Risk of control failure to insecticides malathion, profenophos + cypermethrin mixture, and fipronil in boll weevil (Coleoptera:Curculionidae) populations from Bahia, Brazil
COELHO Beatriz S., LEITE Suzany A., DOS SANTOS Mateus P., GUEDES Raul N.C., BASTOS Cristina S.,MOREIRA Aldenise A., BONFIM João E.V. and CASTELLANI Maria A.,5*
Abstract Background To control the boll weevil Anthonomus grandis grandis (Coleoptera: Curculionidae), a key pest of cotton in the Americas, insecticides have been intensively used to manage their populations, increasing selection pressure for resistant populations.Thus, this study aimed to detect insecticide resistance and assess insecticide control failure likelihood of boll weevil populations exposed to malathion, profenophos + cypermethrin, and fipronil insecticides.
Keywords Anthonomus grandis grandis, Cotton pests, Pest insect resistance management, Insecticide control failure
Introduction
Brazilian cotton production has been growing considerably over the past few years.By 2023, the perspective of Brazilian planted area is 1.64 million hectares, with a production of 2.97 million tons (t), and an export estimate of 2.05 million tons, representing a 19.7% growth compared with the 2020/2021 harvest season (Companhia Nacional de Abastecimento 2022).The scenario of cotton production in Brazil is promising, being 75%cultivated in the state Mato Grosso, which has made the country one of the largest exporters in the world (Associação Brasileira dos Produtores de Algodão 2023).In the Northeast region, Bahia, Piauí, and Maranhão stand out as the main cotton-producing states (Ximenes and Coêlho 2021).Bahia is the second largest national producer, with a production of 1.3 million tons of cotton seed and 0.50 million tons of lint cotton (Companhia Nacional de Abastecimento 2021).
Among the challenges faced by the Brazilian cotton producers, the damages caused by the boll weevil,Anthonomus grandis grandis(Perkin 2023) (Coleoptera: Curculionidae), are certainly worrying and a major threat to cotton production in the country (Azambuja and Degrande 2014).The females of this insect lay their eggs inside reproductive structures such as flowers, buds,squares, and bolls, causing their abscission resulting in production losses (Showler and Cantú 2005).Their oviposition takes several days, allowing for a gradual emergence of the adults and requiring successive insecticide spraying for interruption of the insect life cycle (Showler 2012).Control measures are recommended when 3%-5%of the flower buttons have the presence of adult boll weevils and/or attack signs (oviposition and nutrition) caused by the insect (Bélot 2016).
Chemical control is practically the only method used to reduce the population outbreaks of the boll weevil,with an average of 19 insecticide applications per cultivation cycle in the state of Bahia (Fundo para o Desenvolvimento do Agronegócio do Algodão 2019).The overuse of insecticides in cotton crops without pest resistance monitoring can make chemical control expensive and inefficient (Ullah et al.2021).The use of insecticides may also cause adverse environmental effect,and increases selection of resistant insect populations(Guedes and Cutler 2014).This fact is aggravated by the fact that cotton in Brazil is one of the crops that use more synthetic pesticides (Pignati et al.2017).
Insecticide resistance is a genetic change in response to selection by toxicants that impair field control of the pest species population or, in other words, may lead to insecticide control failure (Guedes 2017; Insecticide Resistance Action Committee 2022).Therefore, resistance monitoring and detection are critical to understanding and addressing existing and developing insecticide resistance problems (Sparks et al.2021).The early detection of resistance allows for a greater chance of delaying the evolution of insecticide resistance, and sometimes reestablishing the susceptibility of pest insects to insecticides and other control methods (Insecticide Resistance Action Committee 2022).Thereby, it is important for the management of boll weevil in cotton areas, thus providing benefits to the integrated pest management (IPM)(Arruda et al.2021).
