Supramolecular complex glycoconjugate vaccine generates self-enhancement effects for carbohydrate antigen delivery
Han Lin,Yanchun Li,Kun Zhou,Dan Li,Hangyan Yu,Jie Zhao,Haofei Hong,Zhifang Zhou ,Zhimeng Wu
Key Laboratory of Carbohydrate Chemistry & Biotechnology,Ministry of Education,School of Biotechnology,Jiangnan University,Wuxi 214122,China
Keywords: Glycoconjugate vaccine Endogenous antibody APC-targeting Supramolecular complex sTn antigen
ABSTRACT Targeting delivery of tumor-associated carbohydrate antigen (TACA)-based vaccine to antigen-presenting cells (APCs) mediated by endogenous antibodies can improve the immunogenicity of TACA.However,an essential requirement of this approach is to generate high titers of endogenous antibodies in vivo through pre-immunization,which complicates the immunization procedure and may cause side effects.Herein,we report a new generation of APC-targeting TACA-based supramolecular complex vaccine,assembled by sialyl Thomsen-nouveau-bovine serum albumin-adamantine (sTn-BSA-Ada) and heptavalent rhamnose (Rha)-modified β-cyclodextrin (β-CD) via host-guest interaction.The complex vaccine retained anti-Rha antibodies recruiting capability and facilitated the APCs uptake of the vaccine via the interaction of the Fc-domain with the Fc receptors on APCs.We demonstrate that direct immunization of complex vaccine elicited anti-Rha and anti-sTn specific immune response synchronously,generating a novel self-enhancement effect that can improve the antigen delivery to APCs in high efficacy.The structure-activity relationship (SAR) study proved that complex vaccine 4 with polyethylene glycol 6(PEG6) linker in host molecule provoked a robust and specific sTn immune response comparable to the pre-immunization approach.The antisera induced by complex vaccine,either through direct immunization or pre-immunization,exhibited equal potency of cytotoxicity against the sTn expression cancer cells.This study provides a general platform for TACA-based vaccines with self-enhancement effects without the need for pre-immunization.
Tumor-associated carbohydrate antigens (TACAs) overexpressed on the surface of tumor cells,are promising targets for anticancer vaccine development [1,2].To improve the immunogenicity of TACAs,TACAs were conjugated to immunogenic carrier protein[3-5],virus-like particles [6],zwitterionic polysaccharide [7],and immunostimulatory small molecules [8-12].Of them,carrier proteins,including keyhole limpet hemocyanin (KLH),tetanus toxoid(TT),and diphtheria toxin (CRM197),were the most studied platform for TACA-based vaccine development [8].However,a carrier protein with strong immunogenicity can also induce high carrierspecific responses that may compete with and suppress the TACAspecific responses [13].These undesired immune responses,also called carrier epitope suppression and bystander interference,can attenuate the vaccine efficacy and result in clinical benefits [14].
Antigen-presenting cells (APCs) are a group of immune cells,such as macrophages,which are responsible for the uptake and proteolytic digestion of the vaccine,followed by presenting peptides to the major histocompatibility complex (MHC) to activate Tcells [15].Targeting vaccine delivery to APCs has approved to be an effective strategy to augment the immune responsesinvitroandin vivo.APC-targeting strategy mediated by natural endogenous antibodies is an unique approach that have been explored to improve the immune responses of bacterial,virus,and cancer vaccines[16-18].Mechanism studies demonstrated that a vaccine modified with haptens could recruit endogenous antibodies,such as the antibodies against galactose-α-1,3-galactose (α-Gal),rhamnose (Rha),and subsequently form aninsituimmune complex (IC) that can facilitate the APCs uptake the vaccine activelyviathe interaction of endogenous antibodies Fc-domain with the Fc receptors,such as Fcγreceptors (FcγRs) on APCs [15,19].This strategy enabled APCs to process,transport,and present more antigenic peptides for activating helper and cytotoxic T cells,leading to a more potent cellular and humoral immune response than non-targeting vaccination.However,this strategy suffers from the short supply of endogenous antibodies that usually account for approximately 1% of total IgG in human serum and the diverse binding of polyclonal antibodies [20].To overcome this shortcoming,a pre-immunization procedure,such as a three-shot of vaccination using hapten-ovalbumin(OVA) conjugate,is generally required to generate a high level of endogenous antibodiesinvivofor subsequent vaccine APCtargeting [17,21].This additional pre-immunization complicates the immunization procedure and may cause side effects.Thus,developing a next-generation APC-targeting vaccine mediated by endogenous antibodies without needing pre-immunization is an urgent need.
