Effect of single Si1-xCxcoating and compound coatings on the thermal conductivity and corrosion resistance of Mg-3Sn alloy
*
College of Materials Science and Engineering,Sichuan University,Chengdu 610064,China
Effect of single Si1-xCxcoating and compound coatings on the thermal conductivity and corrosion resistance of Mg-3Sn alloy
Chunming Wang,Sufen Xiao*,Yungui Chen,Xia Liu
College of Materials Science and Engineering,Sichuan University,Chengdu 610064,China
A single Si1-xCxcoating and compound coatings were deposited on Mg-3Sn matrix alloy by magnetron sputtering method.Compound coatings included Mg or Mg/AlTi intermediates between Mg-3Sn substrate and Si1-xCxcoating.The thermal conductivity of the Mg-3Sn alloy after coating was enhanced at room temperature.The results showed that the Mg-3Sn alloy coated with Mg/AlTi/Si1-xCxdisplayed higher thermal conductivity,its thermal conductivity after corrosion was 90.1 W/(m K)and 108.4 W/(m K)at 25°C and 100°C,respectively. Meanwhile,it was revealed that the Mg/Si1-xCxand Mg/AlTi/Si1-xCxcompound coatings had nobler Ecorrand much lower icorr,higher Rp, compared with the bare Mg-3Sn and Mg-3Sn/Si1-xCxsystem,and improved the corrosion resistance of the magnesium substrate.
Si1-xCxcoating;Compound coatings;Thermal conductivity;Corrosion resistance
1.Introduction
Magnesium alloys are potential heat dissipation materials for their suitable mechanical properties,electromagnetic shielding performance,light weight and high thermal conductivity.However,the low corrosion resistance of magnesium alloys limits their practical applications in humid environment. In order to protect magnesium alloys from corrosion,many surface treatments for magnesium alloys were provided[1-7]. The corrosion resistance and tribological property of the coatings for magnesium alloys were fully studied.However, few papers concerned about thermal properties[8],especially the high heat transfer characteristic.It is well-known that pure magnesium possesses a high thermal conductivity of 156 W/ m K at room temperature.However,the magnesium alloys are easily to be corroded in wet environment,causing some products,such as Mg(OH)2(4.5 W/m K),MgO(36 W/m K), SnO2(40 W/m K)[9-12].These corrosion products cover the surface of magnesium alloys,forming a barrier layer to lower the heat-conducting property.Therefore,it is necessary to study on thermal conductivity of coated magnesium alloy or search for high heat-conducting coatings.
SiC is well known for the high thermal conductivity and electrical resistivity,good corrosion resistance and optical performance,it may counterbalances the disadvantages of magnesium alloy by depositing SiC flm on magnesium alloys. J.S.Goela et al.[13]reported the thermal conductivity of CVD-SiC reached up to 300-374 W/m K at room temperature.To maintain or improve the thermal conductivity of magnesium alloys used in severe environment,a high heatconducted SiC flm was deposited on Mg-3Sn alloy by RF magnetron sputtering in this work.Nevertheless,adhesion between ceramic flm and metallic substrate was poor due totheir different structure,elastic modulus and line-expansion coeffcient[14].And the poor adhesion performance may decrease the thermal conductivity of coated magnesium alloy. In order to enhance the adhesion performance between Mg-3Sn substrate and SiC ceramic flm and understand the effect of intermediate between top ceramic coating and magnesium alloy substrate on the thermal conductivity of coated magnesium alloy,a intermediate of Mg and AlTi was produced.
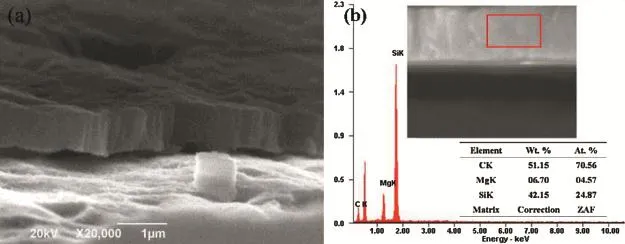
Fig.1.The cross section SEM micrograph and EDS result of Si1-xCxflm deposited on the Mg-3Sn substrate.
