Serum metabolic profiling of targeted bile acids reveals potentially novel biomarkers for primary biliary cholangitis and autoimmune hepatitis
Zhen-Hua Ma,Xiao-Mei Wang,Rui-Hong Wu, Da-Lin Hao, Li-Chao Sun, Pan Li, Jun-Qi Niu
Abstract
Key Words: Primary biliary cholangitis; Autoimmune hepatitis; Biomarkers; Serum metabolic profiling; Bile acids; Ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry
lNTRODUCTlON
Primary biliary cholangitis (PBC) and autoimmune hepatitis (AIH) are two unexplained immune diseases[1]. Although advanced methods have been presented for diagnosing PBC and AIH, 5%-10% of PBC patients have anti-mitochondrial antibody-negative, and missed diagnosis or misdiagnosis mainly occurs in clinical practice[2]. For some patients with anti-mitochondrial antibody-positive, rather than significant changes in hepatic histology and function, long-term follow-up revealed that these patients eventually developed to PBC. Thus, early diagnosis of these patients is a clinical challenge. Clinical manifestations of AIH may have similarities to other autoimmune liver diseases, such as drug-induced hepatitis, alcoholic liver disease, inherited metabolic disorders, and hepatitis C virus infection, such as regardless of the cause of liver disease, patients may present with fatigue, abdominal distention, skin and sclera yellow staining, laboratory test show liver dysfunction. Because of the complexity and difficulty of diagnosing, leading to the delayed diagnosis of several AIH patients. Liver biopsy remains the golden standard for the diagnosis of autoimmune liver diseases, while it is an invasive, painful, and costly method that is associated with the possibility of sampling error and variability in interpretation.Therefore, identification of novel and accurate noninvasive biomarkers for the diagnosis and assessment of severity is of great importance.
As one of the emerging ‘omics’ platforms, metabolomics enables the qualitative and quantitative analyses of metabolites in complex biological samples[3]. As products of cellular adjustment processes,metabolites are regarded as the ultimate readouts that reflect genetic or environmental changes in biological systems[4,5] High-throughput metabolic profiling has been successfully used for the identification of novel diagnostic molecules and disease-related pathways, as well as development of new therapeutic targets for some diseases (e.g., cancer, non-alcoholic fatty liver disease, non-alcoholic steatohepatitis, and PBC)[6-10]. Thus, it is essential to identify specific metabolomic markers, and to establish a diagnostic model for AIH or PBC.
In the present study, we aimed to identify serum biomarkers for the differential diagnosis of PBC and AIH using ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry(UPLC-QTOF-MS). UPLC-QTOF-MS is a newly developed technique that provides rapid and efficient access to detailed information pertaining to the nature of specific components within complex multicomponent mixtures. Compared with traditional high-performance liquid chromatography(HPLC), UPLC possesses the advantages of ultra-high resolution, high-speed scanning, and high sensitivity. Furthermore, bile acids (BAs) are crucial for the diagnosis, follow-up, and prognosis of liver and intestinal disorders, as well as diseases affecting BA metabolism. We applied a targeted metabolomic approach to quantify and compare 15 BA metabolites in PBC/AIH patients with those in healthy controls (HCs). The findings of the present study may reveal potentially novel biomarkers for the diagnosis of PBC and AIH. This study also aimed to compare metabolic profiles between PBC/AIH patients and HCs.
MATERlALS AND METHODS
Patients and study design
A total of 54 PBC and 26 AIH patients who were admitted to the First Hospital of Jilin University(Changchun, China) between May 2009 and November 2013 were respectively recruited in the present study. The study protocol was carefully reviewed and approved by the Institutional Review Board of The First Hospital of Jilin University. All the eligible patients and HCs signed the written informed consent form prior to enrollment. Patients with AIH were diagnosed according to the revised criteria presented by the International Autoimmune Hepatitis Group in 1999[11]. Patients with PBC were diagnosed according to the criteria released by the American Association for the Study of Liver Diseases[12]. Patients taking medication or supplements, or those with gallstones or other factors that might cause cholestatic liver diseases were excluded. In both groups, patients with primary sclerosing cholangitis (PSC), overlap syndromes (e.g., PBC and AIH or AIH and PSC), hepatitis virus infection,human immunodeficiency virus co-infection, hepatocellular carcinoma, or diabetes were excluded. In total, 30 HCs who were admitted to our hospital for physical check-ups were enrolled. These HCs exhibited normal liver functions and had no evidence of disease. No statistically significant differences were found in age and gender among the PBC, AIH, and control groups (Table 1,P> 0.05).
Blood samples at the fasting state were collected from the eligible PBC patients, AIH patients, and HCs, in which 1 mL of serum was collected and stored at -80 °C for subsequent metabolic profiling.Participants’ baseline characteristics are summarized in Table 1.
Reagents
HPLC-grade acetonitrile was purchase from Merck Inc. (Kenilworth, NJ, United States). HPLC-grade formic acid was obtained from Sigma-Aldrich (St. Louis, MO, United States). These two reagents were used for the preparation of mobile phases in HPLC. Milli-Q water was used, and obtained by filtering distilled water through a Milli-Q system (Millipore, Bedford, MA, United States). The chemical standards for the validation of molecular structure were obtained from Sigma-Aldrich.
Sample preparation and serum metabolic profiling
In the present study, 100 μL of each serum sample was mixed with 400 μL of cold acetonitrile for protein precipitation, followed by centrifugation at 14000 g for 10 min at 4 °C. Then, 400 μL of the supernatant was subsequently collected and lyophilized, and the residue was resolved in 100 μL of 20% acetonitrile.Equal aliquot of each serum sample was pooled together and mixed thoroughly by vortex for 1 min,which was used as the quality control (QC) sample. A QC sample was prepared after preparation of 10 real samples, and QC samples served to assess the repeatability of sample pretreatment and to monitor the stability of the UPLC-QTOF-MS system at the sequence analysis.
The UPLC-QTOF-MS approach was employed to perform serum metabolic profiling of samples obtained from PBC patients, AIH patients, and HCs, as previously described[13]. In brief, 5 μL of the reconstituted solution was carefully injected into the ACQUITY-UPLC system (Waters Corp., Milford,MA, United States) for separation using chromatography. Then, MS signals were acquiredviathe QTOF-MS system (Micromass, Manchester, United Kingdom), which was equipped with an electrospray source operating in both positive and negative ion modes. During the acquisition of MS signals, the m/z scan was set to a range of 100-1000[14]. Then, the AC18 column (2.1 mm × 100 mm, 1.7 μm), which was purchased from Waters Corp., was used for the separation of small molecular compounds at an elution speed of 0.35 mL/min. The gradient was set to 95% formic acid (0.1%, V/V),and maintained for 1 min. Subsequently, elution strength linearly increased to 100% acetonitrile for 22 min, and was kept for 3 min. The total duration was 30 min, which included equilibration for 1 min.
Analysis of BAs
All samples prepared with GCA-d5 as the internal standard and blood samples were resolved in 100 μLof 25% ACN aqueous solution. The LC-MS parameters were as follows: 20 μL of the reconstituted solution was carefully injected into an ACQUITY UPLC C8 column with a particle size of 1.7 μm(Waters Corp.), and the SRM signals were obtained using an Agilent 6460 Triple Quadruple MS system(Agilent Technologies, Inc., Chicago, IL, United States), which was equipped with an electrospray source operating in the negative ion mode. The column was eluted with 10 mmol/L NH4HCO3(solution A) and acetonitrile (solution B) in a linear gradient, in which the initial gradient was set to 75% solution A. Subsequently, after 9.0 min of elution, the strength was linearly elevated to 90% solution B, which lasted for 4 min. Then, this was returned to the initial gradient after 13.5 min of elution. Along with an equilibration of 1.5 min, the total running time was approximately 15 min. The following MS parameters were set in this study: Gas flow rate, 8 L/min; gas temperature, 350 °C; sheath gas temperature, 400 °C;nebulizer gas pressure, 40 psi; capillary voltage, 3500 V; sheath gas flow rate, 8 L/min; nozzle voltage,400 V. The precursor and product ion pairs were acquired as follows: Cholic acid (CA) (407.5→407.5),glycocholic acid (GCA) (464.2→74.1), taurocholic acid (TCA) (514.2→80.1), ursodeoxycholic acid(UDCA) (391.4→391.4), glycoursodeoxycholic acid (GUDCA) (448.3→74.1), tauroursodeoxycholic acid(TUDCA) (498.3→80.1), chenodeoxycholic acid (CDCA) (391.4→391.4), glycochenodeoxycholic acid(GCDCA) (448.3→74.1), tauroursodeoxycholic acid (TCDCA) (498.2→80.1), glycochenodeoxycholic sulfate (GCDCS) (528.3→448.3), deoxycholic acid (DCA) (391.2→391.2), glycodeoxycholic acid (GDCA)(448.2→74.1), taurodeoxycholic acid (TDCA) (498.3→80.2), lithocholic acid (LCA) (375.3→375.3),taurolithocholic acid (TLCA) (482.1→80.1), and GCA-d5 (469.2→74.1).
