Synthesis,Structure and Characterization of Pb1-xTbxTi1-xMnxO3(0≤x≤0.10)Solid Solutions
FAN Min LI Guo-Bao WANG Dong-Wei JIN Tou-Nan,*LIAO Fu-Hui LIN Jian-Hua
(1Beijing National Laboratory for Molecular Sciences,State Key Laboratory of Rare Earth Materials Chemistry and Applications,College of Chemistry and Molecular Engineering,Peking University,Beijing 100871,P.R.China;2College of Material Science and Engineering,Beijing University of Technology,Beijing 100022,P.R.China;3National Center for Nanoscience and Technolgy,Beijing 100190,P.R.China)
Synthesis,Structure and Characterization of Pb1-xTbxTi1-xMnxO3(0≤x≤0.10)Solid Solutions
FAN Min2LI Guo-Bao1,*WANG Dong-Wei3JIN Tou-Nan2,*LIAO Fu-Hui1LIN Jian-Hua1,*
(1Beijing National Laboratory for Molecular Sciences,State Key Laboratory of Rare Earth Materials Chemistry and Applications,College of Chemistry and Molecular Engineering,Peking University,Beijing 100871,P.R.China;2College of Material Science and Engineering,Beijing University of Technology,Beijing 100022,P.R.China;3National Center for Nanoscience and Technolgy,Beijing 100190,P.R.China)
Abstract: Solid solutions of Pb1-xTbxTi1-xMnxO3(0≤x≤0.10)were synthesized by a traditional solid state reaction and characterized by powder X-ray diffraction.The solutions crystallize in theP4mmspace group at room temperature.Differential scanning calorimetry(DSC)measurements were performed to obtain phase transition temperatures(Tc)for the samples,and these were found to decrease with an increase in the amount of doped Tb and Mn.The temperature dependent dielectric constant shows a peak close to theTc,indicating that the corresponding phase transition is a ferroelectric phase transition.Magnetic measurements indicate that a paramagnetic to antiferromagnetic phase transition occurs at 25 and 29 K for Pb1-xTbxTi1-xMnxO3withx=0.08 andx=0.10,respectively.
Key Words:Solid-state reaction;PbTiO3;Phase transition;Ferroelectrics;TbMnO3
1 Introduction
The perovskite ferroelectric PbTiO3related materials have been extensively studied for their applications in the field of modern sensors,transducers,ferroelectric random access memories.1-3The properties of PbTiO3are usually modified by doping with other elements.4-16For example,the ferroelectric phase transition of PbTiO3occurs around 490°C from cubic phase(high temperature paraelectric phase)to tetragonal phase(low temperature ferroelectric phase).By doping some ions into the Pb sites and/or Ti sites of PbTiO3,the above phase transition temperature can be modified to meet the needs of certain application.In order to introduce multiferroic to the PbTiO3base materials,several attempts have been made to study the system of PbTiO3-BiFeO317-19because BiFeO3is a well known multiferroic.20Then we focus our attentions on the PbTiO3-TbMnO3system,where TbMnO3is also a well known multiferroic.21Our study shows that it is not easy to get pure phases in PbTiO3-TbMnO3system although hard efforts have been performed for a long time.Here,the synthesis,structure,dielectric and magnetic properties for the(1-x)PbTiO3-xTbMnO3with 0≤x≤0.10,are firstly presented.
2 Experimental
Pb1-xTbxTi1-xMnxO3(x=0,0.02,0.04,0.06,0.08,and 0.10,named as S1,S2,S3,S4,S5,and S6)has been synthesized from stoichiometric amounts of Tb4O7(99.99%),PbO2(A.R.),TiO2(A.R.),and MnCO3(A.R.).The weighted powders were mixed into a paste with acetone using an agate mortar and pestle,dried and heated in Al2O3crucibles at 850°C for 12 h.Finally,the samples were pressed into pellets and heated at 900°C in Al2O3crucibles in an electric muffle furnace for 5 days with several intermediate pressing and grinding steps to make sure that the obtained sample is a single phase confirmed by X-ray diffraction method.The mass of the samples was monitored before and after heat treatments.The maximum difference was about 4 mg for the 6 g samples.Therefore,the compositions of the samples were considered to be the same as the initial ones.
