Assessment of phytotoxic,genotoxic and enzyme inhibition potential of Sterculia diversifolia
Fazle Rabbi,Amir Zada,Amna Nisar
Fazle Rabbi,Department of Pharmacy,Abasyn University Peshawar,Peshawar 25000,Khyber Pakhtunkhwa,Pakistan
Amir Zada,Amna Nisar,Department of Pharmacy,University of Peshawar,Peshawar 25120,Khyber Pakhtunkhwa,Pakistan
Abstract OBJECTIVE:To evaluate Sterculia diversifolia stem bark and leaves for phytotoxic,genotoxic and enzymes inhibition potential.METHODS:Phytotoxic activity of both stem bark and leaves were screened using Lemna minor.The genotoxic activity of Sterculia diversifolia stem bark and leaves extracts were tested using comet assay protocol while enzyme inhibition activity of crude extract and various fractions of both stem bark and leaves were evaluated using acetyl cholinesterase,lipoxygenase,β-glucuronidase,urease,xanthine oxidase and carbonic anhydrase.RESULTS:Phytotoxic activity showed significant results in dose dependant manner in both stem bark (ethyl acetate and n-butanol) and leaves (ethyl acetate,nbutanol and n-hexane) fractions.In genotoxic activity,dichloromethane fraction showed significant activity followed by ethyl acetate fraction.Acetyl cholinesterease inhibitory activity showed significant results in both stem bark and leaves fractions,while significant lipoxygenase inhibition was shown by ethyl acetate,dichloromethane,crude extract and n-hexane fractions of both stem bark and leaves.β-glucuronidase,urease and carbonic anhydrase inhibitory activity showed highly significant results in ethyl acetate fraction of both stem bark and leaves,while xanthine oxidase inhibition was shown by dichloromethane fraction of stem bark and leaves extracts.CONCLUSIONS:This study emphasizes the important phytotoxic,genotoxic and enzyme inhibition effects of Sterculia diversifolia stem bark and leaves.Hence,it is clear that Sterculia diversifolia stem bark and leaves possess phytotoxic,genotoxic and enzyme inhibitory agents.
Keywords:Sterculia;plant extracts;phytotoxic;mutagenicity tests;enzymes
1.INTRODUCTION
Nature is a serving man for treating diseases since paleolthic ages.Medicinal plants are rich sources of therapeutic moieties and are used for curing diseases.1A number of plants and their extracts have been used in traditional medicines.2,3Drugs used in current practices have been derived from natural sources either directly or indirectly.4Excellent therapeutic moieties could be discovered by detailed extensive research on medicinal plants.These moieties should be effective,less toxic and highly potent in resistant pathological conditions.5
Sterculiaceaefamily comprised of 60 genera and 1500 species belonging to tropical as well as sub-tropical region.6This family is a source of bioactive constituents of various chemical classes e.g.polyphenols,flavonoids,glycosides,alkaloids,steroids,terpenoids,triterpenes,saponins,sterols,apigenins,tannins,essential oils,carbohydrates and proteins.7,8Medicinally,Sterculia diversifoliabears antimicrobial,immune-omodulatory,cytotoxic,anti-glycation,larvicidal,anthelmintic,leishmanicidal,insecticidal,antioxidant,anticonvulsant,CNS depressant,analgesic,antipyretic,anti-inflammatory,laxative,anti-diarrheal,hepat-oprotective and diuretic activity.9-13Various secondary metabolites have been reported fromSterculia diversifoliae.g.Stercularin,luteolin,isoquercitrin,ursolic acid,gossypetin,taxifolin,methyl 4-hydroxycinnamate and β-sitosterol-Dglucoside.9,12,14This plant also bears metal contents and fatty acid constituents as reported in the literature.15,16The present study was designed to explore phytotoxic,genotoxic and enzyme inhibitory potential of medicinally important plant,Sterculia diversifolia.
2.MATERIALS AND METHODS
2.1.Plant material
Stem bark and leaves were collected in September,2014 from Pakistan Forest Institute botanical garden (34 00'50.6"N,71 29'03.0"E),University of Peshawar,Peshawar,Pakistan.Ghulam Jelani (taxonomist) at the Department of Botany,University of Peshawar identified the plant and deposited a specimen in their herbarium under the reference No.Bot.20098 (PUP).
2.2.Extraction and fractionation
Sterculia diversifoliastem bark and leaves (17 and 13 kg respectively) were dried under shade at room temperature.After grinding to powder,macerated for two weeks with solvent (hydromethanolic:10%-90%).Filtration was conducted with filter paper (Whatman-I)after maceration.Rotary evaporator was used for the concentration of obtained crude extracts under reduced pressure at 40 ℃.17The obtained crude extracts of both parts were 950 g and 1.2 kg respectively.Methanolic extract ofSterculia diversifolia(MESD) stem bark and leaves were then soaked overnight after mixing with 2.5 L distilled water.Various organic solvents were used in fractionation i.e.n-hexane (3 × 5 L),DCM (3 × 5 L),ethyl acetate (3 × 5 L) and n-butanol (3 × 5 L).The remaining residue was considered as aqueous fraction.
