CuxCo1-x/Al2O3/FeCrAl整体式催化剂上甲苯催化燃烧
赵福真 曾鹏辉,2 季生福,* 杨 肖 李成岳
(1北京化工大学化工资源有效利用国家重点实验室,北京 100029;2中国石油大学(北京)重质油国家重点实验室,北京 102200)
CuxCo1-x/Al2O3/FeCrAl整体式催化剂上甲苯催化燃烧
赵福真1曾鹏辉1,2季生福1,*杨 肖1李成岳1
(1北京化工大学化工资源有效利用国家重点实验室,北京 100029;2中国石油大学(北京)重质油国家重点实验室,北京 102200)
以FeCrAl合金薄片为基底,Al2O3浆料为过渡胶体,不同摩尔比的Cu、Co为催化活性组分,制备了一系列CuxCo1-x/Al2O3/FeCrAl(x=0-1)新型整体式催化剂.采用X射线粉末衍射(XRD),扫描电子显微镜(SEM),X光电子能谱(XPS)和程序升温还原(TPR)等手段对催化剂的结构进行了表征.在微型固定床反应器上评价了催化剂的催化甲苯燃烧性能.研究结果表明:在所制备的整体式催化剂上,当Cu含量比较低时,形成了Cu-Co-O固溶体;当Cu含量比较高时,可以测得CuO的衍射峰.催化剂表面颗粒大小和形貌与Cu、Co摩尔比密切相关.在催化剂表面,Co以Co2+和Co3+价态存在,而Cu主要以Cu2+价态存在.催化剂中的Cu可以改善Co的氧化还原性,从而有利于催化剂活性的提高.在所制备的催化剂中,Cu0.5Co0.5/Al2O3/FeCrAl催化剂具有最好的活性,甲苯在374oC可以完全催化燃烧消除.
FeCrAl; 整体式催化剂; 甲苯催化燃烧; X射线粉末衍射; X光电子能谱
Abstract:A series of CuxCo1-x/Al2O3/FeCrAl(x=0-1)catalysts were prepared using an FeCrAl alloy as support,a boehmite primer sol as the first washcoat layer and copper as well as cobalt oxides as the active washcoat layer.The structure of the catalysts was characterized using X-ray powder diffraction(XRD),scanning electron microscope(SEM),X-ray photoelectron spectroscopy(XPS)and temperatureprogrammed reduction(TPR).Toluene was chosen as the model compound to evaluate the catalytic activity in a conventional fixed-bed quartz reactor.Results indicate that a Cu-Co-O solid solution phase was present when the content of Cu in the catalysts was low and a CuO phase was present when the content of Cu was high.Both Co2+and Co3+were present on the surface of the obtained monolithic catalysts while Cu2+was the main Cu species.The addition of a proper amount of copper oxide improved the reducibility of the cobalt oxide,which enhanced the catalytic activity of the catalysts.All the obtained catalysts showed good activity for the catalytic combustion of toluene.The Cu0.5Co0.5/Al2O3/FeCrAl catalyst had the best catalytic activity,and toluene was totally oxidized at 374°C over it.
Key Words:FeCrAl; Monolithic catalyst; Toluene catalytic combustion; X-ray powder diffraction;X-ray photoelectron spectroscopy
Volatile organic compounds(VOCs)produced from many chemical and petrochemical industries are one of the major air pollutants.Besides their own malodorous and carcinogenic nature,they act as main precursors of ozone and smog formation[1-3].Among all the methods for treatment of VOCs,catalytic combustion has been recognized as one of the most effective and economic technologies.This method has many advantages such as low temperature required for combustion ignition,higher selectivity,and high energy efficiency[4-5].At present,the catalysts investigated for catalytic combustion of VOCs are mainly noble metals and metal oxides[6].Although noble metal catalysts show good activity for the complete oxidation of VOCs,the high cost and the sensitivity to poisoning have led to the development of base metal catalysts for this application.Recently,the development of transition metal oxides for catalytic oxidation of VOCs has been widely reported,and copper-containing catalysts show high activity for the combustion of VOCs.Antuneset al.[7]reported catalytic oxidation of toluene over Cu-NaHY zeolites and found that a higher copper loading led to a higher catalytic activity.The toluene could be completely oxidized to CO2at 350℃,when the copper loading was 5%.Kovandaet al.[8]also concluded that the oxidation activity in toluene combustion increased with the increasing amount of easily reducible copper component.Luet al.[9]prepared a series of transition-metal catalysts using activated carbon(AC)as the support material,and copper,cobalt,iron,and nickel as the catalytic active phases.In their work,they observed that the activity of metal/AC followed a particular order:Cu>Co>Fe>Ni.