In this study,based on the action mechanism of APC-targeting vaccine mediated by antibodies,we designed a new glycoconjugate vaccine functionalized with multivalent Rha haptens.The advantage of such a design is to promote IC formation with anti-Rha antibodies in cluster form,leading to multivalent Fc-FcγRs interactions for efficient antigen uptake,as demonstrated in other studies [22-24].Specifically,a supramolecular complex TACA vaccine,assembled by sialyl Thomsen nouveau-bovine serum albumin-adamantine (sTn-BSA-Ada) and heptavalent Rha-modifiedβ-cyclodextrin (β-CD)viahost-guest interaction [25-27],was designed to achieve APC-targeting mediated by anti-Rha antibodies.sTn-BSA-Ada was synthesized as a guest molecule,where sTn was a classic TACA that is widely overexpressed on various carcinomas,including breast,ovarian,and colon cancer [28].At the same time,Ada on the protein was utilized to assembly with Rha-modifiedβcyclodextrin.BSA was chosen as the carrier protein due to its low immunogenicity.Heptavalent Rha-modifiedβ-CD derivatives with polyethylene glycol (PEG) linkers in different lengths betweenβ-CD and Rha were synthesized for structure-activity relationship(SAR) studies.As shown in Fig.1,supramolecular complex TACA vaccine,sTn-BSA-Ada6/CD-PEGn-Rha72-4(n=1,3,6),were conveniently assembled by simply mixing sTn-BSA-Ada and Rha-CD in aqueous solution.Biological studies demonstrated that the complex vaccine maintained an excellent ability to recruit anti-Rha antibodiesinvitroand elicited potent and specific anti-sTn immune responsesinvivocomparable to the traditional pre-immunization approach.

Fig.1.Design of APC-targeting glycoconjugate vaccine mediated by anti-Rha endogenous antibody via supramolecular complex sTn-BSA-Ada6/CD-PEGn-Rha7 vaccine.
β-CD was selected as the host molecule because of its strong binding affinity with varies guest molecules and biocompatibility [29,30].The synthesis of the heptavalent Rha-modifiedβ-CD derivatives6-8started from peracetylated rhamnose10,as shown in Scheme 1.The glycosylation of azido-substituted PEG alcohol derivatives11-13with10in the presence of BF3·Et2O gave14-16in a yield of 46%-68%.After the removal of the acetyl group and reduction the azido group under hydrogenation conditions,20-22with an amine group at the reducing end were obtained in good yield and then transformed to24-26using bis(4-nitrophenyl)carbonate23.The activated ester24-26coupled with commercially available heptakis-(6-amino-6-deoxy)-β-CD5to affordβ-CD-PEGn-Rha76-8(n=1,3,6) in a yield of 41%,47%,and 39%,respectively.
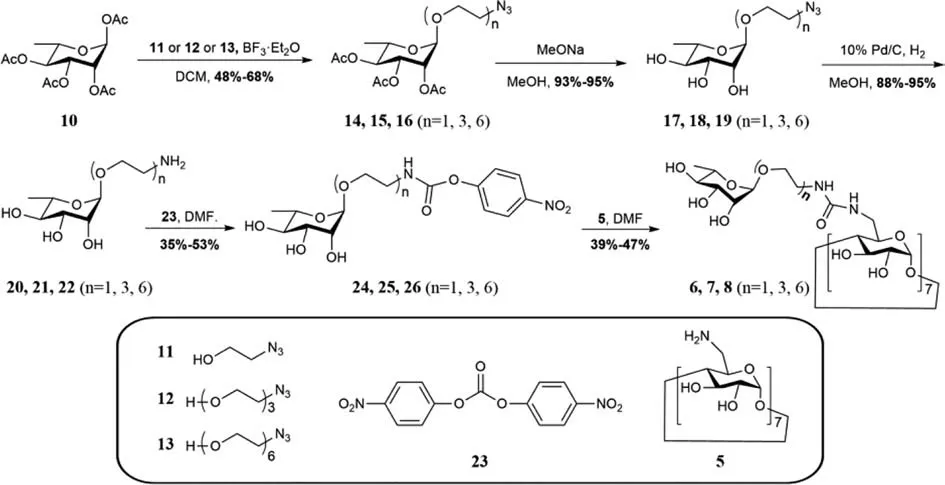
Scheme 1.Synthesis of host molecules 6-8.
