Active sham cupping therapy technique:identification of limitations and suggesting enhancements
Tamer Aboushanab,Monear Qoqandi,Saad Basalom
1Department of Complementary Medicine,Saudi German Health Group,Jeddah 23521,Saudi Arabia.
Abstract
Keywords:cupping therapy; sham;placebo; standardization; clinical trial
Introduction
Cupping therapy is an ancient healing modality that has been used for thousands of years [1]. Cups are the main devices used to perform cupping therapy and a classification of cupping sets was developed and published [2]. A classification of cupping therapy types was developed that included many types of cupping therapy, such as dry cupping, wet cupping, massage (gliding) cupping, and flash cupping[2].
There are two types of sham cupping techniques: the passive type,which uses no suction, and the active type, which uses suction that is rapidly released through a hole on the top of the cup [3].
The active sham cupping device was suggested by a group of researchers in 2010 [3]. The authors suggested a cup (diameter =35.7) with a hole on the top side of the cup. The function of this hole was to gradually and completely release pressure inside the cup [3].
In this narrative review, the active sham cupping technique[3] was the main technique of concern. Various characteristics of the active sham cupping device and technique were explored, and protocols for the sham cupping technique were performed to identify the limitations. This review will have an impact on further clinical trials that use sham cupping device. It also highlighted the importance of enhancement and developing new sham cupping devices. One of the most important aims of this review is to recommend enhancements of the current active sham cupping technique.
To the best of our knowledge, this review is the first review about active sham cupping technique.
Materials and methods
Three medical databases (PubMed, Cochrane, and TRIP) were searched for the identification of clinical trials that used sham cupping device. The search Boolean was “Sham cupping”, “Sham cupping therapy”, “Cupping” OR “cupping therapy” AND “Sham”. The search strategy was modified according to the searched database. Clinical trials were selected after applying inclusion and exclusion criteria by two researchers. The inclusion criteria were as follows: (1) studies published in English,(2)studies that could be retrieved in full articles,(3) clinical trials only studies, and (4) studies that used the active sham cupping technique[3].
The exclusion criteria were as follows: (1) non-English studies, (2)studies rather than clinical trials and(3) full articles that could not be retrieved.
Results
A total of 201 published papers were retrieved. A total of 195 papers were excluded (duplication = 33, not relevant = 143, not legible =19). Finally, 6 clinical trials were selected for this review (Figure 1)[3-8].
Basic characteristics of the included clinical trials
Six trials were included in this review. All six trials recruited 340 participants, 69 males(20%)and 271 females(80%). The distribution of participants according to the treated condition was as follows:fibromyalgia = 141, chronic nonspecific low back pain = 108,nonspecific neck pain = 32, and healthy adults = 59. The six trials were conducted in four countries (USA = 2, Brazil = 2, Korea= 1,Germany = 1).
The first trial was from Korea. This was a pilot study that validated and evaluated the active sham cupping device.It was a small trial that recruited 34 participants who were divided into 2 groups (real cupping group = 17 participants and sham cupping group = 17 participants). Participants received one session of dry cupping on the back. More than 88% of participants in each group had never experienced cupping therapy, but most of them saw it [3]. The cup used for the sham cupping technique was 35.7 mm in diameter and 47.9 mm in height [3]. The diameter of the hole on the cup was 0.2 mm[3].
The second trial was from Germany.The trial investigated the use of cupping therapy for fibromyalgia patients. The study recruited 141 participants who were divided into three groups: real cupping, sham cupping, and usual care groups. The study did not recommend cupping for fibromyalgia despite the superior efficacy of real cupping and sham cupping in comparison with usual care due to the insignificant results between sham and real cupping [4].
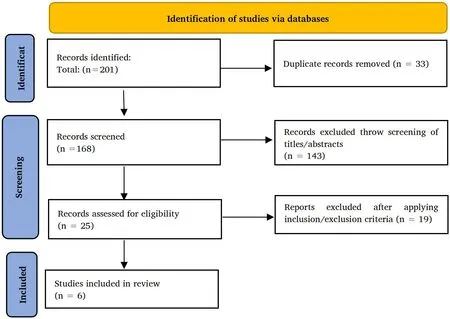
Figure 1 PRISMA chart of the identification of studies
The third trial was from Brazil. The study investigated the efficacy of cupping therapy in the treatment of nonspecific low back pain. The study had 18 participants who were divided into two groups: the real cupping and placebo (sham) cupping groups. The study reported the efficacy of a single cupping session to reduce pain and disability [5].
