Characterization of two novel heat shock protein 70s and their transcriptional expression patterns in response to thermal stress in adult of Frankliniella occidentalis (Thysanoptera: Thripidae)
QIN Jing, GAO Peng, ZHANG Xiao-xiang, LU Ming-xing, DU Yu-zhou
School of Horticulture and Plant Protection & Institute of Applied Entomology, Yangzhou University, Yangzhou 225009, P.R.China
1. Introduction
Insects are poikilotherm, whose activities are affected by many biotic and abiotic factors, including temperature,humidity, light, radiation, heavy metals, food, parasitic,predation and so on. Temperature is one of the most common environmental factors which affects the insect ecology and evolution directly or indirectly within time and space (Hoffmannet al. 2003). Since global warming has led to more frequent fluctuations of the extreme high and low temperatures, insects have developed many behavioral and physiological abilities to adapt to various environmental temperatures by searching for ideal shelters, experiencing cold or heat hardening, and synthesizing heat shock proteins and antioxidant enzymes (Joanisse and Storey 1998; Lalouetteet al. 2011; Liet al. 2011b; Luet al. 2014).
Heat shock proteins (HSPs) are widely found in various organisms. They are special proteins that are highly conserved in evolution and have high homology in all organisms. HSPs can be synthesized rapidly to help organism adapt to adverse environments (Lindquist 1986;Sǿrensenet al. 2003; Zhao and Jones 2012; Sunet al.2014), such as functioning as molecular chaperones in organism and help denatured protein refolding (Hartl 1996).HSPs can be divided into three categories based on the molecular weight and homology, including high molecular weight HSPs (HMW HSPs, 90–110 kDa), medial molecular weight HSPs (MMWHSPs, 62–72 kDa), and low molecular weight HSPs (LMWHSPs, 15–30 kDa) (Lindquist and Craig 1988; Morimoto 1990). Among these, heat shock protein 70 (HSP70) is one of the most conservative and important family and is much more sensitive than other HSPs when exposed under various stress (Glick 1995; Kimet al. 1998). The HSP70 family consists of two proteins,the non-inducible heat shock cognate protein 70 (HSC70)which exists in almost all cells under normal conditions, and stress inducible HSP70 (Welch 1992). The classic structure of HSP70 contains 44 kDa fragment (amino acid residues 1–386) from the N-terminus, 18 kDa peptide-binding domain(amino acid residues 384–543), 10-kDa C-terminus (amino acids residues 542–646), to the highly conserved EEVD terminal sequence glycine/proline-rich aperiodic segment(Flahertyet al. 1990; Borket al. 1992; Hightoweret al.1994; Morshauseret al. 1995). These four terminal amino acids are ubiquitous in all eukaryotic HSP70 and affect the amount of mRNA translation during heat shock stress(Denisenko and Yarchuk 1990). In addition, HSP70 also plays important roles in anti-apoptotic function, immune response, antioxidant and stress tolerance of cells.
Western flower thrips,Frankliniella occidentalis, is a worldwide economically detrimental insect pest to a wide range of crops.F.occidentalisis native to western North America and damages plants by both direct feeding and transmitting plant viruses (Kirk 2002; Reitzet al. 2011). In 2003,F.occidentaliswas recorded in a greenhouse on pepper plant in Beijing, China for the first time (Zhanget al.2003). Afterwards,F.occidentalishas been found in 14 provinces across China (Chenet al. 2011). The spread ofF.occidentalisis in large part due to its ability in tolerance of temperature extremes. Previous studies have shown that the accumulation of inducible HSP70 determined the thermotolerance of cells (Parsell and Lindquist 1994).F.occidentalisdid not diapause and tolerate extreme temperature through cold or heat hardening or by producing heat shock proteins (e.g.,FoHSP90,FoHSP70,FoHSP60)and antioxidase (e.g., catalase) (Ishidaet al. 2003; Li 2011b;Wanget al. 2014; Luet al. 2016; Qinet al. 2017a, b). In this study, to evaluate the thermal response ofF.occidentalisadults, we analyzed the sequence characteristics and expression patterns of two novel full-length HSP70 protein genes obtained from the male adults (AM) and female adults (AF) under various temperature treatments. The discussion of thermal tolerance related to the two novel genes in this study provided fundamentals for further study ofF.occidentalisadaptation.
