Impact of 5-Fu/oxaliplatin on mouse dendritic cells and synergetic effect with a colon cancer vaccine
Xinqiang Hong, Tiangeng Dong, Tuo Yi, Jianwei Hu, Zhen Zhang, Shengli Lin, Weixin Niu
Department of General Surgery, Zhongshan Hospital, Fudan University, Shanghai 200032, China
Introduction
Colon cancer is one of the most common malignant tumors and its incidence in China is increasing rapidly (1). Tumor immunotherapy has attracted considerable attention due to its specific killing effect, although its efficacy in colorectal carcinoma has not been satisfactory (2). Currently, immune therapy alone is insufficient to obtain clinical benefit, and proven effective chemotherapy, radiotherapy and other traditional treatment are considered indispensable.
Conventional chemotherapeutic drugs usually exert their antitumor effects by disrupting DNA and/or interfering with DNA synthesis. Patients who are treated with chemotherapy usually suffer from leucopenia and suppressed lymphocyte function. Therefore, chemotherapy has often been considered to possess an immunosuppressive effect. However, subsequent studies demonstrated that chemotherapeutic drugs occasionally exhibit inconsistent antitumor effectsin vitroandin vivo(3), suggesting that the immune system may participate in the process of antitumor effect of chemotherapy. Therefore, considerable interest was shown in the development of chemoimmunotherapy(4). Recent studies indicated that the effects of chemotherapeutic drugs on the immune system might include the following: regulation of the immunogenicity of tumor cells(5), reduction of the number of regulatory T cells (Tregs)and suppression of their function (6), reduction and/or transformation of myeloid derived suppressor cells (7),induction of the maturation of dendritic cells (DCs) (8) and differential homeostatic proliferation of T-cell subsets (9).
Our previous study showed that the combined application of Toll-like receptor 3 (TLR 3) and TLR7/8 ligands could stimulatein vitrocultured DCs in order to achieve better maturation and effectively activate the cellular immune responses on tumor cells. In addition, a mouse model of colon cancer expressing carcinoembryonic antigen (CEA) was constructed. The combined TLRs activated DC tumor vaccine exhibited optimum preventive effect on the subcutaneous implanted tumors, although it was ineffective for the treatment of tumor bearing mice(10). The explanation for this outcome may be attributed to the lack of immunogenicity of the CEA epitope of our DC tumor vaccine. Moreover, the tumor may evade the immune response by reducing tumor antigen and other mechanisms.
Therefore, we developed a tumor cell/α-galactosylceramide (α-GC) complex tumor vaccine, using the innate immune killing tumor cells to provide tumor associated antigens for DCs. Thein vivopathway of TLR9 ligand was also utilized in order to stimulate DC maturation, and to induce effective anti-tumor immunity via iNKT and DC interaction. The results indicated that the TLR ligand could promote the optimum maturation of DCs, which in turn effectively activated iNKT cells. The activated iNKT cells could concomitantly induce further maturation of DCs in order to exert a potent T-helper 1 (Th1) antitumor immune response. The tumor vaccine has also been shown to produce immune memory effect against colon cancer (11).
The majority of the reports of tumor immunotherapy have demonstrated that the immunotherapy of colon cancer alone cannot be applied effectively in the clinic, and it is contradictory to the comprehensive treatment principle of tumors (12). However, based on the large number of evidence-based medicine, chemotherapy was critically important in the treatment of colon cancer. Its combination with immunotherapy was undoubtedly a very meaningful attempt (13). The major advantage of immunotherapy of our previous study has been shown to be the loading of the antigenin vivothat aims to the activation of the DCs. Therefore, the effects of conventional chemotherapeutic drugs on the immune system, notably DCs, are critical to the success of the combined therapeutic treatment.
Currently, 5-fluorouracil (5-Fu) and oxaliplatin are widely used as a first-line regimen in adjuvant chemotherapy for colon cancer. Clinical studies have used the DC vaccine in patients with colon cancer who received oxaliplatin and capecitabine chemotherapy. It was shown that chemotherapeutic drugs could enhance the proliferation of non-specific T cells without weakening the tumor antigen specific T cell responses (14). However, the effects of their combined use on DCs have not yet been reported. The present study used a mouse colon cancer model in order to investigate the effects of the chemotherapeutic drugs oxaliplatin and 5-Fu on DCs.Oxaliplatin and 5-Fu were combined with our previously developed tumor cell/α-GC complex + TLR ligand in order to derive a treatment that could provide the optimum synergy of the immunotherapy and chemotherapy, and establish the foundation for the application of chemoimmunotherapy in colon cancer.
Materials and methods
Animals and cell lines
Male and female SPF C57BL/6 mice aged 6 to 8 weeks were purchased from Shanghai SLAC experimental animal Co., Ltd. All procedures during the experiments were in accordance with the requirements of the Ethics Committee for laboratory animals in Fudan University.
The MC38 colon cancer cell line was kindly provided by Dr. Steven A. Rosenberg (the National Cancer Institute,Bethesda, MD, USA). MC38 cells were cultured in high glucose DMEM medium (Hyclone, GE Healthcare,Boston, MA, USA) with 10% fetal bovine serum (FBS)(Gibco, Thermo Fisher Scientific, Waltham, MA, USA), 1×HEPES (Gibco), 100 units/mL penicillin and of 100 μg/mL streptomycin (Invitrogen, Thermo Fisher Scientific, MA, USA).
