A Robust Heterometallic Cd(II)/Ba(II)-Organic Framework with Exposed Amino Group and Active Sites Exhibiting Excellent CO2/CH4 and C2H2/CH4 Separation①
FU Hong-Ru JIANG Yu-Ying LUO Jia-Hua LI Ting②
a (College of Chemistry and Chemical Engineering, Luoyang Normal University, Luoyang 471934, China)
b (College of Materials and Chemical Engineering, China Three Gorges University, Yichang 443002, China)
ABSTRACT A microporous and robust bimetal-organic framework [Cd2Ba(NH2-BTB)2]·2(DMA)·2(H2O) (1)was constructed by mixing the heterometallic nodes and an tridentate carboxyl ligand with amino group, benefiting from the synergistic effect of active sites, exposed amino groups and ultramicroporous structure. This compound displays an extraordinary selectivity of CO2/CH4 and C2H2/CH4 (16.7 and 146.3) at 298 K and 1 atm, which can exceed those of many reported MOFs under the same conditions. This work provides an important model to design metal-organic frameworks for the adsorption and separation of small gas molecules.
Keywords: heterometallic-organic framework, C2H2/CH4 separation, exposed sites
1 INTRODUCTION
Achieving purified CH4is of great significance to improve the utilization of natural gas. Commonly, relatively small amounts of impurities (such as CO2, C2H2, C2H6) can significantly interfere with its combustion and catalytic conversion[1]. Up to now, many methods like distillation and pressurized condensation have been exploited to accomplish the efficiency separation of CH4from other small molecule gases. Comparably, the adsorption and separation of porous adsorbents, especially zeolites, show huge advantages and greatly save the energy consumption[2].
In recent years, metal-organic frameworks (MOFs), constructed through the organization of various metal nodes and organic ligands, exhibit enormous potential for gas storage and separation because of the high crystallization, rich structure, high porosity, as well as the functionalization of pore surface[3-12]. Many porous MOFs have been employed for the separation and purification of light hydrocarbons and CO2. It has demonstrated that the high selectivity and high storage of small gas molecules based on MOFs depend on the high porosity, proper pore width and pore functionality[13-16],and simultaneously, the introduction of Lewis basic sites(such as -NH2, and pyridyl groups) into the organic ligands can sharply increase the host-guest interactions[17-19], thus further promoting the separation and purification of light hydrocarbons.
However, the instability of MOFs limits the application for gas storage and separation. Up to now, many effects have been made to enhance the stability and rigidity of MOFs. An important approach is to construct dual or multi-metal ions.Compared with single metal ions as nodes, usually, the heterometallic nodes not only have higher coordination modes to enhance the stability of ligand-metal bonds[20], but also generate the exposed active sites, which can effectively strengthen the framework to the target gases[21].
On the basis of the aforementioned considerations, a tridentate rigid molecule 1,3,5-tri(3-hydroxyl-4-amino)phenylbenzene (NH2-H3BTB) with an exposed amino functional group becomes our preferable selection because (i) the robustness of the large ligands can endow the large pores and the rigidity of framework; (ii) the amino group can enhance the affinity between framework and gas molecules. A porous heterometallic Cd(II)/Ba(II) compound ([Cd2Ba(NH2-BTB)2]·2(DMA)·2(H2O), 1) was solvothermally synthesized via a one-step method. It exhibits ultrahigh CO2/CH4and C2H2/CH4separations simultaneously.
2 EXPERIMENTAL
2. 1 Materials and physical measurements
The organic ligands, all solvents and metal salts were purchased and used directly. NH2-H3BTB was obtained from Yanshen Technology Co., Ltd. Elemental analyses were measured by using a Vario EL III instrument. Gas adsorption was measured using an ASAP 2020 instrument (Micromeritics, USA). Powder X-ray diffraction (PXRD) measurement was performed on a Bruker D8 with CuKαradiation (λ= 1.5406 Å) in the 2θrange of 5°-50° under ambient temperature. Thermal gravimetric analysis (TGA) was carried out under an air environment at a heating rate of 5 ℃/min using a TA-Q50 thermogravimetric analyzer.