The fourth study was from the United States of America (USA). The study evaluated the efficacy of cupping therapy in the treatment of nonspecific neck pain. The study recruited 32 participants who were divided into three groups: real cupping, sham cupping, and control(no treatment) groups. The study reported the efficacy of a single cupping session to reduce pain in patients suffering from nonspecific neck pain[6].
The fifth trial was also from the USA. The study evaluated the efficacy of cupping therapy in influencing the range of motion of the hamstring muscle of healthy adults. The study recruited 25 participants who were divided into three groups: real cupping, sham cupping and control(no treatment).A study reported that dry cupping for 10 minutes did not influence the range of motion of the hamstring muscle in healthy adults [7].
The last included trial was from Brazil. The study investigated the efficacy of cupping therapy in the treatment of nonspecific low back pain. The study recruited 90 participants who were divided into two groups: real cupping and sham cupping. The study reported that real cupping was not superior to sham cupping [8].
Characteristics of active sham cupping procedures
Methods and procedures that were used in performing the active sham cupping technique were heterogenous in included clinical trials in this review. The dry cupping versus sham cupping technique was evaluated in all included trials.
One cupping session was performed in three clinical trials [3, 5, 6].The course of treatment used by Lauche et al. was 5 cupping sessions[4] and 3 cupping sessions used by Shafer et al. [7]. Furthermore, the course of treatment used by Silva et al. was 8 cupping sessions[8].
The frequency of repeated cupping sessions was no repetition [3-6],1 session per week [7, 8], or 2 sessions per week [4].
Additionally, the duration of cup retention on skin was not constant in selected clinical trials. Stephens et al. retained cups for 8 minutes[6], two studies retained cups for 10 minutes [7, 8], and two studies retained cups for 15 minutes [3, 5]. Lauche et al. retained cups from 10 to 15 minutes [4].
In addition, the number of cups used was heterogenous in selected clinical trials. It ranged between 1 and 8 cups. The included clinical trials used 1 cup[6],2 cups[3],4 cups[7,8],from 4 to 8 cups[4],or from 6 to 8 cups [5].
Furthermore, the cup sizes and diameters used in the selected clinical trials were heterogenous. The diameter of cups used in all included clinical trials ranged between 3.56 cm and 10 cm. The diameters of the used cups were 3.56 cm[6],3.57 cm[3],4.5 cm[8],4.57 cm [6], 5 cm [5], and 5 to 10 cm [4]. Shafer et al. did not mention the diameter of the cups used [7].
The hole on the top side of the cups was the main element in the sham cupping device because it was the main route of pressure release from inside the cup. The hole diameter was also heterogenous in the included clinical trials. It ranged between 0.2 mm and <2 mm. The reported hole diameters were 0.2 mm[3], 0.325 mm[7],0.4 mm[6],<1 mm[4], and <2 mm [8].
The time needed for the release of pressure inside the cup was also heterogonous and was mentioned in only two trials:3 seconds[8] and within 30 seconds[6].
The previous results showed heterogenicity in the procedures and methods used in selected clinical trials.
Safety of the active sham cupping technique
Three trials (50% of included clinical trials) did not report adverse events. The other three included clinical trials reported adverse events. Lee et al. reported adverse events in the sham cupping group,which included feeling of pain (1 participant), redness (5 participants), and ecchymosis (1 participant) [3]. Lauche et al.reported that 3 patients in the sham cupping group had adverse events. Two of them were serious adverse events (torn meniscus and persistent pain after spinal operation) and were referred as appropriate. The third patient had a flu [4]. Furthermore, Silva et al.reported no adverse events in a sham cupping group [8].
Success of blindness of the sham cupping technique
The success of blindness is also an important aspect of using placebo or sham devices. The success of blindness means that the participant could not identify whether she/he was in the sham cupping group or in the real cupping group.
Two trials reported the success of blindness with the sham cupping technique (33% of included clinical trials) [3, 8], while Lauche et al.reported failure of blindness with the sham cupping technique [4].Shafer et al. did not provide information about the success of blindness but reported that some participants noted the sham treatment’s decreased pressure [7]. These results highlighted the importance of reporting the success of blindness in all upcoming clinical trials that will use the sham cupping technique.
A summary of the included trials is presented in Table 1.
