Raman spectroscopy of isolated carbyne chains confined in carbon nanotubes: Progress and prospects
Johannes M.A.Lechner, Pablo Hern´andez L´opez, and Sebastian Heeg
Institut f¨ur Physik and IRIS Adlershof,Humboldt Universit¨at zu Berlin,12489 Berlin,Germany
Keywords: carbyne,linear carbon chains,carbon nanotubes,Raman spectroscopy
1. Introduction
Carbyne by definition is an infinitely long sp1-hybridized linear chain of carbon atoms which manifests the truly onedimensional (1D) allotrope of carbon at the one-atom crosssection limit.[1–3]The scientific interest in carbyne is driven by the fact that it is a missing member in the family of carbon allotropes and the text book example of a 1D model system.Carbyne can exist in two structurally different configurations as shown in Fig. 1(a). In the cumulenic or metallic configuration, the carbon atoms are connected to their neighbours by double bonds. The stable form of carbyne is the polyynic or semiconducting configuration, which is a sequence of alternating single and triple bonds. The anticipated stiffness,strength and thermal conductivity of carbyne exceed any other known material,including diamond,graphene and carbon nanotubes, making carbyne a promising candidate for composite materials.[4,5]Metallic carbyne is expected to feature ballistic thermal transport due to long phonon lifetimes and acoustic phonon mean free paths on the µm scale.[6]All of the above properties arise directly from the structural properties of carbyne.
Due to the chemical instability of carbyne, its synthesis in bulk phases has not been achieved to date. A long standing study even postulated that it is impossible to synthesize carbyne.[7]Reports of carbyne were often questioned until the preparation of polyynes(short linear chains with end-capping groups) suggested that a route towards carbyne is feasible.[8]Finite linear carbon chains are expected to exhibit the properties of carbyne if they consist of 100 or more atoms,while the properties of shorter chains are dominated by length effects.With the longest polyynes being comprised of 44 to 48 atoms,the bottom up synthesis of carbyne remains a challenge.[9,10]These obstacles were overcome by synthesizing linear carbon chains inside multiwalled carbon nanotubes starting in 2003.[11–18]The empty space inside the core of the nanotube serves as a nanoreactor that allows for the growth of long chains. The nanotube walls stabilize the chains by protecting them from interactions with the environment. Transmission electron microscopy revealed that the carbyne chains reside at the centre of the inner carbon nanotube as schematically depicted in Fig.1(b).
At this point it is instructive to clarify the terminology that is used for linear chains of carbon inside carbon nanotubes,which we adapt from a recent review on linear carbon chains in general.[19]LCCs@CNTs describes linear carbon chains encapsulated in carbon nanotubes. They are assumed to have a polyynic structure,making them semiconducting,unless specified otherwise. The term linear carbon chain applies both to chains with≤50 atoms,for which length effects dominate the chain’s properties,and for chains with a greater but unknown number of carbon atoms.The terms carbyne,confined carbyne or confined carbyne chains,on the other hand,refer to infinite carbon chains and chains that are long enough such that their properties are not affected by their length.[3,20–22]The latter are the primary focus of this review.
Raman spectroscopy is the primary spectroscopic method to study confined carbyne.[3,14,15,20]While IR spectroscopy and absorption spectroscopy are used to characterise short linear carbon chains and polyynes without a nanotube host,[1,5]nanotubes interfere with these two techniques which renders them unsuitable to study confined carbyne. Similarly, photoluminescence spectroscopy (PL) from confined carbyne is largely quenched by the nanotube host,[23]in particular if isolated confined carbyne chains placed on a substrate are investigated. Until very recently, the study of short linear carbon chains and carbyne confined in carbon nanotubes was primarily conducted on bulk quantities whereby a large number of carbyne chains and nanotubes were probed simultaneously.An overview of these studies is provided in recent review,see Ref.[19]. While these studies provided a broad understanding of the physics of carbyne,several important aspects such as the exact interplay of confined carbyne with their nanotube host are not accessible by bulk measurement. Instead, measurements of isolated carbyne chains and its nanotube host are required,measurement of this sort has been reported in 2016.[14]The importance of single confined carbyne chain studies and the fact that Raman spectroscopy is the primary spectroscopic method to perform such studies is the core motivation of this review.
In this review we discuss recent experimental studies of isolated confined carbyne chains by Raman spectroscopy. We lay out how these measurements help us to understand carbyne confined inside carbon nanotubes as the ultimate 1D carbon allotrope and as a combined material system. We will present a number of open questions that need to be addressed by single chain measurements to further our understanding of confined carbyne,and lay out open challenges in the field.
The rest of this review is arranged as follows.In Section 2 we will review the Raman basics of confined carbyne and discuss how single carbyne chain measurements have contributed to our understanding of this material system and its nanotube host(Section 3). Section 4 provides an overview of the open questions in the research of confined carbyne from tailoring its properties by its nanotube environment or external tuning by doping, strain or pressure. Where possible, we illustrate how these open questions can be addressed by single carbyne chain measurements and finish with conclusions and outlook in Section 5.