Powder X-ray diffraction(PXRD)data for structure refinement were collected on a Bruker D8 Advance diffractometer with Cu Kα1(λ=0.15407 nm)radiation(2θ range:10°-120°;step:0.0197°;scan speed:10 s·step-1)at 50 kV and 40 mA.The X-ray diffraction data were analyzed using GSAS software.22,23The differential scanning calorimetry(DSC)measurement was carried out on SETARAM DSC131EVO equipment from room temperature(RT)to 600°C in Ar atmosphere with a heating rate of 10 °C·min-1.The dielectric properties of the samples were measured using Precision Impedance Analyzer 65120B(Wayne Kerr Electronics).The magnetic properties were investigated with a Quantum Design physical property measurement system(PPMS)from 2 to 300 K.
3 Results and discussion
3.1 X-ray diffraction analysis
The powder X-ray diffraction patterns of Pb1-xTbxTi1-xMnxO3(x=0.00,0.02,0.04,0.06,0.08,and 0.10)are similar to each other as shown in Fig.1(a).This implies that the structures of this series samples are the same.The structure of PbTiO3was used as the beginning model for the Rietveld refinement of the X-ray diffraction data of these samples.Acceptable fittings between the experimental data and the proposed models are obtained with Rp<11%,Rwp<15%(the R factor)for all the data.During the refinement,Pb and Tb atoms are at the sites 4a(0,0,z)for Pb in PbTiO3with an occupancy of(1-x):x,Ti and Mn atoms are at the sites 4b(0.5,0.5,z).Table 1 lists the refinement details of the sample S4(Pb1-xTbxTi1-xMnxO3,x=0.06)as a representative example.The corresponding Rietveld plot is shown in Fig.1(b).Linear relationship between the volume of the unit cell and the value of x in Pb1-xTbxTi1-xMnxO3is found as shown in Fig.1(c),which agrees well with Vigard′s law.24,25Therefore,it can be confirmed that a solid solution Pb1-xTbxTi1-xMnxO3(0≤x≤0.10)forms.
3.2 DSC measurement
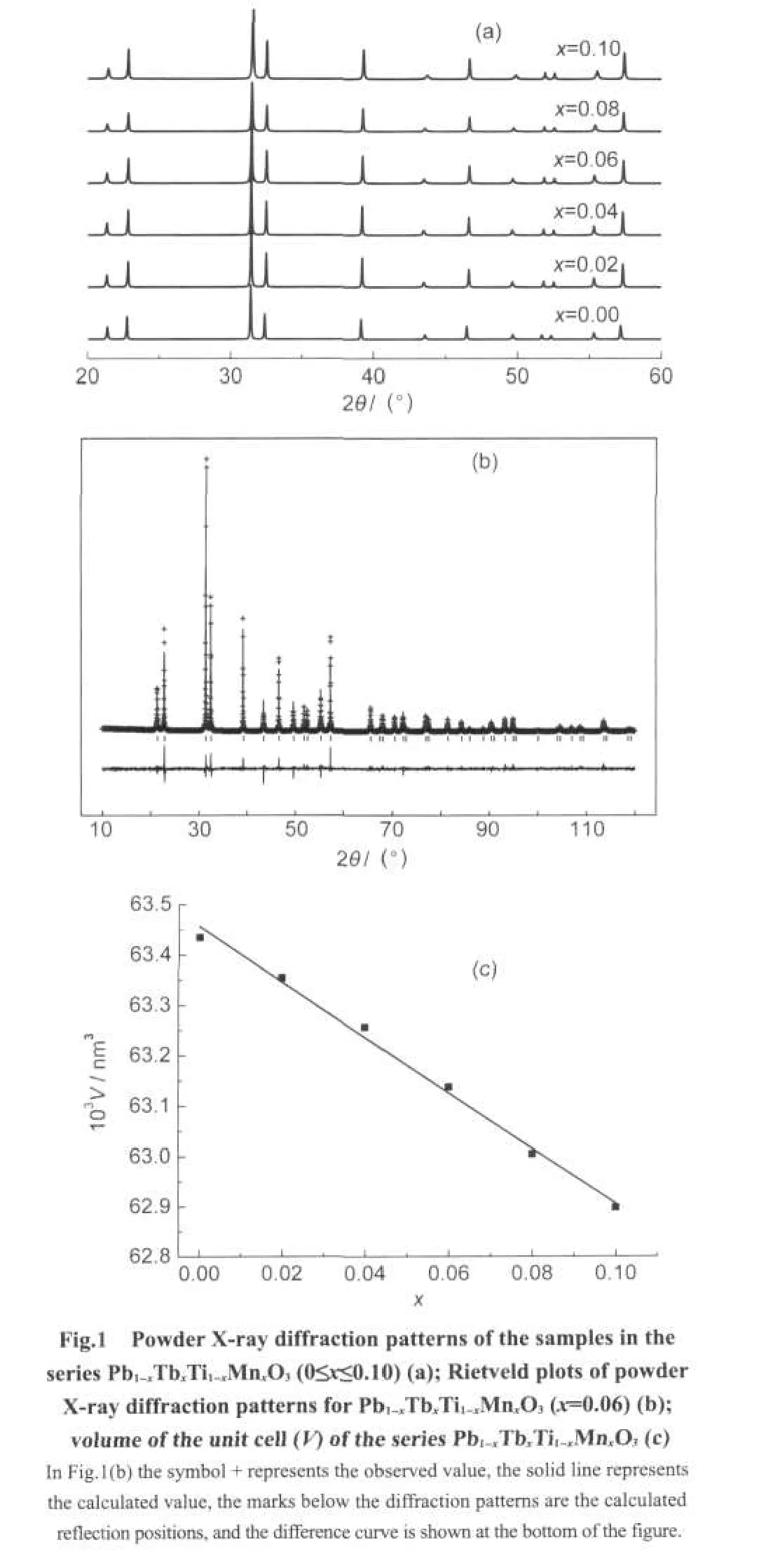

Table 1 Refinement details of Pb1-xTbxTi1-xMnxO3(x=0.06)ain space groupP4mm
As mentioned above,a solid solution Pb1-xTbxTi1-xMnxO3(0≤x≤0.10)has been synthesized.They all crystallize in the space groupP4mmat room temperature,which means that a ferroelectric phase transition may occurs above room temperature.As it is well known that the ferroelectric phase transition for PbTiO3is a first order phase transition,which should show thermal effect during the phase transition.Therefore,the DSC