两种由萘二酰亚胺衍生物构建的配位聚合物的合成、结构和光致变色性能
李 策 郗既明 卢振中*, 张 锐 黄 岭,3
(1南京工业大学柔性电子(未来技术)学院,先进材料研究院,南京 211800)
(2宿州学院化学化工学院,宿州 234000)
(3新疆大学化学学院,乌鲁木齐 830046)
Photochromic materials of reversible color change and appropriate photo-generated color lifetime are one of the core issues for the development of inkless and erasable printing technology[1-4], which is a new concept of printing technology aiming at tackling the issue of extensive use of printing ink and printing paper[5-7].Conventional photochromic materials are usually not suitable for inkless and erasable printing because they return to the initial color within a few minutes[6].
Coordination polymers (CPs) are a new class of porous solids consisting of metal-based nodes bridged by organic linking groups, which have attracted much recent attention in many applications, such as carbon capture,molecular separation,and heterogeneous catalysis[8-13]. For some photochromic CPs, the polymeric network of CPs can prevent the accumulation of photoactive organic motifs and prolong the photo-generated color lifetime[14-17].Naphthalene diimide (NDI) and their derivatives possess redox-active cores and reversible photochromism. Thus, CPs built from NDI - derived ligands are excellent choices for obtaining valuable photochromic materials[18-20]. For example, a CP-based material, [Ca6(BINDI)3(DMA)2(H2O)4]·guest (H4BINDI=N,N′-bis(5-isophthalic acid)naphthalenediimide,DMA=N,N-dimethylacetamide), containing NDI core and 3D network was reported, which exhibited reversible photochromic behavior and photo-generated color lifetime of 72 h[16]. Another NDI-based CP material,Mg-NDI,showed reversible photochromism via an electron transfer pathway due to the presence of electrondeficient NDI moiety[21].
Here, we combined 1,4,5,8-naphthalenetetracarboxylic dianhydride with methionine and synthesized a di-topic NDI-based ligand (H2ncm). Two CPs, namely[Zn(ncm)(H2O)4]·2DMF (1) and [Zn2(ncm)2(H2O)4] (2),were synthesized with Zn2+and H2ncm through solvothermal reactions, which showed interesting structures and reversible photochromism and photo-induced color lifetime up to three weeks in air.
1 Experimental
1.1 Materials and methods
All reagents and solvents for the synthesis and analysis were of analytical grade and were used without further purification. The NMR experiment was recorded on the Bruker Advanced Triple HD 400 Spectrometer. The X-ray single crystal diffraction data was collected on a Bruker SMART APEX Ⅱdiffractometer with molybdenum-calorie radiation (λ=0.071 073 nm).Powder X-ray diffraction (PXRD) data were collected over a 2θrange of 3°-50° on a Bruker Advance D8 diffractometer using CuKα1radiation (λ=0.154 056 nm, 40 kV, 40 mA). The thermogravimetric analysis(TGA) was performed on the PerkinElmer Diamond Thermogravimetric - Differential Thermal Analysis Analyzer, and the heating rate was 5 ℃·min-1. The electron paramagnetic resonance (EPR) spectra were tested using the Bruker Magnetech ESR5000 model EPR spectrometer.
1.2 Synthesis
1.2.1 Synthesis of ligand H2ncm
H2ncm was synthesized according to the literature with some minor modifications[22]. A mixture of 1,4,5,8-naphthalenetetracarboxylic dianhydride (3.0 g, 11.2 mmol)andL-methionine(3.5 g,23.5mmol)was refluxed in 80 mL acetic acid for 3 d.The reaction was cooled to room temperature. The resulting brown mixture was concentrated under reduced pressure and diluted with an excess amount of deionized water.The resulting suspension solution was filtered and dried under a vacuum to obtain the desired product. Yield: 5.1 g (86.5%).1H NMR (400 MHz, CDCl3):δ8.72 (s, 4H, Ar—H), 5.95-5.86 (m, 2H, N—CH—COOH), 2.78-2.69 (m, 2H,CHCOOH—CHH—CH2), 2.62-2.57 (m, 4H, CH2—CH2—S), 2.43-2.35 (m, 2H, CHCOOH—CHH—CH2),2.05(s,6H,—CH3).