In vitro culture and maturation of mouse DCs
The method used for the culture of mouse DCs was reported by previous study (15). Briefly, the bone marrow cells were obtained from the long bones of the mice. After erythrocyte lysis, bone marrow cells were cultured in a 24-well plate containing RPMI1640 medium (Hyclone) with 10% FBS, 100 units/mL of penicillin, 100 μg/mL of streptomycin, 20 ng/mL of rmGM-CSF (Peprotech, Rocky Hill, USA), and 10 ng/mL of recombinant murine interleukin-4 (rmIL-4) (Peprotech). The cells were cultured at 37 °C with 5% CO2and saturated humidity.The medium was changed every 2 d and 75% of the medium was discarded in order to remove the suspended cells. On d 6, 20 μg/mL of TLR3 ligand Poly (I:C) and 3 μg/mL of TLR7/8 ligand CL097 (Invitrogen) (10) in combination with 500 μg/mL of 5-Fu and/or 10 μg/mL of oxaliplatin (OX) (Sigma-Aldrich, St Louis, MO, USA) were added into the culture. The semi-adherent DCs were harvested on d 8.
Detection of the DC phenotypes and the expression of the secreted cytokines
The DCs in each group were suspended with flow cytometry buffer [phosphate buffered saline (PBS)containing 1% bull serum albumin (BSA)] following harvesting. The antibodies for flow cytometry or the corresponding isotype controls were added and incubated at 4 °C for 30 min in dark. The suspension was detected by flow cytometry (BD FACScan flow cytometry system, San Jose, CA, USA). The phenotypes of DCs for the detection included CD11c, CD80, CD86, CD40, major histocompatibility complex I (MHC I), MHC II,programmed death ligand 1 (PD-L1) and PD-L2. All of them were purchased from the eBioscience company. The results were analyzed by the FlowJo 7.6.2 software (FlowJo,LLC, USA).
A small amount of DC culture supernatant was retained and stored at -20℃ following harvesting of DCs. The cytometric bead array (CBA) Mouse Inflammation Kit was purchased from BD Biosciences. The detection of cytokines including IL-12p70, tumor necrosis factor(TNF), IL-10, IL-6, interferon-γ (IFN-γ) and monocyte chemotactic protein 1 (MCP-1) was conducted according to the instructions.
Injection of MC38 cells in mice and administration of chemotherapeutic drugs
The MC38 cells were suspended into 1×107/mL cell suspensions. The cells were subcutaneously injected in the right flank of C57BL/6 mice that were 6 to 8 weeks old.The number of injected cells was 1×106for each mouse.The tumor xenograft was observed every 2 or 3 d. The maximum and minimum diameters of the tumor were measured and the size of tumor was calculated according to the following formula: tumor volume = length of the maximum diameter × length of the minimum diameter2×0.52.
When the volume of the tumor reached 500 mm3, the chemotherapeutic drugs were intraperitoneally injected into the mice. The doses of oxaliplatin and 5-Fu were 5 mg/kg and 50 mg/kg, respectively.
Preparation of single cell suspension of spleen cells, lymph node cells and tumor tissue cells from tumor bearing mice
Following chemotherapy treatment for 5 d, the mice were sacrificed, and the spleen, tumor tissues and lymph nodes(inguinal and axillary lymph nodes) were collected. The spleen was placed on a cell filter (BD Falcon, Franklin Lakes, NJ, USA) with a pore diameter of 70 μm. A syringe needle was used to collect the cells from the spleen tissue.Following erythrocyte lysis, the cells were suspended, and the splenocyte suspension was obtained.
Following stripping of the tumor tissues, the surrounding fat and connective tissues were removed, and the tumor tissues were cut into small pieces with an approximate volume of 1 mm3. The tumor tissues were mixed with Hanks solution containing 1 mg/mL type II collagenase and digested at room temperature for 2 h.Following digestion, the cells were filtered using a filter with a pore diameter of 100 μm. The cell filtrate was washed with PBS twice for subsequent experiments.
The lymph nodes of mice were cut into small pieces with an approximate volume of 1 mm3and mixed with Hanks solution containing 1 mg/mL type II collagenase. The tissues were digested at room temperature for 20 min and the cells were filtered using a filter with a pore diameter of 70 μm. The cell filtrate was washed with PBS twice for subsequent experiments.
Magnetic sorting of DCs and flow cytometry detection
The single cell suspensions of the spleen, lymph nodes and tumor tissues were sorted by CD11c magnetic beads(Miltenyi Biotec, Auburn, CA, USA) according to the instructions provided by the manufacturer. The sorted CD11c+cells were considered as DCs and then were detected by flow cytometry for the markers CD40,CD80/86, MHC I/II and PD-L1/L2.
Specific activation of lymphocytes by 5-Fu and oxaliplatin in vivo
The tumor bearing mice were sacrificed 5 d following chemotherapeutic treatment. The tumor tissues were cut into small pieces with an approximate volume of 1 mm3.The cells were separated and filtered into single cell suspension using a cell filter and subsequently washed with PBS once. A total of 5 mL of single cell suspension was added on the surface of 5 mL leukocyte separation buffer(Sigma) and the samples were centrifuged at 1,500 r/min for 20 min. The cells of the second layer were collected as tumor infiltrating leukocytes, and washed twice with PBS for subsequent experiments.
The tumor infiltrating leukocytes, splenocytes and lymph node cells were co-cultured with dead MC38 tumor cells (180 Gy irradiation for 30 min) according to a cell ratio of 2:1 in 96-well round-bottom plates pre-coated with anti-mouse CD3ε antibody (eBioscience Company,Thermo Fisher Scientific, MA, USA). The cells were cultured in RPMI 1640 medium containing 10% FBS and 10 ng/mL IL-2 (eBioscience) for 16 h. A small amount of supernatant was collected, and the secretion content of IFN-γ was detected by the mouse IFN-γ ELISA Kit (R"D company, Lorton, VA, USA).