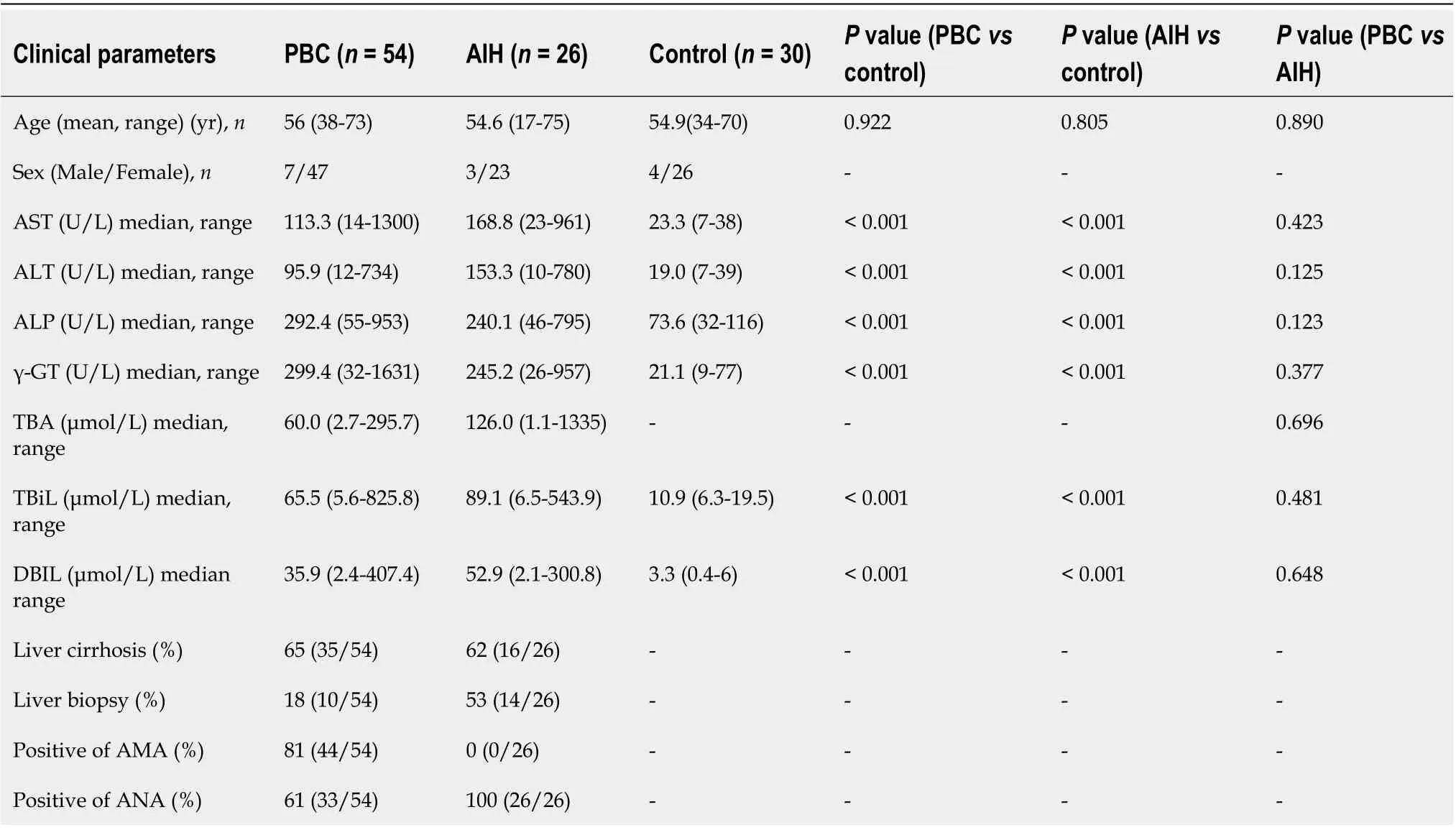
Table 1 Characteristics of enrolled population in the metabolic profiling study
Bioinformatics and statistical analyses
The raw data were imported into Databridge (MassHunter Quantitative Analysis software; Agilent Technologies, Inc.), followed by the peak extraction and alignment on the obtained NetCDF files using XCMS 18.0 software. The alignment parameters were set as follows: The retention time window was 7,the full width at half maximum was 14, and the remaining parameters were set as default. Subsequently, the peaks with the paired m/z, as well as their corresponding peak intensities and retention time were exported into the Excel software. Prior to univariate and multivariate logistic regression analyses, each peak area was initially normalized to the total peak area. In the multivariate logistic regression analysis, the principal component analysis (PCA) in combination with the partial least squares-discriminant analysis (PLS-DA) was conducted by SIMCA-P 11.0 software (Umetrics AB, Umea,Sweden) using the prepared data. After scaling for PCA to unit variance, the data provided an overview of the repeatability of the QC samples. Additionally, the data were Pareto scaled for PLS-DA to assess the performance of the classification models, and to identify variables for the corresponding model.
In the univariate analysis, data were statistically analyzed by SPSS 18.0 software (IBM, Armonk, NY,United States). The biochemical data and the concentrations of BAs were log-transformed to approximately normalize their distributions.P< 0.05 was considered statistically significant. Nonparametric statistical analysis was conducted using GraphPad Prism 5.0 software (GraphPad Software Inc., San Diego, CA, United States) for making comparison between two groups.
RESULTS
Patients’ baseline clinical characteristics
Patients’ baseline clinical characteristics are summarized in Table 1. Previous epidemiological studies have demonstrated that women were more frequently affected by PBC and AIH than men. Consistently,the incidence rates of PBC and AIH were higher in women than in men in our study. Furthermore, to avoid the influences of drugs on the metabolomics analysis, no patient had received any treatment,including traditional Chinese medicine. The mean age of patients with PBC and AIH, and HCs was 56(range, 38-73), 54.6 (range, 17-75), and 54.9 (range, 30-76) years old, respectively. There were no significant differences in age, parity, and gender among patients with PBC and AIH, and HCs (P> 0.05).Besides, 10 cases from the PBC group and 14 cases from the AIH group were newly diagnosed by biopsy. Other cases from the PBC group were diagnosed by M2-positive, and other cases from the AIH group were diagnosed by pathological scores (> 12).
There were 26 cases of Child-Pugh class A, 19 cases of Child-Pugh class B, and 9 cases of Child-Pugh class C in PBC patients. There were 17 Child-Pugh grade A and 9 Child-Pugh grade B patients with AIH. The levels of globulin, transaminases, and specific autoantibodies in the sera are presented in Table 2.
Serum metabolic profiling
A total of 110 serum samples obtained from 54 patients with PBC, 26 patients with AIH, and 30 HCs were analyzed using UPLC-QTOF-MS in both positive and negative ion modes. As shown in Supplementary Figures 1 and 2, a typical base peak chromatogram was detected by MS in positive and negative ion modes, respectively. After the peaks were aligned, 1133 peaks of positive ions and 963 peaks of negative ions were identified using MassLynx and the same acquisition method. The data were transformed into SIMCA-P11 software for PCA. Plots of the PCA scores in positive and negative ion modes are illustrated in Figure 1A, Figure 2A, Figure 3A, Figure 4A, Figure 5A and Figure 6A. Distinct clustering was observed between PBC patients and HCs, and between AIH patients and HCs. No distinct clustering was found between PBC patients and AIH patients. The QC samples were tightly clustered (Figure 1A, Figure 2A, Figure 3A, Figure 4A, Figure 5A and Figure 6A), ensuring the repeatability of the information[13].
Identification of serum metabolites specific to PBC and AIH
To find out the differentially expressed metabolites,P-values (P< 0.05) in the t-test were combined with variable importance in the projection (VIP) values in the PLS-DA model. The PLS-DA score charts are shown in Figure 1B, Figure 2B, Figure 3B, Figure 4B, Figure 5B and Figure 6B. We also conduct sorting verification on the model to check whether the model is "over-fitting". The results are shown in Figure 1C, Figure 2C, Figure 3C, Figure 4C, Figure 5C and Figure 6C. As can be seen from the sorting test figure, Figure 1C, Figure 2C, Figure 5C and Figure 6C, there is no "over-fitting" in these models.Figure 3C and Figure 4C show that the two models are "overfitted". We only established the discriminant analysis model of multivariate analysis between PBC/Control and AIH/Control, but failed to establish the discriminant analysis model of PBC/AIH. The PBC and AIH samples of the two groups overlaps on the PCA score plot (as shown in Figure 3A and Figure 6A), and the supervised PLS-DA (as shown in Figure 3B and Figure 6B) still failed to distinguish them significantly, indicating that there was little difference in metabolic profile between the two groups of different autoimmune liver diseases.