2.Experimental details
As-cast Mg-3Sn alloy(10 mm×10 mm)which contain 3 wt.%Sn was used as substrate in this study.Before sputtering,the polished samples were ultrasonic cleaning in acetone and alcohol for 5 min,respectively,and dried in warm air.All flms were deposited using RF magnetron sputtering method.A SiC,Mg and TiAl target were used for top Si1-xCxceramic flm and intermediate layers.The sputtering time was2.5 h for each layer.The flms were deposited using the following process parameters:base pressure is 4.2×10-3Pa, gas pressure is 1.5 Pa,RF power is 200 W,substrate temperature is 180°C.
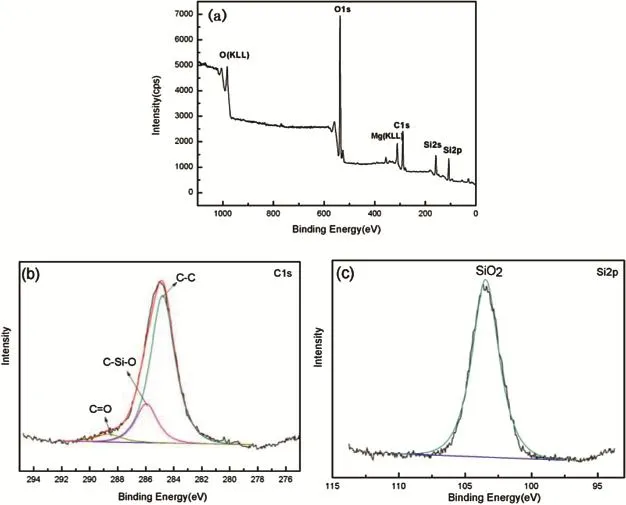
Fig.2.XPS spectra of Si1-xCxflm(a)on Mg-3Sn substrate and C1s(b)and Si2p(c)XPS region scan patterns.
The microstructure and chemical analyses of the sputtered flm were performed using a scanning electron microscopy (SEM)with an energy dispersive spectrometer(EDS).The chemical bindings within the Si1-xCxflm were employed using X-ray photoelectron spectroscopy(XPS)with a monochromatic AlKα(1486.6 eV)line of an X-ray source,and the measurements were taken in an ultra-high vacuum environment.Binding energy positions were verifed using the C 1s peak at 284.8 eV,which was associated to C-C and/or C-H bindings.The peaks were adjusted using Gaussian curves and the background was determined by the Shirley method.Besides,the thermal conductivity measurements of bare or coated magnesium alloy before and after salt spray test were performed with a NETZSCH model LFA447 Flash Analyzer. In order to research the difference of thermal conductivity between bare and coated magnesium alloys and the changes of thermal conductivity in different corrosion condition and temperatures.The bare and coated magnesium alloy samples were made into Φ12.9 mm×2.9 mm,and some of them (exposure area:Φ12.9 mm)were corroded for 4 days using salt spray test in intermittent hand spraying model(Spray 2 h; Pause 22 h).The thermal conductivity of samples was measured at 25°C and 100°C,respectively.In addition, potentialdynamic polarization experiments were done using an EG&G potentiostat model 2273.Three-electrode system was used with the samples as working electrode,platinum as counter electrode and saturated calomel electrode(SCE)as reference electrode.Work electrodes were embedded into epoxy resin with an exposure area of 10 mm×10 mm,the scan rate for potentiodynamic polarization curves was 1 mV/s. The corrosive medium for salt spray test and polarization experiments was 5 wt.%and 3.5 wt.%NaCl solution (analytical reagent),respectively.
3.Results and discussion
3.1.Characteristics of Si1-xCxflms and intermediate layers
Fig.1 shows the cross section morphology(a)and EDS(b) result of Si1-xCxflm directly deposited on the Mg-3Sn matrix.To get the cross section of Si1-xCxflm,the sample was breaking off.As can be seen from Fig.1(a),the cross section of Si1-xCxflm is dense without any porous,and the coating's thickness is about 1 μm.However,the adhesion between Mg-3Sn substrate and thin flm is poor.The coating'sEDS result shows the flm mainly contains C and Si apart from Mg element,and the atom quantity ration between C and Si is approximate 3:1.