However,the conventional pellet or granule catalysts loaded in fixed-bed reactors have high pressure drops and high gradient of temperature thus depressed the activity of the catalysts.Recently,monolithic catalysts have increasingly attracted the attention of researchers.Comparing to the conventional catalysts,the monolithic catalysts have distinct advantages:better mechanical strength,lower capacity of mass and heat transfer,lower thermal expansion coefficient leading to a higher thermal shock resistance[10].The most widely used monolithic substrate is the monoliths made of ceramic or metallic materials.The metallic monoliths present a smaller wall thickness and higher thermal conductivity than the ceramic monoliths[11],thus for the reactions with high space velocity and heat exchange,such as toluene combustion reaction,the metallic monolithic catalysts have a promising application.
In this work,a series of copper-cobalt mixed oxide catalysts with different Cu/Co ratios washcoated on metallic monoliths were reported.The performance of the obtained catalysts for the toluene catalytic combustion was evaluated and the catalysts were characterized by XRD,SEM,XPS,and TPR techniques.
1 Experimental
1.1 Catalyst preparation
The metallic monolithic catalysts were prepared using the FeCrAl alloy foils(OC404,Sandvik Steel,Sweden)as supports.The alloy foils were rolled into several cylinders in different diameters and 50 mm in length.The supports were pretreated successively in basic,acidic and ethanol solution,and then thoroughly rinsed in de-ionized water,and finally the pretreated supports were calcined at 950℃for 15 h in air.In order to improve the adhesion between the washcoat layers and the heat-treated metallic supports,a boehmite primer sol was used as first washcoat layer,then dried at room temperature in air and thereafter at 120℃for 3 h and calcined at 500oC for 4 h,and the monolithic support(Al2O3/FeCrAl)was formed.
The CuxCo1-x/Al2O3/FeCrAl(x=0-1)catalysts were prepared as follows:the solutions of Cu(NO3)2·3H2O(analytical reagent)and Co(NO3)2·6H2O(analytical reagent)were mixed in appropriate ratios.The mixture was added into the boehmite primer sol with a total 40%(w)metal content to obtain the slurry.The monolithic support(Al2O3/FeCrAl)was inserted into the above slurry and held for 1 min,taken out at the constant speed of 3 cm·min-1,then dried at room temperature in air and thereafter at 120℃for 3 h,and then calcined at 500℃for 4 h.The coating procedure should be repeated to achieve the desired coating amount.Finally,the CuxCo1-x/Al2O3/FeCrAl metal support monolithic catalysts were obtained.The compositions of the catalysts were listed in Table 1.
1.2 Catalytic activity measurements
Catalytic combustion experiments were performed in a conventional fixed-bed quartz reactor(i.d.,8 mm;length,300 mm)at atmospheric pressure.The reactant gas mixture was prepared by passing air through a conical flask filled with liquid toluene,which was kept at 0oC.The reaction feed consisted of 1000 μL·L-1of toluene in dry air and the space velocity was 5.6×104mL·g-1·h-1.The reaction was stabilized for 20min at the required temperature,and the outlet products were measured with online gas chromatography(Beijing East and West Electronics Institute,GC-4000A)equipped with a flame ionization detector(FID).The catalyst temperature was controlled with a K-type thermocouple placed in the vicinity of the catalyst bed.In every case,carbon dioxide and water were the only reaction products detected along the whole experiment.