The METLIN metabolomics database (http://metlin.scripps.edu/) was used to facilitate metabolite annotation through MS analysis. The data of differentially expressed are presented in Tables 3 and 4.Fold-change (FC) was used to indicate changes in potential PBC- and AIH-specific biomarkers, and the chosen FC values were > 2 and < 0.5.
Identification of serum metabolites specific to PBC
As presented in Table 3 the levels of 17 of 26 potential biomarkers identified were elevated in the serum samples of patients with PBC, while the levels of 9 of these 26 potential biomarkers were reduced in the serum samples of patients with PBC compared with those in HCs. Among these biomarkers, the levels of TDCA, GUDCA, tetracosahexaenoic acid, bilirubin, sphinganine, phytosphingosine, L-phenylalanine,L-proline, TCA, LysoPC [18:3 (6Z, 9Z, 12Z)], TUDCA, GCA, LysoPE [0:0/18:4 (6Z, 9Z, 12Z, 15Z)],LysoPE [20:3 (11Z, 14Z, 17Z)/0:0], L-urobilinogen, L-urobilin, and DCA significantly increased in patients with PBC compared with those in HCs (P< 0.5). The levels of 12-ketodeoxycholic acid, α-ketoisovaleric acid, pyroglutamic acid, lactic acid, hypoxanthine, LysoPE [0:0/20:2 (11Z, 14Z)],ketoleucine, LysoPE [0:0/22:4 (7Z, 10Z, 13Z, 16Z)], and MG [0:0/18:4 (6Z, 9Z, 12Z, 15Z)/0:0] in patients with PBC were significantly reduced compared with those in HCs.
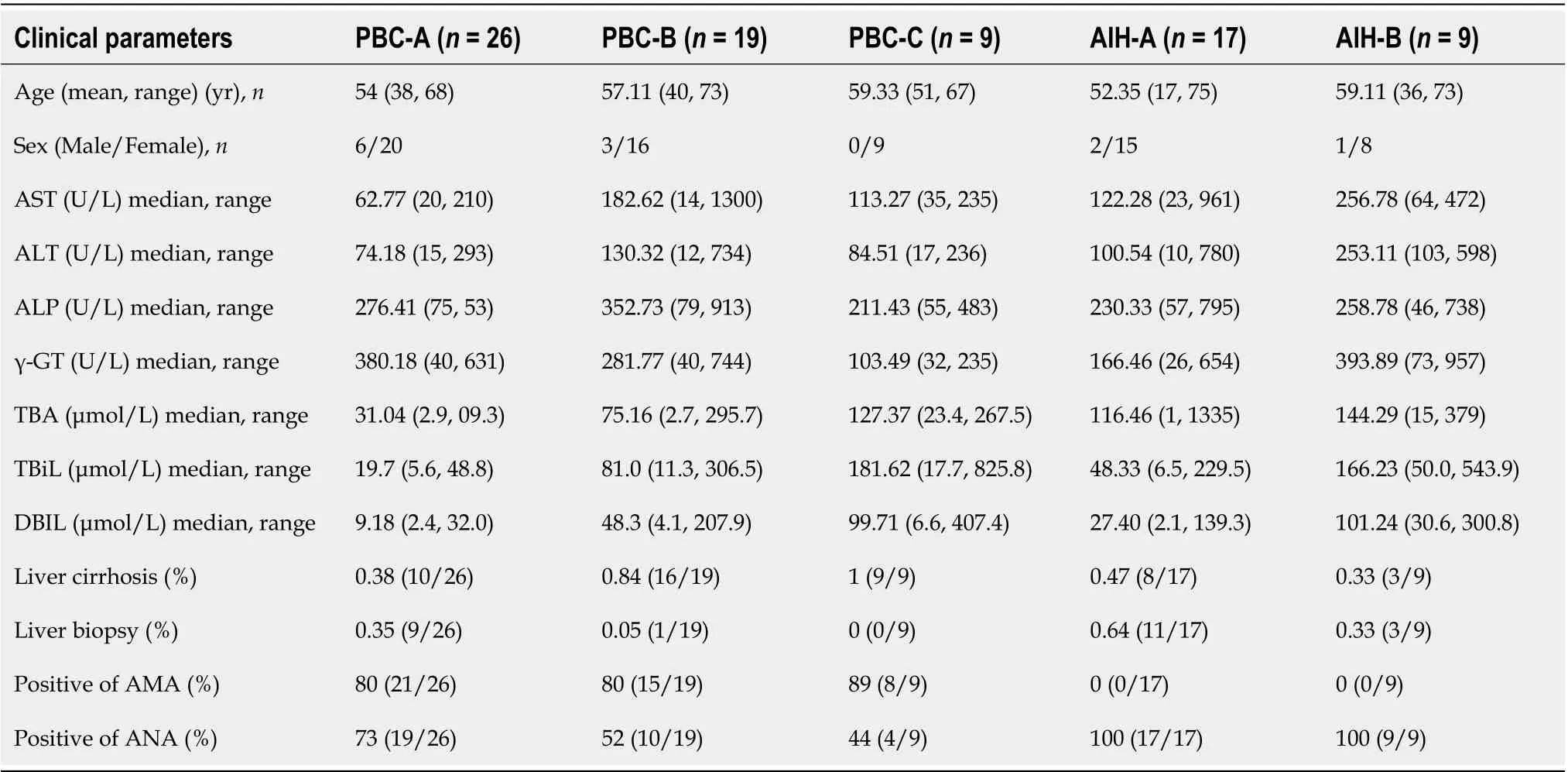
Table 2 Characteristics of classified enrolled population according to Child-Pugh in the metabolic profiling study
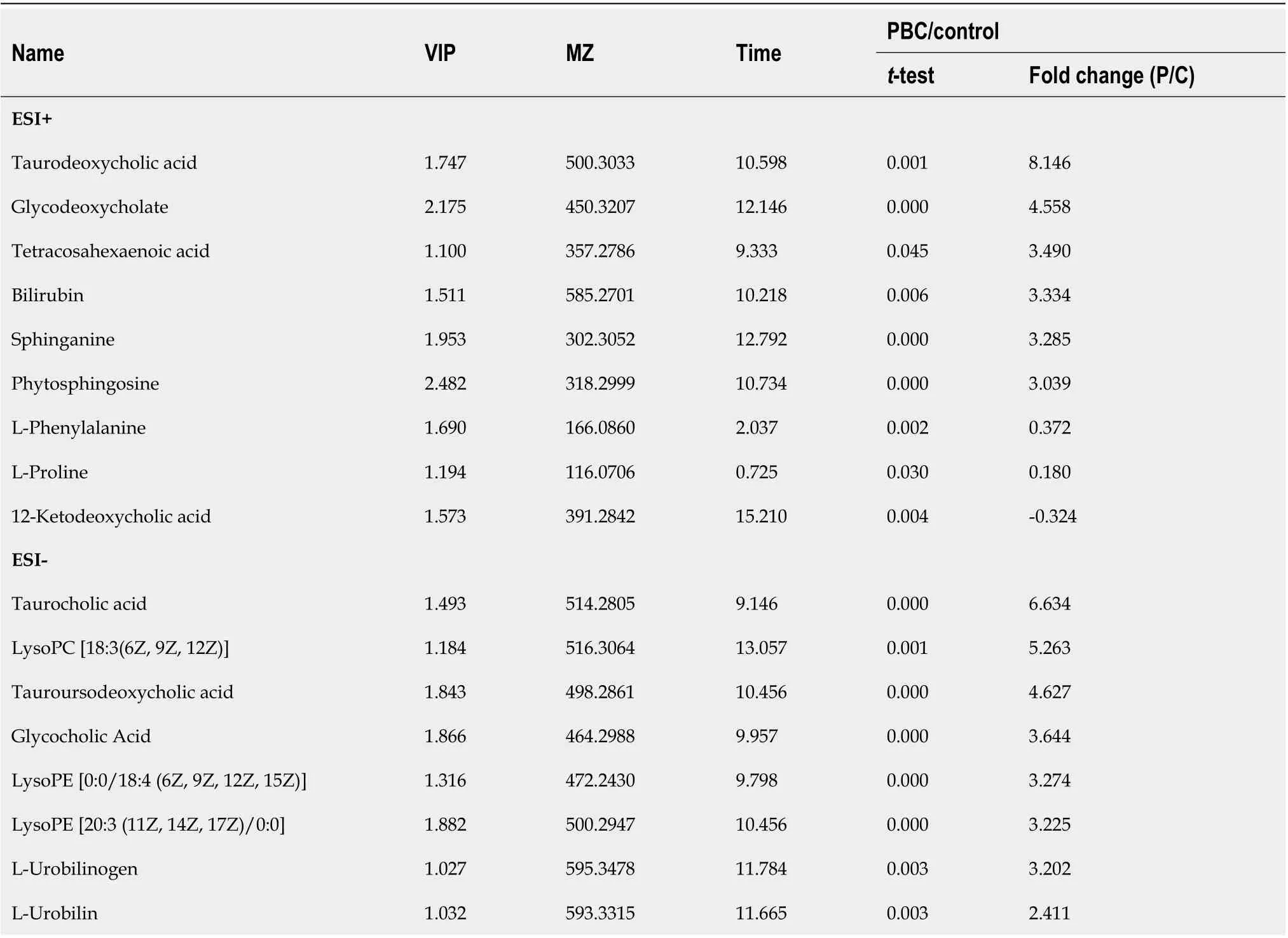
Table 3 Potential serum biomarkers for primary biliary cirrhosis compared to healthy control in positive and negative ions model

Deoxycholic acid 1.032 391.2833 11.641 0.003 2.121 α-ketoisovaleric acid 1.040 115.0399 1.831 0.003-0.348 Pyroglutamic acid 1.252 128.0350 0.931 0.000-0.392 Lactic acid 1.548 89.0242 0.938 0.000-0.402 Hypoxanthine 1.384 135.0308 0.886 0.000-0.431 LysoPE [0:0/20:2 (11Z, 14Z)]1.335 504.3072 14.333 0.000-0.453 Ketoleucine 1.392 129.0555 4.110 0.000-0.486 LysoPE [0:0/22:4 (7Z, 10Z, 13Z, 16Z)]1.333 528.2850 13.617 0.000-0.544 MG [0:0/18:4 (6Z, 9Z, 12Z, 15Z)/0:0]2.060 349.2373 8.969 0.000-2.181 PBC: Primary biliary cirrhosis; VIP: Variable importance in the projection.