2. 2 Synthesis of the title complex
[Cd2Ba(NH2-BTB)2]·2(DMA)·2(H2O) (1): A mixture of NH2-H3BTB (50.0 mg), Cd(NO3)2·8/3H2O (50.0 mg) and Ba(NO3)2(46.0 mg) was added in DMA (N,N-dimethylacetamide, 2 mL) and H2O (2 mL), and then heated at 120 ℃ for 24 h. Colorless polyhedral crystals were collected. Elemental analysis calcd. (%) for 1, C54H32N2O12BaCd2: C, 51.34; H,2.53; N, 1.90. Found: C, 51.57; H, 2.50; N, 2.01.
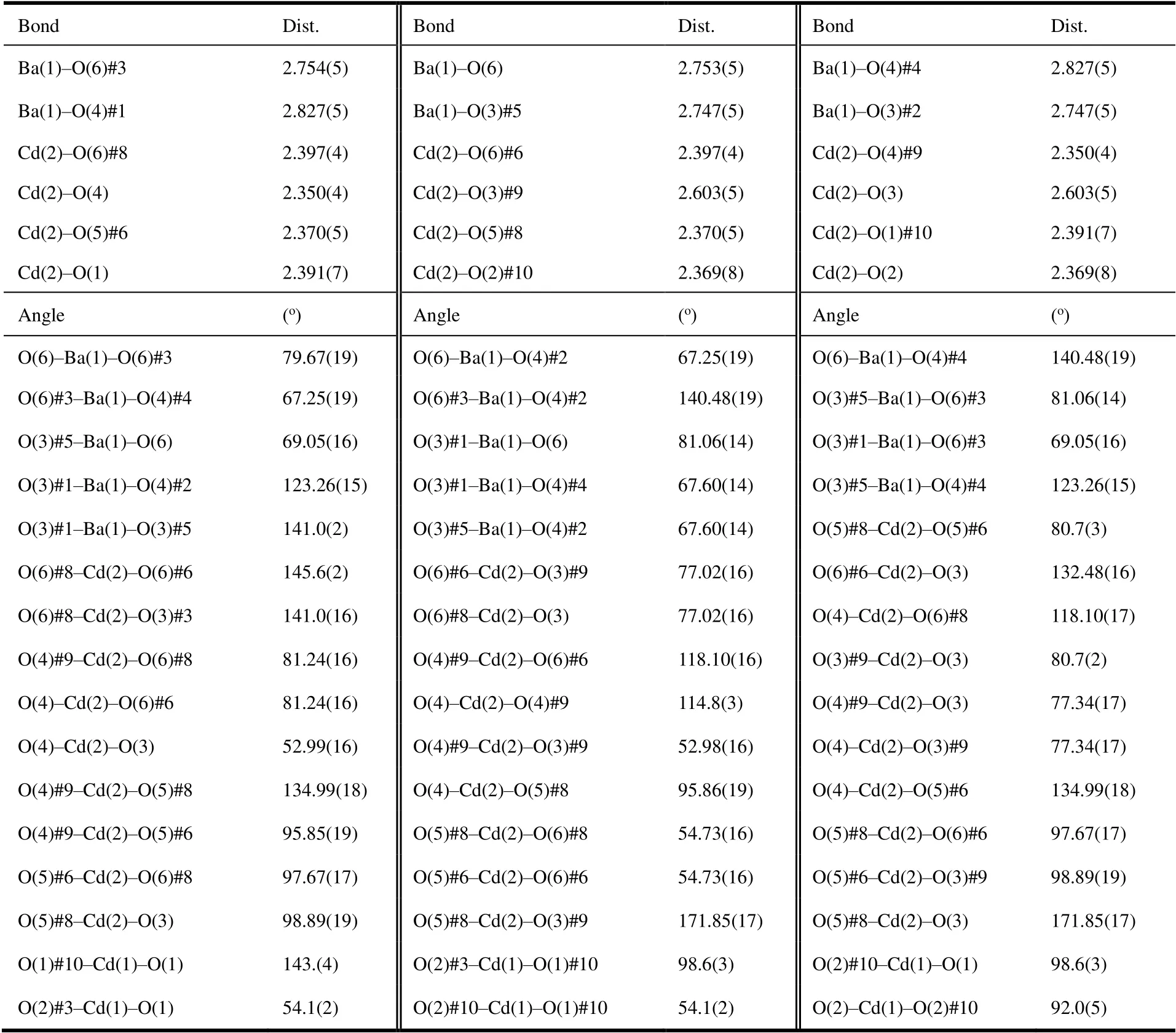
Table 1. Selected Bond Distances (Å) and Bond Angles (o) for Compound 1
2. 3 Crystal structure determination
Single crystal data were collected at room temperature via a Supernova diffractometer (Rigaku, Japan) with a graphitemonochromatized MoKαradiation (λ= 0.71073 Å). Structure was solved and refined through the software OLEX2.0[22]and SHELXTL[23]. The hydrogen atoms of organic ligands were added in the theoretical model directly. The disorder and residual guests were removed thoroughly with SQUEEZE instruction by PLATON soft[24]. The selected bond lengths and bond angles are listed in Table 1.
Complex 1 crystallizes in cubic with space groupI41/acd,a= 32.5028(3),b= 32.5028(3),c= 26.7504(4) Å,V=28260.0(7) Å3,Z= 16,Dc= 1.187 g/m3,F(000) = 9888 andμ= 1.193 mm-1. A total of 31551 reflections including 7793 unique ones (Rint= 0.0361) were collected in the range of 3.87≤θ≤27.25° via anωscan type, and 6488 were used for refinement. The finalR= 0.0751 (I> 2σ(I)),wR= 0.2564 (all data).w= 1/[σ2(Fo2) + (0.1672P)2+ 0.0640P], whereP=(Fo2+ 2Fc2)/3, andS= 1.161.
3 RESULTS AND DISCUSSION