For the control of boll weevil in Brazil, 137 commercial insecticide formulations are registered, counting 27 commercialized active ingredients belonging to the chemical classes of pyrethroids, phenyl pyrazoles,organophosphates, carbamates, neonicotinoids, and others (Sistema de Agrotóxicos Fitossanitário 2022).Among these, the organophosphate malathion is one of the most used insecticides (Rolim et al.2019).In a recent survey, it was found that the most commonly used insecticides for boll weevil control in the Brazilian cerrado were malathion, fipronil, carbosulfan,thiamethoxam + lambda-cyhalothrin, amd profenofos + cypermethrin (Torres 2022).Studies on the detection of boll weevil insecticides resistance have focused on malathion, beta-cyfluthrin, and zeta-cypermethrin in insect populations of the state of Mato Grosso,but insecticide resistance was detected at the time(Oliveira-Marra et al.2019).More recently, populations of boll weevils from the states of Pernambuco,Ceará, and Mato Grosso exposed to malathion and beta-cyfluthrin exhibited high levels of resistance to beta-cyfluthrin (Rolim et al.2021).In boll weevil populations from Texas, USA, malathion resistance was not detected, but temporal and qualitative differences in gene expression were identified in isoforms of detoxifying esterase B1 and glutathione-S-transferases, which can be associated with organophosphate resistance(Perkin et al.2023).
The constantly high population levels of boll weevils in the cotton fields of Bahia, associated with the intensive use of insecticides, raise the concern that some populations of this pest are likely resistant to the main insecticides used against them.Thus, this study aimed to detect and determine the resistance levels in Bahia populations of the boll weevil to the malathion, profenophos + cypermethrin, and fipronil, and to assess the associated risk of insecticide control failure.
Materials and methods
Insects and insecticides
The boll weevil populations were collected in 12 properties located in two cotton-producing meso-regions in the state of Bahia, Brazil – west (2) and southwest (10)(Table 1) in the 2021/2022 season.The VCA1 code refers to the susceptible population collected from the Agricultural Field Station of the State University of Southwest Bahia, in the county of Vitória da Conquista (Bahia).This population has been maintained without exposure to insecticides and is approximately 320 km away from the commercial cotton production farms in the state.Each sampling site was georeferenced using a receptor of the global positioning system (GPS) (Garmin E-Trex Vista HCx, Olathe, Kansas, USA).

Table 1 Identification and geographic coordinates of the sampling sites of the boll weevil (Anthonomus grandis grandis) populations in the state of Bahia, Brazil
To collect adults of known ages to be used in the bioassays, we gathered cotton flower buds with signs of oviposition following the methodology proposed by Oliveira-Marra et al (2019).The buds were packed in rectangular plastic trays (30 cm × 45 cm × 50 cm) and closed with organza fabric until the emergence of the adults, which were daily collected and packed in transparent plastic pots (volume of 500 mL) containing cotton moistened in water and cotton flower buds (for nutrition).The preservation of the boll weevils, as well as the performance of the bioassays, were conducted in an acclimatized room at a temperature of 25 °C ± 2 °C, photoperiod of 12 h/12 h (light/dark), and relative humidity from 50% to 70%.The bioassays were performed with newly emerged boll weevils (between 1 and 3 days of age).
A survey was conducted on the most commonly used products by local producers in cotton cultivation to control the boll weevil in the state of Bahia, and the consequent risk of failure of these products: malathion(active ingredient 1 000 g·L-1, emulsifiable concentrate;produced by FMC Chemical of Brazil Ltd., Campinas,SP, Brazil), profenophos + cypermethrin (active ingredient 400 g·L-1+ 40 g·L-1, emulsifiable concentrate;produced by Syngenta Crop Protection Ltd., São Paulo,SP, Brazil), and fipronil (active ingredient 600 g·L-1,emulsifiable concentrate; produced by Ouro Fino Chemical Ltd., Uberaba, MG, Brazil).
According to the IRAC classification (2022), malathion belongs to the chemical subgroup of organophosphates(1B), which are acetylcholinesterase (ACE) inhibitors;profenofos + cypermethrin consists of the combination of an organophosphate (1B) and a pyrethroid (3A), acting as acetylcholinesterase inhibitor and modulator of sodium channels, respectively; and fipronil is included in the subgroup of phenylpyrazoles (2B), which acts in the blockage of chloride channels mediated by gamma-aminobutyric acid (GABA).