2. Raman basics of confined carbyne
Raman spectroscopy, which probes the inelastic scattering of light by molecular vibrations or phonons, is a powerful characterization technique to study confined carbyne. It is particularly useful to investigate all-carbon materials, since it directly reveals the frequency of the carbon-carbon vibrations,which in turn carry information on the material’s structural,mechanical,electronic and optical properties.[24–26]
Polyynic or semiconducting carbyne has one Ramanactive vibration termed C-mode orR-mode,[27,28]a longitudinal optical phonon from theΓ-point that corresponds to the in-phase stretching of the triple bonds along the chain axis as indicated by the red arrows in Fig.1(a). Cumulenic or metallic carbyne, on the other hand, is Raman inactive. The frequencyωCof the C-mode of semiconducting carbyne is directly proportional to the bond-length alteration(BLA)which describes the change in bond length between the cumulenic and polyynic configuration as indicated by the dashed lines in Fig. 1(a). Typical calculated values for the bond length alteration are of the order of 10 pm with a cumulenic bond length of 128 pm.[14]
One of the special properties of carbyne and linear carbon chains in general is that the bond-length alteration of semiconducting carbyne determines both the bandgap and Cmode frequency. This is generally not found in other lowdimensional materials. On a practical level, this means that the C-mode frequency as seen in a Raman spectrum directly reports the band gapEGenergy of the corresponding carbyne chain. Evidence of this behavior was already found in early wavelength dependent Raman studies of bulk carbon nanotube sample with encapsulated carbyne chains[17]which we show in Fig. 1(c). The C-mode peaks associated with carbyne occur around 1800 cm−1, well separated from the well-known G-modes of the encasing carbon nanotubes.[29]Depending on the excitation energy, different sets of distinct C-mode frequenciesωCand relative intensities appear in the Raman spectra. A systematic study mapped out the fundamental band gap of confined carbyne for differentωCthrough resonance Raman profiles, and showed that the bandgap and the C-mode frequency are connected as shown in Fig. 1(c).[15]This direct link between band gap and C-mode makes Raman spectroscopy a particularly powerful tool to characterize confined carbyne. Resonant Raman studies on carbyne chains further showed that carbyne chains are resonant Raman scatterers,which means that the energy of the laser used for excitation must match the fundamental band gap of carbyne or higher electronic or vibronic transitions to obtain an appreciable Raman response.[15,17,30]
All Raman studies on bulk quantities of confined carbyne report a set of distinct C-mode frequencies and hence different bond-length alterations. It remained unclear, however,whether this variation arose from short chains with different
lengths,the interaction with different host nanotube chiralities(diameter, metallicity), or a combination of both effects. The main reason for this ambiguity was the fact that bulk measurements—in contrast to single chain measurements —cannot establish a correspondence between the chirality of the host nanotube and the encapsulated carbyne. This is only one example of a long-standing open question in the research on carbyne confined inside carbon nanotubes which was eventually addressed by single carbyne chain Raman spectroscopy, see Section 3.
3. Single carbyne chain Raman spectroscopy
In this section we will discuss the growing number of single confined carbyne chain Raman studies and how they contributed to our understanding of the combined chain-nanotube material system. A major leap forward in confined carbyne research was achieved in 2016 when isolated carbyne chains inside double-walled carbon nanotubes (DWCNTs)were measured and imaged by tip-enhanced Raman spectroscopy (TERS) for the first time by Shiet al.as shown in Figs. 2(a) and 2(b). The Raman spectrum in Fig. 2(a) shows a singular C-mode peak of Lorentzian shape with one central frequency which we regard as typical for an isolated carbyne chain. The corresponding TERS image in Fig.2(b),in which the integrated C-mode intensity is displayed,suggests a chain length of up to 800 nm.
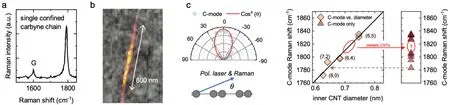
Fig. 2. Single confined carbyne chain Raman characteristics (a) Raman spectrum of a single confined carbyne chain. (d) Overlayed AFM and C-mode tip-enhanced Raman images of a single confined carbyne chain whose spectrum is shown in panel (a). (c) Polarization dependent C-mode intensity of a single confined carbyne chain(top)as a function of the angle θ between the chain axis the polarization of both the excitation laser and the Raman scattering.From Ref. [20].(d) Correlation between diameter of encasing inner carbon nanotube of DWCNTs and the C-mode frequency of the encapsulated carbyne chain. The C for DWCNTs. (e)Collection of single chain Raman spectra. The red circle indicates the general absence of confined carbyne with 1810 cm−1≤ωC ≤1825 cm−1 and how it corresponds to metallic carbon nanotubes in the C-mode versus diameter plot in panel (d). The grey arrow indicates the estimated diameter of the encasing nanotube from ωC. Panels(a)and(b)are adapted from Ref.[14]. Panels(d)and(e)are adapted from Ref.[3].
Polarization dependent Raman measurements on a single carbyne chain are shown in Fig. 2(c), whereby the polarization of the excitation laser and the Raman scattered light were kept parallel and recorded as a function of the angleθrelative to the chain axis.[20]The C-mode intensity scales∝cos4θ, which is the typical behaviour of 1D nano-systems and is called antenna-effect.[31]The Raman intensity arises exclusively from the projection of the excitation and Raman scattered light onto the chain axis,and is suppressed for all other polarizations. A carbyne chain’s C-mode Raman intensity is hence maximized if the wavelength and the polarization of the excitation laser matches the chain’s fundamental bandgap and orientation. Tschannenet al.performed such measurements and additionally recorded the length of the corresponding carbyne chains by TERS.[21]This allowed the authors to quantify the Raman scattering cross section of confined carbyne to a value of 10−22cm2·sr−1per atom — exceeding that of any other known material by a factor of 100 or more — making confined carbyne the strongest Raman scatterer ever reported.