2. Materials and methods
2.1. Insects
TheF.occidentalisadults were originally collected in Hangzhou, China in 2008 and reared in the laboratory according to Liet al. (2011a). The climate chamber conditions were maintained at (25±0.5)°C, with a photoperiod of 16 h L:8 h D forF.occidentaliscolony. Newly emerged AF and AM were used in our experiment.
2.2. Thermal treatments
Two hundred newly emerged adults (1-day-old) (and)were collected and placed in a glass tube and exposed to certain temperature treatments for 1 h. Temperature treatments included exposure to cold from –14 to –6°C at 2°C intervals, and heat from 33 to 41°C at 2°C intervals using a temperature controller (DC-3010, Jiangnan Instrument Factory, Ningbo, China). After treatments,F.occidentalisadults were allowed to recover at (25±0.5)°C for 1 h and then were frozen in liquid nitrogen and stored at 70°C.F.occidentalismaintained at 26°C was treated as control.Each treatment was replicated four times.
2.3. RNA extraction and cDNA synthesis
Total RNA was extracted fromF.occidentalisadults using the SV Total RNA Isolation System (Promega, USA).The concentration and quality of RNA were analyzed by spectrophotometry (Eppendorf Bio Photometer Plus,Germany) and agarose gel electrophoresis. A total of 1 μg of total RNA was used as template and the oligo(dT)18primer was also used to generate the first strand complementary DNA (cDNA) according to the protocol of First Strain cDNA Synthesis Kit (Clontech, USA).
2.4. Cloning of HSP transcripts
Gene-specific primers (Table 1) were designed based on sequences fromde novotranscriptome inF.occidentalisto amplify the partial cDNA fragments of HSP70. The PCR reaction conditions were set as follows: 94°C for 3 min,19 cycles of 94°C for 30 s, 65–45°C (decreasing by 1°C/cycle) for 30 s, 72°C for 1 min, and then 25 cycles of 94°C for 30 s, 45°C for 30 s, and 72°C for 1 min, with extension at 72°C for 10 min. Purified products were cloned into the pGEM-T Easy vector (Promega, USA) and transformed into competentEscherichia coliDH5α cells for sequencing.
Gene-specific primers (Table 1) were designed to obtain 5´ and 3´ regions using the SMART RACE cDNA Amplification Kit (Clontech, USA) based on the sequence of partial fragments. PCR parameters were set as follows:94°C for 3 min, 35 cycles of 94°C for 30 s, 68°C for 30 s, and 72°C for 3 min, followed by extension at 72°C for 10 min.Bands of the expected size were cloned and sequenced as described above.
2.5. Bioinformatic analysis
Nucleotide and amino acid sequence similarities were evaluated using the BLAST Program available at the NCBI website (http://www.blast.ncbi.nlm.nih.gov/Blast).Open reading frames (ORFs) were identified using ORF Finder Software (http://www.ncbi.nlm.nih.gov/projects/gorf/). The molecular weights of predicated proteins were calculated by the SWISS-PROT (ExPASy server)“Compute pI/Mw” Program (http://au.expasy.org/tools/pi_tool.html). The online tool ScanProsite was used to identify the features of HSP families (http://www.expasy.org/). The sequence alignment and identity analysis were carried out using the DNAMAN Software Package (Lynnon Corporation, Canada). Phylogenetic trees were constructed with the neighbor joining (NJ) method using MEGA 7(Kumar 2016).

Table 1 The primers used in the study
2.6. Ampli fication of genomic DNA
Genomic DNA was extracted from adults ofF.occidentalisby using AxyprepTMmultisource Genomic DNA Kit (Axygen,USA). Specific primers (Table 1) flanking the ORF were designed to amplify genomic sequences of the three genes.Touch-down PCR was performed as follows: 94°C for 5 min,14 cycles of 94°C for 30 s, 60–45°C (decreasing by 1°C/cycle) for 30 s, 72°C for 2 min 30 s, and then 25 cycles of 94°C for 30 s, 45°C for 30 s, and 72°C for 2 min 30 s, with a final extension of 72°C for 10 min. PCR products were cloned and sequenced as described above.