Time-mortality bioassays
The insecticides were used in their commercial formulation with the maximum concentration prescribed in the package insert for field use considering the spraying rate of 300 L·ha-1for malathion and fipronil, and 200 L·ha-1for profenophos + cypermethrin, according to the label rates, as registered at the Brazilian Ministry of Agriculture (Sistema de Agrotóxicos Fitossanitário 2022)following the manufacturer’s recommendations.Distilled water was used as the diluent.Thus, the dosages of active ingredients used were 2 000 g·ha-1of malathion,400 g·ha-1, 40 g·ha-1of profenophos + cypermethrin, and 78 g·ha-1of fipronil.Petri dishes (9.0 cm diameter, 1.5 cm height) received 1.0 mL of the insecticide coatings and were subsequently dried out at room temperature for 24 h.Each of the three replicates consisted of 10 adult boll weevils (unsexed) from the same population, which were placed on the Petri dish treated with the respective insecticide.The sides of the plates were impregnated with a layer of neutral talc to prevent the insects from crawling the container and escaping.Mortality assessments were performed every 24 h after insecticide exposure,and then daily until the mortality of all adults.A control treatment (witness) maintained only with distilled water was applied to determine the natural mortality of the insects.Mortality was registered by transferring each adult to glass plates that were placed on a hot plate(35 °C) to stimulate the movement of any boll weevil that would pretend to be dead (Rolim et al.2021); Torres 2022).Adults that would not move in such conditions over an observation period of one minute were considered dead.
Statistical analyses
The survival results of the time-mortality bioassays were subjected to survival analysis using Kaplan–Meier estimators to generate the respective median survival times(lethal time,LT50) for each population and insecticide.The survival curves to each population for each insecticide were compared using the Holm-Sidak test (P< 0.05)(SigmaPlot 12.5, 2013, Systat, San Jose, CA, USA).The resistance levels or resistance ratios (RR) to the referred insecticides were estimated by dividing theLT50of a given population by theLT50of the susceptible population.The resistence ratios (RR) and their 95% confidence intervals were calculated following the method described by Robertson et al.(2007), and these were considered significant when the confidence interval did not include the value 1.0 (Robertson et al.2007).The significance of the estimated resistance levels was determined as previously described (Tuelher et al.2018; Oliveira-Marra et al.2019;Leite et al.2020).
The mortality of the boll weevils within 48 h was used to estimate the control efficacy, duly corrected by the natural mortality recorded in the (untreated) controls(Abbott (Abbott 1925)).The control failure likelihood(CFL) was determined using the 48 h mortality data, following Guedes’ (2017) formula:
A practical discriminatory concentration (the fielduse label rates) was used to monitor control failure of the insecticides tested [based on the minimum efficacy required for insecticides (80% mortality)] (Ministério da Agricultura Pecuária e Abastecimento 1995).The expected (80%) is the minimum efficacy limit expected for insecticide registration in Brazil (Ministério da Agricultura Pecuária e Abastecimento 1995).The results of the risks of control failure were compared through aZtest (P< 0.05) with continuity correction to verify the populations with significant risk of control failure (Dângelo et al.2018).
The relationship between the insecticide resistance levels and their respective control failure likelihood was tested through regression analysis using the curve-fitting procedure of TableCurve 2D (Systat, San Jose, CA, USA).The model selection was based on parsimony, highFvalues (and reduced error), and steep increase inR2with the model complexity.
Results
LT50 and survival of boll weevil the insecticides
LT50varied between 24 and 77 h for malathion and profenophos + cypermethrin, and from 24 to 61 h for fipronil(Table 2).Eleven populations exhibited resistance to at least one insecticide, while 7 populations, LEM1, LEM2,BJL1, CAN1, PMA1, PMA2, and PIN2, exhibited resistance to these three insecticides.Resistance to malathion was found in 10 populations (LEM1, LEM2, BJL1,CAN1, CAN2, GBI1, PMA1, PMA2, PIN2, and PIN3),11 populations were resistant to profenophos + cypermetrin (LEM1, LEM2, BJL1, CAN1, CAN2, GBI1, PMA1,PMA2, PIN 1, PIN2, and PIN3); seven populations were resitant to fipronil ( LEM1, LEM2, BJL1, CAN1, PMA1,PMA2, and PIN2) (Tables 2 and 3).
The LEM1, PMA2, GBI1, and PIN2 populations were capable of surviving for up to 96 h after being exposed to malathion.The similarLT50was found for the BJL1,PMA2, CAN, and PIN3 populations when exposed to profenophos + cypermethrin.For fipronil, the highest survival time reached 72 h for the LEM1 and LEM2 populations (Tables 2 and 3).