2.3.Phytotoxic activity
The MESD (stem bark and leaves) and its fractions were evaluated for phytotoxic potential using standard protocol.18The MESD and its fractions were added with sterilized E-medium at various concentrations (10,100 and 1000 µg/mL) in methanol.Inoculation of sterilized conical flask with desired concentrations fraction prepared from the stock solution and evaporated overnight.The inoculation of each flask was conducted with sterilized E-medium (20 mL).Ten Lemna minor were selected which were placed on media,each containing three frond rosette.Methanol and Parquet were inoculated in other flasks serving as negative and positive control.Treatment was conducted in triplicate and incubated the flasks for a week at 30 ℃ in growth cabinet (Fisons Fi-Totron 600H),intensity (9000 lux),relative humidity (56+10 rh) and at a day length of 12 h.Lemna minor growth in fraction containing flask was calculated by counting fronds number per dose.The determination of growth inhibition was achieved with reference to negative control.Percent (%) growth inhibition was determined by the given formula;% inhibition=100- (Number of fronds in test sample)/ (Number of fronds in control) × 100
2.4.Genotoxic activity
Genotoxic potential of crude extracts and various subsequent fractions of both stem bark and leaves extract(20,100,200 mg/L) was determined by comet assay.19Clean glass slides (n=36) were taken and agarose gel(0.75%) of low melting point was poured (80 µL) on them.Then agarose gel was allowed for solidification for about 45 min.Lymphocytes were poured (100 µL) into 96 well plate which was previously separated from sheep blood and then incubated for 2 h with selected concentration at 37 ℃.Each well (100 µL) containing suspension,which was pipetted out and poured on prepared agarose gel slides.Then on each slide agarose gel (0.5%) of low melting point was poured (80 µL) and allowed for solidification.Lyses solution (500 mL) was kept under refrigeration for 12 h in which slides were dipped.Lyses solution was removed on the 2nd day and placed the slides for 25-45 min in alkaline buffer (500 mL).Slides were kept in electrophoresis chamber having buffer [boric acid MP biomedicals,LLC (5.5 g),Tris base (10.8 g) and disodium EDTA (0.93 g) were dissolved in double distilled water (700 mL) and made the final volume upto 1000 mL] and ran for 45 min at 25 v.From electrophoresis chamber slides were removed and washed two times with neutralizing buffer [24.25 g Tris base was mixed with double distilled water (400 mL)and made the final volume upto 500 mL] and then stained with ethidium bromide solution (50-80 µL).Under fluorescent microscope slides were observed at 100 ×magnification with 590 nm barrier filter and 560 nm excitation filter.Doxorubicin hydrochloride and lymphocytes were used as positive control and negative control respectively.The classes of comets were;Class 0(without tail nucleus),Class 1 (tail less than nucleus diameter),Class 2 (tail one to two times the nucleus diameter) and Class 3 (tail >2 time the nucleus diameter).The experiments were conducted in triplicate and per treatment,300 cells were analyzed.The trypan blue exclusion method determination of cell viability was conducted,where only treatments with >80% viability were considered.The number of cells multiplication in each class by the damage class gave DNA damage score for each sample.DNA damage score was calculated according to the formula:
Total DNA damage score=(0 × n0)+(1 × n1)+(2 × n2)+(3 × n3)
wheren=each class cells number.Thus,the total score could range from 0 to 300.
2.5.Enzyme inhibition activity
2.5.1.Acetyl cholinesterase inhibitory assay
Modified spectrophotometric method was used for determining the acetyl cholinesterase (AChE) inhibitory activity.20Electric-eel AChE (type VI-S,Sigma,St.Louis,MO,USA) was used as source of cholinesterases and the substrate for AChE was acetylthiocholine iodide to perform reaction.Cholinesterase activity was measured and monitored using 5,5-Dithiobis (2-nitrobenzoic acid) (DTNB,Sigma,St.Louis,MO,USA).Both the tested sample and the standard were dissolved in ethanol.Reaction mixture contained 10 μL of DTNB,150 μL of sodium phosphate buffer (100 mM,pH 8.0),20 μL of acetylcholinesterase and 10 μL of sample solution were mixed and incubated at 25 ℃ for 15 min.The reaction was initiated by dissolving 10 mL of acetylthiocholine.A yellow 5-thio-2-nitrobenzoate anion was formed by the enzymatic hydrolysis of acetythiocholine which results by the reaction of DTNB and thiocholine at 412 nm,while kept for 15 min.These reactions took place in 96-well micro-plate in triplicate.The percentage inhibition of AChE was determined by using the formula given below,
% inhibition=(E − S)/ E × 100
Where,E and S is the activity of the enzyme without test sample and with test sample respectively.The IC50values were measured by EZ-Fit Enzyme Kinetics program and expressed in mean ± standard error of mean(SEM) (μM).