2.7. Quantitative real-time reverse transcriptase PCR(qRT-PCR) analysis
The mRNA level of the twohsc70s was measured by comparative quantitative real-time PCR amplification. The 18S rRNA and GAPDH were used as internal standard for cold and heat treatments respectively (Zhenget al. 2014).PCR reactions were performed in 20-μL reaction volumes that included iTaqUniversal SYBR Green Supermix (2×) (Bio-Rad,USA), 1 μL of each forward and reverse primer (10 μmol L–1)(Table 1), 2 μL of cDNA template (2.5×10–4μg μL–1), and 6 μL of ddH2O. The PCR reaction was set as follows: 95°C for 30 s,40 cycles of 95°C for 30 s, and incubation at the Tm value of primer pairs (Table 1, see qRT-PCR) for 15 s, and then the melting curve analysis was carried out to determine the specificity of PCR products. Reactions were conducted using a CFX-96 Real-Time PCR System (Bio-Rad, Berkeley, USA).Each PCR reaction included three replicates.
2.8. Statistical analysis
The results of qPCR were analyzed by using the 2–ΔΔCtmethod (Livak and Schmittgen 2001). The statistical significance of difference between treatments was analyzed by eithert-test or one-way analysis of variance (ANOVA)followed by a Tukey’s honesty significant difference test for multiple comparisons. For the ANOVA,hspexpression levels were log- and cosine-transformed according to Tomanek and Somero (1999) to assure homogeneity of variances among different groups. One-way ANOVA was used to detect significant differences in mRNA levels among treatments,followed by Tukey’s honesty significant difference test withP<0.05. All statistical analysis were performed using SPSS ver. 16.0 (SPSS, Chicago, USA)
3. Results
3.1. Cloning and sequence analysis of HSPs
The complete cDNA of theFohsc704andFohsc705was submitted to GenBank (accession no. KY914547 and MF377626, respectively). The full-length cDNA ofFohsc704andFohsc705consisted of 2 073 and 1 476 bp ORF which encoded 690 and 491 aa with calculated molecular weights of 75 and 54.5 kDa, respectively. The polyadenylation signal(AATAAA) was present at 13 bp upstream from the ployA inFohsc704(Tabaska and Zhang 1999). Three canonical signatures that defined the HSC70 family were found inFoHSC704, including IDLGTTnS (aa residues 56–63),VYDLGGGTfdiSIL (241–254), and VlLvGGmSRMPkVqQ(382–396). TheFoHSC705 protein contained typical feature of the HSP70 family at aa residues 341–355(VeIvGGsSRVPaIkQ). The conserved motif EKKN and GIFL was found at the C-terminal inFohsc704andFohsc705,respectively (Appendices A and B).
3.2. Sequence alignment and phylogenetic analysis
The deduced amino acid sequences ofFoHSC704andFoHSC705were aligned with otherinsect species. A high degree of conservation was observed among these sequences (Appendices C and D).FoHSC704 displayed 85, 86, 84, 84, and 85% identity with theZootermopsis nevadensis(KDR08641.1),Athalia rosae(XP_012264348.1),Lasius niger(KMQ94354.1),Eufriesea mexicana(OAD62568.1), andEpicauta chinensis(AHK26790.1), respectively. But there were some highly variable regions, like the region (1–51 aa) located at N-terminal, 613–638 aa and 659–688 aa regions located at C-terminal. We found a putative ATP/GTP-binding site,AESYLNT (Sarasteet al. 1990) and a EF-hand (EFh) domain(EIQKGVFEVKSTNGDTFLGGEDFDNTLVNF, 255–284 aa)in these sequences.FoHSC705displayed 74, 74, 73,and 73% identity withMyzus persicae(XP_022174838.1),A. rosae(XP_020709814.1),Melipona quadrifasciata(KOX73871.1), andLiposcelis bostrychophila(AKP54258.1)respectively. There was a thymopoietin domain (271–303 aa)in these sequences.
Twenty-seven full-length HSP70 proteins from Lepidoptera, Hymenoptera, Thysanoptera, Hemiptera,Coleoptera, and Psocoptera were used for phylogenetic analysis and tree construction using the NJ method (Fig. 1).The tree was divided into four branches, andFoHSP70 was distributed in all branches. TheFoHSC704 andFoHSC705 in this study were found at the top and bottom of the phylogenetic tree, respectively. TheFoHSC704 has a close evolutionary relationship with HSP70s ofLaodelphax striatellaandNilaparvata lugens, which belong to Hemiptera.FoHSC705 was the closest toLygus hesperus, which also belongs to Hemiptera. The other twoFoHSP70s were closed toTribolium castaneum.