Therapeutic effects of chemotherapy combined with the CD1d-MC38/α-GC tumor vaccine on MC38 colon cancer
The CD1d-MC38/α-GC tumor vaccine was prepared by the previously reported methodology of our research group(11). A total of 1×106MC38 cells were subcutaneously injected into the right flank of C57BL/6 mice that were 6 to 8 weeks old. The injected tumors were observed every 2 or 3 d. The maximum and minimum diameters of the tumors were measured, and the sizes of tumors were calculated. There were 6 mice in each group.
When the volumes of the tumors reached 500 mm3, the chemotherapeutic drugs were intraperitoneally injected into the mice. The immunotherapies were carried out twice on d 3 and d 7 following injection of chemotherapeutic drugs. The methodology included intravenous injections of 50 μg of TLR9 ligand CpG-ODN1826 (synthesized by Shanghai Sangon) and CD1d-MC38/α-GC tumor vaccine(1×106α-GC-loaded CD1d-MC38 cells) (Table 1). The mouse tumor growth and survival curves were drawn. The mice were sacrificed when the volumes of the subcutaneous tumors were over 5,000 mm3.
Statistical analysis
The data are presented asand thet-test and analysis of variance were used to compare the differences between two or more groups, respectively. Survival analysis was carried out using the Kaplan-Meier method and the Log-rank test was used to compare the survival curves between two or more groups, respectively. P<0.05 was considered statistically significant.
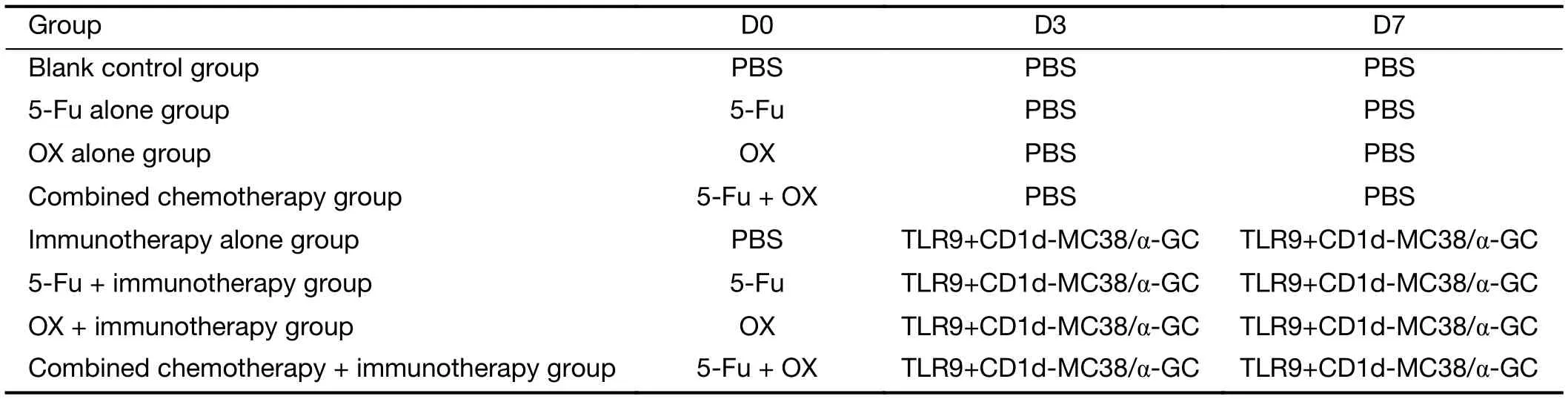
Table 1 Grouping and time points of chemotherapy and immunotherapy
Results
5-Fu/oxaliplatin reduced the expression of PD-L1/L2 in DCs
The murine myeloid-derived DCs were culturedin vitroand 500 μg/mL of 5-Fu and/or 10 μg/mL of oxaliplatin were added at the last 48 h. Flow cytometry detection indicated that the expression levels of the markers CD11c,CD40, CD80/86 and MHC I/II were significantly upregulated following addition of TLR3 and TLR7/8 ligands (P<0.01), which indicated that the combined activation of TLRs could promote DC maturation.However, there was no significant effect on the expression of the aforementioned DC markers following the addition of 5-Fu/oxaliplatin, demonstrating that 5-Fu/oxaliplatin did not affect the maturation effect of the TLR ligands on the DC phenotypes (Figure 1).
In addition, the combined administration of the TLR ligands and 5-Fu did not reduce the expression of the inhibitory receptor PD-L1. However, its expression was significantly decreased following oxaliplatin administration(Figure 2A, P<0.05). Similar findings were shown for the PD-L2 expression levels (Figure 2B) that were not affected by the combined application of the TLR ligands, whereas the addition of 5-Fu significantly decreased the expression of this ligand (P<0.05). Moreover, oxaliplatin caused a highly significant reduction in PD-L2 expression levels(P<0.01).
Effect of 5-Fu/oxaliplatin on DC cytokine secretion
The supernatant was retained during DC harvesting, and the CBA Mouse Inflammation Kit was used to detect the secretion of cytokines. The results are shown inFigure 3.5-Fu exhibited no effect on IL-12 secretion, while oxaliplatin could significantly increase IL-12 secretion. For Th1 cytokines, both 5-Fu and oxaliplatin increased the secretion of TNF but not IFN-γ. As for Th2 cytokines,oxaliplatin decreased the secretion levels of IL-6, while 5-Fu increased the secretion of IL-10 and oxaliplatin increased it at higher levels compared with 5-Fu. Both 5-Fu and oxaliplatin reduced significantly the secretion of the chemokine MCP-1.