2.5.2.Lipoxygenase inhibition assay
The modified spectrometric method of Babatundeet al21was used for lipoxygenase assay.The linoleic acid and Lipoxygenase (EC 1.13.11.12) type I-B i.e.,Soybean were taken from Sigma (St.Louis,MO) in addition to all other chemicals.Reaction mixture consists of 160 μL of sodium phosphate buffer (0.1 mM;pH 7.0),sample solution of 10 mL and lipoxygenase solution of 20 μL.At 258 ℃,the mixture was incubated for 5 min.Reaction was intiated by linoleic acid substrate solution (10 μL)addition.Absorption changes with the formation of (9Z,11E)-13S)-13-hydroperoxyoctadeca-9,11-dienoate after 10 min.In 50% ethanol,test sample (i.e.extract,fractions) and the standard were dissolved and then analysed in triplicate.The standard used for lipoxygenase inhibition was Baicalein.
2.5.3.β-Glucuronidase inhibition assay
MESD (stem bark and leaves) and its fractions were screened for β-Glucuronidase enzyme inhibition using spectrophotometric method of Medina-Perez with minor modification.22A substrate p-nitrophenyl-β-D-glucuronide (250 μL) was used for this assay during which p-nitrophenol formed from substrate,was confirmed by absorbance at 405 nm and added to 96-well micro plate.The reaction mixture containing test sample (10 μL),enzyme solution (10 μL) and acetate buffer (0.1 m,185 μL).Reagents were mixed with the help of DMSO.The incubation period for the reaction mixture was 30 min at 37 ℃.p-nitrophenyl-β-D-glucuronide (0.4 mM,50 μL) was added to the reaction mixture and then change in absorbance was measured for 30 min continuously at 405 nm with Spectra Max spectrophotometer.Following formula was used for the determination of percent inhibition.
% inhibition=100– (Optical Density test sample/Optical Density control) x 100
The IC50 values were determined using the EZ-Fit enzyme kinetic program
2.5.4.Urease inhibition assay
MESD (stem bark and leaves) and its fractions were screened using the procedure of Weatherburn for urease inhibitory assay.22The reaction mixture [enzyme solution (Jack bean 25 μL) and buffer solution (55 μL)containing urea (100 mM)] along with each test sample(5 μL,1 mM) was incubated for 15 min at 30℃ in 96-well plate.Ammonia produced during reaction was measured using indophenol’s procedure to determine urea activity.Alkali reagent (70 μL) and phenol reagent(45 μL) were added to each well.Alkali reagent was prepared from NaOH (0.5% w/v) and active chloride NaOCl (0.1%),while phenol reagent was prepared from phenol (1% w/v) and sodium nitroprusside (0.005 % w/v).Absorbance was measured at 630 nm after 50 min with the help of micro plate reader.Change (per min) in absorbance was recorded with Soft-Max Pro software.EDTA (1.0 mM),LiCl2(0.01 M) and K2HPO4.3H2O(0.01 M) was added to maintain the pH at 8.2.Thiourea was used as control while whole experiment was performed in triplicate.Following formula was used for the determination of percent inhibition.
Percent inhibition=100 -(Optical Density test well/Optical Density control) ×100
2.5.5.Xanthine oxidase (XO) inhibition assay
This assay was used for MESD (stem bark and leaves)and its fractions screening using spectrophotometric method reported by Samahaet al.with minor modification.23Methanol-phosphate buffer (1%) was used for the dissolution of sample (i.e.extracts,fractions)to make up 100 μg/mL final concentration.Reaction mixture was prepared containing test samples (100 μL),enzyme solution (100 μL) and phosphate buffer (300 μL,0.2M,pH 9),then it is incubated (2 min) at room temperature.The reaction was initiated by adding xanthine oxidase solution (500 μL) prepared in phosphate buffer (0.15 mM).Spectrophotometer was used for the determination of absorbance (295 nm) at room temperature for 2 min.In this assay allopurinol(100 μg/mL) was used as a positive control.Following formula was used for the determination of percent inhibition.
% inhibition=(Change in Abs.control– Change in Abs.test sample) × 100)/ Change in Abs.control
2.5.6.Carbonic anhydrase inhibition assay
Carbonic anhydrase assay was performed for MESD(stem bark and leaves) and its fractions screening.Production of yellow colour 4-nitrophenol and colourless 4-nitrophenylacetate upon hydrolysis was measured in this assay.24Reaction was carried out in buffer solution(HEPES-tris,20 mM,pH 7.2-7.9) at 25-28 ℃.Buffer(140 μL) and purified bovine erythrocyte (20 μL) was added in each sample tube solution.4-nitrophenylacetate substrate (20 μL,0.6-0.8 mM) and extracts were dissolved in DMSO (10%) and then diluted in ethanol.The reaction was carried out using 96-well micro plate.Product formation rate was monitored for 30 min keeping 1 min interval by using microplate readers.Percent inhibition was determined by following formula;
% inhibition=100– (Optical Density test sample/Optical Density control) × 100
3.RESULTS
3.1.Phytotoxic activity
Crude MESD and various fractions of stem bark and leaves were evaluated for phytotoxic activity at 1000,100 and 10 μg/mL concentrations.The paraquat was used as standard phytotoxic drug (0.015 µg/mL).The results of test samples were concentration dependent.In crude MESD and subsequent fractions of stem bark,ethylacetate and n-butanol fractions showed maximum percent growth inhibition at 1000 μg/mL concentration i.e.100% and 80% respectively.While in case of crude MESD and its fractions of leaves,EtOAc,n-butanol and n-hexane fractions showed maximum percent growth inhibition at 1000 μg/mL concentration i.e.85%,85%and 80% respectively as presented in Table 1.