3.3. Genomic structure analysis
Pairs ofFohsc704andFohsc705cDNA-specific primers were designed to amplify 3 114 and 1 870 bp sequence fromF.occidentalisgenome (accession no. MF377632 and MF377627, respectively). Comparison of the genomic and cDNA sequence revealed the presence of four introns inFohsc704, including 87- and 71-bp introns in the coding region, a 962-bp intron across coding region and 3´-UTR,and a 70-bp intron in the 3´-UTR.Fohsc705contained six introns in the coding region and the lengths were 633,200, 145, 95, 696 and 73 bp, respectively.Fig. 2 showed the difference of introns of HSP70 family among the same insect species. InF.occidentalis, no introns were found inFohsp703, and four, seven, four and six intorns were found inFohsc701,Fohsc702,Fohsc704andFohsc705,respectively. The size of introns ranged from 76 to 962 bp,and all locations were almost not conservative, there were only two similar locations inFohsc702andFohsc704.
3.4. Expression of Fohsc70s at different temperatures
To determine the expression ofFohsc704andFohsc705in AF and AM at different heat and cold temperatures, the relative mRNA levels of the two genes were quantified by using real-time quantitative PCR. GAPDH and 18S were used as candidate reference genes in heat and cold stress,respectively (Zhenget al. 2014).
ForFohsc704,when exposed toheat stress, the expression level inF.occidentalisadults was low and similar, except for those of AF at 39°C, whose level was significantly decreased (Fig. 3-A). When exposed to cold stress, the expression ofFohsc704reached to the highest level (2.28-fold) in AM at –12°C. No significant difference of the expression level was found in AF (Fig. 3-B).When exposed to high temperature, the expression level of AF is slightly higher than that of AMat 41°C (heat temperature:F=4.732,P<0.001, cold temperature:F=2.930,P=0.01).

Fig. 1 Phylogenetic tree of FoHSC704 and FoHSC705 in Frankliniella occidentalis and other insect species based on neighborjoining method using Mega 7.0. The percentage bootstrap values obtained from 1 000 re-samplings were shown at the nodes and percentages lower than 50 were deleted. The FoHSP70 in our study was marked with black triangles, and FoHSP70s in previous studies were marked with black square.
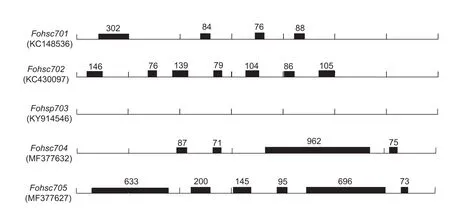
Fig. 2 Comparison of genomic structure of Fohsp70s in Frankliniella occidentalis. The lines represented the cDNA sequences of hsp70s and GenBank accession numbers of species were given on the left of the line. Intron positions were indicated by black rectangles, and the number above the boxes represented the length of introns (bp).
The expression ofFohsc705in AF and AM was found inducible by heat and cold stress. At high temperatures,this gene expression level reached its peak in AF at 33°C and decreased significantly with the temperature increasing.However, no significant difference was found in expression level of AM (Fig. 4-A). In the cold treatment, the relative expression level ofFohsc705was induced significantly to increase in both AF (1.83-fold) and AM (4.18-fold) at–8°C (Fig. 4-B). However,Fohsc705exhibited different expression patterns in different genders ofF.occidentalisunder the same cold stress. For example, the expression level of AM is higher than that of AF at –8 and –12°C,respectively (heat temperature:F=3.217,P=0.007; cold temperature:F=16.875,P<0.001).

Fig. 3 Relative mRNA expression level of Fohsc704 in Frankliniella occidentalis adults under heat (A) and cold (B) stress. All statistics indicate means±SE. Columns labeled with different letters indicate significant differences using one-way ANOVA followed by Tukey’s multiple comparison analysis.
4. Discussion

Fig. 4 Relative mRNA expression level of Fohsc705 in Frankliniella occidentalis adults under heat (A) and cold (B) stress. All statistics indicate means±SE. Columns labeled with different letters indicate significant differences using one-way ANOVA followed by Tukey’s multiple comparison analysis.