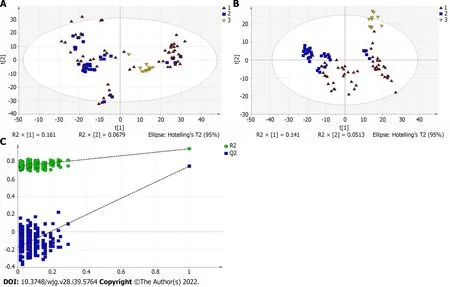
Figure 1 Multivariate statistical analysis on serum profiling data in positive ions between primary biliary cholangitis and control. A: Plots of principal component analysis (PCA) in positive ion mode. (1) Primary biliary cholangitis (PBC); (2) Control; and (3) Quality control (QC); B: Scatter plots of partial least squares-discriminant analysis (PLS-DA) with a positive model of serum from patients with PBC, autoimmune hepatitis and healthy controls. (1) PBC; (2) Control; and(3) QC; C: Validation plot of the original PLS-DA with a positive model, strongly indicating that the original model is valid and shows signs of overfitting. The permutation test was repeated 200 times in the cross-validation plot.
Identification of serum metabolites specific to AIH
As shown in Table 4. The levels of 17 of 25 potential biomarkers identified were elevated in the serum samples of the patients with AIH, while the levels of 8 of these 25 potential biomarkers were reduced in the serum samples of patients with AIH compared with those in HCs. Among these biomarkers, the levels of TDCA, GUDCA, L-Urobilin, sphinganine, phytosphingosine, I-Urobilin, bilirubin, stearamide,kynurenine, L-threonine, L-phenylalanine, urea, TCA, LysoPC [18:3 (6Z, 9Z, 12Z)], TDCA, GCA, and LysoPE [20:3 (11Z, 14Z, 17Z)/0:0] significantly increased in patients with AIH compared with those in HCs. The levels of 12-ketodeoxycholic acid, uric acid, pyroglutamic acid, LysoPE [0:0/20:2 (11Z, 14Z)],lactic acid, pyroglutamic acid, hypoxanthine, CPA (16:0/0:0), and MG [0:0/18:4 (6Z, 9Z, 12Z, 15Z)/0:0]significantly decreased in patients with AIH compared with those in HCs.

Table 4 Potential serum biomarkers for autoimmune hepatitis compared to healthy control
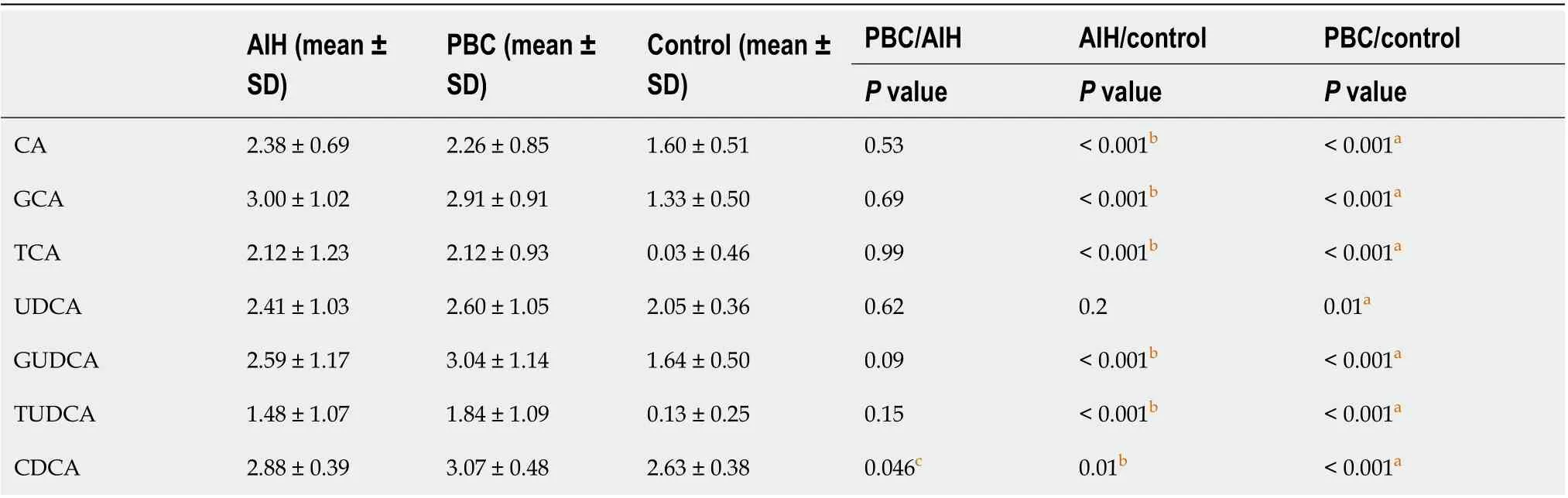
Table 5 Changes of the serum bile acid profile between primary biliary cirrhosis, autoimmune hepatitis and controls

GCDCA 3.78 ± 0.80 3.79 ± 0.67 2.93 ± 0.33 0.96< 0.001b < 0.001a TCDCA 2.73 ± 0.40 2.84 ± 0.44 2.49 ± 0.57 0.27 0.09 0.004a GCDCS 2.39 ± 0.82 2.42 ± 0.70 1.08 ± 0.39 0.95< 0.001b < 0.001a DCA 2.76 ± 0.35 2.75 ± 0.40 2.76 ± 0.18 0.87 0. 子.59 GDCA 3.18 ± 0.58 3.14 ± 0.53 2.64 ± 0.32 0.8< 0.001b < 0.001a TDCA 3.32 ± 0.64 3.16 ± 0.50 2.57 ± 0.19 0.33< 0.001b < 0.001a LCA 0.94 ± 0.68 1.28 ± 0.66 0.72 ± 0.43 0.04c 0.14< 0.001a TLCA 0.30 ± 0.41 0.48 ± 0.47 0.02 ± 0.06 0.07< 0.001b < 0.001a LCA + TLCA 1.25 ± 0.18 1.75 ± 0.14 0.74 ± 0.07 0.034c 0.031b < 0.001a CDCA/CA 1.30 ± 0.35 1.56 ± 0.65 1.74 ± 0.42 0.26< 0.001b < 0.001a pBA 3.11 ± 0.45 3.17 ± 0.51 2.67 ± 0.38 0.98< 0.001b < 0.001a sBA 3.14 ± 0.52 3.19 ± 0.61 2.87 ± 0.17 0.75 0.003b 0.002a pBA/sBA 1.02 ± 0.18 1.01 ± 0.17 1.08 ± 0.13 0.85 0.087< 0.001 sBA 2.77 ± 0.35 2.79 ± 0.40 2.77 ± 0.18 0.81 0.02b 0.003a G-BA 4.14 ± 0.70 4.12 ± 0.64 3.16 ± 0.31 0.97< 0.001b < 0.001a T-BA 3.58 ± 0.54 3.47 ± 0.46 2.89 ± 0.27 0.34< 0.001b < 0.001a G-BA/T-BA 0.55 ± 0.34 0.65 ± 0.34 0.27 ± 0.30 0.25< 0.001b < 0.001a total BA 36.31 ± 6.00 37.69 ± 7.40 24.80 ± 3.12 0.41< 0.001b 0.026aaP < 0.05 Primary biliary cirrhosis (PBC) vs control.bP < 0.05 Autoimmune hepatitis (AIH) vs control.cP < 0.05 PBC vs AIH Bile acid (BA) levels are expressed in log10 concentrations. Statistically significant differences in BA concentrations between controls and patients were determined by the rank sums Mann-Whitney test. CA: Cholic acid; CDCA: Chenodeoxycholic acid; DCA: Deoxycholic acid; GCA: Glycocholic acid;GCDCA: Glycochenodeoxycholic acid; GCDCS: Glycochenodeoxycholic sulfate; GDCA: Glycodeoxycholic acid; GUDCA: Glycoursodeoxycholic acid; LCA:Lithocholic acid; TCA: Taurocholic acid; TCDCA: Taurochenodeoxycholic acid; TDCA: Taurodeoxycholic acid; TLCA: Taurolithocholic acid; TUDCA:Tauroursodeoxycholic acid; UDCA: Ursodeoxycholic acid; PBA: Primary bile acid; PBC: Primary biliary cirrhosis; AIH: Autoimmune hepatitis.