Glycoconjugate sTn-BSA-Ada69was designed as a guest molecule in which Ada is selected as the guest molecule due to its high affinity toβ-CD (K=105L/mol) [31].To install Ada onto carrier protein BSA,a bifunctional maleimide-modified Ada derivative32was synthesized,where the maleimide group will be used to conjugate with sulfhydryl groups on BSA through thiol-ene chemistry.As shown in Scheme 2,32was incubated with sTn-BSA conjugate33,prepared according to our previous procedure (Scheme S1,Figs.S1,S2,S4,and S5 in Supporting information),in phosphate buffered saline (PBS) buffer (pH 7.4),to generate sTn-BSA-Ada glycoconjugate9after purification by a 10 kDa centrifugal filter device.Matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS) analysis demonstrated that glycoconjugate9was modified with approximately 6 Ada moieties and 7.5% of sTn antigen (Fig.S3 in Supporting information).
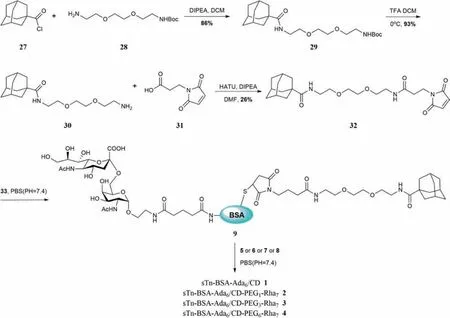
Scheme 2.Synthesis of glycoconjugate sTn-BSA-Ada6 9.
With the host and guest molecules in hand,we measured the ability ofβ-CD-PEGn-Rha76-8to include the sTn-BSA-Ada69using the isothermal titration calorimetry (ITC) method.As shown in Figs.S8-S12 (Supporting information),sTn-BSA-Ada69reacted exothermically withβ-CD orβ-CD-PEGn-Rha76-8(ΔH=-14.9,-26.1,-17.3,and -20.5 kJ/mol),whereas no heat was generated whenβ-CD was titrated to sTn-BSA devoid of Ada modification.This result demonstrated that sTn-BSA-Ada69could successfully assemble with Rha-modifiedβ-CD and the specific intermolecular interaction was driven by the Ada on sTn-BSA-Ada69and the CD moiety.In addition,the N values were 5.34,4.81,5.05,and 4.84,respectively,indicating that the binding stoichiometric ratio of host molecules and guest molecules was about 5:1.Therefore,approximately five of the heptavalent Rha-modifiedβ-CD-PEGn-Rha76-8could form supramolecular complex with one molecule of sTn-BSA-Ada69,generating as high as nearly 35 copies of Rha hapten non-covalently displaying on the surface of conjugate sTn-BSA-Ada69.Based on the above results,supramolecular complex vaccines1-4were prepared by spontaneously self-assemble guest molecule glycoconjugate9with host molecules5-8viathe hostguest interaction in solution.Then,the mixture was dialyzed with a 7 kDa dialysis membrane for 3 days to remove excess free host molecules,affording the supramolecular complex vaccines1-4(Table S1 in Supporting information).