The strong intrinsic Raman response of confined carbyne upon resonant excitation naturally applies to anti-Stokes Raman scattering as well. A recent study reported the observation of the C-mode’s anti-Stokes Raman signal of single carbyne chains, and showed how it can used to read out the local temperature, making confined carbyne a candidate for nanoscale temperature monitoring.[22]Using the strong local field-enhancement provided by tailored Au-pyramids as TERS-tips in combination with carbyne’s intense Raman scattering cross section,Tschannenet al.found indications of correlated Stokes–anti-Stokes emission from carbyne.[32]In this process, a C-mode phonon generated through a Stokes Raman scattering event is instantaneously annihilated in an anti-Stokes event. The two emitted photons are hence correlated through the phonon,which may find use as a read-write protocol for solid state quantum memory.[32]For further insight into TERS on single carbyne chains,we refer to a recent review on the topic.[33]
On top of investigating confined carbyne, Raman spectroscopy is a well-established technique to characterize carbon nanotubes,i.e.to identify their metallicity, diameter, and chirality.[26]Raman studies that probe both the confined carbyne chain and the nanotube host can therefore provide insight into their interaction. A recent seminal study on individual confined carbyne chains and their double-walled nanotube host showed that(i)the chains are long enough such that their properties are not affected by their length — which justifies calling them confined carbyne — and that (ii) the nanotube chirality determines the properties (BLA,ωC,EG) of the encapsulated carbyne chains.[3]Experimentally,it was observed that the radial breathing modes (RBM) of the inner carbon nanotubes, a breathing-like vibration of the nanotube that reveals its chirality, are in a fixed relation with the C-mode of the encapsulated chain. In other words, carbyne chains that reside in the same type of carbon nanotubes have the same Cmode frequency and hence the same properties. On top, the C-mode shows a strong dependence on the host nanotube diameter, Fig. 2(d), indicating that van der Waals forces dominate the interaction. These interactions increase with decreasing CNT diameter, which corresponds to a decreasing BLA as the energetically favourable configuration of the confined carbyne chain. The observation of fixed nanotube/C-mode pairings further suggests that the distinct set of C-mode frequencies between 1760 cm−1and 1880 cm−1observed in the Raman spectra of confined carbyne in bulk as well as single chain measurements across literature reflects the finite number of carbon nanotube chiralities with diameters that allow for the formation of confined carbyne.
Studying confined carbyne chains inside double-walled carbon nanotubes requires a careful analysis of the nanotube’s Raman signatures since the outer nanotube may shift the RBM frequency of the inner tube by to 35 cm−1, which needs to be taken into account when assigning chiralities.[3,34]Using single-walled carbon nanotubes that host carbyne chains will greatly simplify this assignment and first results towards this type of sample have been reported recently.[35]Furthermore,single-walled host nanotubes will facilitate studying charge transfer processes(see Section 4.2.1)and may help shine light on the open question of whether metallic nanotubes host confined carbyne,see detailed discussion in Section 4.1.
It is important to note that not all studies have adopted the interpretation that the carbon nanotube host determines the properties of the encapsulated carbyne chains. Recent pressure and temperature studies on the single/few chain level(encapsulated inside double-and multiwalled carbon nanotubes),for instance,assign different C-mode frequencies to linear carbon chains of different lengths between 40 and 85 atoms.[36,37]This interpretation,however,appears very unlikely when challenged by the correlation between nanotube chirality and Cmode frequency presented in Ref. [3], since it would require chains of a certain length to be present only in one specific nanotube chirality.
4. Open questions
In this section we will highlight some of the open questions in confined carbyne research and how they may be addressed by single carbyne chain measurements. First we will focus on tailoring the interaction of confined carbyne and its nanotube host, growth yields and the transition regime between short linear carbon chains and confined carbyne (Section 4.1). In Section 4.2 we review routes to actively tune carbyne by doping, strain or pressure and discuss experimental works on the topics where controversy exists regarding their interpretation. The discussion of open questions closes with experimentally observed and predicted effects of temperature on the properties of confined carbyne in Section 4.3.