Quantification of targeted BAs specific to PBC and AIH
Recently, BAs have been shown to be potentially more effective biomarkers for PBC and AIH.Regarding the most abundant BAs in humans, the following 15 BAs were selected: CA, GCA, TCA,UDCA, GUDCA, TUDCA, CDCA, GCDCA, TCDCA, GCDCS, DCA, GDCA, TDCA, LCA, and TLCA.The levels of these 15 BAs were measured using the UPLC-QTOF-MS. The levels of all BAs in the disease group were higher than those in the control group, and the levels of glycine-bound cholic acid and tauro-bound cholic acid were elevated, shown in Supplementary Table 1.
Furthermore, the levels of BAs in patients with PBC and AIH were compared with the corresponding levels in HCs, show in Table 5. It was revealed that the levels of BAs were elevated in patients with PBC and AIH. The levels of CDCA, LCA, TLCA, and LCA + TLCA in PBC patients were higher than those in AIH patients, in which a significant difference was found in the levels of CDCA and LCA (P< 0.05; for TLCA,P= 0.0767).The differences in the levels of CDCA, LCA, and LCA + TLCA among the three groups were statistically show in Figure 7. The levels of CDCA, LCA, and LCA + TLCA significantly increased in PBC patients compared with those in AIH patients (P< 0.05). Moreover, the CDCA-to-CA ratio decreased in PBC and AIH patients compared with that in HCs.
The receiver operating characteristic curve analysis of BAs with differences between the PBC and AIH groups showed that the area under the curve values of CDCA, LCA, and TLCA were greater than 0.7, and sensitivity was higher than 70%, indicating a high sensitivity, while a low specificity was noted in identification of patients with PBC and AIH (Figure 8 and Table 6). Compared with sensitivity and specificity of the traditional biochemical indicators, such as alanine transaminase, aspartate transaminase, gamma glutamyl transpeptidase, alkaline phosphatase, total bilirubin, direct bilirubin, and total bile acid, sensitivity and specificity of CDCA, LCA and TLCA were higher than the traditional markers of liver injury, which are of great significance for clinical diagnosis and can be further verified by enlarging the sample size. Thus, BAs can be potentially considered as markers for the diagnosis of PBC and AIH.
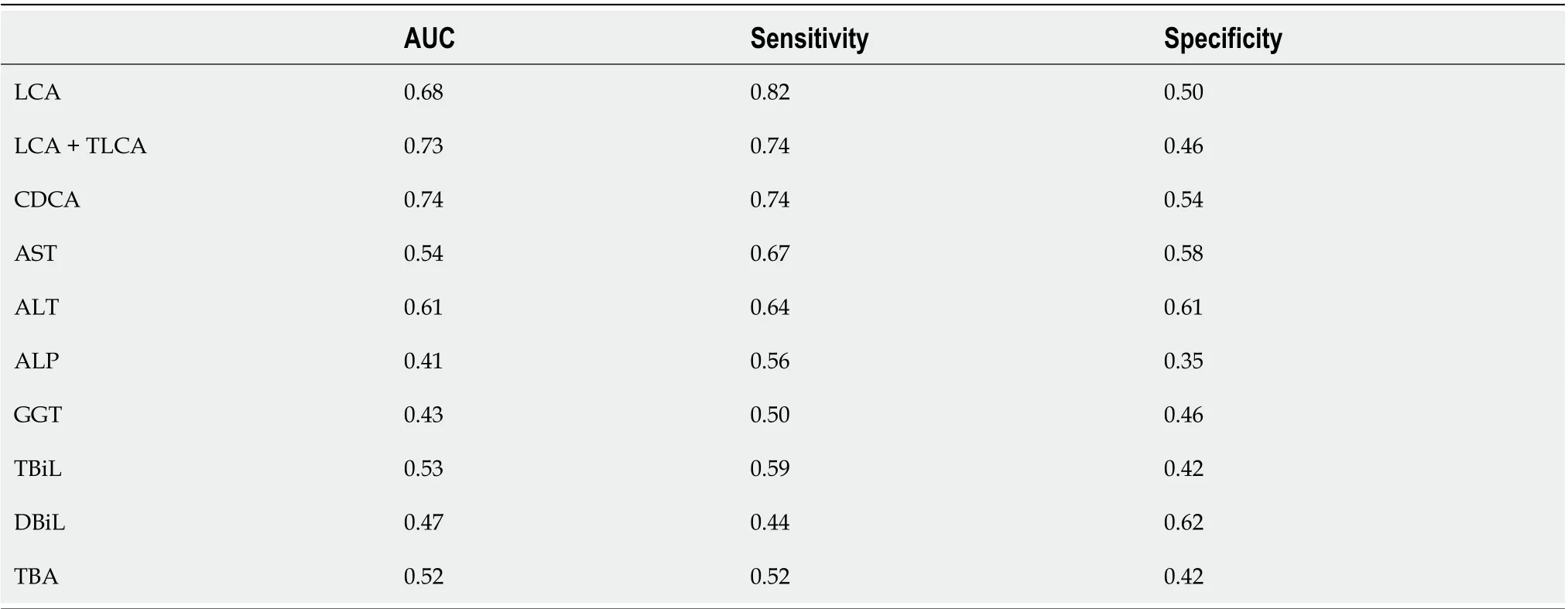
Table 6 Area under the curve, sensitivity and specificity of difference bile acids, conventional biochemical indicators in primary biliary cirrhosis, autoimmune hepatitis and control group

Figure 2 Multivariate statistical analysis on serum profiling data in positive ions between autoimmune hepatitis and control. A: Plots of principal component analysis in positive ion mode. (1) Autoimmune hepatitis (AIH); (2) Control; and (3) Quality control (QC); B: Scatter plots of partial least squaresdiscriminant analysis (PLS-DA) with a positive model of serum from patients with primary biliary cholangitis (PBC), AIH and healthy controls. (1) PBC; (2) Control; and(3) QC; C: Validation plot of the original PLS-DA with a positive model, strongly indicating that the original model is valid and shows signs of overfitting. The permutation test was repeated 200 times in the cross-validation plot.
DlSCUSSlON

Figure 3 Multivariate statistical analysis on serum profiling data in positive ions between primary biliary cholangitis and autoimmune hepatitis. A: Plots of principal component analysis in positive ion mode. (1) Autoimmune hepatitis (AIH); (2) Primary biliary cholangitis (PBC); and (3) Quality control; B: Scatter plots of partial least squares-discriminant analysis (PLS-DA) with a positive model of serum from patients with PBC, AIH and healthy controls. (1)PBC; and (2) AIH; C: Validation plot of the original PLS-DA with a positive model, strongly indicating that the original model is valid and shows signs of overfitting. The permutation test was repeated 200 times in the cross-validation plot.
In the present study, we established a diagnostic model for PBC and AIH using the UPLC-QTOF-MS.Besides, VIP values from PLS-DA were calculated to describe a quantitative estimation of the discriminatory power of each individual feature. We found changes in the levels of amino acids, BAs, organic acids, phospholipids, sugar, and sugar alcohols in patients with PBC and AIH, and in HCs. These substances are mainly involved in lipid metabolism, BA metabolism, and bilirubin metabolism, which are related to metabolic functions of the liver and inflammatory reactions. These compound classes are also associated with key hepatic metabolic pathways. Importantly, our findings are consistent with those reported previously; for instance, BAs have been identified as a significant factor contributing to PBC[14-16]. When liver injury occurs, intrahepatic clearance rate of BAs decreases and serum BA level increases. BAs have long been used as markers of liver dysfunction. In recent studies, elevated serum levels of BAs have been found to be closely associated with liver diseases[17-21].