3. 1 Structural description of compound 1
X-ray structure analysis reveals that 1 crystallizes in the tetragonalI41/acdspace group. The asymmetrical unit of 1 contains two independent Cd(II) ions, one independent Ba(II)ion and one fully deprotonated NH2-BTB ligand. The central Cd(II) ions show two coordinate models (Fig. 1a). one is that the Cd(II) ions adopt the high connecting approach. Each Cd(II) ion is coordinated by eight oxygen atoms from four different carboxylate groups, generating a [Cd(COO)4] cluster.The other coordination type of Cd(II) ions show that each Cd(II) ion is coordinated by four oxygen atoms from four different carboxylate groups to form a [Cd(COO)2] node (Fig.1b), namely, two side arms of NH2-BTB (benzoic acid)coordinate with the Cd(II) ion to get the V-type configuration.The carboxylate anions of the ligand adopt a chelate mode.These nodes are further linked by the organic ligands to afford a two-fold interpenetrated framework, formulated as[Cd2(NH2-BTB)3]n(Fig. 1d). With the introduction of Ba(II)ions, a [CdBa(COO)4] chain is formed (Fig. 1d). The Ba(II)atom is six-coordinated by six carboxyl oxygen atoms coming from six different ligands. One Ba(II) ion is inserted into two Cd(II) ions, alternately, resulting in the packing of Cd⋅⋅⋅Ba⋅⋅⋅Cd.However, the whole framework remains unchanged. Compound 1 has large porosity with the rhombus windows of~7.5×7.5 Å2(Fig. 1f) and a void volume of 42.8% (12101.1 Å3/28260.0 Å3, accessible free volume/unit cell volume calculated by PLATON analyses[24]) after removing solvent guests.