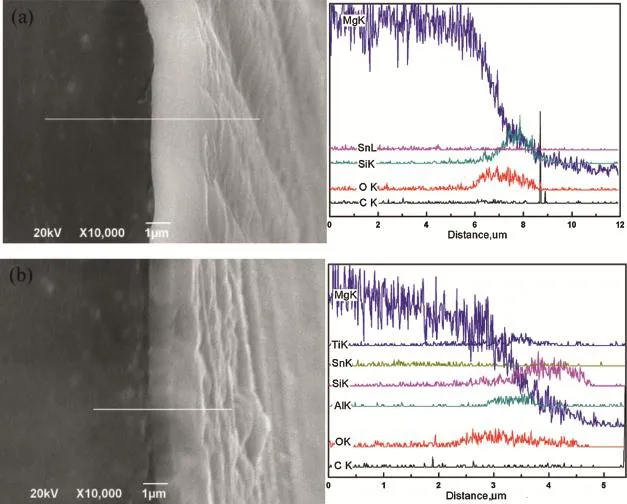
Fig.3.Cross-sectional SEM images and EDS results of(a)Mg-3Sn/Mg/Si1-xCxand(b)Mg-3Sn/Mg/AlTi/Si1-xCxsystems.
The binding nature of the Si1-xCxflm was investigated using XPS analysis.Fig.2(a)shows the XPS spectra of the coating.It is found that the Si1-xCxflm composed of C,Si, Mg and O.The element Mg in the flm formed through the matrix diffusion.Besides,there is a high concentration of O content due to the formation of oxide on the active surface of Si1-xCxflm after sputtering deposited.The C 1s spectra of the Si1-xCxflm is presented in Fig.2(b),three binging energy peaks of C 1s are 284.79 eV,285.95 eV and 288.65 eV,corresponding to the one of C 1s in C-C,C-Si-O[15],and C= O[16]chemical bond,respectively.In this spectrum,the C-C binding predominated and this result expected these chemical bonding comprised major of amorphous carbon[16,17].In addition,Fig.2(c)shows the Si 2p spectrum,the only binding energy peak of Si 2p is 103.46 eV,corresponding to SiO2formed when exposing in air.The XPS result shows the flm contains C and Si,while there isn't crystal SiC formed in the flm at the sputtering condition.Consequently,it is found that the Si1-xCxflm is a rich carbon and amorphous structure flm, which corresponds to the result of EDS in Fig.1(b).
In order to improve the adhesion and corrosion resistance of coatings,Mg and AlTi intermediate layers were sputtering deposited on the Mg-3Sn substrates.As shown in Fig.3(a), the Mg layer,as an interlayer,combined closely with Mg-3Sn substrate and Si1-xCxflm.Especially,no obvious adhesion interface of between Mg intermediate and Si1-xCxflm was observed while the difference of composition shown in EDS result can deduce the interface of them.And it means that there is a strong interface bonding.The relative EDS result shows us a gradual decline of Mg element at 6.2-8.5 μm distance,and the Si element was arisen at 7-8.5 μm while the O element was arisen at the range of 6.2-8.5 μm.The thickness of total coating is about 2.3 μm. Fig.3(b)shows the cross section micrograph and EDS analysis result of Mg-3Sn/Mg/AlTi/Si1-xCxsystems.The adhesion between substrate and coatings was improved and the interfaces of each coating were not clear.From relative EDS result,the Mg,Al,Ti,Si elements were found and changed gradually.The Si,O element was arisen at the range of 3.2-4.6 μm and 2.1-4.6 μm,respectively.The thickness of coating is about 2.5 μm.It can be inferred that an element diffusion phenomenon occurred among the interfaces of coatings from Fig.3.Meanwhile,it is found that the adhesion between compound coatings and Mg substrate is obviously improved,compared with the single SiCxcoating in Fig.1.
3.2.The thermal conductivity of bare and coated magnesium alloys
Fig.4(a)shows the thermal conductivities of Mg-3Sn alloy substrate and coated Mg-3Sn alloy with different flms at 25°C and 100°C,respectively.The thermal conductivities of coated alloys are higher than that of bare alloy at 25°C, however,the thermal conductivities decrease after coating single or compound flms at 100°C.The thermal conductivity of bare Mg-3Sn alloy is 87.3 W/(m K)at 25°C,while the thermal conductivities of the coated magnesium alloys can reach to 89.0,99.1,99.8 W/(m K)after coating with Si1-xCxflm,Mg/Si1-xCxand Mg/AlTi/Si1-xCxcompound flms, respectively.Correspondingly,the thermal conductivities of uncoated and coated alloys are 114.2,109.1,102.1,110.2 W/ (m K)at higher temperature of 100°C,respectively.