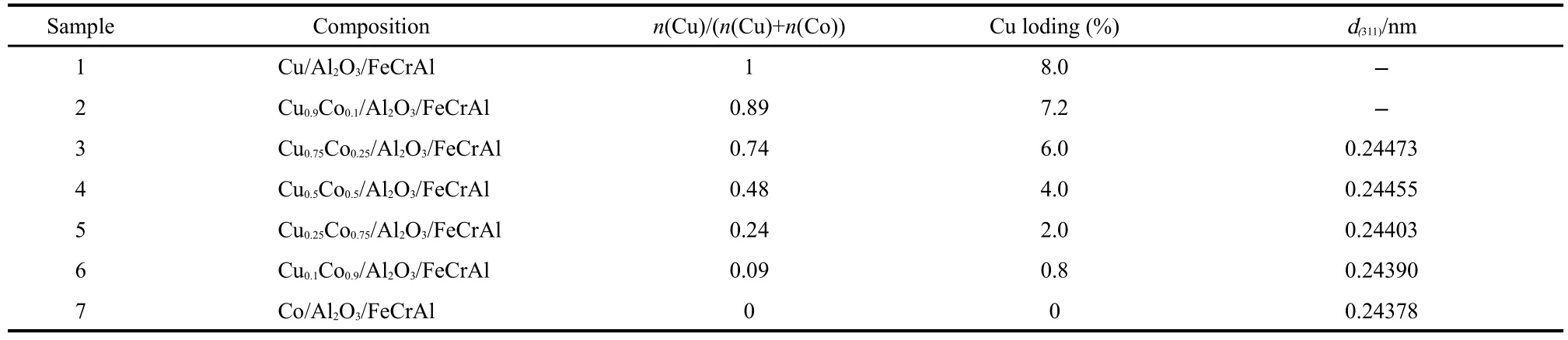
Table 1 Compositions of CuxCo1−x/Al2O3/FeCrAl catalysts
1.3 Catalysts characterization
The phase structure of the samples was characterized by X-ray diffraction(XRD)using a Japan Rigaku D/Max 2500 VB2+/PC diffractometer with a CuKαradiation operating at 200 mA and 40 kV.The morphology of the samples was observed by a Japan HITACHI S-4700 scanning electron microscope(SEM).X-ray photoelectron spectroscopy(XPS)experiments were carried out on an UK ESCALAB250 instrument(Thermo Electron Co.)using MgKαas the exciting radiation at the constant pass energy of 30 eV.Binding energies were calibrated using the carbon present as a contaminant(binding energy of C 1s is 285.0 eV).The surface atomic compositions of all samples were calculated from photoelectron peak areas for each element after correction for instrument parameters.The temperature-programmed reduction(TPR)measurements were carried out using America Thermo Electron Corporation TPD/R/O 1100 series catalytic surfaces analyzer equipped with a TCD detector.0.08 g of the sample was loaded and preheated with 10%(volume fraction,same as below)O2/He mixture at 500℃for 1.5 h.After cooling down to room temperature,5%H2/N2mixture was introduced into the instrument and the temperature was ramped to 800℃at a heating rate of 5℃·min-1.Water produced by the sample reduction was condensed in a cold trap before reaching the detectors.Only H2was detected in the outlet gas confirming the effectiveness of the cold trap.