4.1. Tailoring confined carbyne
We have already established in Section 2 that a nanotube of a specific chirality hosts confined carbyne with a specific Cmode/BLA.The choice of nanotube host can hence be viewed as a way of obtaining confined carbyne with tailored properties. Several aspects of nanotube–carbyne interaction, however, are not understood to date. One example is that single chain[3,20]and bulk studies do not report C-modes with∼1812 cm−1to∼1825 cm−1, which corresponds to a gap in the linear relation between diameter and phonon frequency,Fig. 2(e). When inspecting the linear relation between host nanotube diameter and C-mode frequency,depicted as the diagonal line in Fig.2(d),one finds that only metallic nanotube chiralities occupy the corresponding diameter range and to our knowledge,no confined carbyne chain has been correlated with a metallic host nanotube by single chain spectroscopy so far.[3]
There are several possible explanations for the fact that confined carbyne chains in metalllic nanotubes do not seem to appear at the expected C-mode frequencies. One explanation would be that metallic CNTs only host also metallic cumulenic carbyne, which is not expected to feature optical phonons[24]and can thus not be detected with Raman spectroscopy. A second possibility would be that confined carbyne chains do not grow inside metallic CNTs at all. However,both of these explanations are not consistent with bulk Raman spectroscopy conducted by Shiet al.in a study which indicated no influence of the metallicity of the host nanotubes on the growth of confined carbyne.[35]A third explanation of these missing C-modes is that C-mode frequencies and hence the BLAs of confined carbyne chains in metallic host nanotubes do not follow the same linear frequency dependence as confined carbyne chains in semiconducting hosts, and the actual C-mode phonon frequencies are hidden among the many frequencies corresponding to carbyne chains in semiconducting chiralities. A thorough mapping of all carbyne hosting nanotube chiralities and the corresponding C-mode frequencies can thus provide further insight into this open question.
Measurements of single chains also allow the study of the influence of different growth conditions of confined carbyne,and thus are vital for testing improved synthesis methods. So far, control over the properties of confined carbon chains has been limited, but is improving through the selection of specific host CNTs.[35]Once the correspondence between the single carbyne chain and the host nanotubes is fully established through single chain measurements,we will be able to obtain an overview of the total composition of confined carbyne samples with statistical analysis. The determination of the Raman cross section of carbyne by Tschannenet al.through single chain measurements will enable the accurate estimation of filling factors with Raman spectroscopy.[21]Combined with a precise knowledge of which nanotube chiralities are present and their relative abundance, one may even be able to reveal chirality-or diameter-specific growth rates.
Another open question in confined carbyne research are the exact characteristics of the transition from the regime of short chains with length-dependent properties to the host nanotube-dominated regime of confined carbyne. Single confined carbyne chain measurements are crucial here to characterize the properties of suitable carbon chains, most importantly their length, because, as mentioned before, bulk synthesis of homogeneous confined chains with predetermined lengths has not been conducted so far. However, since theoretical ab initio calculations predict this transition to be taking place in a range of 50–100 atoms of length,corresponding to chains 7 nm to 13 nm long,[14]tip-enhanced Raman spectroscopy with a resolution limit around 20 nm is not suitable to identify such LCCs and other experimental techniques have to be considered.
4.2. Active control
Active tunability of carbyne’s physical properties represents a crucial step towards a better understanding of the physics of carbyne and for future applications. In particular,the external tuning of the bandgap of semiconducting polyynic carbyne and control over the phase transition between its semiconducting and its metallic state are keystones for exploiting the anticipated high-precision sensing capabilities[36–38]of carbyne. This level of control is also a prerequisite for implementing a carbyne transistor.[39]Despite the vast number of theoretical works about the expected mechanical and electrical properties of carbyne,[4,40–42]experiments that verify and actively tune these properties are scarce due to the technical challenge of interacting deterministically and nondestructively with carbon chains. Measurements on single confined carbyne chains allow us to induce changes controllably and monitor the resulting change in the physical properties of carbynein-situ. Moreover, the encapsulating nanotube provides a controlled and well understood environment for confined carbyne, which is parametrically modulated for different host nanotube chiralities.
In this section we will first focus on the changes in the physical properties of confined carbyne induced by doping mediated by charge transfer between the confined chains and the encasing nanotubes.Secondly,we will look at the expected changes caused by mechanical deformation of the chains. Finally, we will go through the different interpretations of the experimental data on one of the first approaches to active tuning of carbyne: the study of confined chains under hydrostatic pressure. This example illustrates the complex interplay between electrostatic and mechanical phenomena in the combined chain-nanotube system.
4.2.1. Charge carrier doping
The confinement of carbyne chains inside carbon nanotubes offers an excellent platform to investigate the effects of electron or hole doping on carbyne chains. In confined carbyne, the chain and the host nanotube establish a common Fermi level through charge transfer.[18,41,43,44]The electrostatic interaction between the encasing nanotube and the carbyne chain offers a way to tune the doping level of carbyne by electrically addressing the nanotube, which transfers electrons to the inner chain upon gating. Adding or substracting electrons to and from the carbyne chain in this way induces changes in its atomic structure and realizes active tuning.
