基于GEO数据库基因表达数据探索影响卵巢癌患者预后及临床分期的基因
侯娟 蒋树立 滕长财

【摘要】 目的 利用基因表达综合征(gene expressed omnibus,GEO)公共数据库筛选与卵巢癌分期相关的基因,并分析其临床意义。
方法 从GEO数据库的数据集GSE9891中筛选与临床分期相关差异基因,通过DAVID数据库对差异基因进行基因本体论(gene ontology,GO)和基因组百科全书(Kyoto encyclopedia of genes and genomes,KEGG)富集分析。并且通过肿瘤基因组图谱(the cancer genome atlas,TCGA)对差异基因进行验证。
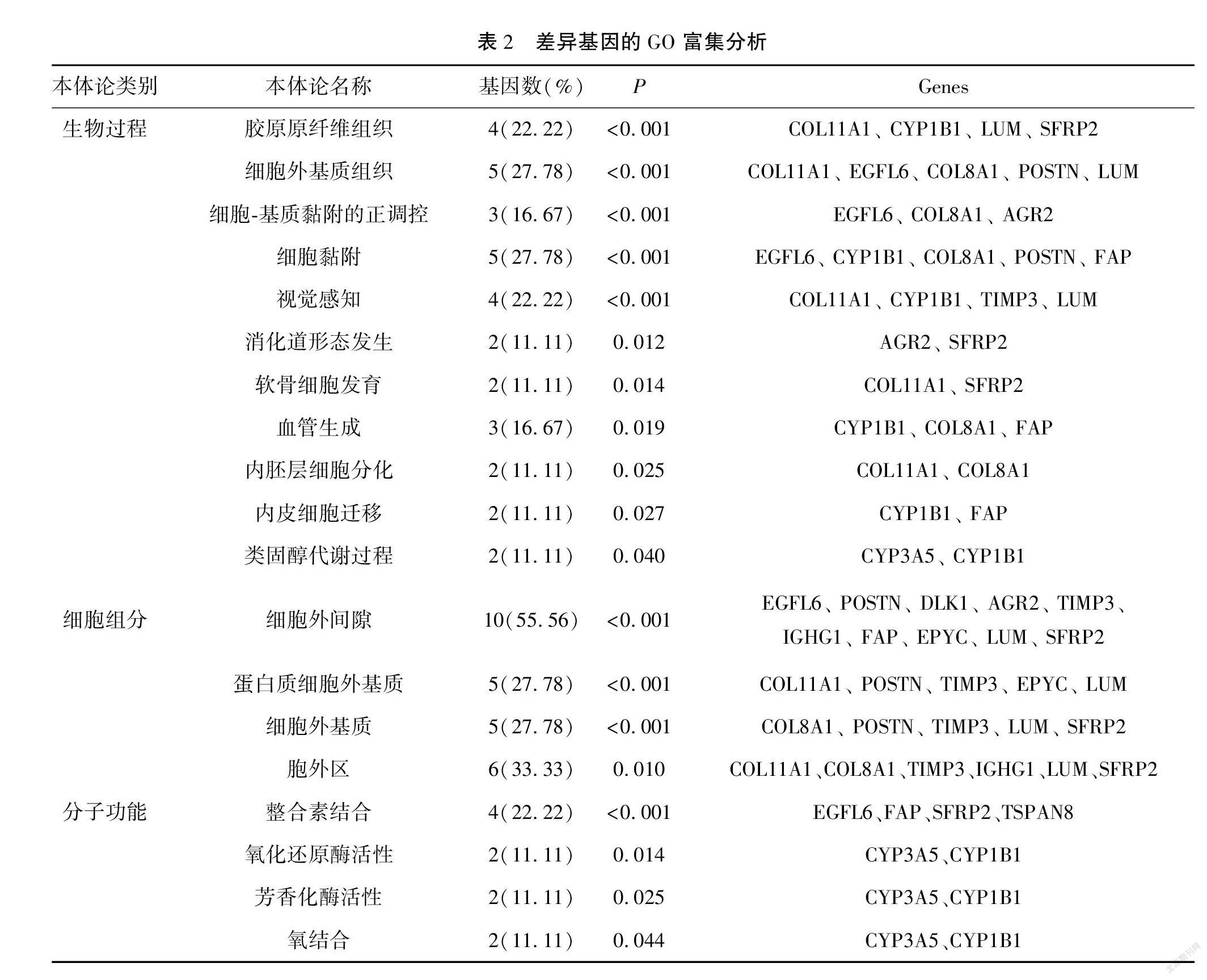
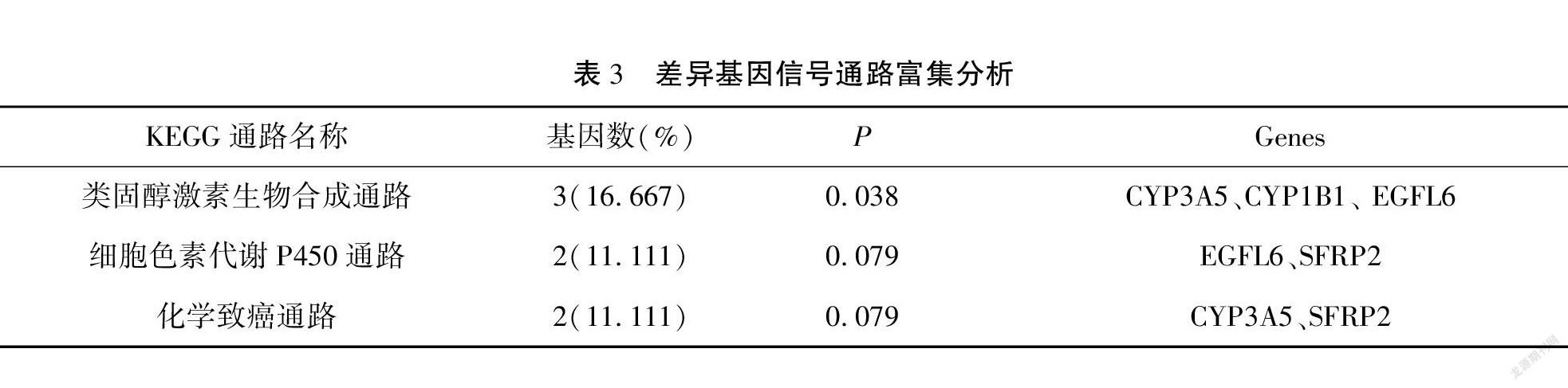
结果 分析数据集GSE9891得到23个与临床分期相关差异基因;GO富集分析发现有效靶基因参与胶原组织和细胞外基质合成;KEGG通路富集分析发现主要通过类固醇合成信号通路影响卵巢癌进展过程。临床分期相关差异基因在TCGA数据库中验证得到LUM、COL11A1、IGF1、TIMP3、COL10A1、FAP、COL8A1、EPYC、SFRP2、FABP4与临床分期相关(P<0.05,|log2FC|>1.5),其中COL8A1、COL11A1、COL10A1、FABP4和SFRP2等五个基因与卵巢癌的预后相关(P<0.05)。
结论 COL8A1、COL11A1、COL10A1、FABP4和SFRP2可能是影響卵巢癌进展的生物标志物。
【关键词】 卵巢癌;临床分期;基因本体论;通路富集分析;预后
中图分类号:R715.5 文献标志码:A DOI:10.3969/j.issn.1003-1383.2022.03.003
Exploration on genes affecting the prognosis and clinical stage of patients with ovarian cancer based on expression data of GEO database gene
HOU Juan, JIANG Shuli, TENG Changcai▲
(Department of Clinical Laboratory, Affiliated Hospital of Binzhou Medical University, Binzhou 256603, Shandong, China)
【Abstract】 Objective To screen genes related to ovarian cancer stage by gene expressed omnibus (GEO) public database, and to analyze their clinical significance.