The survival curves of the boll weevil populations varied according to the insecticide tested (Fig.1A).When exposed to water, after 96 h, 80% of the individuals of each population were alive.The survival time was longer for malathion and profenophos + cypermethrin, reaching total death within 96 h.For fipronil,the survival time was 72 h.The survival probabilities for the boll weevils of the PIN3, CAN2, PMA2, and BJL1 populations after 72 h were superior to 38% when exposed to malathion (Fig.1B).Similar time probabilities were found for the LEM1, PMA2, GBI1, and PIN2 populations when exposed to profenophos + cypermethrin (Fig.1C).For fipronil, 60% of the boll weevils of the LEM1 and LEM2 populations survived until 72 h(Fig.1D).
The resistance ratio and risk of control failure
The RR values varied between 1.00 and 3.21 times for malathion, from 1.00 to 3.23 times for profenophos + cypermethrin, and from 1.00 to 2.54 times for fipronil (Table 3).The resistance levels were low(< 10-fold) for all insecticides.
The final mortality of the boll weevil populations also varied according to the insecticide tested, between 10.00% and 100.00% for malathion and from 16.7% to100.00% for profenophos + cypermethrin and fipronil.Ten and eleven populations exposed to malathion and profenophos + cypermethrin, respectively, did not reach the minimum efficacy limit of 80% of mortality (Ministério da Agricultura Pecuária e Abastecimento 1995).However, fipronil proved more efficient at controlling the boll weevil since 8 populations (PMA1,GBI1, CAN1, CAN2, PIN1, PIN2, PIN3, and VCA1)reached mortality levels above the minimum limit of 80% (Table3).
