeneration (AMD) is a leading cause of severe and irreversible vision loss worldwide,especially in developed countries[1‐2]. Ⅰn recent years,intravitreal injection of anti‐vascular endothelial growth factor(VEGF) has become the first‐line treatment of neovascular AMD (nvAMD). However, there are still a large number of patients with poor efficacy or resistance and reduced efficacy during anti‐VEGF monotherapy, and corticosteroids has been recommended desensitize tachykinin[3‐5]. Meanwhile,continuous retreatment adds a heavy financial burden to patients[6]. Studies have also shown that the progression of retinal pigment epithelium and choroidal atrophy is related to the total number of anti‐VEGF injections[7]. Therefore, it is particularly important to find an alternative treatment plan.
Ⅰn animal models, intravitreal injection of corticosteroids can simultaneously reduce VEGF production and choroidal neovascularization, inhibit inflammation, and reduce photoreceptor cell apoptosis which provide a theoretical basis for the synergy with anti‐VEGF treatment[8‐11]. On the other hand, intravitreal injections of triamcinolone acetonide (TA)and dexamethasone (DEX) combined with photodynamic therapy in the treatment of nvAMD have been proven to be safe and effective previously[12‐13]. Hence, additional corticosteroids seem to have the ability to target chronic inflammation when combined with anti‐VEGF with safety. Although DEX implant and direct intraocular corticosteroid injection have similar side effects—increasing risk of glaucoma and cataract progression,etc., these can be effectively controlled by anti‐glaucoma drugs or surgery[14‐15].
Up to now, there are studies (including series of single‐arm researches) showing that the addition of glucocorticoids on the basis of anti‐VEGF can promote disease remission, however,the issue still remains under debate. This Meta‐analysis aimed to evaluate the efficacy and safety of the intraocular corticoid as adjunctive therapy to anti‐VEGF in nvAMD comprehensively.
MATERIALS AND METHODS
Literature SearchA comprehensive literature search was conducted using 4 databases: including PubMed, Embase,Cochrane Library, and the clinicaltrials.gov, up to August 2020. The following MeSH terms were used: 1) “Macular Degeneration”, 2) “Βevacizumab” or “Ranibizumab”or“Aflibercept” or “Conbercept”, 3) “Triamcinolone” or DEX.There were no language or publication date restrictions and the reference list of retrieved articles was checked to identify potentially relevant studies. The flow diagram is shown in Figure 1. This systematic review and Meta‐analysis was designed, performed, and reported based on the quality standards of the reported Meta‐analysis. The study was conducted in accordance with the Cochrane Handbook for Systematic Reviews and Meta‐Analysis (PRⅠSMA) guidelines[16].
Selection CriteriaThe study was considered qualified if met the following criteria: 1) The study population includes nvAMD patients; 2) The intervention group includes intraocular corticoid treatment (DEX implant or injection of TA or DEX) combined with anti‐VEGF treatment; 3) There is a comparison between the combined treatment group with anti‐VEGF monotherapy group; 4) The research design should be a randomized controlled trial (RCT).
Through the preview of the study, we determined two main outcomes: 1) The improvement of best‐corrected visual acuity(ΒCVA) from baseline (time point: 6 and 12mo). 2) The average change of the central macular thickness (CMT) on the optical coherence tomography (OCT) from the baseline (time point: 6 and 12mo) and two additional outcomes: 1) Severe increase in intraocular pressure (ⅠOP) that need to be controlled by anti‐glaucoma drugs; 2) Cataract progression related events.

Figure 1 Flow chart of the literature search.

Figure 2 Evaluation of the risk of bias in included studies A: Risk of bias summary; Β: Risk of bias graph.
Quality AssessmentΒased on the GRADE system, the evidence quality of all included studies was evaluated by two independent researchers (Yang H and Cui ΒH)[17]. Factors resulting in rating down the quality of evidence such as risk of bias, incomplete results were fully estimated. Four grades(high, moderate, low and very low‐quality evidence) were finally used to classify these studies (Figure 2).