Methods Differential genes related to clinical stage were screened from the data set (GSE9891) of the GEO database. Enrichment analysis of differential genes were performed by gene ontology (GO) and Kyoto encyclopedia of genes and genomes (KEGG) through DAVID database. In addition, the differential genes were verified by the cancer genome atlas (TCGA).
Results By analyzing the data set GSE9891, 23 differential genes related to clinical stage were obtained, GO enrichment analysis found that effective target genes were involved in collagen tissue and extracellular matrix synthesis, and KEGG pathway enrichment analysis was found to influence ovarian cancer progression mainly through the steroid synthesis signaling pathway. The differential genes related to clinical stage were verified in TCGA database that LUM, COL11A1, IGF1, TIMP3, COL10A1, FAP, COL8A1, EPYC, SFRP2 and FABP4 were related to clinical stage (P<0.05, |log2FC|>1.5), and five genes including COL8A1, COL11A1, COL10A1, FABP4 and SFRP2 were related to the prognosis of ovarian cancer (P<0.05).
Conclusion COL8A1, COL11A1, COL10A1, FABP4 and SFRP2 genes may be biomarkers affecting the progression of ovarian cancer.
【Key words】 ovarian cancer; clinical stage; gene ontology; pathway enrichment analysis; prognosis
卵巢癌是致死率最高的妇科肿瘤,在2019年的肿瘤统计资料中显示全球新增卵巢癌患者22 240人,死亡14 170人[1]。卵巢癌患者的5年生存率只有45.6%,但是早期卵巢癌患者的5年生存率有70%,目前卵巢癌尚无令人满意的早期诊断和治疗的方法[2]。卵巢癌由早期向晚期的进展离不开多种基因的协同调控,而对于影响卵巢癌进展的基因的研究目前尚不明确。有研究显示,CirPUM1、YTHDF1和miR-363-3p均可以通过调节不同基因表达影响卵巢癌的进展[3~4]。本文通过基因表达综合征(gene expressed omnibus,GEO)公共数据库中的数据集GSE9891筛选差异基因,对差异基因进行基因本体论(gene ontology,GO)和信号通路富集分析,为研究卵巢癌的进展和治疗提供新的治疗靶点和理论支持。同时用肿瘤基因组图谱(the cancer genome atlas,TCGA)数据库对差异基因进行验证,发现与卵巢癌患者生存相关的差异基因。
1 材料与方法
1.1 资料来源
使用GEO2R(https://www.ncbi.nlm.nih.gov/geo/geo2r)分析GEO数据库中GSE9891数据集中卵巢癌患者的基因表达数据,筛选阈值P<0.05和|log2FC|>1.5,卵巢癌患者相关TCGA数据由cBioPortal(http://www.cbioportal.org/)下载,根据性别、年龄、总生存(overall survival,OS)、肿瘤分级、临床分期以及差异基因表达量数据筛选得到485例卵巢癌患者的数据。基因功能富集分析利用DAVID(database for annotation,visualization and integrated discovery,https://david.ncifcrf.gov/)在线工具对差异表达基因进行GO功能和基因组百科全书(Kyoto encyclopedia of genes and genomes,KEGG)通路富集分析,显著的GO功能和KEGG通路筛选标准为P<0.05。