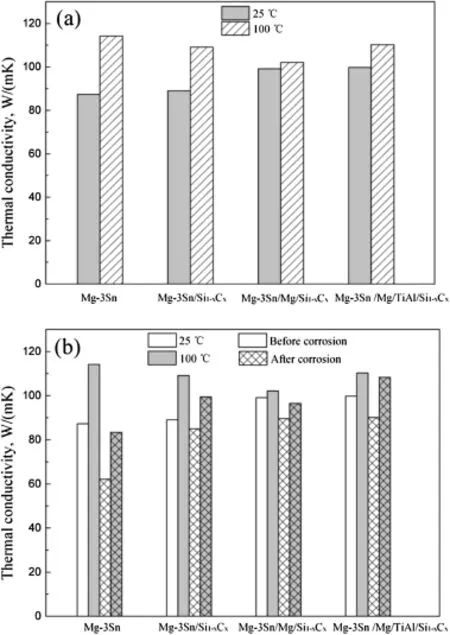
Fig.4.Thermal conductivities of uncoated and coated Mg-3Sn alloys at different temperatures(a),before and after(b)corrosion.
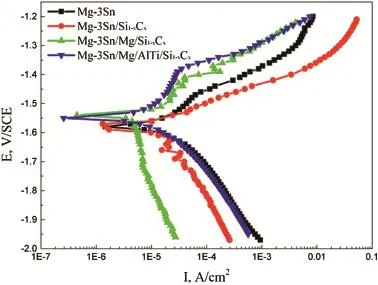
Fig.5.Polarization of bare and coated magnesium alloys with different coatings.
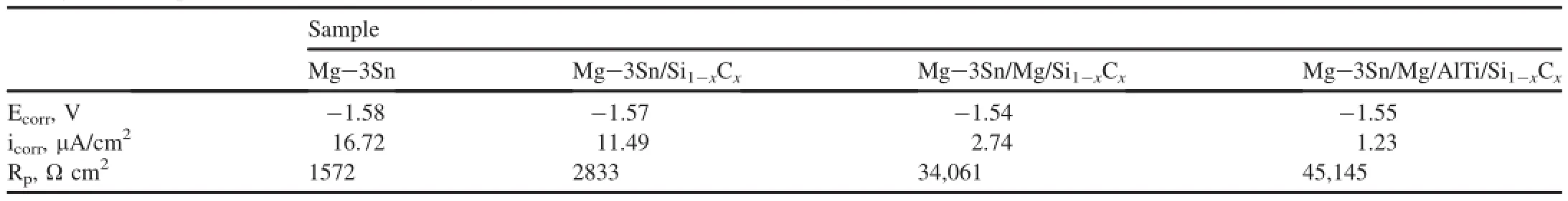
Table 1Fitting results of polarization curves of bare Mg-3Sn alloy substrate,different coatings systems in 3.5 wt.%NaCl solution.
The heat-conducting ability of the Si1-xCxflm or pure Mg, TiAl intermediates is higher than Mg-3Sn matrix,therefore the high thermal conductive coatings become a fast track which the quantity of heat can be quickly passed,and improve the thermal conductivities of coated alloys at room temperature.While the poor adhesion between magnesium alloy substrate and Si1-xCxflm(Fig.1(a))and added interfaces impede the heat transfer strongly at higher temperature, leading to decrease thermal conductivities of coated alloys, consequently.
Fig.4(b)shows the thermal conductivities of bare and coated Mg-3Sn alloys before and after salt spray test. In Fig.4(b),it is found that the thermal conductivities reduced after salt spray test for 4 days,because of the formation of corrosion products with low thermal conductivity in samples. But all thermal conductivity values of coated magnesium alloys after corrosion are higher than that of uncoated alloy (62.1 W/(m K)at 25°C and 83.4 W/(m K)at 100°C),that is because the corrosion products of coating are different from uncoated Mg-3Sn alloy.Meanwhile,it also suggests that the more the quality of coating is,the better the thermal conductivity and corrosion resistance are.After corrosion,the thermal conductivity of magnesium alloy coated with Mg/ TiAl/Si1-xCxcoating is the highest,and reaches up to 90.1 W/ (m K)at 25°C and 108.4 W/(m K)at 100°C,respectively.