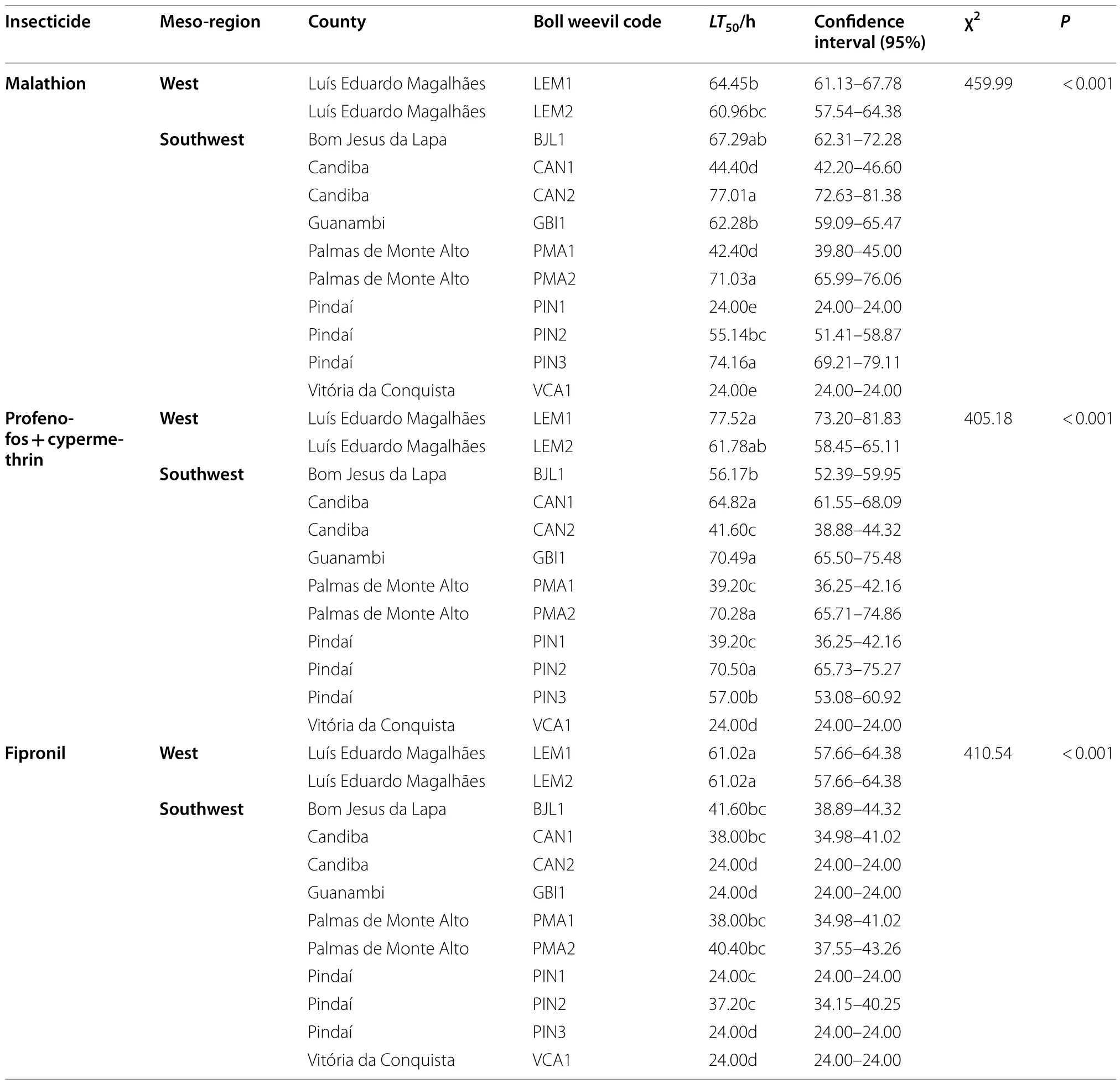
Table 2 The median lethal times (LT50) of the boll weevil (Anthonomus grandis grandis) populations exposed to the malathion,profenophos + cypermethrin, and fipronil insecticides.Values of LT50 followed by the same letter do not differ significantly according to the Holm-Sidak test (P < 0.05).The values of χ 2 and P refer to the difference between the populations concerning the same insecticide
The risk of control failure was significant for malathion and profenophos + cypermerin in 7 populations.For fipronil, only 2 boll weevil populations exhibited a significant risk of control failure (Table 3).
Relationship between resistance and risk of control failure
The relationship between the resistance ratio and the probability of control failure was significant for malathion (Fig.2A), profenophos + cypermethrin (Fig.2B),and fipronil (Fig.2C).The risk of control failure for these insecticides is correlated to the respective resistance level.
Discussion
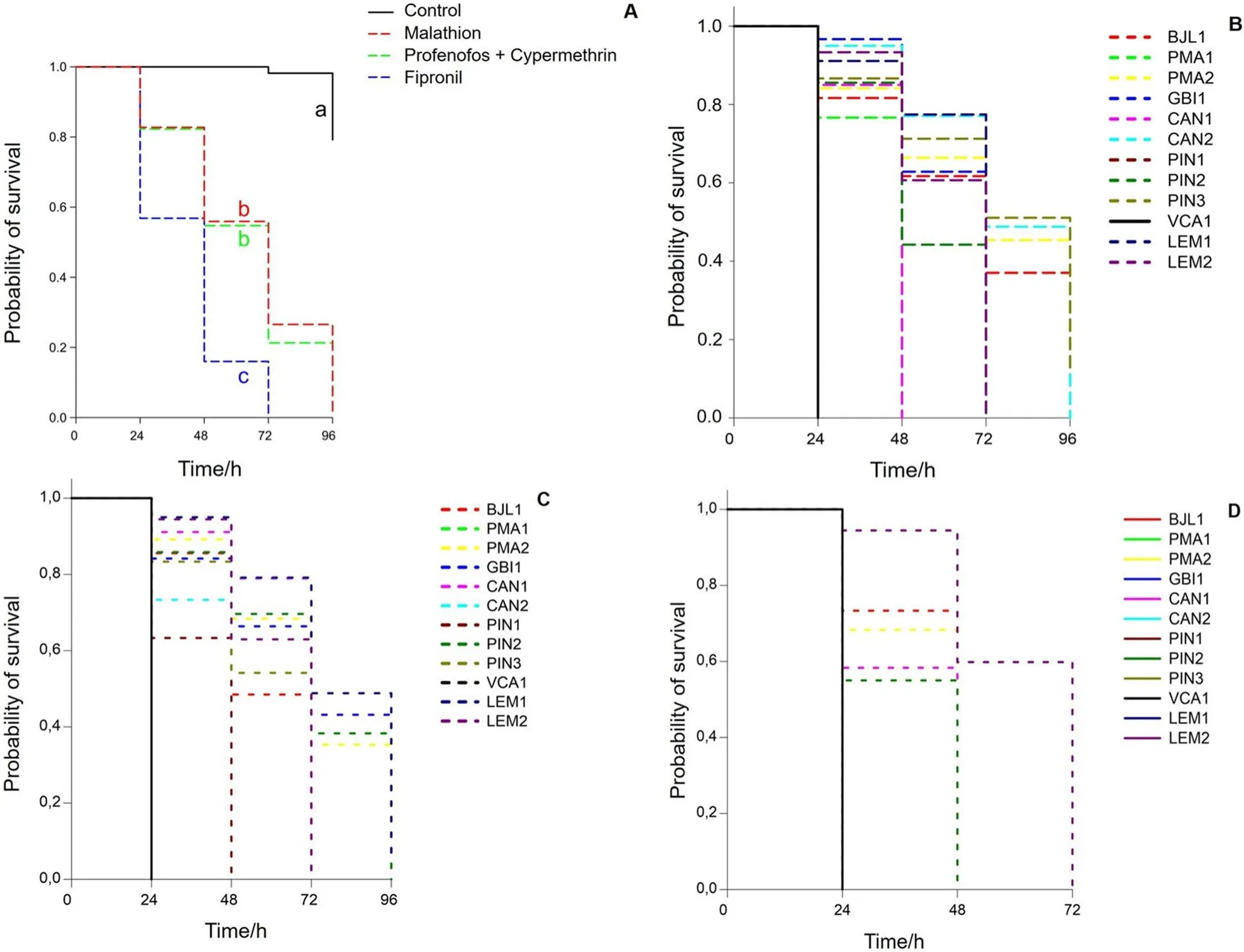
Fig.1 Survival curves for the insecticides assessed, regardless of the boll weevils (Anthonomus grandis grandis) population (A) exposed to malathion (B), profenophos + cypermethrin (C), and fipronil (D)
Despite the possibilities of integrating cultural, biological, and behavioral tactics for the management of the boll weevil, cotton farmers worldwide remain extremely dependent on the use of insecticides for this pest species suppression (Anderson et al.2019).The boll weevil can be present throughout the reproductive phase of the cotton cycle, thus requiring several