1.2 统计学方法
用IBM SPSS Statistics 19.0、Graph Pad Prism 8、Excel 2010對数据进行统计分析。采用K-M法分析相应基因表达量与OS和无进展生存(progression free survival,PFS)间的关系;早期组和晚期组的表达量差异分析采用独立样本t检验。检验水准:α=0.05。
2 结 果
2.1 分析数据集获取的差异表达基因
利用NCBI网站的在线分析工具GEO2R,分析数据集GSE9891获取与卵巢癌患者分期相关的差异表达基因,共得到25个差异表达基因,其中8个基因表达下调和17个基因表达上调,|log2FC|>1.5,P<0.05。见表1。
2.2 差异基因功能富集分析和KEGG通路分析
与卵巢癌分期相关的差异基因富集于19个GO terms中。见表2。另外,差异表达基因主要富集在类固醇激素生物合成(steroid hormone biosynthesis)的信号通路中,而卵巢癌患者激素水平的变化可能与卵巢癌进展相关(P<0.05)。见表3。
2.3 TCGA数据库验证差异基因及预后分析
TCGA数据库验证得到的差异基因,发现10个与卵巢癌分期相关的基因。见表4。笔者对10个差异表达基因进行Cox生存分析,发现COL11A1、COL8A1、SFRP2和FABP4与卵巢癌患者的总生存和无进展生存相关,COL10A1只与卵巢癌患者的总生存相关,而FAP和EPYC只与卵巢癌患者的无进展生存相关(P<0.05)。见表5。
3 讨 论
卵巢癌是一种以晚期为特征且预后极差的妇科肿瘤。卵巢癌患者具有较多的风险因素,比如哺乳期时间、服用避孕药以及生产次数等均与卵巢癌相关[5]。研究显示,卵巢癌的分期直接影响卵巢癌的预后,晚期卵巢癌患者的5年生存率只有45.6%,但是早期卵巢癌患者的5年生存率却有70%[2],因此对于卵巢癌分期相关的分子机制研究十分重要。笔者利用生物信息学方法和工具,筛选出与卵巢癌分期相关的差异基因,对差异基因进行生存分析得到COL8A1、COL11A1、COL10A1、FABP4和SFRP2与卵巢癌的预后相关。研究显示,COL8A1是细胞外基质的组成成分,可以促进平滑肌细胞和肝癌细胞的增殖和降低肝癌细胞对化疗药物的敏感性[6~7]。在乳腺癌和胃癌中也有类似报道,高表达COL8A1肿瘤患者预后较差[6,8~10],这与笔者的研究结果相一致,COL8A1是卵巢癌患者总生存和无进展生存的保护因素。COL11A1、COL10A1与COL8A1同属于胶原蛋白家族,具有相似的生物学功能[4,11~12]。FABP4最初在脂肪细胞和巨噬细胞中表达,调节肥胖诱导的胰岛素拮抗[13],在癌细胞与周围细胞间质进行特异性双向通讯进而影响肿瘤微环境的代谢[14]。SFRP2是褶皱蛋白家族成员,是典型的负反馈Wnt通路调节蛋白[15],SFRP2可以通过调节Wnt信号通路,抑制非小细胞肺癌的生存和转移[15~16],同时,作为Wnt通路的拮抗蛋白可以导致β-catenin和MITF的降低,使氧化还原关键酶APE1失活,使其对靶向治疗更具有抵抗力[17]。笔者对数据集GSE9891的分析发现,差异基因的信号通路富集于激素的生物合成通路,因此推断激素的合成水平可能与卵巢癌的进展相关。目前,激素应用于乳腺癌和前列腺癌的治疗已经有十余年,激素及类固醇类物质对癌症的进展和治疗均具有特殊作用。激素在生理状态和病理状态下均可以发挥作用,但其作用机制却不同,仍需要进一步的机制研究。
综上所述,本研究分析发现,COL8A1、COL11A1、COL10A1、FABP4和SFRP2这5个基因的表达水平与卵巢癌患者的分期有关,并且参与调节代谢,细胞凋亡和激素合成等多种生物过程。因此,当这些基因发生改变时,影响卵巢癌患者体内的激素水平,导致卵巢癌的发生和进展,进而对卵巢癌患者的生存率产生影响。因此及时注意观测卵巢癌患者体内激素水平的变化十分重要,为激素治疗卵巢癌提供了理论支持。
参 考 文 献
[1] DESANTIS C E, MILLER K D, DALE W, et al. Cancer statistics for adults aged 85 years and older,2019[J].CA Cancer J Clin,2019,69(6):452-467.