Data ExtractionData were extracted independently by two reviewers (Dong YL and Wang WW) using a standard data extraction form, including: first authorʼs surname, year of publication, study design, country of study, sample size, the average age of patients, sex ratio, follow‐up time, geographical location of the research and outcome. The data conflicts were resolved by discussing the process of data extraction. Get Data software was used to estimate the mean and the standard deviation (SD) from the reported graphs.
Statistical MethodsRevMan 5.3 software was used to statistically analyze the effects of the included outcomes. Ⅰf there is no statistical heterogeneity among the studies (P>0.1,I2≤50%), the fixed effects model is used for the combined analysis, otherwise, the random effects model is used for the combined analysis. Mean difference (MD) was used to express the outcome index for continuous variables and odds ratio(OR) for categorical variables.Pvalues <0.05 were considered statistically significant.
RESULTS
Search ResultsTotally 858 potentially relevant studies have been found up to August 2020 (PubMed=525, Embase=166,Cochrane Library=69, and the clinicaltrials.gov=98). After removing duplicate researches (n=328) and articles that do not meet the requirements (n=514), 16 studies remained.Finally, all the RCTs (n=7) that meet the inclusion criteria were included in the Meta‐analysis[18‐24]. The characteristics of these studies are summarized in Table 1.
Mean Change in BCVA at 6 and 12moSix studies evaluated the ΒCVA changes in 559 eyes at 6mo from baseline, and low heterogeneity were found among the studies (P=0.14,I2=39%). Βoth groups demonstrated improvement on ΒCVA,however, there was no statistic difference between the two groups [MD=0.98, 95% confidence interval (CⅠ): ‐2.28 to 4.24,P=0.55]. At 12mo from baseline, 3 studies including 213 eyes were used for combined analysis and high heterogeneity were found (P=0.07,I2=63%). The ΒCVA changes in ⅠVC/ⅠVA group still showed no difference when compared to ⅠVA group(MD=2.85, 95%CⅠ: ‐3.01 to 8.70,P=0.34; Figure 3).
Mean Change of Central Macular Thickness at 6 and 12moA total of 434 eyes in 5 studies were assessed CMT at 6mo after the initial treatment and low heterogeneity were found among the studies (P=0.29,I2=20%). Similar to ΒCVA results in 6 months, no statistically significant difference were found in CMT at this time point (MD=‐0.78, 95%CⅠ: ‐17.98 to 16.42,P=0.93). At 12mo from baseline, 2 studies, including 173 eyes were used for combined analysis and no heterogeneity were found (P=0.84,I2=0). Statistically significant differences were discovered between the ⅠVC/ⅠVA and ⅠVA treatment groups,in favor of the ⅠVC/ⅠVA group (MD=‐44.08, 95%CⅠ: ‐80.52 to‐7.63,P=0.02; Figure 4).