sprayings with insecticides, which may lead to the selection of resistant populations.The resistance phenomenon consists of a genetic change in response to the selection that can compromise the efficacy of insecticides, leading to control failure(Guedes 2017; Leite et al.2020).For the boll weevil, there are currently 41 reported cases of resistance to insecticides in cotton-producing regions in the United States of America, Mexico, Venezuela (Arthropod Resistance and Database 2022), and Brazil (Rolim et al.2021).
Our results indicated high risk of control failure and low efficacy, the minimum expected threshold (80%) for malathion and mixture profenophos + cypermethrin against the studied boll weevil populations.The boll weevil populations from the west mesoregion (LEM1 and LEM2) exhibited a high risk of control failure and low mortality for the three insecticides.Since this mesoregion encompasses the greatest commercial cotton cultivation area in the state of Bahia, with 300–320 thousand hectares cultivated, the intensive use of chemical control is common for pest population suppression with an average of 19 applications per harvest (Fundo para o Desenvolvimento do Agronegócio do Algodão 2019).
The results obtained for malathion differ from those findings for the boll weevil populations in the state of Mato Grosso in 2016/2017, which were susceptible to this insecticide (Oliveira-Marra et al.2019).A similar study with boll weevil populations in 2016–2018 from the states of Pernambuco, Ceará, and Mato Grosso found a high efficacy of malathion and high resistance ratios for beta-cyfluthrin (Rolim et al.2019).