The levels of lysophosphatidylcholine (LPC) and lysophosphatidylethanolamine (LPE) significantly changed in patients with PBC and AIH. To date, no study has used lysophospholipids in the diagnosis of PBC and AIH. Lysophospholipids are biologically active lipids that are involved in a variety of important processes, including cell proliferation, cell migration, angiogenesis, and inflammation[22].Our results also provided important clues to further explore the pathogenesis of PBC and AIH.
A discriminatory diagnostic model of PBC/AIH could not be established using the UPLC/MS/MS,suggesting that the changes of terminal metabolites in serum samples of patients with PBC and AIH were no special differences. Failure in the establishment of a discriminatory diagnostic model of PBC/AIH could be related to the sample size. Therefore, BAs were quantitatively analyzed according to the differences found between PBC/control and AIH/control groups.
BA is the general term used for a class of bisexual molecules produced by the metabolism of cholesterol. The liver has an effective clearance effect on BAs, and BAs are kept at low levels, confirming the low levels of BAs in the human peripheral blood plasma. In the human liver, cholesterol is metabolized into primary BAs, including CA and CDCA, and then into the intestine, followed by into the corresponding secondary show in Figure 9.
The results of our targeted metabolomic study of BAs showed that the levels of BAs increased in patients with PBC and AIH compared with those in HCs, and the levels of CDCA, LCA, and LCA +TLCA in PBC patients were significantly higher than those in AIH patients. It is noteworthy that CA and CDCA, the two major human BAs, are synthesized from cholesterol in a series of reactions catalyzed by enzymes located in the endoplasmic reticulum, mitochondria, cytosol, and peroxisomes,suggesting that there were significant differences in the levels of BAs in PBC patients, providing clues for the future study on the pathogenesis of PBC. In autoimmune liver diseases, the dysfunction of BA metabolism occurs after liver injury, which may be related to bile stasis after liver injury, especially in PBC, which is more drastic, and is related to the pathogenesis of PBC. After bile duct obstruction and sclerosis, BAs cannot be transported and metabolized normally. Patients may present with jaundice and itchy skin.

Figure 4 Multivariate statistical analysis on serum profiling data in negative ions between primary biliary cholangitis and control. A: Plots of principal component analysis in negative ion mode. (1) Primary biliary cholangitis (PBC); (2) Control; and (3) Quality control (QC); B: Scatter plots of partial least squares-discriminant analysis (PLS-DA) with a negative model of serum from patients with PBC, autoimmune hepatitis and healthy controls. (1) PBC; (2) Control; and(3) QC; C: Validation plot of the original PLS-DA with a negative model, strongly indicating that the original model is valid and shows signs of overfitting. The permutation test was repeated 200 times in the cross-validation plot.
Therefore, determination of the levels of BAs in plasma can reflect the synthesis, ingestion, and secretion of hepatocytes. Abnormalities in the levels of BAs not only reflect the extent of liver damage,but also indirectly indicate the conditions of blood-bile barrier in the liver.
To our knowledge, this is the first study that used serum metabolic profiling for diagnosing patients with PBC and AIH.
LCA is an endogenous compound associated with hepatic toxicity during cholestasis. A previous study[23] revealed that LCA induced disruption of phospholipid/ sphingolipid homeostasis through the transforming growth factor-β signaling pathway and serum LPC could be a biomarker for biliary injury.
The hepatic level of LCA was reported to elevate in patients with cholestatic liver disease[24,25]. This result is consistent with our finding, in which the levels of CDCA, LCA, TLCA, and LCA + TLCA were higher in PBC patients than those in AIH patients.
Previous studies[26,27] indicated that the activation of cytochrome P450 is correlated with Farnesoid X receptor (FXR). Mammalian FXR, which is a transcription regulatory factor in bile salt synthesis, is activated by BAs, such as CDCA or LCA[28,29]. The derangements of lipid metabolism are weakened in FXR-null mice compared with those in wild-type mice after LCA exposure[30,31]. As a cholestatic liver disease, the high levels of BAs may induce FXR gene transcription in PBC patients. Therefore, we hypothesized that these pathways may lead to LCA poisoning in PBC patients, and LCA metabolic pathway plays an important role in the incidence of PBC. Lianet al[14] used the untargeted metabolomic method of UPLC-MS, and clarified the relationship between LCA level and PBC incidence, as well as the relationship between LCA level and the incidence of lipid metabolism disorders. Our study also revealed the abnormal levels of LPC and LPE in PBC patients.
However, the retention of hydrophobic BAs in pathophysiological conditions, such as cholestatic diseases, plays an important role in liver injury by inducing apoptosis or necrosis of hepatocytes[32].The retention and accumulation of hydrophobic Bas (e.g., CDCA and DCA)inside hepatocytes during cholestasis have long been implicated as a major cause of liver dysfunction[32].

Figure 5 Multivariate statistical analysis on serum profiling data in negative ions between autoimmune hepatitis and control. A: Plots of principal component analysis in negative ion mode. (1) Autoimmune hepatitis (AIH); (2) Control; and (3) Quality control (QC); B: Scatter plots of partial least squaresdiscriminant analysis (PLS-DA) with a negative model of serum from patients with Primary biliary cholangitis, AIH and healthy controls. (1) Autoimmune hepatitis(AIH); (2) Control; and (3) QC; C: Validation plot of the original PLS-DA with a negative model, strongly indicating that the original model is valid and shows signs of overfitting. The permutation test was repeated 200 times in the cross-validation plot.
The hydrophobicity of BAs is an important determinant of the toxicity and protection of BAs. Under normal conditions, the levels of BAs undergoing further biotransformations to dianionic glucuronidated or sulfated derivatives are negligible, although they may become important in cholestasis[33].
Several mechanisms may be involved in the cytotoxicity associated with the most hydrophobic BAs in cholestatic liver diseases[32]. BAs could disrupt cell membranes through their detergent action on lipid components[34] and promote the generation of reactive oxygen species that, in turn, oxidatively modify lipids, proteins, and nucleic acids, and eventually cause hepatocyte apoptosis[35].
As shown in Figure 9 CDCA/LCA/TLCA are all related to the decomposition and hydrolysis of bacteria in the intestine. CDCA is decomposed into LCA through bacteria in the intestine, and then,synthesizes TLCA through the intestinal bacteria. Intestinal bacteria may play a key role in this process.Therefore, we can hypothesize that dysfunction of intestinal bacteria may increase the incidence of autoimmune liver diseases, including PBC. Lvet al[36] and Zhenget al[37] found that the interaction of intestinal microflora with metabolism and immunity is crucial for the occurrence or development of PBC.
The Child-Pugh scoring system was used to classify PBC and AIH patients according to their Child-Pugh scores, show in Supplementary Table 2 and the levels of BAs in these patients with Child-Pugh scores were statistically show in Table 7. It was found that the levels of GCA, TCA, GCDCA, GCDCS,TDCA, and tauro-conjugated BAs were gradually elevated with the increase of Child-Pugh scores. The levels of BAs in PBC patients with Child-Pugh class C were significantly different from those in PBC patients with Child-Pugh class A (P< 0.05). The levels of BAs in AIH patients with Child-Pugh class B were significantly different from those in PBC patients with Child-Pugh class A (P< 0.05). The levels of GCA, GCDCS, and TDCA significantly differed in PBC and AIH patients with Child-Pugh class B(Figure 10). These BAs are all conjugated BAs, suggesting that the levels of conjugated BAs are elevated in patients with severe liver injury. The determination of BA level can not only reflect liver damage, but also indicate the degree of liver damage, which is similar to the results of our previous study on druginduced liver injury (DILI)[38]. The increase in the levels of GCA, TCA, TUDCA, GCDCA, GCDCS, and TDCA was corresponded to a higher degree of DILI. Tanget al[15] used UPLC/Q-TOF-MS to analyzethe metabolic groups of blood and urine in 32 pairs of PBC patients and HCs. It was found that the BA level increased with the PBC progression, while the higher accuracy of our findings was confirmed.Elevated levels of BAs are correlated with severity of a variety of diseases. BAs can be used as a factor to judge the severity of the disease and as a basis for the diagnosis of the disease. It is necessary to further expand the sample size for research.