measurements can be performed to find the phase transition temperature(Tc)for the samples S1 to S6.Fig.2 shows the corresponding results for these samples.All the samples undergo an endothermic phase transition above room temperature.Apart from the endothermic peak atTc,no other peak/transition occurred in the measured temperature region.For PbTiO3,the present data show that there is an endothermic phase transition occurs atTc=485°C,which is consistent with the reported value26for the ferroelectric phase transition of PbTiO3.Therefore,it is reasonable to assume that the phase transition occurring atTcis a ferroelectric phase transition.When the Tb3+ion is substituted at the A-cation lattice site and the Mn3+ion is substituted at the B-cation lattice site,theTcshifts towards the lower value.It is observed that the gradual increase in the concentration of Tb3+ion at the A-cation site and Mn3+ion at the B-cation site of the host lattice decreases the transition temperature,which is shown in Fig.2.This result is different from the reported data for PbTiO3-BiFeO3system19,where when Bi and Fe are substituted into PbTiO3the ferroelectric phase transition temperature increases.
3.3 Dielectric property
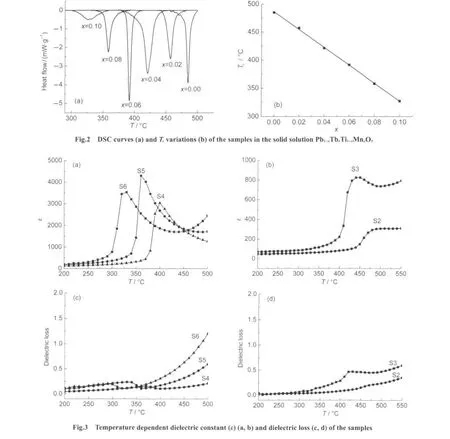
The dielectric property of the samples was measured using Precision Impedance Analyzer 65120B.The temperature dependent dielectric constants and losses of S2 to S6 are shown in Fig.3.There are peaks around the temperature corresponding to Tc,which confirms that the phase transition is a ferroelectric phase transition.The measured dielectric constants of S4 to S6 are comparable to the reported values of other doped PbTiO3,such as in the system(1-x)PbTiO3-xBiAlO3(x=0,0.05,0.10,0.15).27However,the dielectric constants of S2 and S3 are very low,which is attributed to the fact that the samplesare not very compact.The relative density of the samples is just about 0.65 for S2 and S3,and about 0.80 for S4,S5,and S6.In fact,the temperature dependent dielectric constant for PbTiO3is not given in Fig.3 because the pellet usable for dielectric measurement for PbTiO3has not been obtained,which is a known difficulty for the synthesis of PbTiO3.27The dielectric losses of these samples are similar to that reported for other similar system such as BiFeO3doped Bi(MgTi)O3-PbTiO3,18which are little higher than that for practical use.