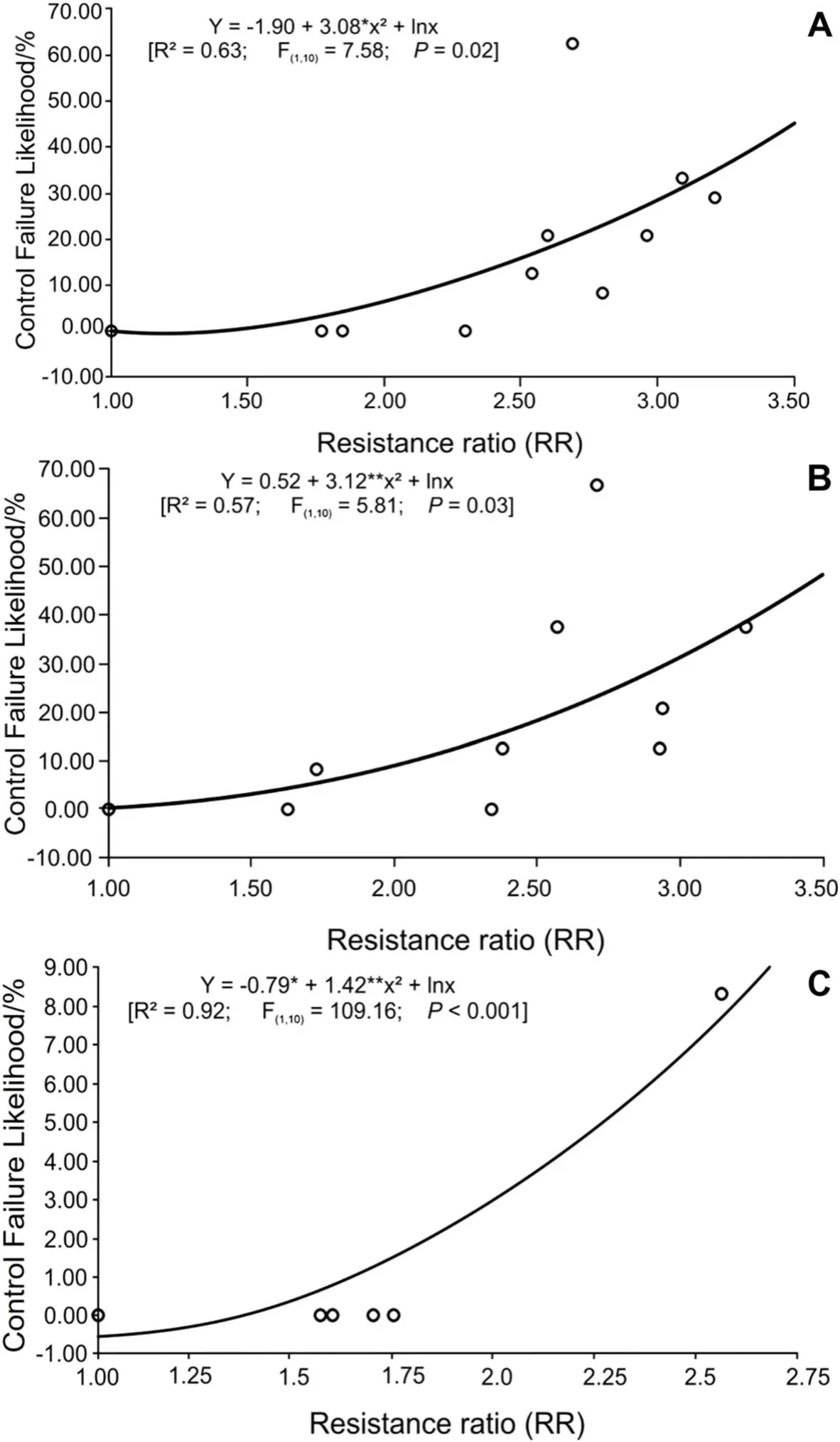
Fig.2 Relationship between the resistance ratio and control failure likelihood in boll weevil (Anthonomus grandis grandis) populations to malathion(A), profenophos + cypermethrin (B), fipronil (C)
More recently, the high toxicity of phenylpyrazoles(fipronil and ethiprole) insecticides was reported in the boll weevil (Torres et al.2022).The same study also found moderate risks of control failure for profenofos + cypermethrin and low risks for malathion.Organophosphate and pyrethroid insecticides are among the most used for pest control in agriculture(Yao et al.2020).Malathion, an organophosphate inhibitor of acetycholinesterase, is the insecticide most used against the boll weevil, while pyrethroidsact on the sodium channel of the axon membrane(Costa 2015).Of the insecticides used, fipronil showed the greatest efficiency against the surveyed boll weevil populations.This phenylpyrazole insecticide blocks the passage of chlorine ions in the pre- and post-synapse of the nerve cell membrane and in the gamma-aminobutyric acid channel (GABA) (Gant et al.1998; Singh et al.2021).