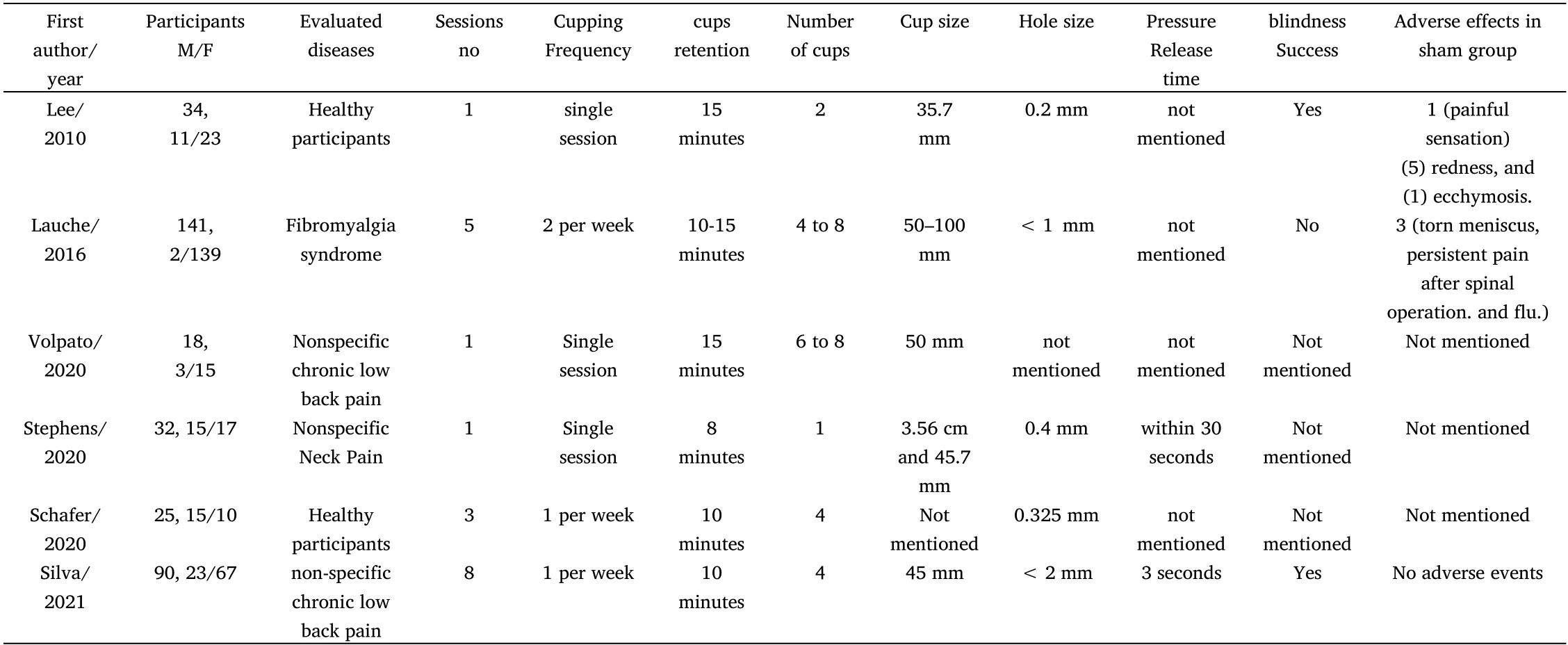
Table 1 Summary and characteristics of included clinical trials
Discussion
This review found that the reporting of the characteristics of cups that were used in the active sham cupping technique in the selected clinical trials (cup size, hole size, the time needed to release the pressure) was heterogenous. Furthermore, various cupping therapy protocols were used in the included clinical trials. In addition, the number of cups used, the duration of each session, and the time space among sessions were heterogonous. Additionally, the success of blindness was not provided in half (50%) of the included clinical trials.
The safety of cupping therapy was discussed in previously published systematic reviews [9, 10]. Previously published systematic reviews reported that cupping therapy was a relatively safe practice.Furthermore, previous systematic reviews recommended reporting adverse events in all cupping therapy clinical trials [9, 10]. A classification of cupping therapy adverse events was published in 2016 [9], updated in 2018 [2], and updated again in 2021 [11].Cupping therapy adverse events were classified into preventable and non-preventable and subcategorized into systemic and local adverse events [11]. In contrast, there was no published review about the safety of sham cupping device and techniques. This review reported the adverse events related to the sham cupping therapy technique in all included clinical trials. The overall incidence in the included trials that reported adverse events was 10/109 (0.09%). This highlighted the importance of reporting cupping therapy related adverse events,which included sham cupping therapy techniques in all clinical trials.Furthermore,these results were raising a question about the similarity of sham cupping and real cupping technique and whether sham cupping could represent a gentle type of real cupping therapy.