[2] CHANDRA A, PIUS C, NABEEL M, et al.Ovarian cancer:current status and strategies for improving therapeutic outcomes[J].Cancer Med,2019,8(16):7018-7031.
[3] GU S Q, LUO J H, YAO W X. The regulation of miR-139-5p on the biological characteristics of breast cancer cells by targeting COL11A1[J].Math Biosci Eng,2019,17(2):1428-1441.
[4] LI C S, SHAO T, BAO G C, et al. Identification of potential core genes in metastatic renal cell carcinoma using bioinformatics analysis[J].Am J Transl Res,2019,11(11):6812-6825.
[5] ORONSKY B, RAY C M, SPIRA A I, et al.A brief review of the management of platinum-resistant-platinum-refractory ovarian cancer[J].Med Oncol,2017,34(6):103.
[6] ZHOU J, SONG Y N, GAN W, et al. Upregulation of COL8A1 indicates poor prognosis across human cancer types and promotes the proliferation of gastric cancer cells[J].Oncol Lett,2020,20(4):34.
[7] LI X F, WANG Z, TONG H L, et al. Effects of COL8A1 on the proliferation of muscle-derived satellite cells[J].Cell Biol Int,2018,42(9):1132-1140.
[8] MA Z H, MA J H, JIA L, et al. Effect of enhanced expression of COL8A1 on lymphatic metastasis of hepatocellular carcinoma in mice[J].Exp Ther Med,2012,4(4):621-626.
[9] ZHAO Y F, JIA L, MAO X Y, et al. siRNA-targeted COL8A1 inhibits proliferation,reduces invasion and enhances sensitivity to D-limonence treatment in hepatocarcinoma cells[J].IUBMB Life,2009,61(1):74-79.
[10] PENG W, LI J D, ZENG J J, et al. Clinical value and potential mechanisms of COL8A1 upregulation in breast cancer:a comprehensive analysis[J].Cancer Cell Int,2020,20:392.
[11] LI A Q, LI J, LIN J P, et al. COL11A1 is overexpressed in gastric cancer tissues and regulates proliferation,migration and invasion of HGC-27 gastric cancer cells in vitro[J].Oncol Rep,2017,37(1):333-340.
[12] WU Y H, CHANG T H, HUANG Y F, et al. COL11A1 promotes tumor progression and predicts poor clinical outcome in ovarian cancer[J].Oncogene,2014,33(26):3432-3440.
[13] MUKHERJEE A, CHIANG C Y, DAIFOTIS H A, et al. Adipocyte-induced FABP4 expression in ovarian cancer cells promotes metastasis and mediates carboplatin resistance[J].Cancer Res,2020,80(8):1748-1761.
[14] ZHANG Y Q, ZHAO X T, DENG L L, et al. High expression of FABP4 and FABP6 in patients with colorectal cancer[J].World J Surg Oncol,2019,17(1):171.
[15] LIN H, ANGELI M, CHUNG K J, et al. sFRP2 activates Wnt/β-catenin signaling in cardiac fibroblasts:differential roles in cell growth,energy metabolism,and extracellular matrix remodeling[J].Am J Physiol Cell Physiol,2016,311(5):C710-C719.
[16] LIU X Y, FU J M, BI H R, et al. DNA methylation of SFRP1,SFRP2,and WIF1 and prognosis of postoperative colorectal cancer patients[J].BMC Cancer,2019,19(1):1212.
[17] KAUR A, WEBSTER M R, MARCHBANK K, et al. sFRP2 in the aged microenvironment drives melanoma metastasis and therapy resistance[J].Nature,2016,532(7598):250-254.
(收稿日期:2021-07-04 修回日期:2022-02-09)
(編辑:潘明志)