3.4 Magnetic property
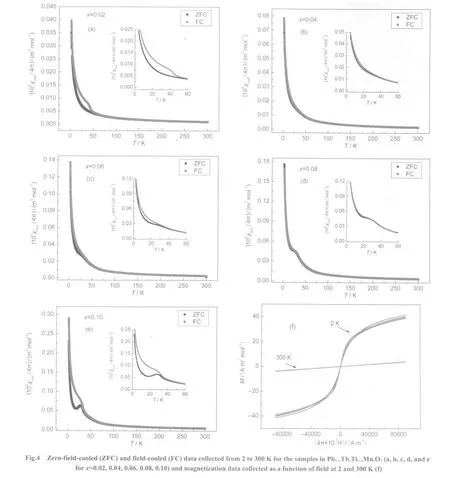
The temperature dependence of the magnetic susceptibility of the samples S2 to S6 measured in a field of 7957 A·m-1(100 Oe)is shown in Fig.4.The departure of the zero-fieldcooled and field-cooled curves for the samples S2 and S6 occurred around 42 K is attributed to the impurity of Mn3O4,28although it is difficult to find the diffraction peaks of Mn3O4in the powder X-ray diffraction patterns of S2 and S6 because the amount of Mn3O4is too less.This departure is not observed for the samples S3,S4,and S5.Therefore,it is reasonable to believe that this departure is not the property of the solid solution Pb1-xTbxTi1-xMnxO3itself.Furthermore,it is reasonable to believe that zero-field-cooled and field-cooled data are overlapped for the solid solution Pb1-xTbxTi1-xMnxO3.No loops are found in the M-H curves of the samples S2 to S6 at 2 and 300 K as shown in Fig.4(f),which confirms the above suggestion.It should be mentioned that in Fig.4(a-e),the value of the vertical axis is 4π×10-6m3per mole of Pb1-xTbxTi1-xMnxO3for each sample,and that for Fig.4(f)is 10-3A·m2per mole of(Tb+Mn).So the curves for the different samples are overlapped as shown in Fig.4(f),no effort is paid to indicate which line corresponding to S2,S3,S4,S5,or S6.
It is found that with the increase of the amount of Tb and Mn doped into PbTiO3,the peak corresponding to the antiferromagnetic ordering appears more clearly.The magnetic phase transition temperature(TN)increases from 25 K for S5 to 29 K for S6.
4 Conclusions
The samples in the system(1-x)PbTiO3-xTbMnO3(0≤x≤0.10)have been synthesized by traditional solid state reaction at 900°C.They form a solid solution with a decrease of the volume of unit cell when more Tb and Mn in the samples.They all crystallize in the space groupP4mm at room temperature.The ferroelectric phase transition temperature(Tc)decreases with the increase of Tb and Mn in the samples.In the meantime,antiferromagnetic ordering is found for the samples Pb1-xTbxTi1-xMnxO3(x=0.08,0.10)indicating that this system is a potential multiferroic.
(1) Jaffe,B.;Cook,W.R.;Jaffe,H.Piezoelectric Ceramics;New York:Academic,1971.
(2) Uchino,K.Ferroelectric Devices;New York:Marcel Dekker,2000.
(3) Scott,J.F.;Araujo,C.A.Science 1989,246,1400.
(4) Qi,T.T.;Grinberg,I.;Rappe,A.M.Phys.Rev.B 2010,82,134113.
(5) Datta,K.;Walker,D.;Thomas,P.A.Phys.Rev.B 2010,82,144108.
(6) Li,F.;Zhang,S.J.;Xu,Z.;Wei,X.Y.;Luo,J.;Shrouty,T.R.J.Am.Ceram.Soc.2010,93,2731.
(7) Wu,J.G.;Zhu,J.L.;Xiao,D.Q.;Zhu,J.G.;Tan,J.Z.;Zhang,Q.L.Thin Solid Films 2008,517,1005.
(8) Hu,P.H.;Chen,J.;Deng,J.X.;Xing,X.R.J.Am.Chem.Soc.2010,132,1925.
(9)Chen,J.G.;Qi,Y.F.;Shi,G.Y.;Yu,S.W.;Cheng,J.R.IEEE Trans.Ultrason.Ferroelectr.Freq.Control.2009,56,1820.
(10)Huang,W.;Jiang,S.W.;Li,Y.R.;Zhu,J.;Zhang,Y.;Wei,X.H.;Zeng,H.Z.Thin Solid Films 2006,500,138.
(11)Sun,C.;Wang,J.G.;Kang,H.J.;Chen,J.;Kim,M.J.;Xing,X.R.Dalton Trans.2010,39,9952.