Figure 1 Effect of 5-Fu/oxaliplatin (OX) on dendritic cell (DC) phenotypes. The murine myeloid-derived DCs were cultured in vitro, and Toll-like receptor 3 (TLR3), TLR7/8, 500 μg/mL 5-Fu and/or 10 μg/mL oxaliplatin were added at the last 48 h. Flow cytometry was used to detect the phenotypic expression of DCs in each group. imDCs: immature DCs, no TLR ligand and/or chemotherapeutic drugs were added; mDC: mature DCs, combined TLR ligands were added. Data are obtained from three independent experiments and represented as. *, P<0.05; **, P<0.01; ns, no significance.

Figure 2 Effect of 5-Fu/oxaliplatin on the expression of dendritic cell (DC) programmed death ligand 1/2 (PD-L1/L2). The murine myeloid-derived DCs were cultured in vitro, and the expression of the inhibitory receptor PD-L1/L2 on the DC surface was detected. (A)The expression of PD-L1 in each group; (B) The expression of PD-L2 in each group. The flow chart indicates a typical experimental sample derived from three independent experiments. Data are obtained from three independent experiments and represented as . *,P<0.05; **, P<0.01; ns, no significance.
5-Fu/oxaliplatin induced DC phenotypic maturation in tumor bearing mice
Following 5 d of intraperitoneal injection of 5-Fu/oxaliplatin in tumor bearing mice, the spleens, tumor tissues and draining lymph nodes of the mice were obtained and then were made a single cell suspension. Subsequently,DCs were selected by CD11c magnetic beads, and the phenotype expression was detected. The results indicated that 5-Fu/oxaliplatin increased the expression of CD40,CD80/86 and MHC I molecules on splenic DCs.Compared with 5-Fu, oxaliplatin enhanced the expression of the aforementioned phenotypes more effectively. In addition, 5-Fu/oxaliplatin decreased the expression of MHC class II molecules. 5-Fu exhibited no significant effect on the expression of PD-L1/L2, whereas PD-L1 expression was slightly decreased and PD-L2 expression was significantly reduced following treatment of DCs with oxaliplatin. The effects of the combined treatment of 5-Fu/oxaliplatin on the expression of the DC phenotypes in tumor tissues and lymph nodes were roughly similar, as shown inFigure 4.

Figure 3 Effects of 5-Fu/oxaliplatin on the secretion of dendritic cell (DC) cytokines. A small amount of supernatant was obtained during DC harvesting, and the cytokines were detected by cytometric bead array (CBA). The data are obtained from three independent experiments and represented as . *, P<0.05; **, P<0.01; ***, P<0.001; ns, no significance.
5-Fu/oxaliplatin specifically activates lymphocytes in tumor bearing mice
The spleens, tumor tissues and draining lymph nodes were removed from the tumor bearing mice 5 d following the injection of 5-Fu and/or oxaliplatin. The tumor infiltrating leukocytes, splenocytes and lymph node cells were cocultured with the nonviable MC38 cells, respectively, and IFN-γ secretion was detected. With regard to the tumor infiltrating leukocytes, the IFN-γ secretion levels were increased significantly in the 5-Fu group compared with the PBS group, while oxaliplatin further increased IFN-γ secretion (Figure 5A). The results obtained from the splenocytes and the lymph node cells were similar to those of the tumor infiltrating leukocytes, that is, 5-Fu/oxaliplatin significantly increased the IFN-γ secretion levels (Figure 5B, C).
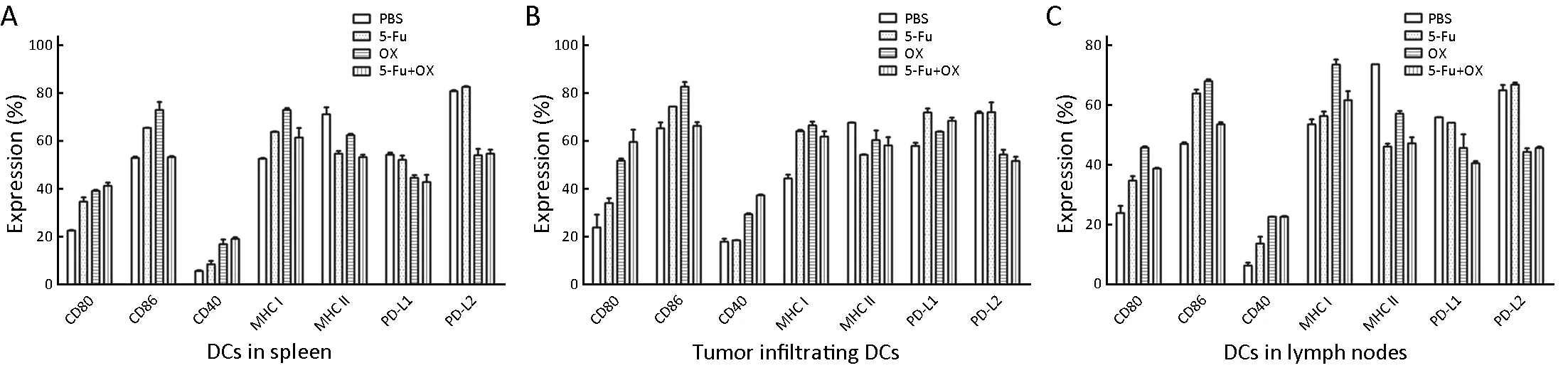
Figure 4 Effects of 5-Fu/oxaliplatin on the phenotypes of dendritic cells (DCs) in tumor bearing mice. MC38 colon cancer cells were subcutaneously inoculated in mice, and when the tumor volume reached 500 mm3, 50 mg/kg 5-Fu and/or 5 mg/kg of oxaliplatin were intraperitoneally injected. Following 5 d of treatment, the DCs of the spleens, tumor tissues and draining lymph nodes were separated, and the phenotype expression was detected by flow cytometry. (A) The phenotypic expression of DCs in the spleens; (B) The phenotypic expression of DCs in the tumor tissues; (C) The phenotypic expression of DCs in the lymph nodes. The data are obtained from two independent experiments and represented the .