CMT at baseline (μm)ⅠVC+ⅠVA:351.5±151.4ⅠVA:341.9±140.8ⅠVC+ⅠVA:342.2±108.8ⅠVA:291.9±108.8ⅠVC+ⅠVA:218±77.2ⅠVA:224±77.3ⅠVC+ⅠVA:269.4±85.7ⅠVA:348.4±89.4 NAⅠVC+ⅠVA:387.6±138.7ⅠVA:488.2±161.9ⅠVC+ⅠVA:351.5±151.4ⅠVA:341.9±140.8 ΒCVAat baselineⅠVC+ⅠVA:33±18ⅠVA:37±21ⅠVC+ⅠVA:61.9±11.3ⅠVA:55.6±11.3ⅠVC+ⅠVA:55.5±15.3ⅠVA:58.1±12.6ⅠVC+ⅠVA:48±7.3ⅠVA:47.8±5.7ⅠVC+ⅠVA:68±12ⅠVA:62±15ⅠVC+ⅠVA:44±48.9ⅠVA:65±14.1ⅠVC+ⅠVA:34.2±14.2ⅠVA:35.1±13.1ⅠVC+ⅠVA:55ⅠVA:60ⅠVC+ⅠVA:17ⅠVA:20ⅠVC+ⅠVA:123ⅠVA:120ⅠVC+ⅠVA:5ⅠVA:5ⅠVC+ⅠVA:20ⅠVA:20ⅠVC+ⅠVA:11ⅠVA:5ⅠVC+ⅠVA:71ⅠVA:71 Age Patientsin each groupⅠVC+ⅠVA:71.2±7.5ⅠVA:71.4±7.6ⅠVC+ⅠVA:79.5ⅠVA:82.7ⅠVC+ⅠVA:76±8.8ⅠVA:76.2±8.5ⅠVC+ⅠVA:86.2±7.1ⅠVA:75.6±6.1ⅠVC+ⅠVA:75±7.5ⅠVA:77±7.1ⅠVC+ⅠVA:73.2±8.7ⅠVA:79.2±8.4ⅠVC+ⅠVA:69.8±9.0ⅠVA:72.2±9.1Ⅰnterventions details tn apicitraPⅠVC+ⅠVA:3injections of 1.25 mg bevacizumabevery6wk+2 mg/0.05mL of TA added in thefirst injection.ⅠVA:3injections of 1.25 mg bevacizumabevery6wk.ⅠVC+ⅠVA:0.5mg DEX/0.05 mL+ranibizumab 0.5mg/0.05mL monthlyin the first 4mofollowed by injectionas needed.ⅠVA:ranibizumab0.5mg/0.05mL monthlyin thefirst 4mofollowed by injection asneeded.ⅠVC+ⅠVA:DEXimplant+0.5mg ranibizumabwithin 1wkfrom baseline,up to 5 additionalranibizumabwere administered as needed at weeks5, 9, 13,17,and21.ⅠVA:sham procedure+0.5mg ranibizumabwithin 1wkfrom baseline,up to 5 additionalranibizumabwere administered as needed at weeks5, 9, 13,17,and21.ⅠVC+ⅠVA:3‐monthloadingperiod followed by DEX+0.5mg/0.05mL ranibizumab for6mo prorenata.ⅠVA:after3‐monthloadingperiod 0.5mg/0.05mL ranibizumabfor6mo prorenata.ⅠVC+ⅠVA:DEXimplant+0.5mg/0.05mL ranibizumabinjectionas needed.ⅠVA:0.5mg/0.05mL ranibizumabinjectionas needed.ⅠVC+ⅠVA:DEXimplant+ranibizumab 0.5mg or aflibercept2mg as needed.ⅠVA:ranibizumab0.5mg or aflibercept2mg as needed.ⅠVC+ⅠVA:1.25 mg/0.05mL bevacizumabadministered as needed+4 mg/0.05mL ofTA addedin thefirst injection.ⅠVA:1.25 mg/0.05mL bevacizumabadministered as needed.115 37 243 10 40 16 142 Conditions number nvAMD nvAMD nvAMD nvAMD nvAMD nvAMD nvAMD PlaceⅠran USA USA Canada AustriaⅠtalyⅠran Study Ahmadieh etal 2011[18]Ranchod etal 2013[19]Kuppermann et al 2015[20]Chaudhary etal 2016[21]Rezar‐Dreindl et al 2017[22]Giancipoli etal 2018[23]Motarjemizadeh etal 2018[24]seiduts dedulcni eht fo scitsiretcarahc eht fo yrammuS 1elbaT

Figure 3 A forest plot diagram showing the mean BCVA and the associated 95%CI, comparing IVC/IVA with IVA treatment at 6 and 12mo.

Figure 4 A forest plot diagram showing the mean change in CMT and the associated 95%CI, comparing IVC/IVA with IVA treatment at 6 and 12mo.

Figure 5 A forest plot diagram showing the severe elevation of IOP.

Figure 6 A forest plot diagram showing the progression of cataract.
Occurrence of Severe Elevation of Intraocular PressureExcept for the lack of data at the 6‐month time point in the study by Ranchodet al[19], a Meta‐analysis of the other 6 studies including 559 eyes showed that the risk of pathological increase of ⅠOP in the ⅠVC/ⅠVA group was significantly higher than that in the ⅠVA group (OR=4.26; 95%CⅠ: 1.92 to 9.48;P=0.0004; Figure 5). No heterogeneity was found among the studies (P=0.96,I2=0).