2 Results and discussion
2.1 XRD measurements
The XRD patterns of the pre-oxidized FeCrAl metal support and CuxCo1-x/Al2O3/FeCrAl monolithic catalysts are presented in Fig.1.For FeCrAl metal support,after the heat treatment at 950℃for 15 h,the characteristic peaks of FeCr(JCPDS 34-0396)and α-Al2O3(JCPDS 88-0826)were observed.The diffraction peaks of Co3O4(2θ=31.20°,36.84°,44.63°,59.46°and 65.35°,JCPDS 42-1467)could be seen when x=0(Co/Al2O3/FeCrAl).With the increase of x,the diffraction peaks had a little shift.Take the most intense peak at 2θ=36.84°as an example:when x=0,the d value for(311)plane is 0.24378 nm(Table 1);when x=0.5,the d value for(311)plane is 0.24455 nm;when x=0.75,the d value for(311)plane is 0.24473 nm.This is because the radius of Cu2+(0.062 nm)is larger than that of Co3+(0.052 nm),which leads to the expansion of Co3O4lattice when Cu2+migrates partially into the cubic structure.Therefore,changes in the d(311)value of the samples indicate that the missing CuO phase at low Culoading formed the Ce-Co-O solid solution in these catalysts[12].Cu and Co can form a Cu-Co-O solid solution that was also been found by Radwan[13].When x>0.5,the most intense peaks of bulk CuO at 35.5°and 38.8°[4]were detected.

2.2 Surface morphology of the samples
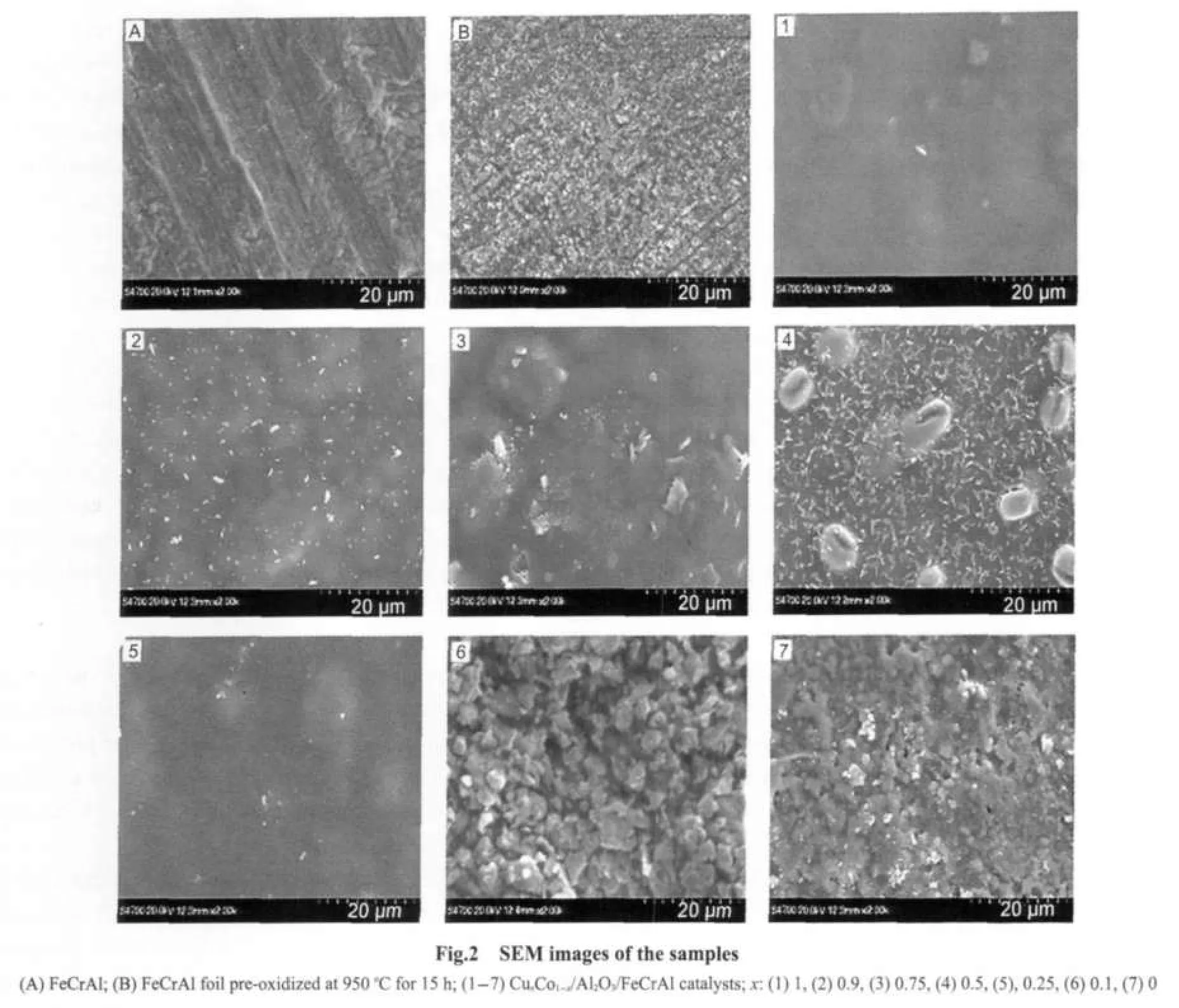
Fig.2 shows the morphology of the FeCrAl support and CuxCo1-x/Al2O3/FeCrAl monolithic catalysts.From the surface view of FeCrAl(Fig.3(A,B)),it can be seen that after pretreatment in air at 950℃for 15 h,the surface became more coarse and had more macropores.This benefited the coating of CuxCo1-x/Al2O3.