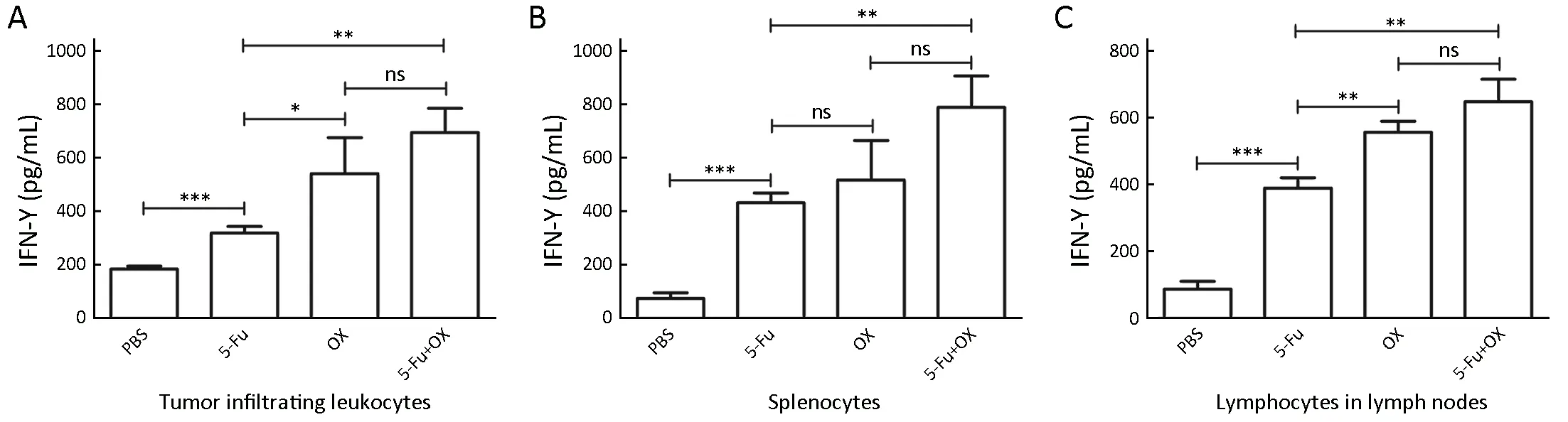
Figure 5 The specific activation of lymphocytes by 5-Fu/oxaliplatin in vivo. When the volume of the subcutaneous tumor reached 500 mm3,50 mg/kg 5-Fu and/or 5 mg/kg oxaliplatin were injected in mice. The spleens, tumor tissues and draining lymph nodes were removed 5 d following the injection. The tumor infiltrating leukocytes, splenocytes and lymph node cells were co-cultured with nonviable MC38 cells for 16 h. Enzyme-linked immunosorbent assay (ELISA) was used to detect interferon-γ (IFN-γ) secretion. (A) Secretion level of IFN-γ from tumor infiltrating leukocytes; (B) Secretion level of IFN-γ from splenocytes; (C) Secretion level of IFN-γ from lymph node cells. The data are obtained from three independent experiments and presented as . *, P<0.05; **, P<0.01; ***, P<0.001; ns, no significance.
Combined administration of 5-Fu/oxaliplatin with CD1d-MC38/α-GC tumor vaccine exerts a synergistic role in the treatment of colon cancer
5-Fu or oxaliplatin monotherapy reduced thein vivotumor growth rate. The tumor growth rate was significantly lower in the oxaliplatin group compared with the PBS group,while there was no significant difference between the 5-Fu and the PBS groups, which might be related to the short observation time of the tumor growth rate. The combination treatment of the 5-Fu and oxaliplatin group could reduce the tumor growth rate in mice at a significantly higher level. The data suggested that the combined administration exhibited higher treatment efficacy compared with either 5-Fu or oxaliplatin alone(Figure 6A).
The tumor growth rate was higher when immunotherapy alone was used on tumor bearing mice compared to the following combination treatments: 5-Fu + oxaliplatin group, immunotherapy + 5-Fu group and immunotherapy+ oxaliplatin group. These combination treatments could significantly reduce the tumor growth rate (P<0.01).However, the differences among the three combination groups were not significant. The combination of immunotherapy with 5-Fu and oxaliplatin exhibited significantly higher reduction in thein vivotumor growth rate compared to the aforementioned three combination groups (Figure 6B).
The tumor bearing mice were observed for 90 d after inoculation. The mice with tumor volume exceeding 5,000 mm3were sacrificed. The survival curves of the tumor bearing mice were drawn by the Kaplan-Meier method(Figure 7). The results indicated that all the other groups could significantly prolong the survival of the mice compared with the PBS group. The 5-Fu and the oxaliplatin monotherapy groups could significantly prolong the survival time of the mice compared with the immunotherapy group, since no significant difference was detected between these two groups. The combination of 5-Fu and oxaliplatin could significantly prolong the survival time compared to the 5-Fu or oxaliplatin monotherapy group.The immunotherapy + 5-Fu and immunotherapy +oxaliplatin groups exhibited higher efficacy in prolonging the survival compared with the 5-Fu + oxaliplatin group. The combined immunotherapy and 5-Fu + oxaliplatin group could significantly prolong the survival compared with the immunotherapy + single chemotherapeutic drug group.