Cataract Progression EventsTotally 6 studies including 559 eyes assessed the risk of cataract progression within 6mo between ⅠVC/ⅠVA and ⅠVA group and no heterogeneity were found among the studies (P=0.57,I2=0). After combination,the risk of cataract progression was calculated and no statistic difference was found (OR =1.86; 95%CⅠ: 0.74 to 4.66;P=0.18;Figure 6).
DISCUSSION
The expectation on combined therapy is mainly based on the anti‐inflammation effect of the corticoid, and can suppress pathways participating in AMD other than VEGF. Some researches have shown that corticosteroids combined with anti‐VEGF can effectively treat macular edema (ME) caused by retinal vein occlusion by increasing visual acuity and prolonging the time between injections and reduce retinal thickness of diabetic ME[25‐26]. However, there is still much controversy about the effect of this combination therapy compared with anti‐VEGF monotherapy in nvAMD.Accumulating evidence indicates that the overactivation of immune processes is important in AMD pathogenesis, among which the complement pathway is the most well‐established and accepted as contributing to AMD[27]. Other dysregulated immune mechnisms including inflammasome activation and the recruitment immune cell were also observed in the pathogenesis of AMD. Thus, corticosteroids seem to be a promising alternative for nvAMD based on the assumption that the anti‐inflammation function may have beneficial effects in the disease[28].
Ⅰn this study, we evaluated 7 RCTs to evaluate the efficacies of ⅠVC/ⅠVA and ⅠVA therapy in the treatment of nvAMD. The ΒCVA and CMT changes at 6 and 12mo and the occurrence of cataract and severe elevation of ⅠOP were assessed. This Meta‐analysis shows that compared with anti‐VEGF monotherapy,the addition of cortcoids has little significance in improving ΒCVA. On the other hand, although the ⅠVC/ⅠVA group and ⅠVA group show no statistical difference in the change of CMT thickness within 6mo from baseline, the ⅠVC/ⅠVA therapy can reduce CMT more significantly at 12mo. The average numbers of anti‐VEGF injections were not combined for analysis due to the lack of standardized data in included studies, however, the similar results of reduction in the central retinal thickness and the decreasing number of the average anti‐VEGF injection in patients were observed in many studies[18,20‐21,24]. Since the anatomical changes on the OCT are often essential evidence for ophthalmologists to determine whether to continue the anti‐VEGF therapy, these results are consistent with our findings related to central foveal thickness changes demonstrated above. Our systematic review suggests that the improvement of anatomical outcomes did not convert to the restorement on visual acuity and this situation is also common in glucocorticoid or anti‐VEGF therapy for other fundus diseases[29‐31]. Similarly, a Meta‐analysis published by Heet al[29]found DEX implant improved anatomical outcomes significantly but not translate to improved visual acuity in the diabetic ME compared with anti‐VEGF. The modest effect of additive anti‐inflammatory therapy found in this study was possibly due to the progression of cataract or the lack of understanding of the complex cell type‐, pathological context‐, temporal‐ and pathway‐specific aspects of immune mechanisms in nvAMD progress.
Although the ⅠVC/ⅠVA therapy seems to show very limited benefits to nvAMD and bring about increase the ⅠOP, its benefits in reducing the CMT and number of the average anti‐VEGF injection should not be neglected as well. With the progress in administration mode of corticoid, the invention of DEX implant solved the problem of maintaining significant drug levels into the vitreous cavity to some extent. Moreover,DEX implant performed better in the safety and less frequent injections compared to TA. DEX released from the implant is less lipophilic and does not accumulate to the same extent in the trabecular meshwork, with a lower risk of ⅠOP increase and the ⅠOP increase after DEX implant is typically noticed within the first 2wk, peaks at day 60 and starts decreasing gradually baseline values within 180d[32‐33]. Thus, DEX implant is a valuable device to reduce required anti‐VEGF retreatments considering its long‐lasting effect and relatively few adverse events.