As for CuxCo1-x/Al2O3/FeCrAl(x=0-1)monolithic catalysts,the surface morphology changed significantly with the x value.For the catalyst with x=1,namely Cu/Al2O3/FeCrAl(sample 1),the surface was quite homogenous and smooth,only several large particles could be found.However,the surface morphology of the catalysts changed greatly with the decrease of Cu content.When x=0.9,although the surface was still smooth,there were some small particles appeared(sample 2).When x=0.75 and x=0.5(samples 3 and 4),the surface became inhomogenous gradually,the particles became bigger and the heap of particles were very loose.When x further decreased to 0.25(sample 5),the surface morphology of the catalyst became smooth again and almost no particles could be found.When x=0.1(sample 6),the surface feature was different from other catalysts,there appeared the big clusters of particles,the dispersion was inhomogenous and the particle size was large.When x=0(sample 7),the particles of the catalyst decreased obviously,the clusters increased and the heap of the particles was compact.
2.3 X-ray photoelectron spectroscopy
XPS spectra of Cu 2pof catalysts 1,4,and 6 are shown in Fig.3(A).The presence of a higher Cu 2p3/2binding energy(933.0-933.8 eV)and the pronounced shake-up peak(940.0-945.0 eV)are the characteristics of CuO,while a lower Cu 2p3/2binding energy(932.2-933.1 eV)and the absence of the shake-up peak are the characteristics of Cu2O[15-16].After the Gauss model fitting,it can be noticed that the given catalysts exhibit the Cu 2p3/2main peak at about 933.2 eV with the char-acteristic shake-up peak,which indicate the existence of Cu2+species in the catalysts.
The Co 2pspectra of catalysts 2,4,and 7 are shown in Fig.3(B).It can be seen that all the Co species in these samples are characterized by two main peaks(2p3/2at about 780.5 eV and 2p1/2at about 796.1 eV)with the spin-orbit splitting of about 15.0 eV.Since the Co 2pbinding energies of Co2+are similar to that of Co3+,satellite peak information must be considered to distinguish the two oxidation states of cobalt.It is generally accepted that energy gaps between the Co 2pmain lines and satellite peaks are highly related to the oxidation states of Co cation.According to the reports,the high spin Co2+compounds show strong satellite peaks and the binding energies are located at about 5-6 eV above the Co 2p3/2main line.While the low spin Co3+compounds exhibit very weak satellite peaks which are shifted about 10-11 eV to higher binding energies from the main peak[17-18].Based on the binding energies of Co 2pmain lines and the energy gaps between the main lines andthe satellite peaks,it can be concluded that both Co2+and Co3+exist on the surface of the obtained monolithic catalysts.