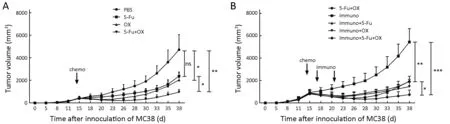
Figure 6 The growth curve of MC38 xenografts in mice following chemotherapy combined with immunotherapy. When the volume of the tumor reached 500 mm3, the chemotherapeutic drugs 5-Fu/oxaliplatin and/or phosphate buffered saline (PBS) were intraperitoneally injected in mice. The immune therapies were conducted twice on d 3 and d 7 following injection of the chemotherapeutic drugs[intravenous injection of Toll-like receptor 9 (TLR9) ligand and CD1d-MC38/α-galactosylceramide (α-GC) compound, respectively]. A total of 6 mice were used in each group. The inoculated tumors were observed every 2 or 3 d. The maximum and minimum diameters of the tumors were measured in order to calculate the tumor volume. (A) Chemotherapy for MC38 xenografts in mice; (B) Chemotherapy combined with immunotherapy for MC38 xenografts in mice. *, P<0.05; **, P<0.01; ***, P<0.001; ns, no significance.
Discussion
DCs are the most potent known antigen presenting cells that play an important role in the activation and differentiation of T cells. Immature DCs are mainly located in the peripheral tissues, and can capture antigens. These cells are stimulated and migrate to lymphoid tissues.Mature DCs could highly express MHC and costimulatory molecules, present antigen peptides to T cells and activate them in order to induce antigen-specific CD8+cytotoxic T cells and/or CD4+helper T cells, thereby mediating a secondary immune response (16,17).
Our previous studies mainly focused on DCs and demonstrated that the combined application of TLR ligands could stimulatein vitrocultured DCs in order to achieve optimal maturity and activate the cellular immune response more effectively. In addition, we developed a tumor cell/α-GC compound tumor vaccine, which could cause DCs loading with tumor antigens and subsequent activationin vivo.

Figure 7 The survival curves of mice bearing MC38 xenografts following chemotherapy combined with immunotherapy treatment. When the volume of the tumor reached 500 mm3, the chemotherapeutic drugs 5-Fu/oxaliplatin and/or phosphate buffered saline (PBS) were intraperitoneally injected in the mice.The immune therapies were conducted twice on d 3 and d 7 following injection of the chemotherapeutic drugs. The survival curves of the tumor bearing mice were drawn by the Kaplan-Meier method. The tumor bearing mice were observed for 90 d after inoculation. The mice with a tumor volume of greater than 5,000 mm3 were sacrificed.
In the present study, 5-Fu and/or oxaliplatin were incubated with DCsin vitroand their effects on the phenotypes of DCs were detected. TLR3 and TLR7/8 ligands stimulated the expression of CD11c, CD40, the costimulatory molecules CD80/86 and MHC molecules. It is in agreement with the results reported in our previous study. However, 5-Fu/oxaliplatin would not adversely affect the phenotypic maturation of DCs. PD-L1 and PDL2 are inhibitory receptors, and their binding with the PD-1 ligand on T cells can reduce the accumulation of antigenspecific T cells in the lymph nodes. In addition, they can inhibit the proliferation and cytokine secretion of CD4+T cells and the transduction of the B7-CD28 signaling pathway (18,19). Immature DCs and DCs with combined activation of TLRs expressed high levels of PD-L1/L2. 5-Fu significantly reduced PD-L2 expression, and oxaliplatin significantly reduced PD-L1 and PD-L2 expression. The effect of the simultaneous application of 5-Fu and oxaliplatin on the expression of PD-L1/L2 was similar to that of oxaliplatin alone, which might aid the function of DCs in activating T cells and inducing cellular immunity,thus contributing to the induction of the anti-tumor immune responses.
DCs secrete a series of cytokines following maturation,which are involved in the activation of T cells and the induction of the immune responses. IL-12 was notably involved in the differentiation process from naive T cells to Th1 cells, and could stimulate T cells and NK cells to produce IFN-γ and TNF in order to enhance the cytotoxicity (20,21). Oxaliplatin significantly increased IL-12 secretion, which was one of the most important characteristics of DC maturation. TNF and IFN-γ are Th1 cytokines and contribute to the Th1 differentiation of T cells. In addition, these cytokines mediate cytotoxicity and favor the antitumor immune responses (22,23). Both 5-Fu and oxaliplatin significantly increased the secretion of TNF by DCs, although the secretion of IFN-γ was not significantly affected. IL-6 and IL-10 are Th2 cytokines,whose main function is to stimulate the proliferation of B cells, promote the production of antibodies, and mediate humoral immunity (24,25). Oxaliplatin significantly reduced IL-6 secretion and was beneficial for Th1 differentiation, while both 5-Fu and oxaliplatin significantly increased IL-10 secretion by DCs. The effect of the IL-10 secretion on the anti-tumor immune response requires further investigation. It has also been reported that IL-10 exhibits an immune stimulating effect and can mediate an IFN-γ and CD8+T cell-dependent anti-tumor immune response (26).