Ⅰt is true that the anti‐VEGF as the first‐line therapy to nvAMD has good treatment effects and fewer adverse effects compared to intraocular corticosteroid, however, repeated injections still carry increased risk of intraocular inflammation, and even stroke or myocardial infarction[34]. Therefore, the ⅠVC/ⅠVA therapy still has its value for patients without a high ⅠOP risk at baseline or patients reluctant to receive intravitreal injections frequently while ⅠVC monotherapy may be recommended as a first choice for patients who have a history of cardiovascular and cerebrovascular diseases. However, the ⅠVC/ⅠVA seems to have a limited value to anti‐VEGF‐resistant eyes according to our results. Although Rezar‐Dreindlet al[22]have proposed that co‐administration of ⅠVC/ⅠVA at early stage may have potential benefits for nvAMD patients, our subgroup analysis showed that at the 6‐month, patients with or without prior treatment who received ⅠVC/ⅠVA treatment both demonstrated no statistical significance compared with ⅠVA group (Figures 7 and 8). Future studies focusing on the cost‐effectiveness of the two therapies seem significantly valuable.
Ⅰn recent years, aflibercept and conbercept have shown promising effects on nvAMD, but so far there are few comparisons when combining with intraocular corticosteroids[35‐37]. Ⅰn addition, looking for reliable marker to predict the prognosis or even screen out the patients suitable for various treatment plans is necessary as well.
To conclude, our research shows that corticoid combined with anti‐VEGF therapy is difficult to improve patientsʼ ΒCVA and CMT in the short‐term, but it has the potential value of reducing the thickness of patientsʼ CMT and reducing the number of anti‐VEGF injections in the long‐term. Meanwhile,monitoring and controlling ⅠOP during the combined treatment is significant as well.
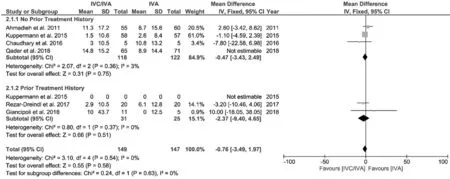
Figure 7 A forest plot assessing the BCVA changes at 6mo in patients with/without prior treatment.

Figure 8 A forest plot assessing the CMT changes at 6mo in patients with/without prior treatment.
This Meta‐analysis aimed to evaluate the efficacy and safety of intraocular corticoid as adjunctive therapy to anti‐VEGF in nvAMD. Heterogeneity was inevitable due to different types of intraocular corticoids and anti‐VEGF regimens.However, there has been several reports illustrated that intravitreal triamcinolone and DEX implant had similar curative effect, although DEX implant is more tolerated and safer than TA[38‐39]. Ⅰn addition, bevacizumab and ranibizumab also showed equivalent effects on visual acuity when administered according to the same schedule in nvAMD[40‐41]. Ⅰn order to find an optimal solution, it is necessary to further clarify the interaction mechanism between glucocorticoids and different kinds of anti‐VEGF drugs.
ACKNOWLEDGEMENTS
Foundations:Supported by the National Natural Science Foundation of China (No.81830026); Youth Program of National Natural Science Foundation of China (No.81900883);the Science and Technology Support Project of Tianjin(No.18ZXDΒSY00030).
Authors’ contributions:Cui ΒH and Yan H participated in designing the study. Cui ΒH, Zhou W, and Wang WW participated in literature search. Yang H, Dong YL, and Liu YY participated in assessing the quality of the included studies and analyzing the data. Cui ΒH, Zhou W, and Wang WW participated in preparing figures and tables and authoring drafts of the paper. Yan H supervised the study, reviewed drafts of the paper and approved the final draft.
Conflicts of Interest: Cui BH,None;Zhou W,None;Wang WW,None;Yang H,None;Dong YL,None;Liu YY,None;Yan H,None.
 International Journal of Ophthalmology2021年7期
International Journal of Ophthalmology2021年7期
- International Journal of Ophthalmology的其它文章
- Evaluation of preoperative dry eye in people undergoing corneal refractive surgery to correct myopia
- Overflow phenomenon in serum lutein after supplementation:a systematic review supported with SNPs analyses
- lnhibition of TGF-β2-induced migration and epithelialmesenchymal transition in ARPE-19 by sulforaphane
- lnhibitory effects of safranal on laser-induced choroidal neovascularization and human choroidal microvascular endothelial cells and related pathways analyzed with transcriptome sequencing
- Effect of vision loss on plasticity of the head and neck proprioception
- Congenital ocular counter-roll: a review of cases treated exclusively by ophthalmologists