1.2.2 Synthesis of single crystals of compound 1
H2ncm (0.1 mmol, 0.053 g) and Zn(NO3)2(0.2 mmol, 0.038 g) were added to a mixed solvent of DMF(1.0 mL) and deionized water (1.0 mL). The mixture was ultrasonically for 5 min and then transferred to a Teflon-lined autoclave. The reaction was conducted at 90 ℃for 2 d.A brown solution was obtained and transferred into a test tube, diethyl ether (2.0 mL) was added on top of the brown solution, and the test tube was sealed. Yellow-brown crystals were obtained in two weeks. Elemental analysis Calcd. for C30H42N4O14S2Zn(%): C, 44.36; H, 5.21; N, 6.90. Found(%): C, 44.56;H,5.31;N,7.06.
1.2.3 Synthesis of single crystals of compound 2
H2ncm (0.1 mmol, 0.053 g) and Zn(NO3)2(0.2 mmol, 0.038 g) were added to a mixed solvent of DMF(2.0 mL) and deionized water (1.0 mL). The remaining steps were the same as the preparation of compound 1.Elemental analysis Calcd. for C24H24N2O10S2Zn(%): C,45.75; H, 3.84; N, 4.45. Found(%): C, 45.53; H, 3.96;N,4.56.
1.3 Single crystal X-ray diffraction analysis
The single crystal X-ray diffraction data was collected on a Bruker SMART APEX Ⅱdiffractometer with MoKαradiation (λ=0.071 073 nm). Using the SHELXS-97 program, the crystal structure was solved by the direct methods and further refined by full-matrix least squares onF2with the SHELXL program.The nonhydrogen atoms were refined anisotropically. C-bound H atoms were positioned geometrically with C—H lengths of 0.093 0-0.097 0 nm, and constrained to ride on their parent atoms withUiso(H)=1.2Ueq(C) or 1.5Ueq(methyl C). The H atoms of water were placed guided by difference maps, with O—H lengths of 0.082-0.088 nm andUiso(H)=1.2Ueq(O).
CCDC:2257324,1;2257325,2.
2 Results and discussion
2.1 Crystal structure
The asymmetric unit of compound 1 is composed of one Zn2+ion, one ncm2-ligand, four coordinated water, and two DMF solvent molecules. The Zn2+ion is in an octahedral coordination environment containing six O atoms from two ncm2-ligands and four water molecules (Fig.1). The coordinated water molecules also form hydrogen bonds with the carboxylate O atom of ncm2-ligand and the DMF solvent molecules (Fig.2),the H…O and O…O separation are in the ranges of 0.182 9(2)-0.186 2(2)nm and 0.260 9(2)-0.270 9(2)nm.
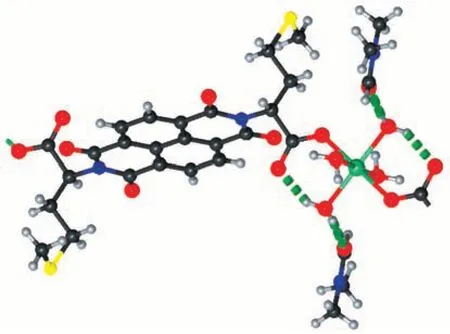
Fig.1 Coordination environment of the Zn2+ion in compound 1
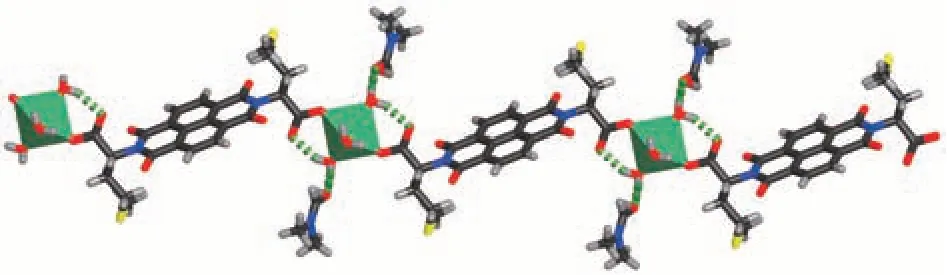
Fig.2 Chain structure of compound 1
The ncm2-ligand bridges two Zn2+ions into a linear chain with the methyl sulfide arms and hydrogenbonded DMF molecules on two sides of the chain(Fig. 2). The chains are further linked by hydrogen bonds between water and acyl O atoms of ncm2-ligands from adjacent chains into a hydrogen-bonded layer structure(Fig.3).
