Reassembly of the axon initial segment and nodes of Ranvier in regenerated axons of the central nervous system
Reassembly of the axon initial segment and nodes of Ranvier in regenerated axons of the central nervous system
Myelinated axons of the peripheral and central nervous system (PNS & CNS) are divided into molecularly distinct excitable domains, including the axon initial segment (AIS) and nodes of Ranvier.e AIS is composed of a dense network of cytoskeletal proteins, cell adhesion molecules, and voltage gated ion channels and is located at the proximal most region of the axon (Kole and Stuart, 2012).e primary functions of the AIS are to both maintain neuronal polarity by regulating vesicular traf fi c across the somatodendritic and axonal domains as well as initiate the action potential when sufficiently depolarized by integrated postsynaptic potentials (Zollinger et al., 2015). Nodes of Ranvier, which are structurally similar to the AIS, are found adjacent to myelin sheaths and are interspersed at regular intervals along the axon. Enriched with voltage gated ion channels, the primary function of the node is to regenerate the action potential as it propagates along the myelinated axon away from the soma (Rasband and Peles, 2016). Thus, AIS and nodes play critical roles in normal neural physiology and their specif i c disruption has been implicated as a common pathology in a wide spectrum of neurological disorders including stroke, epilepsy, and traumatic injury (Schafer et al., 2009; Baalman et al., 2013).
These remarkable observations have set the stage for the development of future therapeutic strategies and provide a platform to answer deeper questions about CNS axon regeneration. In particular, a major unanswered question is whether regenerating CNS axons become remyelinated and reassemble their excitable domains. We recently answered this question by examining myelin, AIS and nodes in an optic nerve crush injury and regeneration model (Marin et al., 2016).
In our study, we fi rst def i ned the time course of excitable domain disassembly after optic nerve crush. We found that loss of AIS and nodes occurs shortly aer crush: within 12 hours of crush, regions within and immediately proximal and distal to the injury site were almost completely devoid of nodes. In the retina we observed a signif i cant decrease in the length of RGCAIS within 3 days, which was then followed by a signif i cant decrease in AIS density by one week aer crush injury. By 30 days aer crush injury, AIS and nodes were almost completely gone in the retina and optic nerve. Previous studies show that loss of AIS and nodes is due to calcium-dependent calpain mediated proteolysis (Schafer et al., 2009).
Following our analysis of excitable domain disassembly after optic nerve crush, we next analyzed AIS and node reassembly in the regenerating retina and optic nerve of the PTENf/f+zymosan+CPT-cAMP regeneration mouse model. Brief l y, RGCs were transduced using an adeno-associated virus (AAV) expressing Cre recombinase 2 weeks prior to optic nerve crush. Immediately aer crush, a single bolus of zymosan and CPT-cAMP was injected intravitreally. For longer survival times, an additional bolus of zymosan and CPT-cAMP was given every 3 weeks. Axon regeneration, and AIS and node reassembly were assessed at 2, 6, and 12 weeks after crush. Remarkably, we found new nodes, paranodal junctions indicative of remyeliantion, and AIS in the regenerating optic nerve and retina 6 weeks aer crush injury (Figure 1). Despite extensive axon regeneration, node reassembly was limited to the proximal optic nerve. Twelve weeks after nerve crush node, reassembly extended into areas distal to the crush site, indicating that node reassembly progresses in a proximal to distal direction down the optic nerve, and that remyelination and reassembly of excitable domains is a very protracted process (Figure 2).
We also determined if AIS were reassembled in the retina aer axon regeneration. Flatmount retinas were immunostained for βIV spectrin and analyzed at 2, 6, and 12 weeks aer optic nerve crush. We found that retinas were mostly devoid of AIS 2 weeks aer crush. However, we found an increase in both AIS density and average AIS length 6 weeks aer injury. Together, these results suggest that neuronal polarity is re-established and that the cytoskeletal and ion channel protein complexes necessary for action potential initiation and propagation are reassembled. However, further studies are required to determine whether RGCs with regenerating axons develop normal electrophysiological properties.
Although reassembly of nodes suggests that regenerating axons become remyelinated, it is dif fi cult to demonstrate this fact using electron microscopy since myelin debris is not ef fi ciently removed aer CNS injury. Instead, we used immunostaining for caspr, a marker for the paranodal junction. Paranodal junctions form between myelinating glial cells and the axon and are an excellent surrogate marker for myelination. In parallel to these immunofluorescence studies, we also analyzed remyelination of regenerated axons defined by labeling with cholera toxin B conjugated to horseradish peroxidase (CTB-HRP) using electron microscopy (EM). Consistent with our studies using antibodies against the paranodal junction, we found myelinated, CTB-labeled axons.is EM analysis shows that regenerating axons may be remyelinated as far as 2.5 mm past the injury site. Together, these data demonstrate that regenerated axons can be remyelinated and that proper axo-glial interactions are reestablished.
Functional recovery is the ultimate goal of axon regeneration. Thus, any therapeutic approach aimed at functional recovery will require the re-establishment of nodes and AIS. Our results are the fi rst to show that this is possible. As we continue searching for genetic and pharmacological mechanisms that bring about functional recovery in axon regeneration models, it is critical that we remain mindful of the physiology of these axons, especially those processes that underlie the electrogenicity of the neuron. Recent evidence suggests that neural activity drives mechanisms central to neuronal repair including axon outgrowth and remyelination (Stevens et al., 2002; Gibson et al., 2014; Lim et al., 2016).us, the reassembly of excitable domains in regenerating axons may underpin downstream repair processes. The discovery and use of optic nerve regeneration models provides new and unprecedented opportunities to beginanswering critical questions about the viability and ef ficacy of strategies to promote regeneration of injured CNS axons.