In this study, we identified newFohsc704andFohsc705genes, members of HSC70sin F.occidentalisadults.We found a putative ATP/GTP-binding site, AESYLNT(Sarasteet al. 1990) and a EF-hand (EFh) domain(EIQKGVFEVKSTNGDTFLG GEDFDNTLVNF, 255-284 aa)inFoHSC704. In normal situations, a balance exists between the generation of reactive oxygen species (ROS)and antioxidant defenses (Poljsaket al. 2013). When cells exposed to extreme temperatures, the balance will be broken and surplus ROS can trigger severe damage to biological molecules, including lipids, proteins, and nucleic acids (Martindale and Holbrook 2002), resulting in damage or genomic instability (Dizdaroglu and Jaruga 2012). EFh can help prevent cell from oxidative damaging (Chen 2007).Thymopoietin domain inFoHSC705 is related to cellular immunity. The motifs at the C-terminus were EKKN and GIFL forFoHSC704 andFoHSC705, respectively. The motif at C-terminus of HSP70 members in insect is specific in which the sequence ended with EEVD located in the cytoplasm,KDEL in endoplasmic reticulum, and PEAEYEEAKK in mitochondrion (Guy and Li 1998). EKKN and GIFL found in our study were different from the description as above,and the motif at C-terminus ofFoHSP70s in previous studies was EEVD or KDEL (Wanget al. 2014; Luet al. 2016). In addition, the full length ofFohsp705was only 1 476 bp which encoded 491 aa, and its molecular weight was only 54.5 kDa which was less than HSPs in previous studies. Thus,we summarized that theFoHSC704 andFoHSC705 found in our study were two novel members in HSP70 family. The specific locations need to be certified in the future study.
The phylogenetic tree showed the decentralized distribution ofFoHSP70s, indicating that HSP70s in insecta are diverse and mutated. TheFoHSC704 andFoHSC705 in this study were not located in the same branch with otherFoHSP70s, but they were close to Hemiptera. The result suguested that the evolutionary history of the twoFohsc70s be parallel to insects in Hemiptera. Meanwhile,the twoFoHSP70s shared high sequence similarity with other insects. These results suggested that the twoFoHSP70s may have other functions. Wang and Lei(2005) reported that the HSP70 proteins from the same cell compartment among species were more similar than those from different organs of one species, due to the specialized function of members in HSP70 family. The analysis of genomic sequence suggested that four of the fiveFohsp70s contained introns, and their number and size differ from each other. Since the introns were the remnants of a process that speeded up evolution, the number and location of introns reflect the evolutionary history to a certain extent (Gilbert 1985). The genomic structure analysis result was consistent with the phylogenetic analysis that theFoHSP70s were different in evolutionary process. Compared to HSP70, HSC70 existed in normal cells and tissues and could not be induced easily. We hereby speculated that the presence of introns improved the stability of HSP70 and have affected the expression ofhsp70genes. Previous studies have shown that the presence of introns offset the efficiency of transcription and affected other molecular biological processes. It can also influence the gene expression in mRNA transport, stability,and translation efficiency (Nottet al. 2003). Comeron(2004) also reported that there was negative correlation between intron size and gene expression level, and the shorter or no intron genes had high expression level.
The induction ofFohsc704andFohsc705by heat and cold treatments in our study indicated the important roles they played in thermotolerance adaptation. There were differences in gene expression in genders ofF.occidentalisin our study, for instance, theFohsc704expression level of AF was significantly higher than that of AM at 41°C,which was consistent with the previous reports that AF was more thermotolerant than AM (Liet al. 2011a). Similarly,Grapholita molesta(Busck) adult genders were found to be correlated with the expression ofhsp70gene (Chenet al. 2014). However, theFohsc705expression in AM at–8 and –12°C was significantly higher than AF. This was inconsistent with previous reports in Liet al. (2011a, b).The difference in thermotolerance may be influenced by various factors including water and ion homeostasis, levels of proline, asparagine and glutamic acid, etc. HSPs only contributed partially to thermotolerance (Fieldset al. 1998;MacMillanet al. 2015). ForFohsc704, the expression difference was only found in AF under the heat stress,and this was opposite when exposed to cold stress. This finding indicated that the sensitivity of different genders to temperature was variable. InF.occidentalis, we found that the relative expression ofFohsc704was lower thanFohsc705in adults in same treatments, particularly under heat stress. We surmised that the polyadenylation signal(AATAAA) in 3´ nontranslated regions inFohsc704resulted in the situation. The signal had greater mRNA stability at normal temperatures (Colgan and Manley 1997; Rubenstein and Lyons 2001), and therefore it contributed to the maintenance and re-establishment of basal levels of gene expression (Lindquist and Petersen 1990).