The amount of charge transfer (CT) between host carbon nanotube and encapsulated carbyne chain, theoretically modelled for short chains in single-walled carbon nanotubes(SWCNTs)[43]and carbyne confined in double-walled carbon nanotubes (DWCNTs),[41]is expected to be strongly dependent on the chirality of the host carbon nanotube without a systematic dependence oni.e. diameter or chiral angle. Interestingly, also chirality-dependent van der Waals interactions between chain and CNT are found to be exceptionally strong in chain-CNT systems.[41]The dependence of CT and van der Waals interactions on the host nanotube chirality makes it problematic to establish general quantitative descriptions of the effects of doping in confined carbyne. Wankoet al.described how the Raman spectrum of confined carbyne reflects the electrostatic equilibrium between chain and host nanotube,[41]predicting a strong shift in the C-mode frequency of polyynes as a consequence of charge transfer and the strong van der Waals interactions between chain and inner tube. As we will explain in more detail in Section 4.2.3, recent measurements on confined LCCs under hydrostatic pressure are regarded as experimental evidence of a CT-induced downshift of the C-mode prompted by the deformation of the compressed host tube. Above a certain threshold of charge transfer,the BLA is expected to vanish,triggering a transition from semiconducting polyyne to metallic cumulene.[44]
The change of the BLA upon gating could be monitored through Raman spectroscopy on the single carbyne chain level by tracking the corresponding shift in the frequency of the Cmode. For the eventual switch to the metallic state,the Raman signal disappears. However, additional evidence, for example with electrical transport measurements, would be necessary to confirm the transition to the metallic state,as a disappearance of the Raman C-mode could also be due to a shift out of resonance. Electrical transport measurements on confined carbyne nanowires are the subject of recent theoretical investigations[45–47]but nevertheless still very challenging experimentally. Their realization would additionally enable the implementation of nanodevices such as a carbyne-based transistor.
4.2.2. Mechanical tuning
Carbyne is expected to be a particularly attractive material for mechanical tuning. Mechanical deformation of carbyne,for instance bending or twisting of the chains,may lead to changes in carbyne’s fundamental properties, as the linear carbon chains’reduced dimensionality makes their atomic structure more sensitive to mechanical changes than their twodimensional (2D) and three-dimensional (3D) counterparts.Following the tradition of newly discovered forms of carbon,carbyne is predicted to be the strongest of all carbon allotropes and therefore of all materials in nature.[4,42]This record strength should allow to stretch carbyne greatly before breaking it,which in turns tunes its electrical and vibrational properties and enables electro-mechanical,[48]sensing,[36,38,40]and even engineering applications.[49]
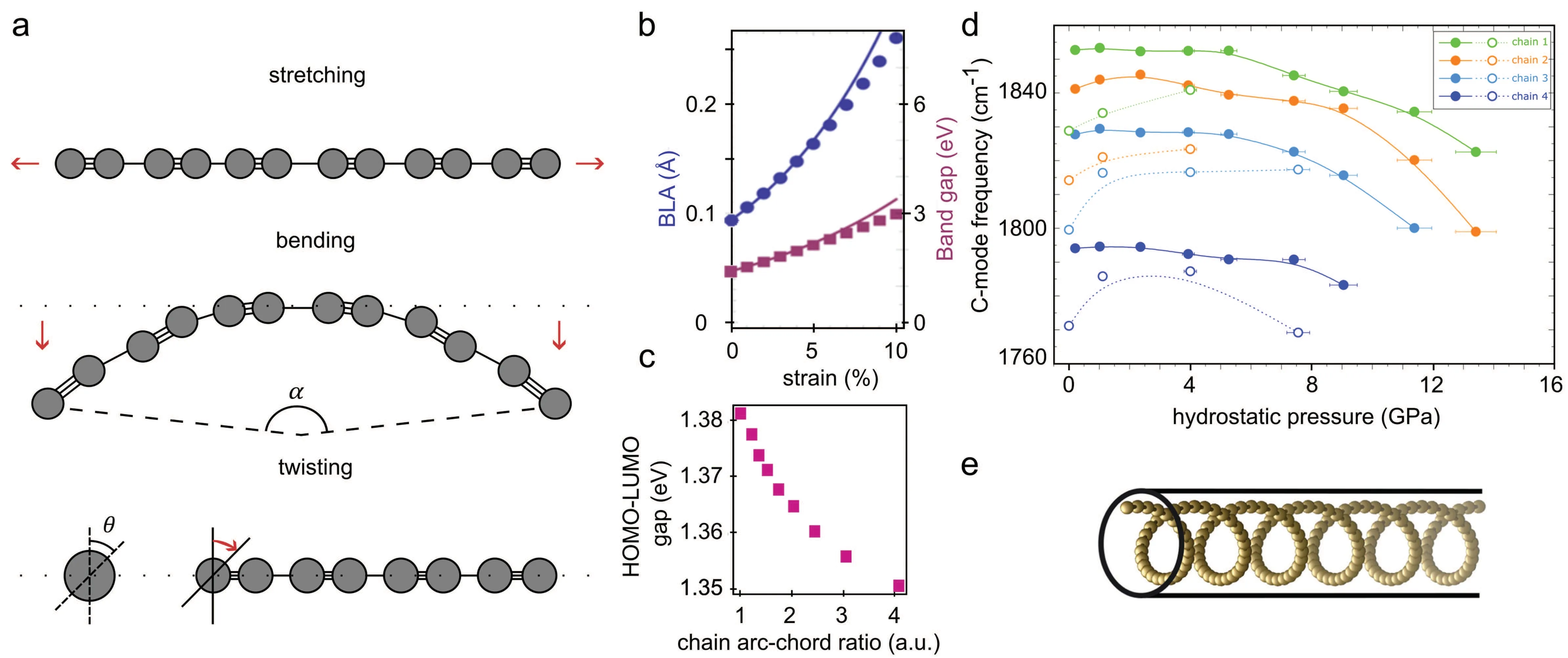
Fig.3. Mechanical tuning of confined carbyne. (a)Schematic depicting the three basic mechanical deformations of a linear carbon chain: tensile straining(stretching/compressing), bending and twisting. (b) Bond length alteration (dots) and bandgap (squares) as a function of increasing tensile strain. Points:DFT calculations. Lines: fitting based on analytical model. Adapted from Ref.[48]. (c)HOMO–LUMO energy gap as a function of increasing bending arcchord ratio. DFT calculation for a 20-atoms polyynic chain. An analogous decrease of the bandgap upon increasing bending is expected for infinite chains.Adapted from Ref.[38]. (d)C-mode frequencies for four MWCNT-encapsulated carbon chains upon increasing(solid dots, compression)and decreasing(empty dots,decompression)hydrostatic pressure. Solid and dotted lines are a guide to the eye. Adapted from Ref.[51]. (e)Depiction of a mechanically stable 3D structure of confined carbyne under compressing pressure suggested by Argaman and Makov.[44] Adapted from Ref.[44]