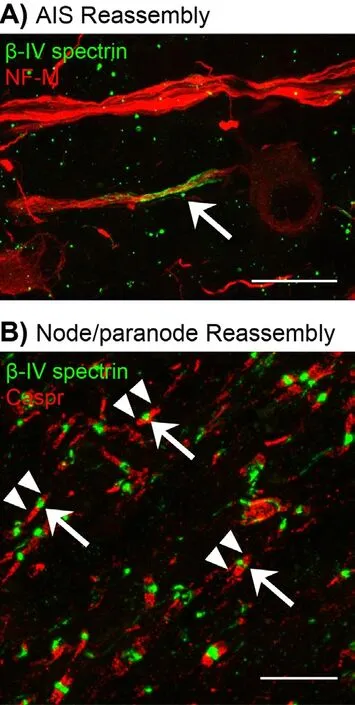
Figure 1 Reassemly of excitable domains in regenerated axons of the CNS.

Figure 2 Disassembly and reassembly of excitable domains in regenerated axons of the optic nerve is a protracted process.
Miguel A. Marin*,†, Matthew N. Rasband
对于配电设施,小农水项目资金只能用于低压电缆设备的购置安装,变压器及高压线需要电力部门配合完成。由于个别县在项目区选择时整合资金项目没有真正落实到位,井灌工程完成后,变压器以上设施配套跟不上,使小农水工程不能及时发挥效益。
Department of Neuroscience, Baylor College of Medicine, Houston, TX, USA
†Present address: Department of Neurology, David Gef f en School of Medicine, University of California Los Angeles, Los Angeles, CA, USA
*Correspondence to: Miguel A. Marin, Ph.D., mamarin87@gmail.com.
orcid: 0000-0002-5812-8715 (Miguel A. Marin)
Accepted:2017-07-26
How to cite this article:Marin MA, Rasband MN (2017) Reassembly of the axon initial segment and nodes of Ranvier in regenerated axons of the central nervous system. Neural Regen Res 12(8):1276-1277.
Plagiarism check: Checked twice by ienticate.
Peer review: Externally peer reviewed.
BaalmanKL, Cotton RJ, Rasband SN, Rasband MN (2013) Blast wave exposure impairs memory and decreases axon initial segment length. J Neurotrauma 30:741-751.
Baldwin KT, Carbajal KS, Segal BM, Giger RJ (2015) Neuroinflammation triggered by β-glucan/dectin-1 signaling enables CNS axon regeneration. Proc Natl Acad Sci U S A 112:2581-2586.
Benowitz LI, He Z, Goldberg JL (2016) Reaching the brain: Advances in optic nerve regeneration. Exp Neurol 287:365-373.
de Lima S, Koriyama Y, Kurimoto T, Oliveira JT, Yin Y, Li Y, Gilbert HY, Fagiolini M, Martinez AM, Benowitz L (2012) Full-length axon regeneration in the adult mouse optic nerve and partial recovery of simple visual behaviors. Proc Natl Acad Sci U S A 109:9149-9154.
Galiano MR, Jha S, Ho TS, Zhang C, Ogawa Y, Chang KJ, Stankewich MC, Mohler PJ, Rasband MN (2012) A distal axonal cytoskeleton forms an intra-axonal boundary that controls axon initial segment assembly. Cell 149:1125-1139.
Gibson EM, Purger D, Mount CW, Goldstein AK, Lin GL, Wood LS, Inema I, Miller SE, Bieri G, Zuchero JB, Barres BA, Woo PJ, Vogel H, Monje M (2014) Neuronal activity promotes oligodendrogenesis and adaptive myelination in the mammalian brain. Science 344:1252304-1252304.
Kole MH, Stuart GJ (2012) Signal processing in the axon initial segment. Neuron 73:235-247.
Lim JH, Staf f ord BK, Nguyen PL, Lien BV, Wang C, Zukor K, He Z, Huberman AD (2016) Neural activity promotes long-distance, target-specific regeneration of adult retinal axons. Nat Neurosci 19:1073-84
Marin MA, de Lima S, Gilbert HY, Giger RJ, Benowitz, L, Rasband MN (2016) Reassembly of excitable domains aer CNS axon regeneration. J Neurosci 36:9148-9160.
Rasband MN, Peles E (2016)e nodes of Ranvier: molecular assembly and maintenance. Cold Spring Harb Perspect Biol 8:a020495.
Schafer DP, Jha S, Liu F, Akella T, McCullough LD, Rasband MN (2009) Disruption of the axon initial segment cytoskeleton is a new mechanism for neuronal injury. J Neurosci 29:13242-13254.
Stevens B, Porta S, Haak LL, Gallo V, Fields RD (2002) Adenosine: a neuron-glial transmitter promoting myelination in the CNS in response to action potentials. Neuron 36:855-868.
Zollinger DR, Baalman KL, Rasband MN (2015)e ins and outs of polarized axonal domains. Ann Rev Cell Dev Biol 31:647-667.
10.4103/1673-5374.213543
- 中国神经再生研究(英文版)的其它文章
- Transcriptional inhibition in Schwann cell development and nerve regeneration
- A progressive compression model of thoracic spinal cord injury in mice: function assessment and pathological changes in spinal cord
- Effects of estrogen receptor modulators on cytoskeletal proteins in the central nervous system
- Optogenetics and its application in neural degeneration and regeneration
- Live-cell imaging: new avenues to investigate retinal regeneration
- Neurotrophic factors and corneal nerve regeneration