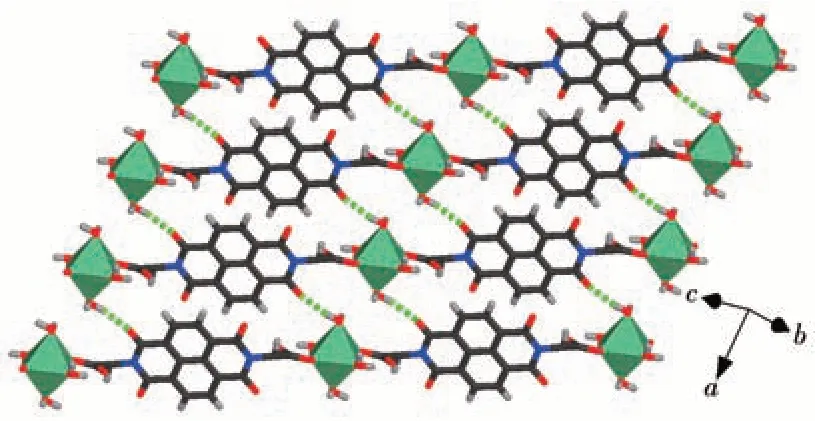
Fig.3 Chains in compound 1 linked by hydrogen bonds between water and acyl O atoms from ncm2-ligand to form a layer structure
Compound 2 contains two Zn2+ions, two ncm2-ligands, and four coordinated water molecules. The two Zn2+ions are bridged by two carboxylate groups into a binuclear Zn2unit. Each Zn2+ion is in a triangular bipyramid coordination geometry with five O atoms from three ncm2-ligands on the equatorial plane and two water molecules in the axial positions (Fig.4). The two ncm2-ligands are of different coordination modes,one ligand bridges two Zn2+ions, and the other ligand bridges four Zn2+ions (Scheme 1). The arms of the two ncm2-ligands were disordered over two positions.
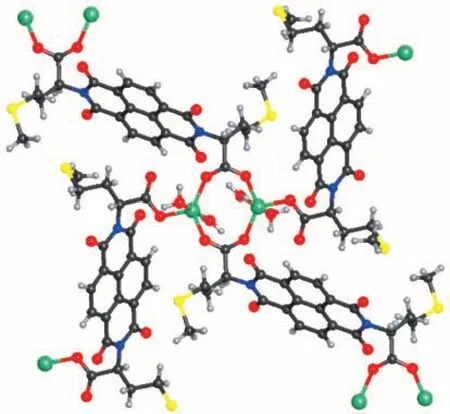
Fig.4 Molecular structure of compound 2
The ncm2-ligands in compound 2 link the Zn2binuclear units into a layer structure(Fig.5a).The adjacent layers interact with each other via hydrogen bond interaction between coordinated water molecules with the acyl O atoms of ncm2-ligands from adjacent layers(Fig.5b).
2.2 PXRD and TGA
The PXRD patterns of as-synthesized samples of compounds 1 and 2 fitted well with the simulated patterns from single-crystal X-ray diffraction results, and indicated that pure phase samples of both 1 and 2 were obtained (Fig.6a and 6b). TGA result of 1 showed a weight loss of 26% below 170 ℃corresponding to the loss of hydrogen-bonded DMF solvent and coordinated H2O molecules, and a weight loss after 270 ℃corresponding to the loss of ncm2-and collapse of the polymeric structure (Fig.6c). TGA result of 2 showed a weight loss of 6% below 130 ℃corresponding to the loss of coordinated H2O molecules and a weight loss after 300 ℃corresponding to the loss of ncm2-and collapse of the polymeric layer structure of 2(Fig.6d).