3.2.Genotoxicity activity
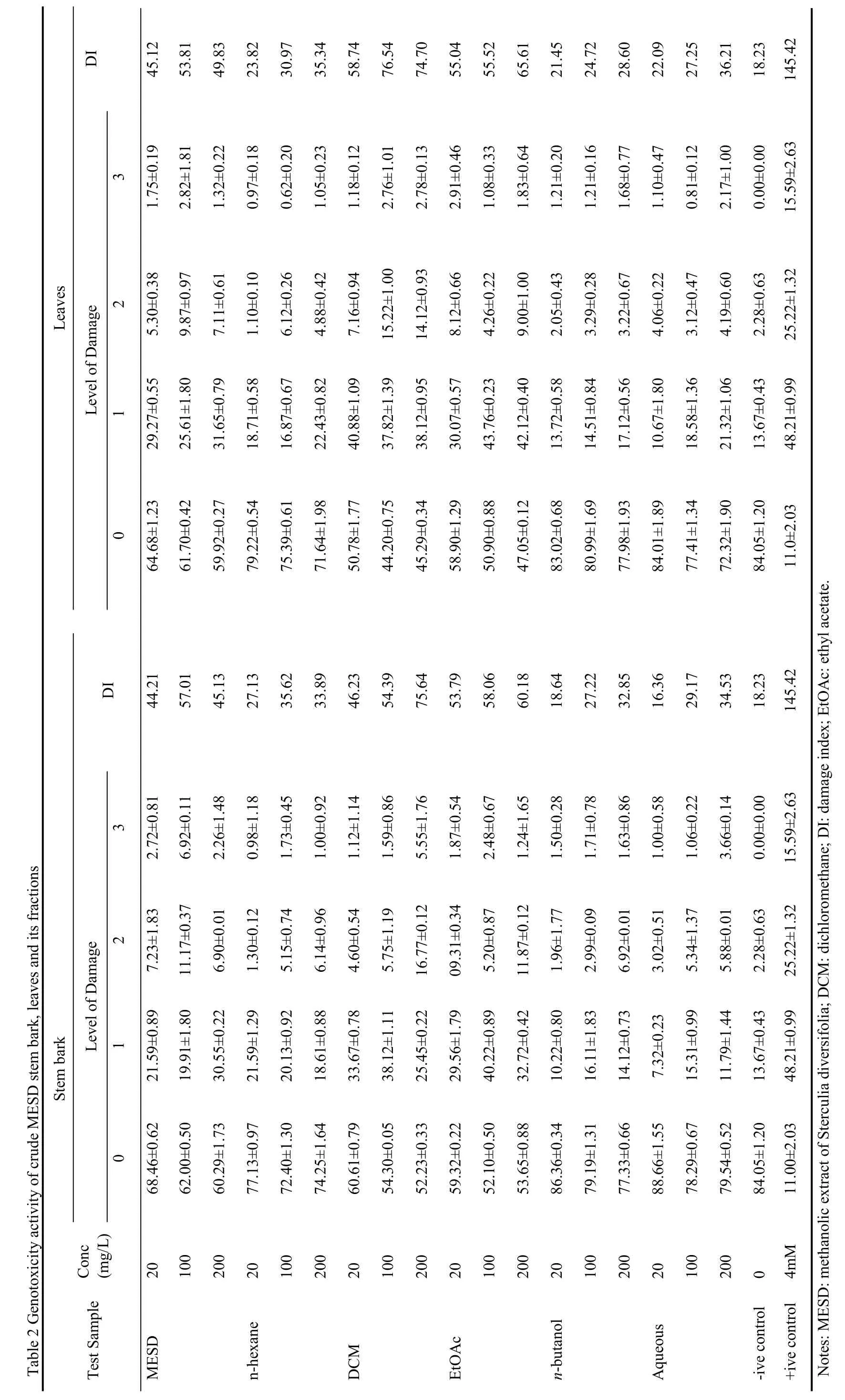
Genotoxic activity of MESD (stem bark and leaves) and its fractions were evaluated.Table 2 showed level of damage as well as damage index (DI) by the tested samples in three different concentrations used.The level of damage was significant for DCM and ethyl acetate fractions,while DCM and ethyl acetate fractions also showed significant damage index at all concentrations especially at higher concentrations.The data of genotoxicity effects were also comparable to those of the doxorubicin hydrochloride (positive control) used in this study.