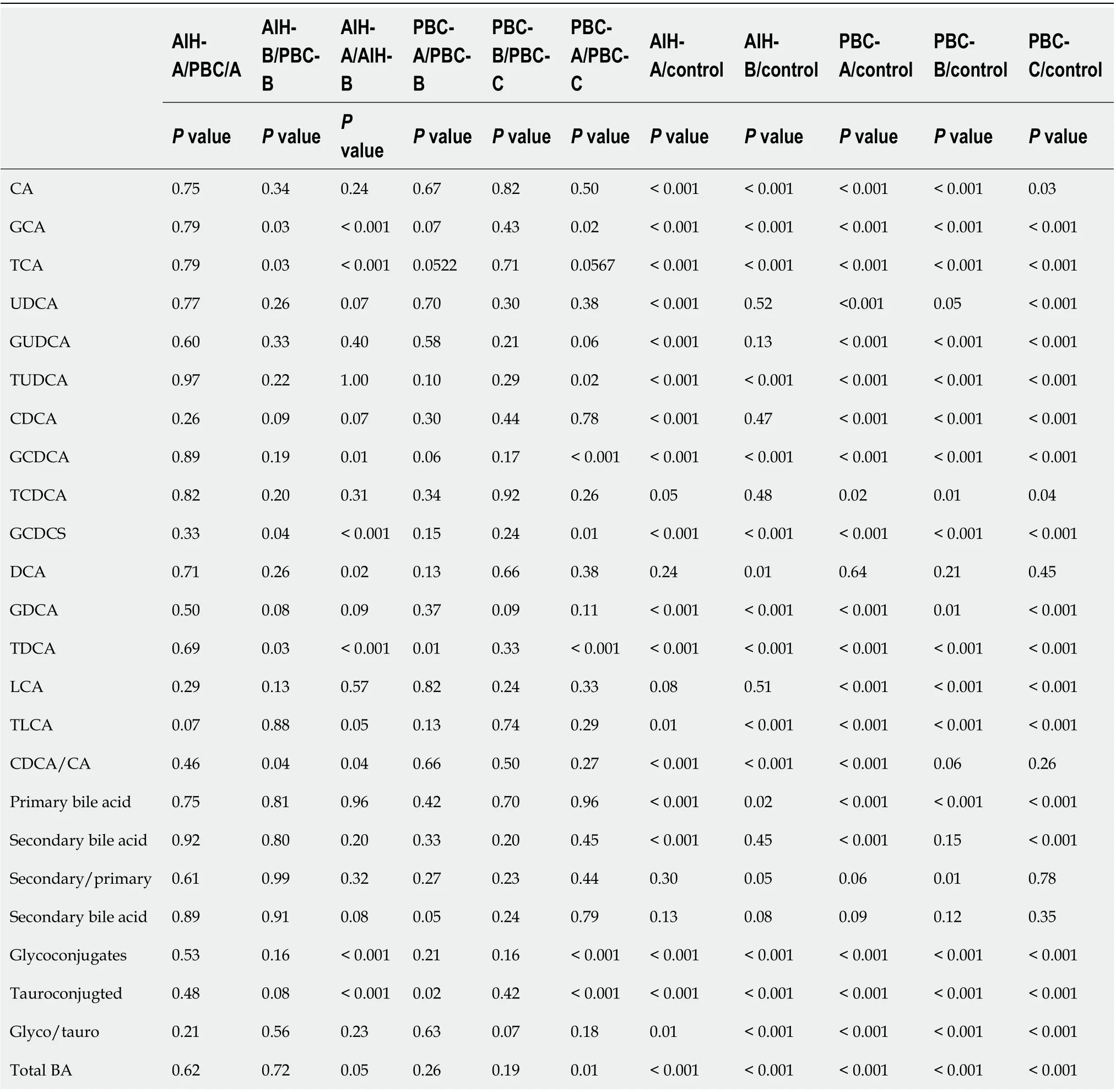
Table 7 Changes of the serum bile acid profile between primary biliary cirrhosis and autoimmune hepatitis in different grade of Child-Pugh
CONCLUSlON
A discriminatory diagnostic model for PBC and AIH using UPLC-QTOF-MS was established. Besides,differential metabolomics analysis was conducted using the PLS-DA model to screen the differentially expressed substances in the different groups. The changes in the levels of BAs, LPC, LPE, bilirubin, and phytosphingosine in PBC and AIH patients and HCs were compared.
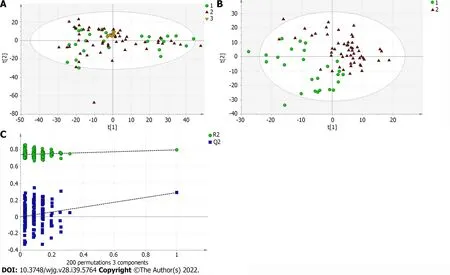
Figure 6 Multivariate statistical analysis on serum profiling data in negative ions between primary biliary cholangitis and autoimmune hepatitis. A: Plots of principal component analysis in negative ion mode. (1) Autoimmune hepatitis (AIH); (2) Primary biliary cholangitis (PBC); and (3) Quality control; B: Scatter plots of partial least squares-discriminant analysis (PLS-DA) with a negative model of serum from patients with PBC, AIH and healthy controls. (1)AIH; and (2) PBC; C: Validation plot of the original PLS-DA with a negative model, strongly indicating that the original model is valid and shows signs of overfitting.The permutation test was repeated 200 times in the cross-validation plot.
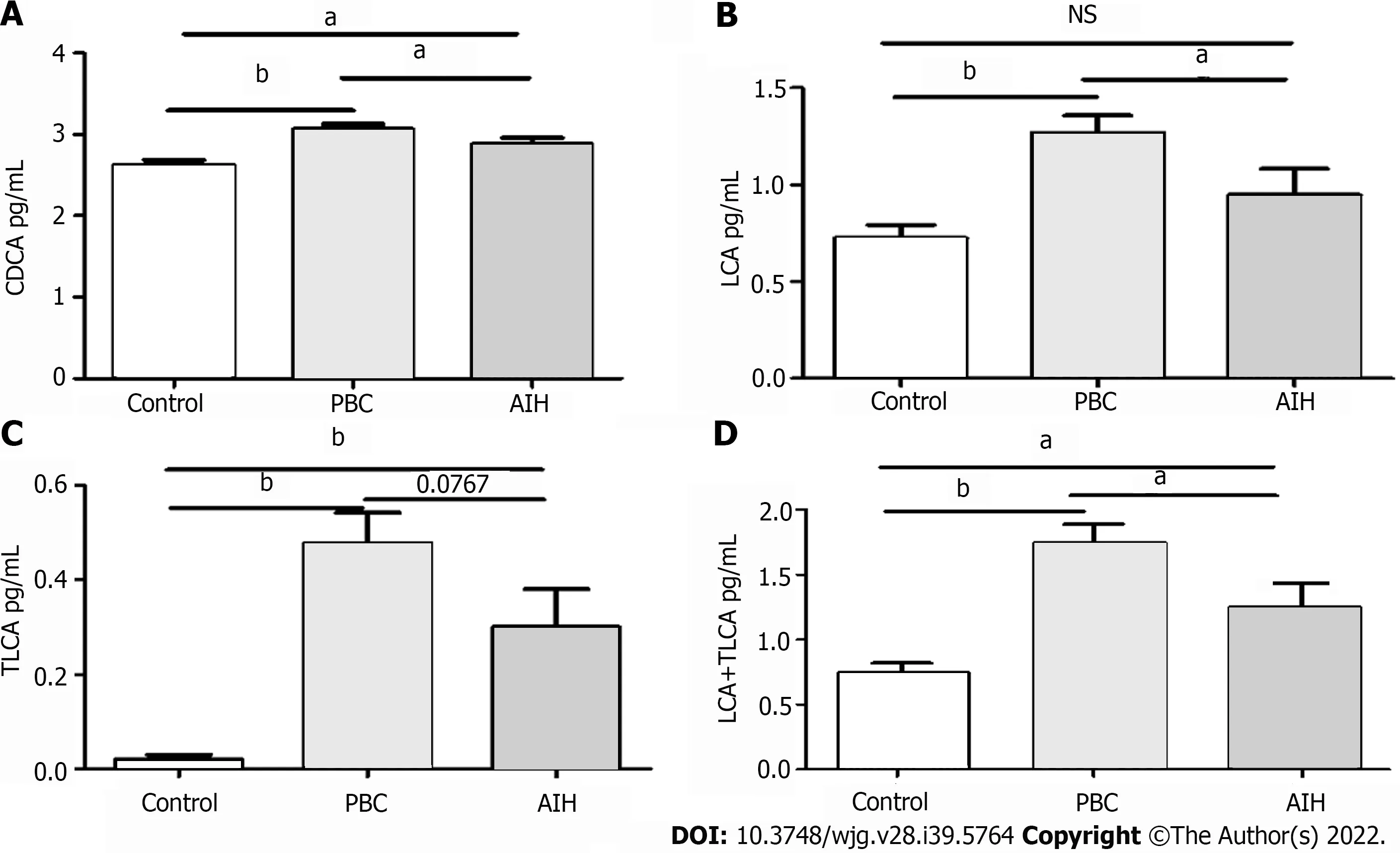
Figure 7 Comparative analysis of alterations in serum bile acid levels in patients in the mild and severe injury groups, and in healthy controls. A: Chenodeoxycholic acid; B: Lithocholic acid (LCA); C: Taurolithocholic acid (TLCA); D: LCA + TLCA; aP < 0.05; bP < 0.0001; NS: Not significant.