Next,enzyme-linked immunosorbent assays (ELISA) were used to measure the IC formation between vaccines1-4and anti-Rha antibodiesinvitro.The plates were coated with PBS buffer,complex vaccines1-4(0.5 μg/mL per well),respectively,and then incubated with anti-Rha IgG antibodies serum,followed by HRPmodified secondary antibodies.As shown in Fig.S13 (Supporting information),no appreciable signal was observed in wells coated with PBS and glycoconjugate1.By contrast,strong signals were observed in the complex vaccines2-4coated wells and their absorption values were 2.2,4.7,and 3.5 times higher than that of glycoconjugate1,respectively.The ability to form IC is associated with the structure of host molecule,where a longer PEG linker between Rha andβ-CD,such as complex vaccines3and4,displayed better anti-Rha antibody recruiting ability than that of complex vaccine2.This result proved that the supramolecular complex vaccine could potentially be used to target delivery of vaccine to APCs.
To determine whether anti-Rha antibodies can mediate APCs uptake of the supramolecular complex vaccine,the macrophage THP-1 cells differentiated by phorbol 12-myristate 13-acetate(PMA) [32],were incubated with fluorescein isothiocyanate (FITC)-labeled sTn-BSA-Ada6/CD (FITC-labeled1) or FITC-labeled sTn-BSAAda6/CD-PEG3-Rha7(FITC-labeled3) in the presence of anti-Rha sera.The fluorescence of THP-1 was then detected by flow cytometry and fluorescence microscope.As shown in Fig.S14 (Supporting information),only 34% and 36.4% of cells were engulfed when treated with FITC-labeled1without Rha-modification (ii) and FITClabeled3in the absence of anti-Rha sera (iii).However,approximately 63.2% of the total cells (iv) were labeled with FITC after incubated with FITC-labeled3cooperated with anti-Rha sera.This result revealed that the anti-Rha antibodies can successfully mediate the antigen uptake.Both Rha-modification and anti-Rha antibodies were necessary to enhance of phagocytosis of supramolecular complex vaccine3by THP-1 cells.A fluorescence microscope further confirmed the result (Fig.S16 in Supporting information).These results suggested that multivalent Rha hapten modification facilitated the uptake of supramolecular complex vaccine by APCs in the presence of anti-Rha antibodies.
To evaluate the immunological activities of supramolecular complex vaccines1-4,the C57BL/6j mice were divided into seven groups (groups 1-7) (Fig.2a).The animal protocol (JN.No.20201030c0800120[276]) for this study was approved by the Laboratory Animal Center (LAC,SYXK (Su) 2016-0044) of Jiangnan University.Mice in groups 5-7 were pre-immunized with Rha-OVA(Figs.S6 and S7 in Supporting information) to establish high levels of endogenous anti-Rha antibodies (Fig.S15 in Supporting information).Then,all groups were immunized with complex vaccines1-4on Day 1 and boosts on Day 7,14,and 21.Mouse blood samples were collected and the antibodies were analyzed by enzyme-linked immunosorbent assay (ELISA).The antibody titers of seven groups in Day 28 antisera were summarized in Fig.2b.Non-APC-targeting sTn-BSA-Ada6/CD glycoconjugate1elicited the weakest anti-sTn IgG antibodies in group 1 due to no Rha modification on the vaccine and the low immunogenicity of carrier protein.APC-targeting complex vaccines2-4in groups 2-7 elicited significantly robust sTn-specific IgG antibodies.In particular,in groups 2-4,the sTnspecific antibody titers induced by sTn-BSA-Ada6/CD-PEGn-Rha72-4alone were 37,526,62,978,and 77,272,respectively,which were 4.2-,7.1-,and 8.8-fold compared of that in group 1.The titers of sTn-specific antibodies in pre-immunized groups 5-7 were 52,583,82,801,and 83,560,which were 6.0-,9.4-,and 9.5-fold of that in group 1,respectively.These results proved that the APC-targeting supramolecular complex vaccine could significantly improve the immunogenicity of sTn antigen,highlighting the importance and efficacy of the APC-targeting strategy mediated by endogenous antibodies.The PEG linker in the host molecule affects the potency of the immune response.Supramolecular complex vaccine4containing PEG6linker induced the most potent anti-sTn immune response,which was 1.2-and 1.4-fold higher than complex vaccines3and2.