3.3.Enzyme Inhibition activity
3.3.1.Acetylcholinesterase inhibitory assay
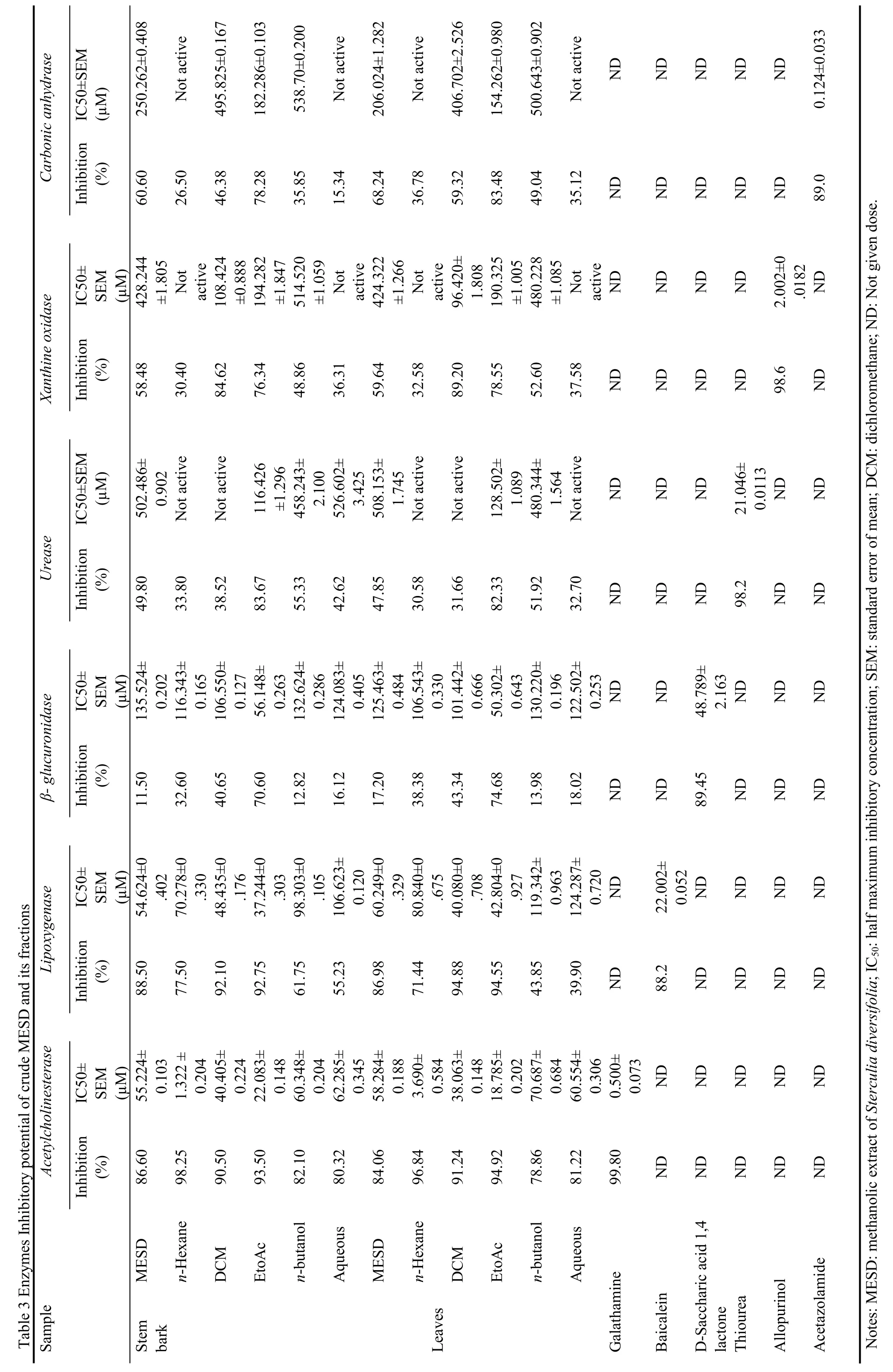
The crude MESD (stem bark and leaves) and its fractions were screened for Acetylcholinesterase inhibitory activity.The maximum inhibitory activity of stem bark extract and fractions were observed for n-hexane(98.25%) with IC50value of (1.32 ± 0.20) µg/mL,followed by EtOAc (93.50%) with IC50value of (22.08± 0.14) µg/mL,DCM (90.50%) with IC50value of (40.40± 0.22) µg/mL,crude MESD (86.60%) with IC50value of (55.22 ± 0.10) µg/mL,n-butanol (82.10%) with IC50value of (60.34 ± 0.20) µg/mL and aqueous fraction(80.30%) with IC50value of (62.28 ± 0.34) µg/mL)respectively as shown in Table 3.The maximum inhibitory activity of leaves extract and fractions were observed for n-hexane (96.84%) with IC50value of (3.69± 0.58) µg/mL,followed by EtOAc (94.92%) with IC50value of (18.78 ± 0.20) µg/mL,DCM (91.24%) with IC50value of (38.06 ± 0.14) µg/mL,crude MESD (84.06%)with IC50value of (58.28 ± 0.18) µg/mL,aqueous fraction (81.22%) with IC50value of (60.55 ± 0.30)µg/mL and n-butanol (78.86%) with IC50value of (70.68± 0.68) µg/mL respectively as shown in Table 3.The Acetylcholinesterase inhibitory activity of standard drugs (Galathamine) was 0.5 µM respectively.