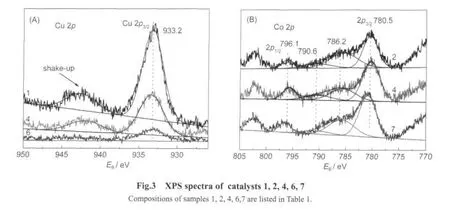
Table 2 presents the binding energies of Cu,Co,Al,O and the surface atomic percentages of all the samples.It can be seen that there is no significant difference among the binding energies of different samples for the same element but the surface compositions vary among different samples.
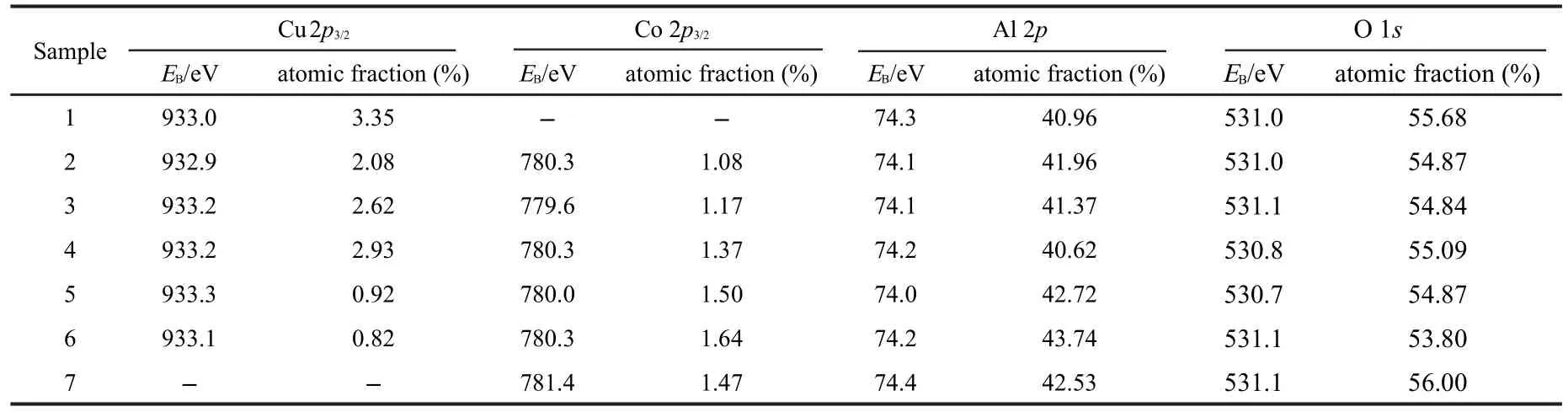
Table 2 XPS results of the CuxCo1-x/Al2O3/FeCrAl catalysts
2.4 Catalytic activity
Fig.4 shows catalytic activities of toluene oxidation with reaction temperatures on the CuxCo1-x/Al2O3/FeCrAl catalysts.It can be observed that the atomic ratio of Cu/Co had a significant influence on the catalytic activity of the catalysts.The catalytic activity of toluene oxidation over either Cu/Al2O3/FeCrAl(sample 1)or Co/Al2O3/FeCrAl(sample 7)was quite low.For sample 1,T10(the temperature for 10%conversion of toluene)andT90(the temperature for 90%conversion of toluene)were 284 and 415oC,respectively;while for sample 7,T10andT90were 313 and 436℃,respectively.Wanget al.[19]reported catalytic combustion of toluene over CuO/γ-Al2O3catalysts.They found that 5%(w)CuO/γ-Al2O3showed the highest catalytic activity andT90was 320 ℃ at the space velocity of 3600 mL·g-1·h-1.Considering the space velocity in our experiment is 5.6×104mL·g-1·h-1,the Cu/Al2O3/FeCrAl monolithic catalyst exhibited the better catalytic performance.The activities of CuxCo1-x/Al2O3/FeCrAl(x=0.1-0.9)catalysts were much higher than that of sample 1 or 7.Whenx=0.5(sample 4),the catalyst exhibited the best catalytic activity with toluene conversion of 10%at 260℃.The toluene can be completely oxidized at 374℃.However,further increasing the Cu content(x>0.5)resulted in the decrease of the catalytic activity.From the results of XRD measurements,the peaks of CuO were observed whenx>0.5,which suggested that the presence of bulk CuO depressed the catalytic activity.These also implied that the Cu-Co-O solid solution was the active phase for toluene oxidation.Liet al.[20]also suggested that the Cu-Mn-O was responsible for the toluene combustion reaction in Cu-Mn/MCM-41 catalysts.