3.3.Corrosion behaviors
The corrosion behaviors of Si1-xCxcoatings are investigated by potentiodynamic curves.Fig.5 shows the polarization of uncoated and coated Mg-3Sn alloy in 3.5 wt.%NaCl solution.It can been seen from Fig.5 that the corrosion potential values of Mg-3Sn/Mg/Si1-xCxand Mg-3Sn/Mg/AlTi/ Si1-xCxsystems are obviously higher than that of Mg-3Sn/ Si1-xCxsystem and Mg-3Sn alloy.For the bare Mg-3Sn alloy and Mg-3Sn/Si1-xCxsystem,an activation-controlled cathodic process occurs in the cathodic branch,and the main reaction is hydrogen evolution[12].When the applied potential increases into the anodic branch,an activationcontrolled anodic process is observed.The polarization current increased by increasing the applied anodic potential and the effect of the passivation is not obviously.But,for the Mg-3Sn/Mg/Si1-xCxandMg-3Sn/Mg/AlTi/Si1-xCxsystems,the phenomenon of clear passivation occurs in the anodic branch,and the passivation range reaches 100 and 200 mV,respectively.It is also explained that the compound coatings present an excellent corrosion resistance.
According to the polarization curves displayed in Fig.5, electrochemical parameters including corrosion current density(icorr)and corrosion potential(Ecorr)calculated using Tafel extrapolation are summarized in Table 1.It is found that the Ecorrfor coated magnesium alloys show a little positive shift than bare Mg-3Sn alloy,but the icorrarises varies remarkably with different coatings.The Mg/AlTi/Si1-xCxcomposite coatings exhibit the nobler Ecorrwith a value of-1.55 V and the lowest icorrwith a value of 1.23 μA cm-2,while bare substrate showed the lowest Ecorrwith a value of-1.58 Vand the highest icorrwith a value of 16.72 μA cm-2.The values of polarization resistance(Rp)calculate with the Stern-Geary formula,the corrosion resistanceofMg-3Sn/Mg/AlTi/ Si1-xCxsystems increases by nearly 30 times,compared with bare Mg-3Sn alloy.
However,corrosion potential value of Mg-3Sn/Si1-xCxsample is very close to the value of bare magnesium alloy due to the thinner coating's thickness of about 1-2.5 μm and the existence of the surface coating defects.In theory,ceramic coatings can separate the substrate from the aggressive environment and hence protect the substrate magnesium alloy. However,in reality,the formation of small structural defects in the coating-pinholes,pores,or cracks is almost impossible to avoid totally[18].Every defect allowing corrosion medium to contact substrate surface leads to the formation of a galvanic cell,and pitting corrosion starts[19],and decreases the corrosion resistance of Si1-xCxflm.
Therefore,from the analysis mentioned above,it can be seen that the Mg/Si1-xCxcompound coatings and Mg/AlTi/ Si1-xCxcomposite coatings both provided excellent protection for the bare substrate.The nobler Ecorrand much lower icorr, higher Rpfor the Mg/Si1-xCxand Mg/AlTi/Si1-xCxcomposite coatings indicate the better protection compared with the bare Mg-3Sn and Mg-3Sn/Si1-xCxsystem.
4.Conclusion
1.The structure of deposited Si1-xCxflm is rich carbon and amorphous.
2.The thermal conductivities of coated Mg-3Sn alloy are higher matrix alloy at room temperature because the high thermal conductive coatings become a fast track which the quantity of heat can be quickly passed,and improve the thermal conductivities of coated alloys.
3.After corrosion,the compound coatings can protect Mgalloy substrate from corrosion effectively,and the Mg/ TiAl/Si1-xCxcompound coating shows the highest heat-conducting property at corrosion condition,and the values of thermal conductivity are reaching up to 90.1 W/(m K), 108.4 W/(m K)at 25°C and 100°C,respectively.
4.The compound coatings consisted of more coatings can improve the quality of coating,and have the nobler Ecorrand much lower icorr,higher Rp,compared with the bare Mg-3Sn and Mg-3Sn/Si1-xCxsystem.So the corrosion resistance is enhanced.