3.3.2.Lipoxygenase Inhibitory assay
The crude MESD (stem bark and leaves) and its fractions were screened for Lipoxygenase inhibitory activity.The maximum inhibitory activity of stem bark extract and fractions were observed for EtOAc (92.75%) with IC50value of (37.2 ± 0.30) µg/mL,followed by DCM(92.10%) with IC50value of (48.4 ± 0.17) µg/mL,crude MESD (88.50%) with IC50value of (54.6 ± 0.40) µg/mL,n-hexane (77.50%) with IC50value of (70.2 ± 0.33)µg/mL and n-butanol (61.75%) with IC50value of (98.30± 0.10) µg/mL respectively as shown in table 3.The maximum inhibitory activity of leaves extract and fractions were observed for DCM (94.88%) with IC50value of (40.08 ± 0.70) µg/mL,followed by EtOAc(94.55%) with IC50value of (42.80 ± 0.92) µg/mL,crude MESD (86.98%) with IC50value of (60.24 ± 0.32)µg/mL,n-hexane (71.44%) with IC50value of (80.84 ±0.67) µg/mL respectively as shown in Table 3.The Lipoxygenase inhibitory activity of standard drugs(Baicalein) was 22.0 µM respectively.
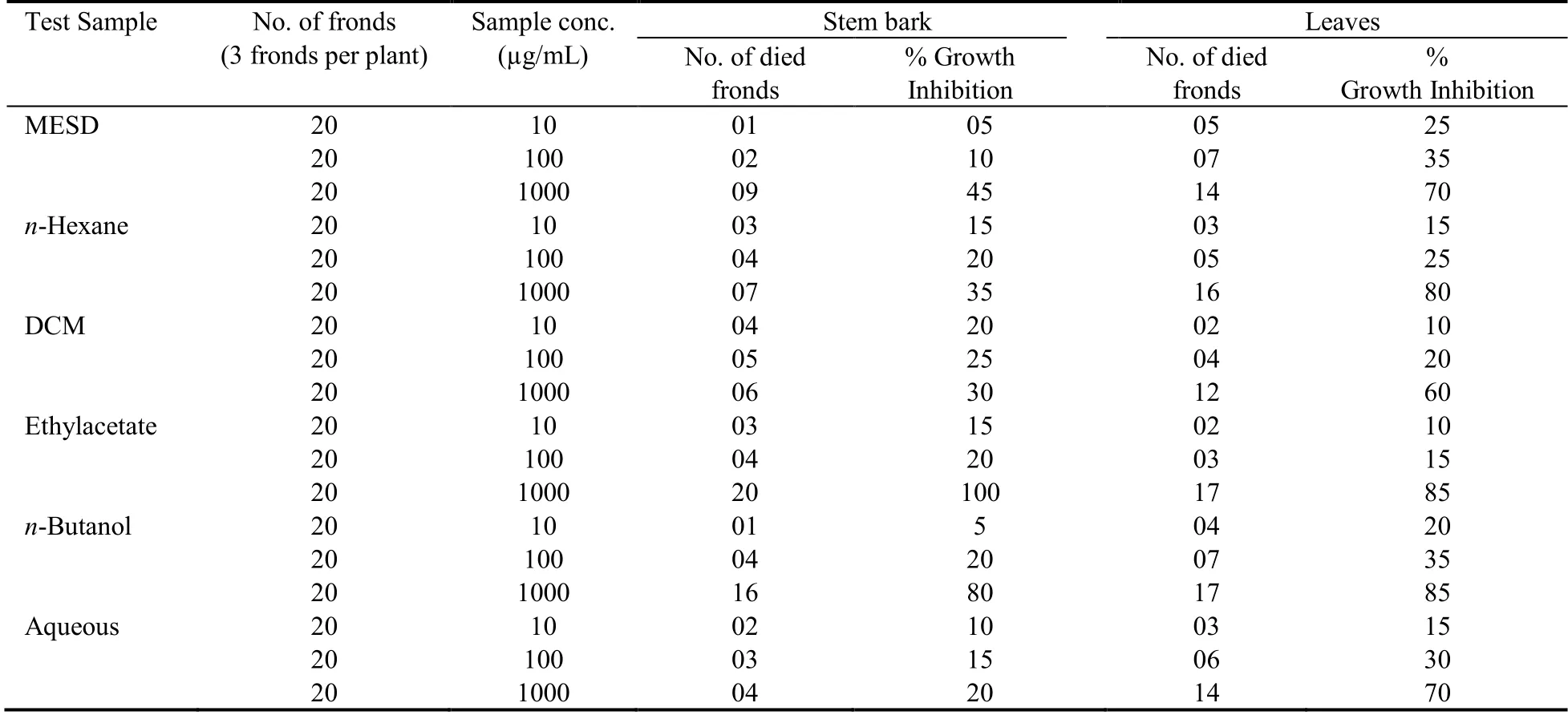
Table 1 Phytotoxic activity of crude extracts (stem bark and leaves) and its fractions
3.3.3.β-glucuronidase inhibitory assay
The crude MESD (stem bark and leaves) and its fractions were screened for β-glucuronidase inhibitory activity.EtOAc fraction of stem bark showed maximum inhibitory activity (70.60%) with IC50value of (56.14 ±0.26) µg/mL,while other fractions showed nonsignificant results.Similarly maximum inhibitory activity of leaves extract and fractions were observed for EtOAc (74.68%) with IC50value of (50.30 ± 0.64)µg/mL,while other fractions showed non-significant results as shown in Table 3.D-Saccharic acid 1,4 lactone(standard drug) showed 89.4% inhibition with IC50values of (48.78 ± 2.16) µg/mL.