2.5 Temperature-programmed reduction measurements
Fig.5 shows the H2-TPR profiles of CuxCo1-x/Al2O3/FeCrAl monolithic catalysts.It can be seen that there were two reduction peaks for Co/Al2O3/FeCrAl catalyst(sample 7).The peak at 470℃should be corresponded to the reduction of surface dispersed Co3O4species to CoO,the peak at 543℃should be attributed to the reduction of CoO to metallic cobalt[21-22].It is obvious that the reduction temperature shifted to the lower temperature with the increase of copper oxide loading,suggesting that the introduction of a proper amount of copper oxide improved the reducibility of the cobalt oxide,which should be related to the strong interaction between surface cobalt oxide and surface copper oxide.For samples 5 and 6,the reduction peaks at lower temperature may be attributed to the reduction of copper oxide,and the higher temperature peaks can be assigned to the reduction of cobalt oxide[23].While for sample 4,the twopeaks at 184 and 217℃were the reduction of surface copper oxide,the third peak at 270℃should be assigned to reduction of surface cobalt oxide.For samples 2 and 3,the two reduction peaks in this area were associated with the stepwise reduction of the dispersed copper oxides[24].It is known that the reduction of pure CuO is characterized by a single peak at about 315℃[25].Our work shows that there are three reduction peaks at 178,197,and 286℃for sample 1,which substantiate the interaction between Cu and the support.The two lower temperature peaks may correspond to the reduction of small CuO clusters and isolated Cu2+ions respectively,while the 286℃can be ascribed to the bulk CuO particles[26].

3 Conclusions
CuxCo1-x/Al2O3/FeCrAl monolithic catalysts were prepared using FeCrAl alloy as support and Al2O3as washcoat.In the CuxCo1-x/Al2O3/FeCrAl monolithic catalysts,the catalytic activities of the catalysts were strongly influenced by the Cu/Co atomatic ratios.The Cu0.5Co0.5/Al2O3/FeCrAl exhibited the highest catalytic activity.Toluene could be completely oxidized at 374 ℃ with a space velocity of 5.6×104mL·g-1·h-1.Using FeCrAl alloy foils as supports benefited the dispersion of Cu.CuO phase was observed at higher Cu contents.The SEM results showed that the surface morphology of the monolithic catalysts changed significantly with the ratios of Cu and Co.The XPS results showed that both Co2+and Co3+existed in the obtained monolithic catalysts,and Cu existed mainly as Cu2+species.The TPR results showed that,when x<0.5,the introduction of copper oxide made the reduction temperature of the catalysts shift to the lower temperature.It was suggested that the introduction of a proper amount of copper oxide improved the reducibility of the cobalt oxide and thus enhanced the catalytic activity of the catalysts.