The expression level ofFohsc705with six introns was induced by temperature, which was similar to the expression level ofFohsp703with no intron in Qinet al. (2017b).Therefore, the situation was consistent with the study that the presence or absence of introns cannot be used to distinguish HSC70 from HSP70, and genes with introns can encode stress-inducible HSP70 (Qinet al. 2003). The highest expression ofFohsc705in adults was 4.18-fold of those in the control, indicating that there might be other members of HSP70 family contributing to temperature response. In addition, thymopoietin domain in the gene is related to cellular immunity, not cell stress. What’s more,development of insects will affect the gene expression level,Luet al. (2016) reported thatFohspc701andFohspc702expression levels were significantly higher in pupae than that in adults. Expression levels ofhsps were also altered by treatment and recovery time. For example, the expression ofhsp90andhsp70inGrapholita molestaincreased with the treatment time (Zhang and Denlinger 2010), and the expression ofFohsp60andFohsp90was the highest at 2-h treatment (Luet al. 2016). Thus, the developmental stages ofF.occidentalisstatus and treatment pattern played important roles in expression ofhsp70s.
5. Conclusion
We obtained two novelFohsc70s with introns and found that there was difference in expression patterns of the twohsc70sin genders ofF.occidentalis. When exposed to heat stress, the two genes in AF were induced whereas not in AM. However, when exposed to cold stress, the expression differences of two genes were found in both AF and AM.Our results indicated that the twoFoHSP70s are associated with thermal adaption and provided useful information in understanding the thermotolerance ofF.occidentalisat the molecular basis.
Acknowledgements
This research was funded by the Special Fund for Agro-Scientific Research in the Public Interest of China(201103026, 200803025) and the Science and Technology Innovation Project of Student in Yangzhou University, China(X20160637). The authors would like to thank the Testing Center of Yangzhou University, China for assistance.
Appendicesassociated with this paper can be available on http://www.ChinaAgriSci.com/V2/En/appendix.htm
Bork P, Sander C, Valencia A. 1992. An ATPase domain common to prokaryotic cell cycle proteins, sugar kinases,actin, and hsp70 heat shock proteins.Proceedings of the National Academy of Sciences the United States of America,89, 7290–7294.
Chen H, Xu X L, Li Y P, Wu J X. 2014. Characterization of heat shock protein 90, 70 and their transcriptional expression patterns on high temperature in adult ofGrapholita molesta(Busck).Insect Science, 21, 439–448.
Chen J Q, Zhang Y Z. 2007. The research progress of one member of the EF-hand superfamily-troponin C.Chinese Journal of Biotechnology, 23, 375–380. (in Chinese)
Chen X, Yuan L, Du Y, Zhang Y, Wang J. 2011. Cross-resistance and biochemical mechanisms of abamectin resistance in the western flower thrips,Frankliniella occidentalis.Pesticide Biochemistry and Physiology, 101, 34–38.
Colgan D F, Manley J L. 1997. Mechanism and regulation of mRNA polyadenylation.Genes and Development, 11,2755–2766.
Comeron J M. 2004. Selective and mutational patterns associated with gene expression in humans: Influences on synonymous composition and intron presence.Genetics,167, 1293–1304.
Denisenko O, Yarchuk O. 1990. Heat shock translational control in cell-free system.Antonie van Leeuwenhoek, 58,163–168.
Dizdaroglu M, Jaruga P. 2012. Mechanisms of free radicalinduced damage to DNA.Free Radical Research, 46,382–419.
Fields P G, Fleurat-Lessard F, Lavenseau L, Febvay G,Peypelut L, Bonnot G. 1998. The effect of cold acclimation and deacclimation on cold tolerance, trehalose and free amino acid levels inSitophilus granariusandCryptolestes ferrugineus(Coleoptera).Journal of Insect Physiology, 44,955–965.
Flaherty K M, DeLuca-Flaherty C, McKay D B. 1990. Threedimensional structure of the ATPase fragment of a 70K heat-shock cognate protein.Nature, 346, 623.
Gilbert W. 1985. Genes-in-pieces revisited.Science, 228,823–825.
Glick B S. 1995. Can Hsp70 proteins act as force-generating motors?Cell, 80, 11–14.
Guy C L, Li Q B. 1998. The organization and evolution of the spinach stress 70 molecular chaperone gene family.The Plant Cell, 10, 539–556.
Hartl F U. 1996. Molecular chaperones in cellular protein folding.Nature, 381, 571−580.
Hightower L E, Sadis S E, Takenaka I M. 1994. Interactions of vertebrate hsc70 and hsp70 with unfolded proteins and peptides.Cold Spring Harbor Monograph Archive, 26,179–207.