(12) Kaneshiro,J.;Uesu,Y.Jpn.J.Appl.Phys.2010,49,09me02.
(13) Pontes,D.S.L.;Longo,E.;Pontes,F.M.;Pereira-Da-Silva,M.A.;da Silva,J.H.D.;Chiquito,A.J.;Pizani,P.S.J.Sol-Gel Sci.Technol.2010,55,151.
(14)Birks,E.;Dunce,M.;Antonova,M.;Sternberg,A.Physica Status Solidi C-Current Topics in Solid State Physics 2009,6,2737.
(15)Amorin,H.;Jimenez,R.;Ricote,J.;Hungria,T.;Castro,A.;Alguero,M.Journal of Physics D-Applied Physics 2010,43,285401.
(16)Mastelaro,V.R.;Mascarenhas,Y.P.;Neves,P.P.;Mir,M.;Doriguetto,A.C.;Michalowicz,A.;Moscovici,J.;Lente,M.H.;Eiras,J.A.J.Appl.Phys.2010,107,114103.
(17) Li,H.M.;Zhou,Y.Y.;Tian,Y.F.;Li,X.D.;Guo,H.L.;Xiao,D.Q.;Zhu,J.G.Appl.Surf.Sci.2010,257,1407.
(18) Rai,R.;Kholkin,A.L.;Sharma,S.J.Alloy.Compd.2010,506,815
(19) Ranjan,R.;Raju,K.A.Phys.Rev.B 2010,82,054119.
(20) Wang,J.;Neaton,J.B.;Zheng,H.;Nagarajan,V.;Ogale,S.B.;Liu,B.;Viehland,D.;Vaithyanathan,V.;Schlom,D.G.;Waghmare,U.V.;Spaldin,N.A.;Rabe,K.M.;Wuttig,M.;Ramesh,R.Science 2003,299,1719.
(21) Kimura,T.;Goto,T.;Shintani,H.;Ishizaka,K.;Arima,T.;Tokura,Y.Nature 2003,426,55.
(22) Larson,A.C.;von Dreele,R.B.Report LAUR 86-748 Los Alamos National Laboratory;1985.
(23) Rietveld,H.M.J.Appl.Crystallogr.1969,2,65.
(24) Vegard,L.Z.Physik 1921,5,17.
(25) Vegard,L.Z.Kristallogr.1928,67,239.
(26) Jaffc,B.;Roth,R.S.;Marzullo,S.J.Res.Nat.Bur.Stand.1955,55,239.
(27) Yu,H.C.;Ren,W.;Ye,Z.G.IEEE Trans.Ultrason.Ferroelectr.Freq.Control.2010,57,2177.
(28) Dwight,K.;Menyuk,N.Phys.Rev.1960,119,1470.
Pb1-xTbxTi1-xMnxO3(0≤x≤0.10)固溶体的合成、结构与表征
范 敏2李国宝1,*王东伟3金头男2,*廖复辉1林建华1,*
(1北京大学化学与分子工程学院,北京分子科学国家实验室,稀土材料化学及应用国家重点实验室,北京100871;2北京工业大学材料科学与工程学院,北京100022;3国家纳米科学中心,北京100190)
用固相反应合成了Pb1-xTbxTi1-xMnxO3(0≤x≤0.10)固溶体,并用X射线粉末衍射进行了表征,室温下其空间群为P4mm.热分析仪测试结果显示,随着Tb和Mn掺杂量的增加,该固溶体的相变温度Tc降低.介电常数在Tc附近出现峰值,表明对应的相变是铁电相变.磁性测量显示,当x=0.08和x=0.10时,Pb1-xTbxTi1-xMnxO3分别在25和29 K附近有顺磁性向反铁磁性的转变.
固相反应;PbTiO3;相变;铁电;TbMnO3
O641
Received:December 27,2010;Revised:February 15,2011;Published on Web:March 11,2011.
∗Corresponding authors.LI Guo-Bao,Email:liguobao@pku.edu.cn.JIN Tou-Nan,Email:tnjinkim@bjut.edu.cn.
LIN Jian-Hua,Email:jhlin@pku.edu.cn.
The project was supported by the National Natural Science Foundation of China(20771008),Scientific Research Key Program of Beijing Municipal Commission of Education,China(KM201010005019),and National Key Basic Research Project of China(2010CB833103).
国家自然科学基金(20771008),北京市教委重点基金(KM201010005019)和科技部重大研究计划(2010CB833103)资助项目