Fig. 1. (a, b) Coordination environment of Cd2+ ions. (c) Singlet of the framework. (d) Two-fold interpenetrated structure.(e) Metal-oxo chains consisting of Cd2+, Ba2+ and the carboxyl groups. (f) 3D framework
3. 2 PXRD and thermal properties
The powder X-ray diffraction (XRPD) pattern is used to evaluate the phase purity of 1 at ambient conditions. As shown in Fig. 2, the experimental results are consistent with the simulated ones, indicating the high purity of the phase.Besides, in order to measure the N2adsorption, compound 1 is soaked in ethanol, and the solvent is changed ten times in three days. Then 1 is degassed at 80 °C under a dynamic vacuum for another 8 h to give fully activated samples. Also,the XRPD peaks of the activated sample are also as same as the simulated one, demonstrating that the activated sample maintains the crystalline state.

Fig. 2. XRPD patterns for complex 1: the as-synthesized patterns,the activated sample, the sample after adsorption behaviors and the simulated one based on single-crystal X-ray data

Fig. 3. TGA plots of 1 and the activated sample
TG curve for complex 1 is shown in Fig. 3. TGA analysis on the as-synthesized sample clearly shows that the free H2O and DMA molecules are removed below ~280 ℃ with a weight loss of 24.3% (calcd: 25.1%). The structure collapses near 400 ℃. In addition, TG analyses of activated frameworks are performed. The framework collapses sharply at 400 ℃, indicating that the guest molecules are almost completely exchanged by ethanol.
3. 3 Adsorption properties
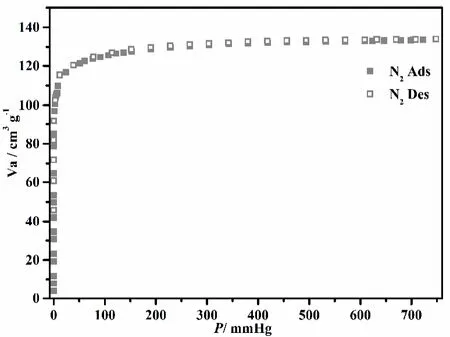
Fig. 4. N2 sorption isotherms of compound 1
The rigidity of the porous framework 1 as well as the high porosity is assessed by solid-gas adsorption experiments with N2, CO2and C2H2as probe molecule. The absorption capacity of saturated N2is 131.25 cm3·g-1, and Brunauer-Emmett-Teller (BET) and Langmuir specific surface areas are calculated to be 432.27 and 510.86 m2·g-1, respectively (Fig. 4). As shown in Fig. 5, the CO2uptake for 1 is 50.7, 33.5 cm3⋅g-1,and for CH4it is 19.8 and 14.3 cm3·g-1at 273 and 298 K. The isosteric heat (Qst) of gases is calculated based on the isotherms recorded at 273 and 298 K to quantify the affinity of compound 1 for each gas. Near zero coverage, the Qst values of CO2, CH4and C2H2are 27.19, 10.83 and 34.32 kJ·mol-1, respectively. Particularly, the high isosteric heat of C2H2is much higher than those for CO2and CH4, which suggests the strong gas-framework interaction. It is easy to understand that the exposed amino polarized functional group as the base group shows larger affinity with C2H2molecule via the hydrogen bonds, and with strong interaction with the acidic CO2[25,26].
To evaluate the gas separation ability of 1, the adsorption selectivity of C2H2/CH4and CO2/CH4is calculated by the ideal adsorption solution theory (IAST)[27]. IAST is a method for predicting the adsorption equilibria for components in a mixture using only single-component adsorption data at the same temperature on the same adsorbent. The adsorption isotherms of the pure component at 298 K are selected to fit with the Langmuir-Freundlich equation. As shown in Fig. 6,the adsorption selectivity of molar mixtures of C2H2/CH4(50% and 50%) reaches 146.3 at atmospheric pressure. By comparison, compound 1 shows more efficient selective separation of CH4than many reported MOFs, such as UTSA-222a (20)[28], ZJU-33a (16)[29], Cu-CPAH (65.5)[30],NbU-9-NH2(29.6)[31]and [Zn4(μ4-O)(μ4-4-pca)3]·2(DEF)·2(H2O) (13.3)[32]under the same conditions. Besides, the selectivity of CO2/CH4(5 and 95%) is 16.7 at atmospheric pressure, confirming that 1 has huge potential for the purification of light hydrocarbons.