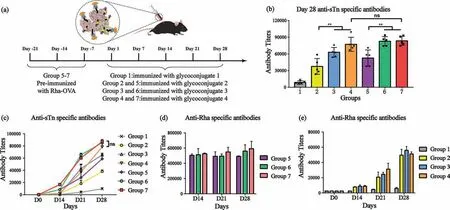
Fig.2.(a) Immunization procedure.(b) Antibody titers of different groups at Day 28.sTn-HSA was the capture antigen to detect anti-sTn specific antibodies.Each spot represents the antibody titer of each mouse.(c) The average titers of anti-sTn specific antibodies in pooled antisera collected on Day 0,14,21 and 28 from mice of groups 1-7.(d,e) Evaluations of anti-Rha specific antibodies in pooled antisera of groups 1-7 collected on Day 14,21 and 28.The mean of three parallel experiments is shown for each sample,and the error bar shows the standard error of three replicate experiments (**P <0.01;ns: no significance).HSA,human serum albumin,purchased from Shanghai yuanye Bio-Technology Co.,Ltd.
Notably,the titers of anti-sTn antibodies elicited in group 4 and group 7 were comparable with no significant difference in statistical analysis.To investigate this intriguing result,we detected the anti-sTn and anti-Rha immune response progresses in groups 2-7.As shown in Figs.2c-e,mice in groups 5-7 maintained high-titers of anti-Rha antibody (approximately 500,000)during the immunization period due to pre-immunization.Consequently,a quick and robust anti-sTn immune response was elicited when mice were vaccinated with a supramolecular complex vaccine containing Rha hapten.Interestingly,the immune responses in mice of groups 2-4 displayed different profiles,where anti-Rha and anti-sTn specific immune responses were produced synchronously.For example,low titers of anti-Rha antibodies and antisTn specific antibodies were produced at the early stage of immunization (Day 14).However,both increased dramatically at the third and fourth boost on Day 21 and Day 28.Of note,although only approximately one-tenth of anti-Rha antibody was produced in group 4 compared with pre-immunization group 7 on Day 28(titers: 51,180vs.592,374),group 4 mice vaccinated with complex vaccine4elicited the highest level of anti-sTn antibody production that is comparable to that of pre-immunization group 7.Clearly,theinsituconcomitantly generated anti-Rha antibody could form IC with complex vaccine4,generating a robust self-enhancement effect for APC-targeting antigen delivery that is as effective as the pre-immunization approach.This result suggested that the supramolecular complex vaccine could induce a self-enhancement effect,which attributes to the following facets.Firstly,as high as 35 copies of Rha hapten were displayed on the supramolecular complex vaccine in cluster form that can bind with anti-Rha antibodies in high affinityviamultivalent interactions.Secondly,such an IC can provide multivalent Fc-FcγRs interactions with APCs accordingly,which leads to an enhanced APC-targeting capability.APC-targeting vaccine delivery mediated by endogenous antibodies has been demonstrated to be a universal approach to improving the efficacy of vaccination.However,the required extra preimmunization will increase the complexity of the immunization procedure and may cause safety concerns in future medical applications.Here,for the first time,a simplified APC-targeting vaccine delivery mediated by the endogenous antibody was achieved by the supramolecular complex vaccine,although the behind mechanisms need further investigation.
The antibody isotypes and subtypes in the D28 antisera of each group were further analyzed by ELISA.As presented in Fig.3a,all groups produced higher titers of IgG over IgM antibodies,indicating that all complex vaccines provoked strong sTn-specific and T-cell dependent immune responses.The assessment of antibody isotypes revealed that mice immunized with complex vaccines1-4produced mainly IgG1 and IgG2b antibodies,suggesting that these vaccine candidates could stimulate Th1 and Th2 immune responses.These results were confirmed by the release of cytokines(interferon gamma (IFN-γ) and interleukin-6 (IL-6)) analysis (Figs.3b and c).

Fig.3.Immunological evaluation of the synthetic glycoconjugates 1-4: (a) Antibody isotypes and subtypes on Day 28.(b) The levels of IFN-γ in different groups.(c) The levels of IL-6 in different groups.Error bars represent the standard deviation (SD)of three parallel experiments (****P <0.001).