Hoffmann A A, Sørensen J G, Loeschcke V. 2003. Adaptation ofDrosophilato temperature extremes: Bringing together quantitative and molecular approaches.Journal of Thermal Biology, 28, 175–216.
Ishida H, Murai T, Sonoda S, Yoshida H, Izumi Y, Tsumuki H. 2003. Effects of temperature and photoperiod on development and oviposition ofFrankliniella occidentalis(Pergande) (Thysanoptera: Thripidae).Applied Entomology and Zoology, 38, 65–68.
Joanisse D R, Storey K B. 1998. Oxidative stress and antioxidants in stress and recovery of cold-hardy insects.Insect Biochemistry and Molecular Biology, 28, 23–30.
Kim K K, Kim R, Kim S H. 1998. Crystal structure of a small heat-shock protein.Nature, 394, 595–599.
Kirk W D. 2002. The pest and vector from the West:Frankliniella occidentalis. In:Thrips and Tospoviruses: Proceedings of the 7th International symposium on Thysanoptera. vol. 2,No. 7. Australian National Insect Collection, Australia. pp.33–42.
Kumar S, Stecher G, Tamura K. 2016. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets.Molecular Biology and Evolution, 33, 1870–1874.
Lalouette L, Williams C M, Hervant F, Sinclair B J, Renault D.2011. Metabolierate and oxidative stress in insects exposed to low temperature thermal fluctuations.Comparative Biochemistry and Physiology(Part A), 158, 229–234.
Li H B, Shi L, Lu M X, Wang J J, Du Y Z. 2011a. Thermal tolerance ofFrankliniella occidentalis: Effects of temperature,exposure time, and gender.Journal of Thermal Biology,36,437–442.
Li H B, Shi L, Wang J J, Du Y Z. 2011b. Rapid cold hardening of Western flower thrips,Frankliniella occidentalis,and its ecological cost.Acta Ecologica Sinica,31, 7196–7202. (in Chinese)
Lindquist S, Craig E A. 1988. The heat-shock proteins.Annual Review of Genetics. 22, 631–677.
Lindquist S. 1986. The heat-shock response.Annual Review of Biochemistry, 55, 1151–1191.
Lindquist S, Petersen R. 1990. Selective translation and degradation of heat-shock messenger RNAs inDrosophila.Enzyme, 44, 147–166.
Livak K J, Schmittgen T D. 2001. Analysis of relativegene expression data using real-time quantitative PCR and the 2–ΔΔCTmethod.Methods, 25, 402–408.
Lu M X, Li H B, Zheng Y T, Shi L, Du Y Z. 2016. Identification,genomic organization and expression pro files of four heat shock protein genes in the western flower thrips,Frankliniella occidentalis.Journal of Thermal Biology, 57,110–118.
Lu M X, Liu Z X, Cui Y D, Du Y Z. 2014. Expression patterns of three heat shock proteins inChilo suppressalis(Lepidoptera:Pyralidae).Annals of the Entomological Society of America,107, 667–673.
MacMillan H A, Andersen J L, Davies S A, Overgaard J.2015. The capacity to maintain ion and water homeostasis underlies interspecific variation inDrosophilacold tolerance.Scienti fic Reports, 5, 18607.
Martindale J L, Holbrook N J. 2002. Cellular response to oxidative stress: Signaling for suicide and survival.Journal of Cell Physiology, 192, 1–15.
Morimoto R I, Tissieres A, Georgopoulos C. 1990. The stress response, function of the proteins, and perspectives.Cold Spring Harbor Monograph Archive, 19, 1–36.
Morshauser R C, Wang H, Flynn G C, Zuiderweg E R. 1995.The peptide-binding domain of the chaperone protein Hsc70 has an unusual secondary structure topology.Biochemistry,34, 6261–6266.
Nott A, Meislin S H, Moore M J. 2003. A quantitative analysis of intron effects on mammalian gene expression.RNA, 9,607–617.
Parsell D A, Lindquist S. 1994. 18 heat shock proteins and stress tolerance.Cold Spring Harbor Monograph Archive,26, 457–494.
Poljsak B, Šuput D, Milisav I. 2013. Achieving the balance between ROS and antioxidants: When to use the synthetic antioxidants.Oxidative Medicine and Cellular Longevity,2013, 956792.