3.3.4.Urease inhibitory assay
Crude MESD and its fractions of both stem bark and leaves were screened for urease inhibitory activity.Among stem bark samples,EtOAc fraction showed significant inhbitory effect (83.67%) with IC50value of(116.4 ± 1.29) µg/mL against urease enzyme while moderate inhibitory activity was shown by the n-butanol fraction (55.33%) with IC50value of (458.24 ± 2.10) µg/mL respectively.Among leaves extract and fractions,significant inhibition was showed by ethyl acetate fraction (82.33%) with IC50value of (128.50 ± 1.08)µg/mL,while moderate inhibitory activity was shown by the n-butanol fraction (51.92%) with IC50value of(480.34 ± 1.56) µg/mL as shown in table 3.The percent inhibition for thiourea (standard drug) was 98.2 % with IC50values of (21.00 ± 0.01) µg/mL.
3.3.5.Xanthine oxidase inhibitory assay
Crude MESD and its various fractions of both stem bark and leaves were screened for xanthine oxidase inhibitory potential.Maximum inhibitory activity of stem bark extract and fractions were observed for DCM fraction(84.62%) with IC50value of (108.4 ± 0.88) µg/mL,followed by EtOAc fraction (76.34%) with IC50value of(194.28 ± 1.84) µg/mL respectively,while among leaves extract and fractions,significant inhibitory effect against urease was also shown by DCM fraction (89.20%) and EtOAc fraction (78.55%) with IC50value of (96.42 ±1.80) and (190.32 ± 1.00) µg/mL.The percent inhibition for allopurinol (standard drug) was 98.6 % with IC50values of (2.00 ± 0.01) µg/mL.
3.3.6.Carbonic anhydrase inhibitory assay
The crude MESD and various fractions of stem bark and leaves were evaluated for carbonic anhydrase inhibitory assay (Table 3).Maximum inhibitory activity of stem bark extract and fractions were observed for EtOAc(78.28%) with IC50value of (182.28 ± 0.10) µg/mL,followed by crude MESD (60.60%) and DCM fraction(46.38%) with IC50value of (250.26 ± 0.40) and (495.82± 0.16) µg/mL respectively.Similarly maximum inhibitory activity of leaves extract and fractions were observed for EtOAc (83.48%) with IC50value of (154.26± 0.98) µg/mL,followed by crude MESD (68.24%),DCM fraction (59.32%) and n-butanol (49.04%) with IC50value of (206.02 ± 1.28),(406.70 ± 2.52) and(500.64 ± 0.90) µg/mL respectively.
4.DISCUSSION
Weed is one of the significant cause for huge economic losses all over the world by reducing the quality and quantity of agricultural crops.It is estimated that weeds causes a loss of about 12% costing to about US 33 billion dollars in United State,while in developing countries the situation is more worst.25There is a problem that resistance arises against these herbicides because of frequent use of these herbicides.The species have the affinity to convert into other species that have much similarity with beneficial plants.The haphazard use of herbicides also produce many environmental and health pollution problems,so there is an immense need of a herbicide showing significant pytotoxic activity with less toxicity to human health.It is evident from results that phytotoxic activity showed significant outcomes in dose dependant manner in both stem bark (ethyl acetate and nbutanol) and leaves (ethyl acetate,n-butanol and nhexane) fractions.Literature showed that plants,their products and different isolated compounds possess phytotoxic potential,so they can be used as herbicide.26The phytotoxicity of the plant has proved that weeds could be controlled without any harmful effect on the crop growth as well as overall yield which results in significant increase in crops production.27On the basis of results it is therefore assumed that the phytotoxic agents of the stem bark and leaves could be a significant natural source for weeds control in a sustainable way for better production of crop.28
Natural products are used as structural models for the novel molecules synthesis and exploited for their pharmacological properties.However,still there is not much in literature about studies on the natural products potential mutagenic and toxicological effects to understand the genotoxic effects of new phytotherapeutic agents.It is crystal clear from the present study that DCM fraction showed significant genotoxic activity followed by ethyl acetate fraction.This activity is performed through comet assay.In recent times,comet assay grip special attention in order to identify substances with genotoxic effect.Comet assay is used for the primary DNA damage detection in individual cells.It is a very fast and sensitive tool for detection.Several classes of DNA injury could be detected with this method like single or double strand breaks,incomplete repair of a basic sites,alkali labile sites and cross links.29This activity showed a little increase in total damaged cells and scores in animal groups treated with theSterculiadiversifoliacrude extracts and its fractions.The results are non-significant except DCM and ethyl acetate fractions at higher concentrations.It is known that many flavonoids and alkaloids have shown to be genotoxic in a variety of prokariotic and eukariotic cells andin vivosystems.30-32For genotoxicity,the mechanistic basis needs full elucidaion,although structure-activity relationship studies have identified as requisite flavonoids structural features.