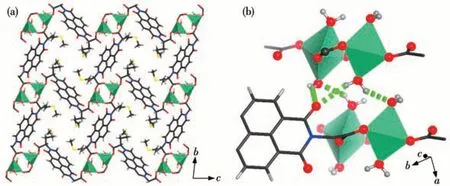
Fig.5 (a)Polymeric layer structure in compound 2;(b)Hydrogen bonds formed between adjacent layers in 2
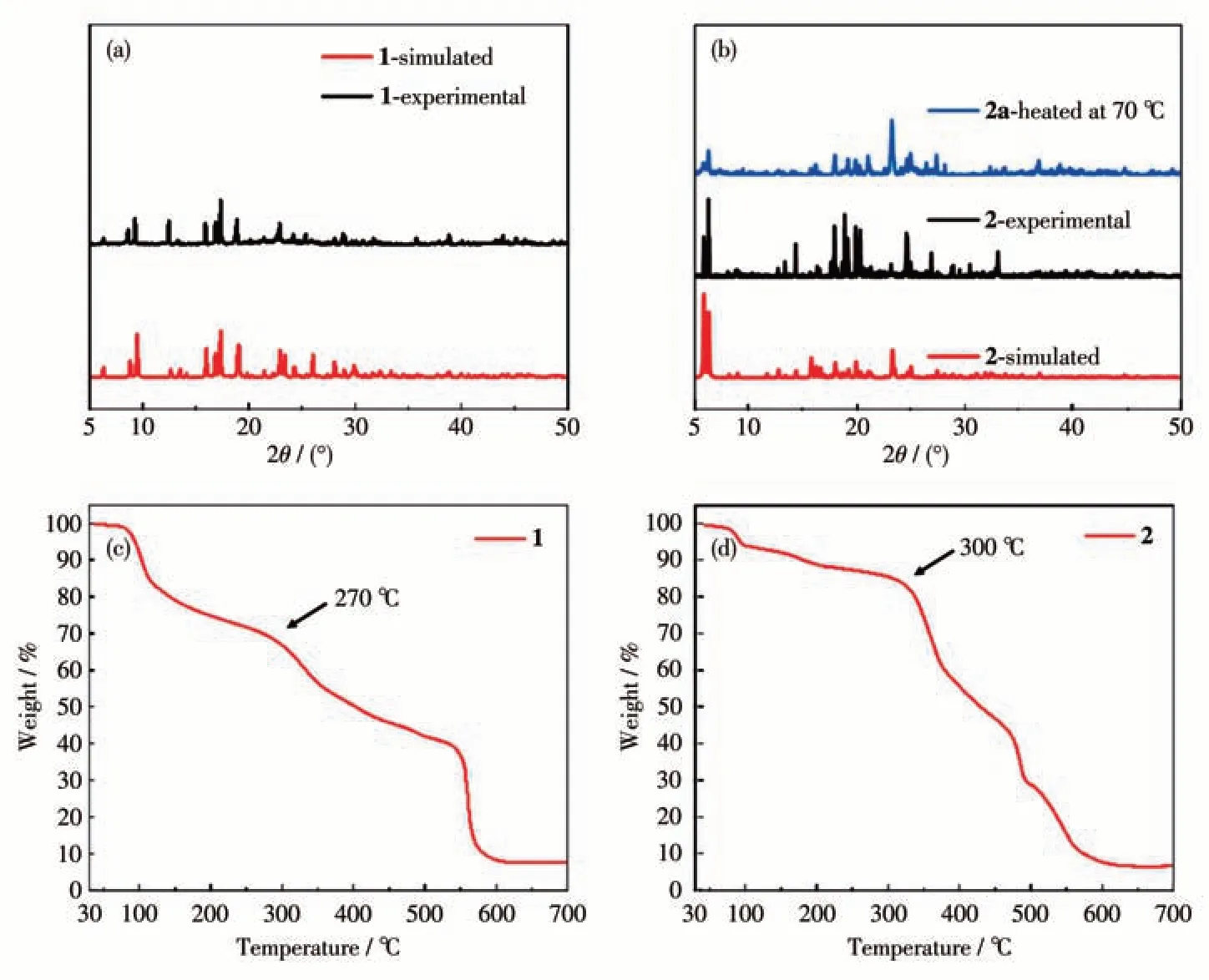
Fig.6 PXRD patterns(a,b)and TGA curves(c,d)for compounds 1 and 2
2.3 Photochromic property of compound 2
Compound 2 exhibited rapid color changes from yellow to brown in 10 min upon continuous irradiation with UV light (365 nm, the brown sample was denoted as 2a hereafter), and the color became saturated in 60 min of light irradiation (Fig.7). UV-Vis diffuse reflection spectra of 2a showed that there were new broad peaks observed at 265 and 575 nm(Fig.8a).The photogenerated brown color of 2a was stable in air for at least three weeks and can be changed back to yellow when 2a was heated at 70 ℃for 5 min (Fig.7, right).The recovered sample could also display color transformation after light irradiation without any significant deterioration, indicating that the photochromic process is reversible and repeatable. Compound 1 showed a similar photo-generated color change from yellow to dark brown,while it lost crystallinity after being heated at 70 ℃for 5 min.