Qin J, Lu M X, Zheng Y T, Du Y Z. 2017a. Molecular cloning,characterization and functional analysis of catalase inFrankliniella occidentalis.Annals of the Entomological Society of America, 110, 212–220.
Qin J, Zhang X X, Gao P, Lu M X, Du Y Z. 2017b. Cloning and expression pro file of a novel, thermal, inducible HSP70 gene in insects.Chinese Journal of Applied Entomology,54, 380–391. (in Chinese)
Qin W, Tyshenko M G, Wu B S, Walker V K, Robertson R M.2003. Cloning and characterization of a member of thehsp70gene family fromLocusta migratoria, a highly thermotolerant insect.Cell Stress & Chaperone, 8, 144–152.
Reitz S R, Gao Y L, Lei Z R. 2011. Thrips: Pest of concern to China and the United States.Journal of Integratiave Agriculture, 10, 867–892.
Rubenstein R C, Lyons B M. 2001. Sodium 4-phenylbutyrate downregulates HSC70 expression by facilitating mRNA degradation.American Journal of Physiology(Lung Cellular and Molecular Physiology), 281, L43–L51.
Saraste M, Sibbald P R, Wittinghofer A. 1990. The P-loop - A common motif in ATP and GTP-binding proteins.Trends in Biochemical Sciences, 15, 430–434.
Sǿrensen J G, Kristensen T N, Loeschcke V. 2003. The evolutionary and ecological role of heat shock proteins.Ecology Letters, 6, 1025–1037.
Sun M, Lu M X, Tang X T, Du Y Z. 2014. Molecular cloning and sequence analysis of the HSP83 gene inSesamia inferens(Walker) (Lepidoptera: Noctuidae).Chinese Journal of Applied Entomology, 51, 1246–1254. (in Chinese)
Tabaska J E, Zhang M Q. 1999. Detection of polyadenylation signals in human DNA sequences.Gene, 231, 77–86.
Tomanek L, Somero G N. 1999. Evolutionary and acclimationinduced variation in the heat-shock responses of congeneric marine snails (genusTegula) from different thermal habitats:Implications for limits of thermotolerance and biogeography.Journal of Experimental Biology, 202, 2925–2936.
Wang H H, Lei Z L. 2005. Current developments of heat shock proteins in insect.Scientia Agricultura Sinica, 38,2023–2034. (in Chinese)
Wang H H, Reitz S R, Wang L X, Wang S Y, Li X, Lei Z R. 2014.The mRNA expression pro files of five heat shock protein genes fromFrankliniella occidentalisat different stages and their responses to temperatures and insecticides.Journal of Integrative Agriculture, 13, 2196–2210.
Welch W J. 1992. Mammalian stress response: Cell physiologt,structure/function of stress proteins, and implications for medicine and disease.Physiological Review, 72,1063–1081.
Zhang Q, Denlinger D L. 2010. Molecular characterization of heat shock protein 90, 70 and 70 cognate cDNAs and their expression patterns during thermal stress and pupal diapause in the corn earworm.Journal of Insect Physiology,56, 138–150.
Zhang Y J, Wu Q J, Xu B Y, Zhu G R. 2003. The occurrence and damage ofFrankliniella occidentalis(Thysanoptera:Thripidae): A dangerous alien invasive pest in Beijing.Plant Protection, 24, 58–59. (in Chinese)
Zhao L, Jones W A. 2012. Expression of heat shock protein genes in insect stress responses.Invertebrate Survival Journal, 9, 93–101.
Zheng Y T, Li H B, Lu M X, Du Y Z. 2014. Evaluation and validation of reference genes for qRT-PCR normalization inFrankliniella occidentalis(Thysanoptera: Thripidae).PLoS ONE, 9, e111369.
 Journal of Integrative Agriculture2018年5期
Journal of Integrative Agriculture2018年5期
- Journal of Integrative Agriculture的其它文章
- Detection and characterization of an isolate of Tomato mottle mosaic virus infecting tomato in China
- Genes encoding heat shock proteins in the endoparasitoid wasp,Cotesia chilonis, and their expression in response to temperatures
- Molecular mechanisms controlling seed set in cereal crop species under stress and non-stress conditions
- Rapid semi-quantification of triacylglycerols, phosphatidylcholines,and free fatty acids in the rice bran of one grain
- lnfluence of different nitrogen application on flour properties,gluten properties by HPLC and end-use quality of Korean wheat
- Evaluation indices of sour flavor for apple fruit and grading standards