The characterization of carbyne’s mechanical properties requires first to identify the linear response parameters to decouple the different kinds of deformation and study them separately. The linear response of carbyne to mechanical deformation can be decoupled in three basic contributions:tensile strain or longitudinal elongation/compression,bending and torsion. Figure 3(a) illustrates the three kinds of mechanical distortion. The anticipated exceptional resistance of carbyne to longitudinal deformation would make it the stiffest known material with a calculated tensile stiffness of 1.5·10−7N/m,[50]two times bigger than that of graphene and carbon nanotubes, and a breaking force of 9.3 nN–11.7 nN,also outperforming graphene and diamond.[4]In contrast to its high stiffness against tensile strain, carbyne is a very soft material against bending, enduring deformation to high curvatures without breaking.[4,38,40]Additionally, carbyne’s torsional stiffness,i.e.its resistance against twisting,can be made non-zero in end-capped chains,switching on magnetism in the material.[4]In the following we will focus on the vibrational and electronic changes induced by tensile strain and bending,underlining how Raman spectroscopy can be used to track these changes. We will not discuss torsion as its relevance for CNT-encapsulated carbyne is considered to be limited.
Tensile strain, the stretching or compressing of the carbon chains,has been predicted to increase/reduce the BLA and bandgap of carbyne, respectively, see Fig.3(b). The increase of the bandgap with increasing strain in carbyne is opposite to the usual response of semiconductors to strain.[48,52]Carbyne’s anomalous bandgap behaviour with strain when compared to other semiconductors can be understood when we consider that the bond lengths in polyynic carbon chains increase non-uniformly,intensifing the Peierls distortion,which in turn increases the BLA and the bandgap. Indeed, by amplifying the Peierls distortion strain can balance off the energetic competition between Peierls dimerization and the vibrational energy of the lattice driving the transition from metallic state to semiconductor.[48]Notwithstanding, the technical realization of strain engineering of confined carbyne by directly stretching the encapsulated carbon chains is still unclear, as the stretching of the host CNT does not guarantee the stretching of the chain inside, which may slip to accommodate to an energetically preferred position. On the other hand, it has been recently argued that an infinite chain of carbon atoms is not dynamically stable under compressive strain.[44]Carbon chains under compressive strain would buckle into new nonlinear configurations,as it is described in Section 4.2.3.
Tensile strain on linear chains of carbon atoms is being intensively investigated[4,36,40,42,44]and there are still discrepancies in the proposed theoretical models and the mechanical constants derived from them.[36,44,50,53]For instance, the often cited record-breaking values for the Young modulus of carbyne[4]are controversial,as the cross-sectional area of carbyne is not well-defined.[44,49]The use of tensile stiffness is therefore recommended to quantify carbyne’s outstanding resistance to longitudinal deformation.[50]The fact that several competing models of the mechanics of carbyne are being put forward in the literature highlights the need to generate experimental data to test the predictions of the corresponding models. So far, the main source of experimental data on the mechanical properties of confined carbyne has focused on the compression of encapsulated chains with hydrostatic pressure,[36,51,54]see Section 4.2.3. In these works,carbyne’s Raman active C-mode tracks the structural changes in the chain, while the G-band of carbon nanotubes reports structural changes in the tubes.These experiments have shown the potential of Raman spectroscopy to track mechanically induced changes in chain-carbon nanotube systems.
External tuning of LCCs through bending of the chains promises to be a straight-forward and powerful strategy for tailoring confined carbyne’s properties. Carbyne has been calculated to have a small but nonzero rigidity against bending,comparable to that of typical polymers and double-stranded DNA.[4,40]Carbyne’s high softness is matched by the high softness of the encapsulating CNTs, which are often found naturally bent.[38]It has been calculated[38]and experimentally measured[55]for short polyynic chains that changing the bond angles between atoms by curving the chains increases the BLA while reducing the bangap,[38]see Fig.3(c). This behavior is also expected for carbyne.[44]The reduction of carbyne’s bandgap upon increasing bending could allow to externally drive the transition of a single chain from the semiconducting configuration to a metallic one. Additionally, carbyne’s soft bending degree of freedom suggests its application to develop sensitive acceloremeters,[40]pressure,[36,38]and mass sensors.[36]Bending of confined single chains could be experimentally realized in situ by AFM nanoindentation,while a comprehensive characterization of carbyne’s Raman response would track any changes in the BLA by measuring the spectral shift of the C-mode.