The levels of CDCA, LCA, TLCA, and LCA + TLCA significantly increased in the PBC group compared with those in the AIH group. These results suggested that the levels of BAs can be used as a marker to differentiate PBC from AIH, and the results may be advantageous to study the pathogenesis of PBC/AIH in the future.
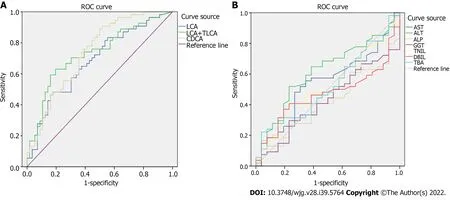
Figure 8 Receiver operating characteristic curve analysis. A: Lithocholic acid (LCA), sum of LCA and taurolithocholic acid, chenodeoxycholic acid; B:Common clinical biochemical indicators.

Figure 9 Cholesterol host cell metabolism. CA: Cholic acid; CDCA: Chenodeoxycholic acid; DCA: Deoxycholic acid; TCA: Taurocholic acid; GCA:Glycocholic acid; TDCA: Taurodeoxycholic acid; GDCA: Glycodeoxycholic acid; TCDCA: Taurochenodeoxycholic acid; TLCA: Taurolithocholic acid; TUDCA:Tauroursodeoxycholic acid.
In conclusion, this study revealed that in patients with PBC and AIH, there were significant differences in serum levels of BAs. However, due to the existence of some limitations (i.e., the small sample size, the lack of staging methods for PBC and AIH, and phenotypic information), further study with a larger sample size is required to eliminate the above-mentioned limitations and to confirm the findings.
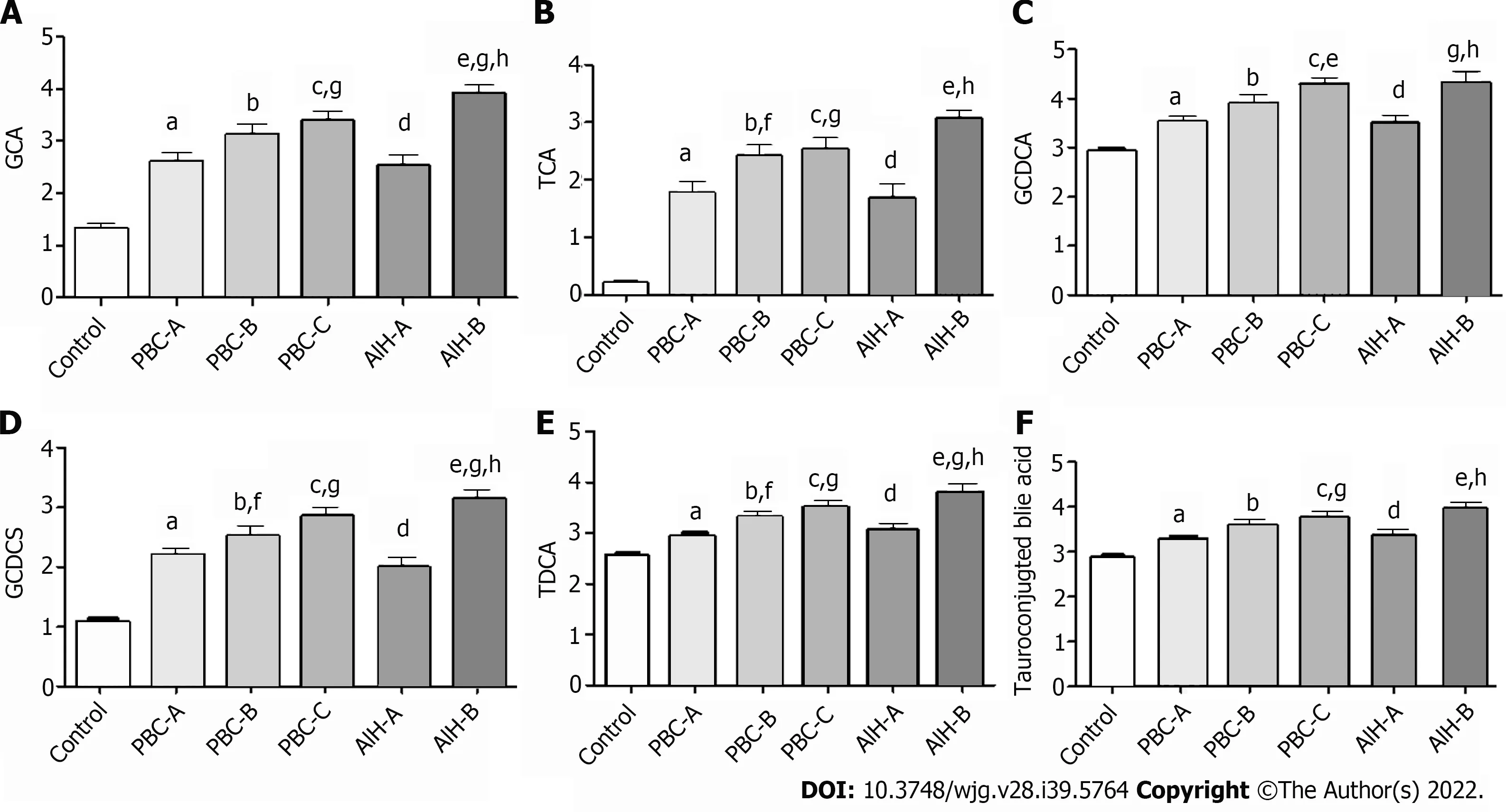
Figure 10 Bile acid levels are expressed in log10 concentrations. Statistically significant differences in bile acid concentrations between controls and patients were determined by the rank sums Mann-Whitney test. Primary biliary cholangitis (PBC)-A vs control, aP < 0.05; PBC-B vs control, bP < 0.05; PBC-C vs control, cP < 0.05; autoimmune hepatitis (AIH)-A vs control, dP < 0.05; AIH-B vs control, eP < 0.05; PBC-A vs PBC-B, fP < 0.05; PBC-A vs PBC-C, gP < 0.05; AIH-A vs AIH-B, hP < 0.05; PBC-A vs AIH-A. A: Glycocholic acid; B: Taurocholic acid; C: Glycochenodeoxycholic acid; D: Glycochenodeoxycholic sulfate; E: Taurodeoxycholic acid; F: Tauroconjugted bile acid. GCDCA: Glycochenodeoxycholic acid; GCDCS: Glycochenodeoxycholic sulfate; TDCA: Taurodeoxycholic acid; GCA: Glycocholic acid; TCA: Taurocholic acid.
ARTlCLE HlGHLlGHTS

ACKNOWLEDGEMENTS
Thanks to Dalian Chemical Institute for providing the test site and technical guidance.
FOOTNOTES
Author contributions:Niu JQ contributed to conception and design of the research, review and editing; Ma ZH, Sun LC, and Li P contributed to investigation and wrote the manuscript; Ma ZH, Wang XM, and Wu RH contributed to acquisition of data and statistical analysis; Hao DL contributed to funding acquisition; all authors have read and approve the final manuscript.
Supported byHealth and Family Planning Commission Project of Jilin Province, No. 2016Q043; and Health and Hygiene Committee Project of Jilin Province, No. 2021LC082.
lnstitutional review board statement:The study protocol was carefully reviewed and approved by the Institutional Review Board (IRB) of the Affiliated Hospital of the Medical School of Jilin University, No. 2009-017.
lnformed consent statement:All the eligible patients and HCs signed the written informed consent form prior to enrollment.
Conflict-of-interest statement:All the authors report no relevant conflicts of interest for this article.
Data sharing statement:No additional data are available.
STROBE statement:The authors have read the STROBE Statement-checklist of items, and the manuscript was prepared and revised according to the STROBE Statement-checklist of items.
Open-Access:This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BYNC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is noncommercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
Country/Territory of origin:China
ORClD number:Xiao-Mei Wang 0000-0003-2606-4013; Jun-Qi Niu 0000-0001-5415-2024.
S-Editor:Fan JR
L-Editor:A
P-Editor:Cai YX
 World Journal of Gastroenterology2022年39期
World Journal of Gastroenterology2022年39期
- World Journal of Gastroenterology的其它文章
- COVID-19 and hepatorenal syndrome
- Interplay between metabolic dysfunction-associated fatty liver disease and chronic kidney disease:Epidemiology, pathophysiologic mechanisms, and treatment considerations
- Pitfalls and promises of bile duct alternatives: A narrative review
- SARS-CoV-2-induced liver injury: A review article on the high-risk populations, manifestations,mechanisms, pathological changes, management, and outcomes
- Immune checkpoint inhibitor-mediated colitis is associated with cancer overall survival
- Receptor of advanced glycation end-products axis and gallbladder cancer: A forgotten connection that we should reconsider