[1]Y.W.Song,D.Y.Shan,E.H.Han,Electrochim.Acta 53(2008)2135.
[2]S.Y.Zhang,Q.Li,X.K.Yang,X.K.Zhong,Y.Dai,F.Luo,Mater. Charact.6(2010)269.
[3]T.Lei,C.Ouyang,W.Tang,L.F.Li,L.S.Zhou,Surf.Coat.Technol.204 (2010)3798.
[4]A.L.K.Tan,A.M.Souter,I.F.Annergren,Y.N.Liu,Surf.Coat.Technol. 198(2005)478.
[5]H.Pokhmurska,B.Wielage,T.Lampke,T.Grund,M.Student, N.Chervinska,Surf.Coat.Technol.20(2008)4515.
[6]C.Sella,J.Lecoeur,Y.Sampeur,P.Catania,Surf.Coat.Technol.60 (1993)577.
[7]G.S.Wu,X.Q.Zeng,G.Y.Li,S.S.Yao,X.M.Wang,Mater.Lett.60 (2006)674.
[8]J.A.Curran,T.W.Clyne,Surf.Coat.Technol.199(2005)177.
[9]W.M.Rohsenow,J.P.Hartnett,Y.I.Cho,Handbook of Heat Transfer, third ed.,McGraw-Hill Professional,United States of America,1998.
[10]Z.X.Shi,Effect of Inorganic Fillers on the Thermal Conductivity and Flame Resistance of Epoxy Resin,Nanjing University of Aeronautics and Astronautics,Nanjing,2011.
[11]L.Gmelin,K.Kraut,A.N.F.Naumann,Handbuch der Anorganischen Chemie,Verlag Chemie,Berlin,1967.
[12]X.B.Liu,D.Y.Shan,Y.W.Song,R.S Chen,E.H.Han,Electrochim.Acta 56(2011)2582.
[13]J.S.Goela,N.E.Brese,L.E.Burns,M.A.Pickering,MRS Bull.26(2001) 458.
[14]M.F.Chen,D.B Liu,C.You,X.J.Yang,Z.D.Cui,Surf.Coat.Technol. 201(2007)5688.
[15]Y.Zou,J.F.Du,H.Y.Dai,D.Ren,N.K.Huang,Vacuum 85(2010)26.
[16]T.Maruyama,H.Bang,N.Fujita,Y.Kawamura,S.Naritsuka, M.Kusunoki,Diam.Relat.Mater.16(2007)1078.
[17]G.Capote,L.F.Bonetti,L.V.Santos,V.J.Trava-Airoldi,E.J.Corat,Thin Solid Films 516(2008)4011.
[18]H.Altun,S.Sen,Mater.Des.27(2006)1174.
[19]F.Hollstein,R.Wiedemann,J.Scholz,Surf.Coat.Technol.162(2003) 261.
Received 1 June 2014;revised 26 November 2014;accepted 3 December 2014 Available online 21 January 2015
*Corresponding author.Tel.:+86 28 85405670;fax:+86 28 85407335.
E-mail address:xiaosf692@sohu.com(S.Xiao).
Peer review under responsibility of National Engineering Research Center for Magnesium Alloys of China,Chongqing University.
http://dx.doi.org/10.1016/j.jma.2014.12.001.
2213-9567/Copyright 2015,National Engineering Research Center for Magnesium Alloys of China,Chongqing University.Production and hosting by Elsevier B.V.All rights reserved.
Copyright 2015,National Engineering Research Center for Magnesium Alloys of China,Chongqing University.Production and hosting by Elsevier B.V.All rights reserved.
 Journal of Magnesium and Alloys2015年1期
Journal of Magnesium and Alloys2015年1期
- Journal of Magnesium and Alloys的其它文章
- Improved mechanical proprieties of“magnesium based composites”with titanium-aluminum hybrids
- Investigation on mechanical properties and creep behavior of stir cast AZ91-SiCpcomposites
- Effect of microstructure evolution on mechanical property of extruded Mg-12Gd-2Er-1Zn-0.6Zr alloys
- Effects of Bi on the microstructure and mechanical property of ZK60 alloy
- Multi-response optimization of process parameters in friction stir welded AM20 magnesium alloy by Taguchi grey relational analysis
- Polyaspartic acid as a corrosion inhibitor for WE43 magnesium alloy