4.2.3. Pressure experiments
A series of works on the change of the Raman signatures of LCCs encapsulated in DW-and MWCNTs under hydrostatic pressure represent the first experimental realization of external tuning of confined carbyne’s electronic and vibrational properties.[16,18,36,51,54]In these experiments, confined carbyne samples are placed inside a diamond anvil cell(DAC)filled with a pressure-transmitting fluid, typically Nujol mineral oil,that transforms the uniaxial pressure from the DAC in uniform hydrostatic pressure. A ruby sensor placed inside the cell allows to calibrate the pressure by tracking the variation of ruby’s luminescence lines. Unlike the RBM, G, and 2D bands of other carbon allotropes, the frequency of carbyne’s C-mode has been extensively reported to decrease upon increasing hydrostatic pressure in encapsulated chains,[36,51,54]see Fig.3(d). The ongoing discussion on the interpretation of confined carbyne’s C-mode softening with compressing pressure illustrates the complex interplay of mechanical and electrostatic effects in the chain-carbon nanotube systems.
The first measurements of the Raman response of encapsulated LLCs were performed by Andradeet al. on bulk samples of MWCNT-encapsulated chains under hydrostatic pressures between 0 GPa–10 GPa. This work would later be extended to higher pressures up to 28 GPa and chains confined in DWCNTs.[18,51]An exponential redshift of the C-mode frequencies of about 30 cm−1under maximum pressures of up to 14 GPa was interpreted as a signature of charge transfer between CNT and chain. Upon increasing hydrostatic pressure the host CNT is deformed, bringing the inner tube and chain closer together and thus increasing their interaction. An increased interaction between host tube and chain results in an increased CT that reduces the BLA and therefore downshifts the C.The deformation of the host DWCNT was backed by an analysis of the evolution of the CNTs G-mode with increasing pressure. CT between chain and nanotube was supported by DFT calculations.
Sharmaet al. proposed an alternative explanation in Ref.[36]for the softening of the C-mode in confined carbyne upon increasing pressure other than charge transfer induced by the deformation of the host inner tube. In Ref.[36]the authors claim to measure isolated LCC@MWCNTs under hydrostatic pressure via resonance Raman scattering,presenting a Raman C-band spectrum consisting of four distinct Lorentzian peaks,in contrast to the criteria suggested in Section 1 for single chains. The four MWCNT-encapsulated chains exhibited reversible C-mode softenings of up to more than 30 cm−1within a range of applied pressures of only 5 GPa, almost a third of the maximum applied pressures in Ref. [18]. The authors argued that within this pressure range no efficient deformation of the inner tube is expected to happen because the inner tube would only be efficient for pressures beyond 9 GPa,[56,57]questioning the role of charge transfer in the C-band redshift.Instead they suggested an alternative force-constant atomistic model that exploited the anharmonicity of the C-C single bond to explain the softening of the C-mode. However, a following work by Argaman und Makov criticized the constant-force assumption in the model in Ref. [36] and backed the charge transfer interpretation with new theoretical evidence.[44]Argaman and Makov found through first-principles calculations that polyynic carbyne is not stable under compression. Instead, the chains would adopt one of the two possible mechanically stable configurations, a 2D sinusoidal or a 3D helical structure,making use of carbyne’s high bending softness(see Section 4.2.2). Such a 3D helical structure is depicted in Fig.3(e). Furthermore, these 2D and 3D structures were formulated in a wave equation formalism, predicting additional metastable excited states of the atomic configurations with an increasing number of nodes. Upon compressing pressure,the encapsulated chain would access the different wave-like excited states, increasing the number of contact points with the CNT and thus promoting charge transfer with the inner tube.This model could be effectively probed by measurements on single chains,where the structural changes leading to 2D and 3D atomic structures would be expected to have their corresponding Raman fingerprint.
4.3. Temperature characteristics
The temperature-dependent characteristics of confined carbyne are an area where single chain Raman spectroscopy can contribute substantially to our knowledge of potential phase transitions and phonon dynamics of confined carbyne.While bulk measurements can be used to derive thermodynamical properties like thermal expansion coefficient and heat capacity,[37]single chain measurements offer further insights through precise tracking of peak width,position and amplitude at different temperatures.
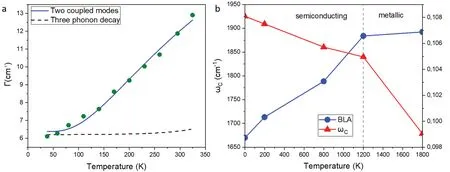
Fig.4. Temperature dynamics of carbyne. (a)Spectral linewidth Γ of bulk Raman measurements of the carbyne C-mode at different temperatures,adapted from Ref.[14]. The dashed line represents a fit with a three phonon decay model, the solid line a fit with a model of coupled LCC and host CNT modes.(b)The ab initio calculations of temperature dependent shifts in C-mode frequency ωC and bond length alteration(BLA),adapted from Ref.[58]. A phase transition from polyynic semiconducting carbyne to polyynic metallic carbyne is expected to occur at a temperature ∼1200 K.