To evaluate the ability of elicited antibodies to recognize sTn antigen on cancer cells,human breast cancer cell line MCF-7 with high expression of sTn was employed as a model to perform fluorescence-activated cell sorting (FACS).As shown in Figs.4a and b,significant fluorescent peak shifts were observed in samples treated with groups 1-7 antisera compared to normal mouse serum without anti-sTn antibodies.Notably,the mean fluorescent intensity (MFI) of MCF-7 cells treated with serum from group 4 (3116) was comparable to that of group 6 (3370) and group 7 (3667),indicating that the antibodies elicited by the complex vaccine with or without pre-immunization displayed nearly equal binding affinity to cancer cells.Competitive FACS analysis of cancer cells incubated with sera from groups 1-7 in the presence of sTn(compoundS10) showed no significant fluorescence shifts were observed (Figs.4c and d).These results proved that the antibodies in sera induced by complex vaccines2-4were sTn-specific.
The anticancer activities mediated by antisera derived from groups 1-7 were evaluated by complement-dependent cytotoxicity (CDC) assay.As shown in Fig.4e,compared with the nonimmunized sera,the antisera of groups 2-7 exhibited significant CDC cytotoxicity against MCF-7 cells.Among them,sera from group 4 induced potent CDC activity comparable to that of groups 6 and 7,where the cell lysis percentage was 48%,51.5%,and 53.3%,respectively.No obvious cell death was observed when the heat inactivated rabbit complement sera (HIRS) were applied in each group,demonstrating that the observed cell-killing effect was indeed induced by antibody-mediated CDC.This result proved that antisera induced by complex vaccines,either through direct immunization or pre-immunization,exhibited similar CDC activities to promote tumor cell death.

Fig.4.Immunological evaluation of the synthetic vaccines: FACS analysis (a) and MFI (b) of the binding of antisera induced by groups 1-7 to MCF-7 cells.Competitive FACS analysis (c) and MFI (d) of antisera from groups 1-7.(e) CDC activities of antisera from each group were measured using the counting kit-8 (CCK-8) assay.Error bars represent the SD of three parallel experiments.RS represents rabbit complement sera,and HIRS represents heat inactivated rabbit complement sera (***P <0.005,****P <0.001).
In summary,a new APC-targeting TACA-based supramolecular complex vaccine was developed for cancer immunotherapy.The immunological investigation demonstrated that direct immunization supramolecular complex vaccine could produce anti-Rha and anti-sTn immune response synchronously,generating a novel and improved self-enhancement effect that can improve the antigen delivery of the complex vaccine to APCs in high efficacy.SAR results revealed that complex vaccine4with PEG6linker in the host molecule provoked a robust and specific sTn immune response comparable to the pre-immunization approach.A significant advantage of the current APC-targeting supramolecular complex vaccine with a self-enhancement effect mediated by endogenous antibodies is that it will significantly simplify the immunization procedure and generate a robust immune response without needing pre-immunization.Considering the wide application of carbohydrate antigens in vaccine research and industry,this strategy will provide a general platform to construct glycoconjugate vaccines for cancer and other disease prevention and treatment.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No.22177040),the Natural Science Foundation of Jiangsu Province (No.BK20200601).The project was partly funded by the 111 Project (No.111-2-06).
Supplementary materials
Supplementary material associated with this article can be found,in the online version,at doi:10.1016/j.cclet.2023.108670.
 Chinese Chemical Letters2024年2期
Chinese Chemical Letters2024年2期
- Chinese Chemical Letters的其它文章
- Hybrid ionic/electronic interphase enabling uniform nucleation and fast diffusion kinetics for stable lithium metal anode
- Multifunctional properties of a polar spin chain compound[N(C3H7)4][Cu(C8H4NO4)]·H2O exhibiting both one-dimensional magnetism and nonlinear optical activity
- Ti3C2Tx MXene wrapped,carbon-coated porous Si sheets for improved lithium storage performance
- Interfacial charge redistribution to promote the catalytic activity of Vs-CoP-CoS2/C n-n heterojunction for oxygen evolution
- Synergy of phosphorus vacancies and build-in electric field into NiCo/NiCoP Mott-Schottky integrated electrode for enhanced water splitting performance
- Ultrathin ternary PtNiGa nanowires for enhanced oxygen reduction reaction