The poor antigen capture capability of theex vivocultured DCs renders the selection of the tumor related antigens required for DC loading considerably difficult. So theex vivomatured DCs are unsuitable to be directly used as tumor vaccines. Therefore, we further detected the phenotypic changes of DCsin vivofollowing the use of chemotherapeutic agents. Since CD11c was expressed mainly on DCs, CD11c+cells were selected from splenocytes, lymph node cells and tumor tissues and were considered as DCs. It was noteworthy that tumor infiltrating DCs played an important role in the tumor microenvironment. The immune responses can be enhanced if the tumor is infiltrated by mature DCs, which recruit immune effector cells to selectively kill tumor cells.However, tumor cells inhibit the functions of DCs in multiple ways (27). Our results indicated that the effect of 5-Fu/oxaliplatin on DCs in the spleens and lymph nodes was similar to that on the tumor infiltrating DCs. The expression levels of CD40 and CD80, CD86 and MHC class I molecules were increased in varying degrees. In addition, oxaliplatin reduced PD-L1/L2 expression on DCs, suggesting that it could promote the phenotypic maturation of DCsin vivo, notably tumor infiltrating DCs.This process may be beneficial for the induction of antitumor immune responses by immunotherapy.
IFN-γ is the major cytokine secreted by Th1 cells, which mediates the cellular immune responses of CTLs and macrophages. Following the restimulation ofin vitrononviable MC38 tumor cells, higher levels of IFN-γ secretion by tumor infiltrating leukocytes, splenocytes and lymph node cells in tumor bearing mice were observed in the 5-Fu and/or oxaliplatin groups. The results indicated that lymphocytes, notably tumor infiltrating lymphocytes,could be activated to specifically identify the tumor cells following chemotherapeutic treatment. This would undoubtedly contribute to the killing effect of CD8+T cells on tumors, suggesting that 5-Fu/oxaliplatin may target tumors via the immune system of the body except the single way that affects DNA synthesis.
At present, 5-Fu combined with oxaliplatin is a common chemotherapeutic regimen used for colon cancer (28). The present study indicated that the chemotherapeutic drugs significantly reduced the growth rate of MC38 xenografts and prolonged the survival time of the mice compared with the PBS group. No significant differences in the tumor growth rate between the 5-Fu and the PBS groups (P=0.0559) were noted, possibly due to the short observation time. In the PBS group, the tumor volumes of half of the mice were over 5,000 mm3on d 38 following tumor inoculation. The mice were sacrificed on that day and in view of the statistical considerations, the tumor growth curve ended up to d 38. The 5-Fu + oxaliplatin group exhibited significant advantages with regard to the tumor growth rate and survival time compared with the 5-Fu and oxaliplatin monotherapy groups, which was also in accordance with typical clinical responses of colon cancer patients.
Our previous work produced a tumor vaccine of CD1d-MC38/α-GC complex combined with the TLR9 ligand that could effectively activate iNKT cellsin vivo, promote DCs maturation, inhibit MC38 tumor growth in mice and prolong survival. This methodological approach was also applied in the present study, although the curative effect was slightly greater compared with that of the blank control group and significantly lower than that of the 5-Fu or oxaliplatin monotherapy group. The reason was mainly related to the time points of the immune therapy. In our previous study, the immunotherapy was conducted on d 3 and d 7 following subcutaneous injection of MC38 tumor cells when the tumor load was small, which was optimum for the induction of anti-tumor immune responses. In the present study, the chemotherapy treatment time point was selected on d 3 and d 7 after the subcutaneous tumor volume reached 500 mm3, and the tumor burden increased significantly compared with our previous study. Therefore,it was difficult to obtain an optimal curative effect solely on immune therapy (29).
The combination of chemotherapy and immunotherapy was used in the present study, and the results indicated that the effect of immunotherapy combined with 5-Fu or oxaliplatin was equivalent to that of 5-Fu + oxaliplatin on the reduction of tumor growth rate. However, the combination treatment could significantly prolong the survival time. The reason may be attributed to the decreased tumor size following the administration of chemotherapeutic drugs. This reduced the tumor burden,which was beneficial for the successful function of the tumor vaccine. Further analysis showed that the 5-Fu +oxaliplatin group reduced the tumor volume faster following the administration compared with the single drug chemotherapy combined with the immunotherapy groups.Nevertheless, the tumor increased in size and exhibited a rapid growth two weeks later. Although the tumor volume reduction in the single drug chemotherapy combined with immunotherapy group was less than that noted in the 5-Fu+ oxaliplatin group in the early stage of treatment, it could effectively inhibit tumor volume rebound and prolong the survival time. The immunotherapy combined with 5-Fu +oxaliplatin group exhibited significantly decreased tumor volume following the injection of chemotherapeutic drugs,and the tumor burden was further reduced compared with the immunotherapy combined with single drug group.Therefore, it was more effective in inhibiting the growth of subcutaneous tumors and prolonging the survival of mice.
Conclusions
The present study demonstrated the effects of 5-Fu/oxaliplatin on DCs with regard to the phenotypic expression, cytokine secretion and specific lymphocyte activation. In addition, chemotherapy may exert a synergetic role in immunotherapy with the CD1d-MC38/α-GC complex + TLR9 ligand. The detailed mechanism and the concrete role of DCs in this process require further studies. Moreover, the effects of 5-Fu/oxaliplatin on the immune system are multifaceted,especially the effects on T cells, which need to be further investigated.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
1.Chen W, Zheng R, Zuo T, et al. National cancer incidence and mortality in China, 2012. Chin J Cancer Res 2016;28:1-11.
2.Koido S, Ohkusa T, Homma S, et al. Immunotherapy for colorectal cancer. World J Gastroenterol 2013;19:8531-42.
3.Schwartz HS, Grindey GB. Adriamycin and daunorubicin: a comparison of antitumor activities and tissue uptake in mice following immunosuppression.Cancer Res 1973;33:1837-44.
4.Chen G, Emens LA. Chemoimmunotherapy:reengineering tumor immunity. Cancer Immunol Immunother 2013;62:203-16.