For example,through the spectral linewidthγ,the phonon lifetimeτcan be extracted asτ= ¯h/γ. The two main suggested mechanisms for the evolution of optical phonons in confined carbyne are a three phonon anharmonic decay process in which the optical phonon splits into two acoustic phonons, and coupling between the modes of the LCC and its inner host nanotube,where the temperature dependence of the linewidth can be described as
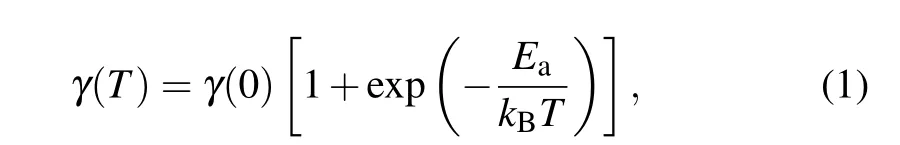
withEaas the activation energy.[14]Research on bulk carbyne appears to support the second theory,as shown in Fig.4(a),[14]however ambiguity remains because it is not clear how many different LCCs contribute to the spectroscopic signal and whether that number changes with temperature due to a potential widening or sharpening of the excitation resonance curve.Additionally, an analysis of the C-mode of single chains by Tschannenet al.revealed a wide range in the linewidth from 8.2 cm−1to 16.7 cm−1, with no apparent correlation to the frequency.[32]Conducting such temperature-dependent analysis on single chains can thus greatly improve our understanding of this dynamic behavior,as these kind of group effects can be eliminated. Due to the strong electron–phonon coupling in carbyne,this also provides insight into the carrier characteristics of carbyne,another area where current research is limited since direct conductivity measurements of carbyne have not been realized so far to our knowledge. As metallic carbyne is expected to feature ballistic thermal transport,carrier dynamics are directly influenced by acoustic phonon lifetimes and their mean free paths,which can be inferred to a good approximation from the corresponding values of optical phonons of semiconducting carbyne like the C-mode.
Another open question are potential temperature-induced phase transitions from polyynic semiconducting carbyne,which is the only form of confined carbyne found so far, to a metallic state. The Peierls theorem,which is applicable to carbyne as a true 1D material, explains the relatively large BLA and bandgap of carbyne,and the 1D structure of carbyne as the origin of its strong electron–phonon coupling. However,other quantum effects like zero-point vibrations and anharmonicity have to be considered and theoretical calculations of carbyne’s electronic states vary massively in their conclusions, speaking to the complexities of the system. Earlier calculations that predicted a stable cumulenic phase at room temperature and a phase transition from cumulenic to polyynic configuration at temperatures of several hundreds of K,[59]could not be confirmed by experiment.
A recent study by Romaninet al.explains this discrepancy as a consequence of strong quantum anharmonicity, as the bond length alteration in carbyne is too big to be considered in the small displacement limit (7%–10% of the total bond length).[58]Thus, the temperature of the transition from polyynic to cumulenic carbyne is predicted to be extremely high and unphysical. However, for confined carbyne surrounded by a high dielectric environment, as it occurs in LCC@CNTs, they predict a transition from semiconducting polyynic carbyne to metallic, but surprisingly also polyynic carbyne atT=1200 K due to the effects of optical vibrations on the gap,with a saturation of the longitudinal optical phonon(C-mode)frequency,as depicted in Fig.4(b).[58]Such a transitions would lift the correspondence of C-mode and bandgap as shown in Fig. 1(d), and is therefore readily detectable by Raman spectroscopy. As bandgap and phonon energies vary massively among LCCs,a shift in excitation resonance would be hard to correctly identify in bulk measurements, while it could be observed conveniently in single chain measurements as an abrupt change in the resonance condition. An additional advantage of using single chain spectroscopy for such an analysis would be the convenient temperature control through laser heating and anti-Stokes Raman thermometry as it was demonstrated by Tschannenet al.[22,32]
5. Conclusion
In conclusion,we have provided an introduction to the basics of investigating single chains of confined carbyne by Raman spectroscopy, and summarized how these types of measurements have contributed to our understanding of confined carbyne. We present avenues to tailor the properties confined carbyne by the choice of its nanotubes host, by charge carrier doping, or by mechanical tuning. A particular focus of this review was to collect and illustrate open questions in the field of confined carbyne research and how some of them can be addressed by Raman spectroscopy of single carbyne chains.The ongoing debate on the interpretation of confined carbyne’s Raman response to hydrostatic pressure particularly illustrates the need for measurements on single confined chains.We hope that this review inspires further research on confined carbyne to address many of the open questions.
Acknowledgement
Johannes M. A. Lechner, Pablo Hern´andez L´opez, and Sebastian Heeg acknowledge funding from the Deutsche Forschungsgemeinschaft(DFG)under the Emmy Noether Initiative(HE 8642/1-1).
- Chinese Physics B的其它文章
- Fault-tolerant finite-time dynamical consensus of double-integrator multi-agent systems with partial agents subject to synchronous self-sensing function failure
- Nano Ag-enhanced photoelectric conversion efficiency in all-inorganic,hole-transporting-layer-free CsPbIBr2 perovskite solar cells
- Low-voltage soft robots based on carbon nanotube/polymer electrothermal composites
- Parkinsonian oscillations and their suppression by closed-loop deep brain stimulation based on fuzzy concept
- Temperature dependence of spin pumping in YIG/NiO(x)/W multilayer
- Interface effect on superlattice quality and optical properties of InAs/GaSb type-II superlattices grown by molecular beam epitaxy