On the other hand, the largest cup sizes (50-100 mm) with a moderate hole size(<1 mm)were used by Lauche et al.Additionally,this trial had the closest frequency of sessions (2 per week), and its maximum number of cups used were (8). Eight cups was the highest number of cups per session used in all included clinical trials. This trial reported the superior efficacy of cupping and sham cupping in comparison with usual care despite the failure of success of blindness[4]. In addition, the study did not recommend cupping for fibromyalgia due to the insignificant results between the sham and real cupping groups[4].Furthermore,the clinical trial that conducted by Silva et al.reported that cupping was not superior to sham cupping in the management of chronic nonspecific low back pain. The trial used 4 cups, 45 mm in diameter, hole ≤2 mm, for 8 sessions, 1 per week (for 2 months), and each cup was retained for 10 minutes. This clinical trial also reported the success of blindness[8].
All the above details were rising the question about the impact of sham cupping and whether it represented a gentle type of real cupping therapy, especially if performed using large cups, delayed pressure releasing time due to small or moderate hole diameter, using a large number of cups, and performing frequent or prolonged cupping sessions. This may affect the results and the conclusion of the clinical trials due to the similar effect of real and sham cupping.
Controversially, Valpato et al. reported a positive effect of cupping on nonspecific low back pain.The study was conducted by performing a single cupping session with 6 to 8 cups, which were retained for 15 minutes [5].In addition, Stephens et al.reported the positive effect of cupping for nonspecific neck pain. The study was conducted by performing a single cupping session with one cup [6]. These results may indicate the lesser effects of sham cupping when performed as a single session and by using moderate to small cup sizes.
Limitations of the active sham cupping technique
This review identified multiple limitations of the current sham cupping technique:
(1) The reported failure of blinding of the sham cupping technique in one clinical trial [4].
(2) The heterogenicity of the characteristics of the cupping methods and procedures used, including the cup hole diameter, which was an important element of the sham cupping device.
(3) The similarity of the sham cupping technique and real cupping technique may be considered a gentle type of cupping (minimal cupping), especially if the release time of pressure was prolonged,using large cup sizes, prolonged course of treatment and increasing frequency of cupping sessions.
(4) The heterogeneity of reporting sham cupping intervention.
(5) The limitation of the current sham cupping device is that it can be used as a comparison in dry cupping clinical trials only and cannot be used with other types of cupping.
Enhancements and standardization of the active sham cupping technique
This review suggested enhancements and standardization for the current active sham cupping technique, which included the following:(1) Using cups with bigger holes ≥2 mm in the side top of the cup,which led to the rapid release of pressure inside the cup before the suction produced significant physiological or mechanical effects.
(2) The hole diameter should be constant on all medium-sized cups(3.5 to 4.5).
(3) Using cups with an inner diameter from 3.5 to 4.5 cm in sham cupping therapy clinical trials. Using large cups should be limited due to the prolonged release of pressure inside the cup.
(3) Patients without any experience with cupping preferred to participate in trials using the sham cupping technique.
(4) Using a less frequent cupping protocol of treatment was suggested beginning with a single cupping session to a single cupping session per week for 4 sessions.
(5) The release pressure inside the cup should not be prolonged to more than 3 seconds.
(6) The size and number of cups used should be standardized in all clinical trials sessions and should not be reported as a range.
Furthermore, this review suggested criteria for reporting clinical trials that involved the sham cupping technique which included the following:
(1)Reporting the success of blindness in all clinical trials that used the sham cupping technique.
(2) The release time of pressure inside the cup should be reported.(3) Size and number of used cups should be reported.
(4) The adverse events of real cupping and sham cupping should be reported and compared.
(5) Details of the intervention, including number of sessions,frequency, method of suction, and cup retention time should be reported for real and sham cupping
Limitations of the review
Limitations of this review included the low number of published clinical trials that used the active sham cupping technique and the language restriction, which may exclude other non-English clinical trials. Additionally, there were many strengths of this review. This review is the first review which provided a narrative analysis of the active sham cupping technique and device. In addition, this review highlighted the limitations of the current sham cupping technique and recommended enhancements and reporting criteria for clinical trials that will use the active sham cupping technique in the future.
Conclusion
This paper identified the limitations of the current sham cupping technique, suggesting enhancements in the active sham cupping technique.
Continued evaluation of the current active sham cupping technique and development of new sham cupping devices and techniques for various types of cupping therapy are highly recommended.
- TMR Non-Drug Therapy的其它文章
- Effects of exercise-based cardiac rehabilitation on inflammatory biomarkers in patients with cardiovascular disease:a systematic review with meta-analysis
- Impact of resistance exercise training on physical performance of patients undergoing hemodialysis
- The role of pain neuroscience education in the management of chronic musculoskeletal pain:a physiotherapeutic approach
- Implementation of alternative methods in the health system through evidence-based medicine