Fig.7 Diagrams showing the photo-induced color change of compound 2
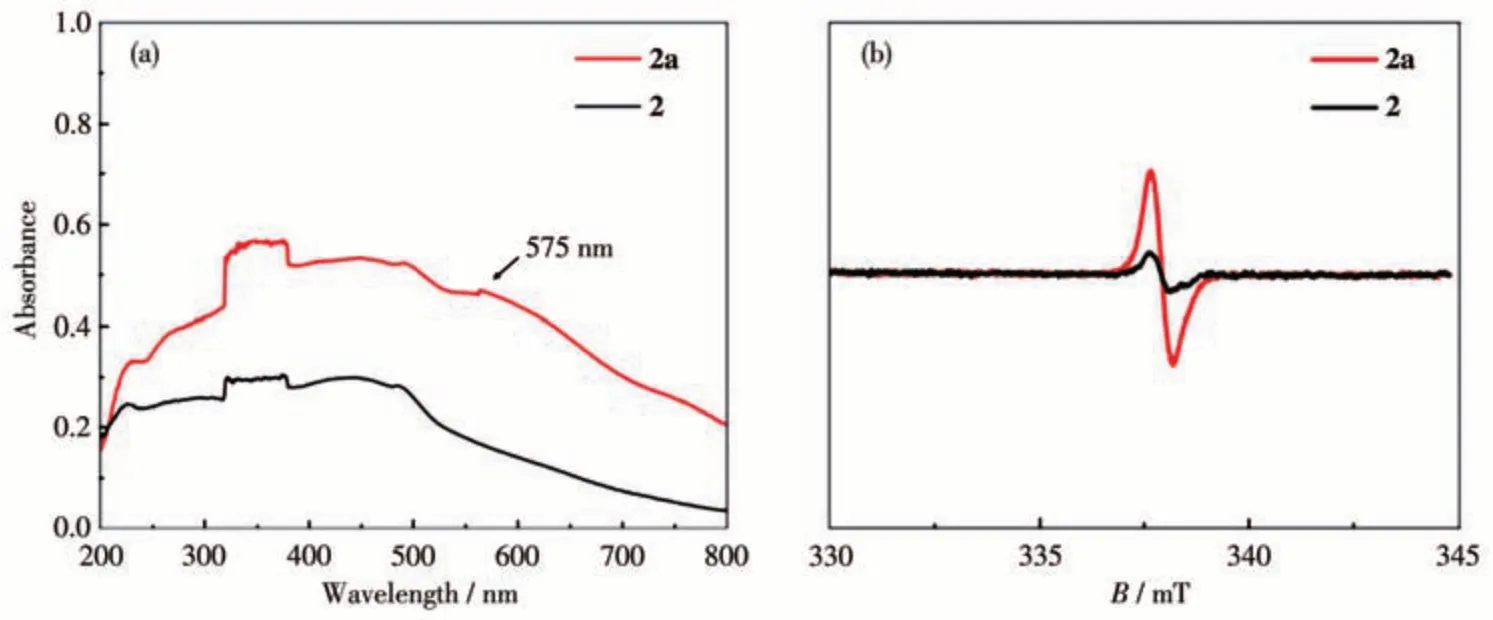
Fig.8 UV-Vis diffuse reflection and EPR spectra for 2 and 2a
It is reported that the NDI core is redox-active and can generate NDI radicals(NDI·)upon light irradiation.Therefore,the photochromic process from 2 to 2a may arise from the photo-induced radical of the ncm2-core.EPR measurement exhibited a remarkable radical signal with agvalue of 2.003 5 of 2a, which was close to the value of unpaired electrons reported in the literature.It confirmed the generation of photo-induced radicals[16,21]. Generally speaking, NDI· radical can easily be quenched by pairing with paramagnetic or single electron species (such as oxygen gas), and returns to the original color. However, the photo-generated color of 2a can be stable in air for at least three weeks. This exceptional stability should be attributed to the loosed packing of the photo-generated NDI· motifs (Fig.5a)and the extended framework in 2, which prevent NDI·radicals from direct contact with quenchers such as oxygen gas in air, and prolong the photo-generated color lifetime[14,16].
3 Conclusions
In summary, we designed a naphthalene diimidebased (NDI-based) ligand (H2ncm) by combining 1,4,5,8-naphthalenetetracarboxylic dianhydride with methionine and synthesized two coordination polymers from Zn2+and H2ncm through solvothermal reactions. One of the coordination polymers, [Zn2(ncm)2(H2O)4], contains an extended 2D layer structure and showed photoinduced color change from yellow to dark brown. The photo-induced brown color can be stable for up to three weeks in the air but change back to yellow within 5 min upon heating at 70 ℃. We showed that this photochromism originates from the generation of NDI· radical upon light irradiation. The long lifetime of the NDI· radical is probably because of the polymeric structure of the CP which effectively protects the photoinduced radical from quenching.