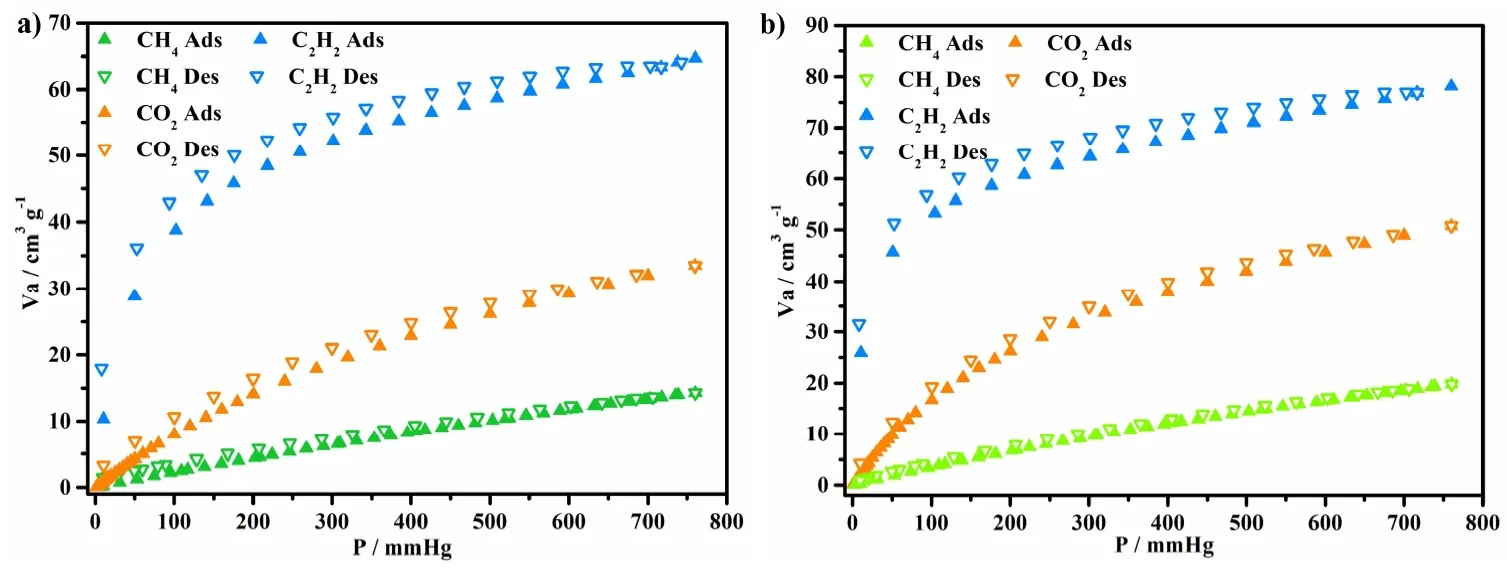
Fig. 5. CO2, CH4, C2H2 sorption isotherms of compound 1: (a) at 298 K, (b) at 273 K
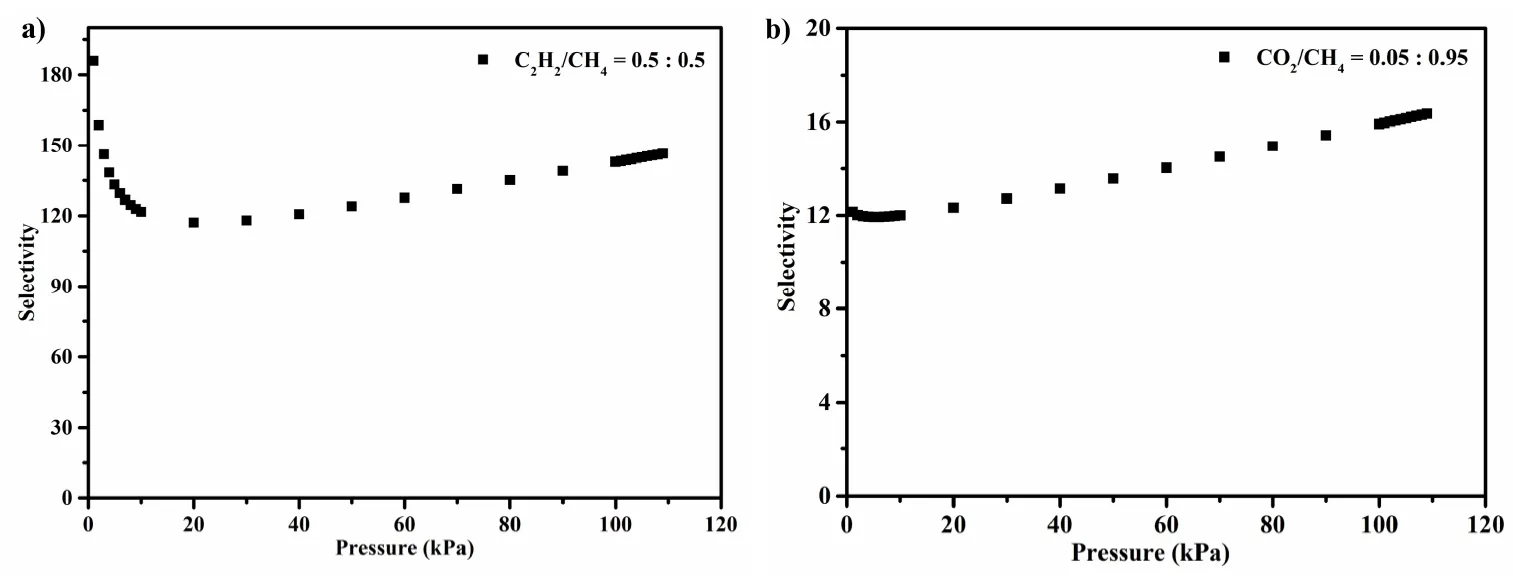
Fig. 6. Adsorption selectivity of dual-component gas predicted by IAST at ambient temperature
4 CONCLUSION
In summary, a heterometallic MOF has been successfully synthesized through one-step synthesis by using NH2-decorated tricarboxylic ligand. Interestingly, this compound displays permanent porosity and abundant active sites on the inter-surfaces, which are conducive to gas adsorption and separation. This compound displays the excellent separation effect for C2H2/CH4and CO2/CH4. Especially, the selectivity of C2H2/CH4is 146.3 at ambient conditions, which can match the C2H2/CH4selectivity ability of many MOFs. The high isosteric value and efficient purification of C2H2over CH4demonstrate that the exposed organic group and the metal nodes can greatly boost the affinity between the framework and C2H2molecules. Thus, this work successfully provides an efficient approach to design heterometallic MOF materials for gas storage and separation.
- 结构化学的其它文章
- Structural and Electronic Properties of Lutetium Doped Germanium Clusters LuGen(+/0/-) (n = 6~19):A Density Functional Theory Investigation①
- Discovery of Benzimidazole Derivatives as Novel Aldosterone Synthase Inhibitors: QSAR, Docking Studies, and Molecular Dynamics Simulation①
- QSAR Models for Predicting Additive and Synergistic Toxicities of Binary Pesticide Mixtures on Scenedesmus Obliquus①
- Preparation, Crystal Structure and Fungicidal Activity of N-(5-(benzofuranol-7-oxymethyl)-1,3,4-thiadiazol-2-yl)amide Compounds①
- Antibiotic Silver Particles Coated Graphene Oxide/polyurethane Nanocomposites Foams and Its Mechanical Properties①
- Planar Tetracoordinate Carbon in 6σ + 2π Double Aromatic CBe42- Derivatives①