The ranges of rasitance ratios are not directly translated into (field) pest control failure (French-Constant and Roush 1990), but they tend to be related, particularly if insecticide resistance is the prevalent cause of control failure.The resistance ratio estimates obtained were relatively low, but the efficacy of these insecticides(especially malathion and profenophos + cypermethrin)is compromised for most of the boll weevil populations studied.Thus, low resistance levels can still lead to high risks of control failure, as observed here, which is useful warning for the evolution of resistance and providing relevant information to guide the implementation of efficient pest management practices (Arthropod Resistance and Database 2022).
The high potential of damage caused by the boll weevil requires an intensive system that includes the use of different pest control methods (Santos et al.2021).It is especially recommended the compulsory disposal of cultural remains respecting a specific time window for seeding (sanitary emptiness), pheromone traps for monitoring, and biological control practices, among other methods (Santos et al.2021).
In summary, the resistance to insecticides is already one of the major challenges for the management of the boll weevil in cotton-producing regions of Bahia.Resistance levels are low but enough to compromise insecticide field efficacy and impose significant risk of future control failure.Thus, continued monitoring and increased use of resistance management practices are necessary for effectively controlling the boll weevil in the region.
Acknowledgements
We would like to thank the CAPES Foundation (Brazilian Ministry of Education)and the Foundation for Research Support of the State of Bahia (FAPESB) for the financial support.We would also like to thank the cotton farmers of West and Southwest Bahia, the ABAPA, and consultants for their support.
Authors’ contributions
Conceptualization, Coelho BS, Leite SA and Castellani MA; Methodology, Coelho BS, Leite SA and Castellani MA; Software, Coelho BS, Leite SA and Santos MP dos; Validation, Coelho BS, Formal analysis, Coelho BS, Leite SA and Guedes RNC; Investigation, Coelho BS and Leite SA; Resources, Coelho BS, Leite SA and Castellani MA; Data curation, Coelho BS; Writing – original draft preparation,Coelho BS; Writing-review and editing, Coelho BS, Leite SA and Castellani MA,Bastos CS, Guedes RNC, Moreira AA; Visualization, Coelho BS, Leite SA and Castellani MA, Bastos CS, Guedes RNC, Moreira AA; Supervision, Castellani MA and Guedes RNC; Project Administration, Castellani MA; Funding acquisition,Bonfim JEV.All authors read and approved the manuscript.
Funding
This research was supported by Foundation for Research Support of the State of Bahia (FAPESB), the CAPES Foundation (Brazilian Ministry of Education;Finance Code 001) for financial support and Bahia Association of Cotton Producers.
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this article.
Author details
1Department of Crop Science and Animal Science, State University of Southwestern Bahia, Vitória da Conquista, BA 45031-900, Brazil.2Department of Entomology, Federal University of Viçosa, Viçosa, MG 36570-900, Brazil.3Faculty of Agronomy and Veterinary, University of Brasília, Brasília, DF 70910- 900,Brazil.4Bonfim Industry and Commerce Ltd, Varzéa Grande Farm, Bom Jesus da Lapa, BA 47600-000, Brazil.5Departament of Crop Science and Animal Science, State University of Southwest Bahia, Vitória da Conquista, BA 45031-900,Brazil.
Received: 14 April 2023 Accepted: 2 October 2023

 Journal of Cotton Research2023年4期
Journal of Cotton Research2023年4期
- Journal of Cotton Research的其它文章
- Biocontrol potential of entomopathogenic nematode, Heterorhabditis indica against pink bollworm, Pectinophora gossypiella (Saunders)(Lepidoptera: Gelechiidae)
- Enhancing waterlogging tolerance in cotton through agronomic practices
- Mepiquat chloride priming confers the ability of cotton seed to tolerate salt by promoting ABA-operated GABA signaling control of the ascorbate–glutathione cycle
- Feasibility study of assessing cotton fiber maturity from near infrared hyperspectral imaging technique
- Effects of mepiquat chloride and plant population density on leaf photosynthesis and carbohydrate metabolism in upland cotton
- Are yarn quality prediction tools useful in the breeding of high yielding and better fibre quality cotton (Gossypium hirsutum L.)?