5.Ramakrishnan R, Assudani D, Nagaraj S, et al.Chemotherapy enhances tumor cell susceptibility to CTL-mediated killing during cancer immunotherapy in mice. J Clin Invest 2010;120:1111-24.
6.Chen CA, Ho CM, Chang MC, et al. Metronomic chemotherapy enhances antitumor effects of cancer vaccine by depleting regulatory T lymphocytes and inhibiting tumor angiogenesis. Mol Ther 2010;18:1233-43.
7.Vincent J, Mignot G, Chalmin F, et al. 5-Fluorouracil selectively kills tumor-associated myeloid-derived suppressor cells resulting in enhanced T celldependent antitumor immunity. Cancer Res 2010;70:3052-61.
8.Salem ML, Díaz-Montero CM, Al-Khami AA, et al.Recovery from cyclophosphamide-induced lymphopenia results in expansion of immature dendritic cells which can mediate enhanced primeboost vaccination antitumor responsesin vivowhen stimulated with the TLR3 agonist poly(I:C). J Immunol 2009;182:2030-40.
9.Gameiro SR, Caballero JA, Higgins JP, et al.Exploitation of differential homeostatic proliferation of T-cell subsets following chemotherapy to enhance the efficacy of vaccine-mediated antitumor responses.Cancer Immunol Immunother 2011;60:1227-42.
10.Hong X, Dong T, Hu J, et al. Synergistical toll-like receptors activated dendritic cells induce antitumor effects against carcinoembryonic antigen-expressing colon cancer. Int J Colorectal Dis 2013;28:25-33.
11.Dong T, Yi T, Yang M, et al. Co-operation of αgalactosylceramide-loaded tumour cells and TLR9 agonists induce potent anti-tumour responses in a murine colon cancer model. Biochem J 2016;473:7-19.
12.Liang J, Liu X, Xie Q, et al. Endostatin enhances antitumor effect of tumor antigen-pulsed dendritic cell therapy in mouse xenograft model of lung carcinoma. Chin J Cancer Res 2016;28:452-60.
13.Medina-Echeverz J, Berraondo P. How can chemoimmunotherapy best be used for the treatment of colon cancer? Immunotherapy 2012;4:1787-90.
14.Lesterhuis WJ, de Vries IJ, Aarntzen EA, et al. A pilot study on the immunogenicity of dendritic cell vaccination during adjuvant oxaliplatin/capecitabine chemotherapy in colon cancer patients. Br J Cancer 2010;103:1415-21.
15.Inaba K, Inaba M, Romani N, et al. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J Exp Med 1992;176:1693-702.
16.Gilboa E. DC-based cancer vaccines. J Clin Invest 2007;117:1195-203.
17.Banchereau J, Palucka AK. Dendritic cells as therapeutic vaccines against cancer. Nat Rev Immunol 2005;5:296-306.
18.Kim ST, Klempner SJ, Park SH, et al. Correlating programmed death ligand 1 (PD-L1) expression,mismatch repair deficiency, and outcomes across tumor types: implications for immunotherapy.Oncotarget 2017;8:77415-23.
19.Patel SP, Kurzrock R. PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther 2015;14:847-56.
20.Del Vecchio M, Bajetta E, Canova S, et al.Interleukin-12: biological properties and clinical application. Clin Cancer Res 2007;13:4677-85.
21.Hernandez-Alcoceba R, Poutou J, Ballesteros-Briones MC, et al. Gene therapy approaches against cancer usingin vivoandex vivogene transfer of interleukin-12. Immunotherapy 2016;8:179-98.
22.Vilcek J, Lee TH. Tumor necrosis factor. New insights into the molecular mechanisms of its multiple actions. J Biol Chem 1991;266:7313-6.
23.Razaghi A, Owens L, Heimann K. Review of the recombinant human interferon gamma as an immunotherapeutic: Impacts of production platforms and glycosylation. J Biotechnol 2016;240:48-60.
24.Anestakis D, Petanidis S, Kalyvas S, et al. Mechanisms and applications of interleukins in cancer immunotherapy. Int J Mol Sci 2015;16:1691-710.
25.Mosser DM, Zhang X. Interleukin-10: new perspectives on an old cytokine. Immunol Rev 2008;226:205-18.
26.Emmerich J, Mumm JB, Chan IH, et al. IL-10 directly activates and expands tumor-resident CD8+T cells withoutde novoinfiltration from secondary lymphoid organs. Cancer Res 2012;72:3570-81.
27.Tran Janco JM, Lamichhane P, Karyampudi L, et al.Tumor-infiltrating dendritic cells in cancer pathogenesis. J Immunol 2015;194:2985-91.
28.André T, Boni C, Mounedji-Boudiaf L, et al.Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med 2004;350:2343-51.
29.Finn OJ. Cancer vaccines: between the idea and the reality. Nat Rev Immunol 2003;3:630-41.
 Chinese Journal of Cancer Research2018年2期
Chinese Journal of Cancer Research2018年2期
- Chinese Journal of Cancer Research的其它文章
- Future of anti-PD-1/PD-L1 applications: Combinations with other therapeutic regimens
- Tumor immunotherapy: New aspects of natural killer cells
- Systemic therapy for cervical carcinoma – current status
- A nomogram to predict adjuvant chemotherapy recommendation in breast cancer patients with intermediate recurrence score
- A multicenter hospital-based diagnosis study of automated breast ultrasound system in detecting breast cancer among Chinese women
- Health-related quality of life among rural residents aged 45−69 years in Hua County, Henan Province, China: Results of ESECC Trial for esophageal cancer screening with endoscopy