1 Scire,S.;Minico,S.;Crisafulli,C.;Satriano,C.;Pistone,A.Appl.Catal.B-Environ.,2003,40:43
2 Mitsui,T.;Tsutsui,K.;Matsui,T.;Kikuchi,R.;Eguchi,K.Appl.Catal.B-Environ.,2008,81:56
3 Li,W.B.;Gong,H.Acta Phys.-Chim.Sin.,2010,26:885 [黎维彬,龚 浩.物理化学学报,2010,26:885
4 Palacio,L.A.;Silva,J.M.;Ribeiro,F.R.;Ribeiro,M.F.Catal.Today,2008,133-135:502
5 Huang,H.;Liu,Y.;Tang,W.;Chen,Y.Catal.Commun.,2008,9:55
6 Kim,S.C.;Shim,W.G.Appl.Catal.B-Environ.,2008,79:149
7 Antunes,A.P.;Ribeiro,M.F.;Silva,J.M.;Ribeiro,F.R.;Magnoux,P.;Guisnet,M.Appl.Catal.B-Environ.,2001,33:149
8 Kovanda,F.;Jiratova,K.;Rymes,J.;Kolousek,D.Appl.Clay Sci.,2001,18:71
9 Lu,C.Y.;Wey,M.Y.;Chen,L.I.Appl.Catal.A-Gen.,2007,325:163
10 Avila,P.;Montes,M.;Miro,E.E.Chem.Eng.J.,2005,109:11
11 Barbero,B.P.;Costa-Almeida,L.;Sanz,O.;Morales,M.R.;Cadus,L.E.;Montes,M.Chem.Eng.J.,2008,139:430
12 Cesar,D.V.;Perez,C.A.;Salim,V.M.M.;Schmal,M.Appl.Catal.A-Gen.,1999,176:205
13 Radwan,N.R.E.;Mokhtar,M.;El-Shobaky,G.A.Appl.Catal.A-Gen.,2003,241:77
14 Zou,H.;Dong,X.;Lin,W.Appl.Surf.Sci.,2006,253:2893
15 Avgouropoulos,G.;Ioannides,T.;Matralis,H.Appl.Catal.BEnviron.,2005,56:87
16 Zhu,P.;Li,J.;Zuo,S.;Zhou,R.Appl.Surf.Sci.,2008,255:2903
17 Khassin,A.A.;Yurieva,T.M.;Kaichev,V.V.;Bukhtiyarov,V.I.;Budneva,A.A.;Paukshtis,E.A.;Parmon,V.N.J.Mol.Catal.AChem.,2001,175:189
18 Zhu,J.;Gao,Q.Microporous Mesoporous Mat.,2009,124:144
19 Wang C.H.Chemosphere,2004,55:11
20 Li W.B.;Zhuang M.;Wang J.X.Catal.Taday,2008,137:340
21 Haneda,M.;Kintaichi,Y.;Bion,N.;Hamada,H.Appl.Catal.BEnviron.,2003,46:473
22 Tang,C.W.;Kuo,M.C.;Lin,C.J.;Wang,C.B.;Chien,S.H.Catal.Today,2008,131:520
23 Zou,Z.Q.;Meng,M.;Zha,Y.Q.J.Alloy.Compd.,2009,470:96
24 Liu,L.;Chen,Y.;Dong,L.;Zhu,J.;Wan,H.;Liu,B.;Zhao,B.;Zhu,H.;Sun,K.;Dong,L.;Chen,Y.Appl.Catal.B-Environ.,2009,90:105
25 Tang,X.;Zhang,B.;Li,Y.;Xu,Y.;Xin,Q.;Shen,W.Appl.Catal.A-Gen.,2005,288:116
26 Moretti,E.;Lenarda,M.;Storaro,L.;Talon,A.;Montanari,T.;Busca,G.;Rodriguez-Castellon,E.;Jimenez-Lopez,A.;Turco,M.;Bagnasco,G.;Frattini,R.Appl.Catal.A-Gen.,2008,335:46
Catalytic Combustion of Toluene over CuxCo1−x/Al2O3/FeCrAl Monolithic Catalysts
ZHAO Fu-Zhen1ZENG Peng-Hui1,2JI Sheng-Fu1,*YANG Xiao1LI Cheng-Yue1
(1State Key Laboratory of Chemical Resource Engineering,Beijing University of Chemical Technology,Beijing100029,P.R.China;2State Key Laboratory of Heavy Oil Processing,China University of Petroleum,Beijing102200,P.R.China)
O643
Received:June 9,2010;Revised:July 29,2010;Published on Web:October 19,2010.
∗Corresponding author.Email:jisf@mail.buct.edu.cn;Tel/Fax:+86-10-64419619.
The project was supported by the National Natural Science Foundation of China(20473009).
国家自然科学基金(20473009)资助项目
--纳米材料与技术论坛例记