Acetyl cholinesterease inhibitory activity showed significant results in both stem bark and leaves fractions.Acetylcholinesterase is an essential enzyme in the human body.33The biological role of AChE is the cessation of impulse transmissions in nervous system at cholinergic synapsesviaquick hydrolysis of neuro-transmitter acetylcholine.34Alzheimer's disease patient central cholinergic function is improved by AChE inhibitors application and as a result of this the deficiency in central nervous system functions is recompense.This is the only approved therapy.35Therefore,this is an important issue to search the natural products that having AChE inhibitory activity.
Lipoxygenase inhibition was shown by ethyl acetate,DCM,MESD and n-hexane fractions of stem bark and leaves.Lipoxygenase is a non-heme iron containing dioxygenase extensively dispersed in nature.Lipoxygenases enzymes converts linoleic,arachidonic and other polyunsaturated fatty acid into biologically active metabolites that are involved in immune and inflammatory responses.In human,lipoxygenase genes have been identified and due the diversity of lipoxygenase genes,the role of lipoxygenase is complex in the development and succession of cancer.36
β-glucuronidase inhibitory activity showed highly significant results in ethyl acetate fraction of both stem bark and leaves.It is reported that β-glucuronidase activity increases in various diseases e.g.AIDS,arthritis,cancer and liver disorders.37Thus natural products which possess β-glucuronidase inhibition also possess cytotoxic effect.In living system glucuronidation leads to detoxification process.38
Urease inhibitory activity showed highly significant results in ethyl acetate fraction of both stem bark and leaves.Urease catalyzes urea hydrolysis to ammonia and carbon dioxide.Ureas enzyme is pathogenic when upregulated,mediating ailments e.g.hepatic coma,peptic ulcer,pyelonephritis and urinary lithiasis.It also facilitatesProteus mirabilis,Helicobacter pyloriandYersinia enterocoliticainfections.Urease increases the stomach pH which helps inHelicobacter pyloricolony formation.This leads to the pathogenesis of peptic ulcer,gastritis and cancer.39Various extract of plants have shown urease inhibitory activity e.g.Aristolachia bracteata,Achillea millefolium,Ginkgo biloba,Taraxacum officinale,Diospyros lotusetc.39,40
Xanthine oxidase inhibition was shown by DCM fraction of both stem bark and leaves.This enzyme plays an important role in humans in the metabolism of purine nucleotide which converts hypoxanthine into xanthine,which is further converted into uric acid.Uric acid excessive production leads to a pathological condition named gout,in which uric acid accumulation occurs in joints.This uric acid accumulation in joints causes inflammation and severe pain.41The gout prevalence is higher in male (>30 years of age) and female (>than 50 years of age).Allopurinol is used worldwide for this pathological condition.42It is reported in the literature that plant extract exhibits its anti-gout activity through xanthine oxidase inhibition.
Carbonic anhydrase inhibitory activity showed highly significant results in ethyl acetate fraction of both stem bark and leaves.Carbonic anhydrase is involved in physiological process,related to transport and respiration of bicarbonate/CO2between metabolizing tissues and lungs.39It has been reported that this enzyme expression leads to colorectal cancer,glaucoma,leukemia,epilepsy and cystic fibrosis.Polyphenolic compounds isolated from plants have been reported as carbonic anhydrase inhibitors.43Both urease and carbonic anhydrase are key enzymes in the living organisms physiology.During homeostasis,urease elevate stomach pH,while carbonic anhydrase plays a vital role in acid-base balance,CO2and ion transport.
In the management of various pathological conditions,plant origin natural products are important.The enzymes inhibitory effect of these agents in human beings has got a special place in the field of drug discovery.Thus various pathological conditions could be treated by enzyme inhibitors.
In conclusion,this study emphasizes the important phytotoxic,genotoxic and enzyme inhibitory activity ofSterculia diversifoliastem bark and leaves.It is concluded thatSterculia diversifoliastem bark and leaves bears phytotoxic,genotoxic and enzyme inhibitory agents.
5.REFERENCES
1.Ur Rahman S,Ismail M,Khurram M,Ullah I,Rabbi F,Iriti M.Bioactive steroids and saponins of the genus Trillium.Molecules 2017;22:2156-71.
2.Noreen I,Iqbal A,Muhammad A,Shah Z,Rahman ZU.Antimicrobial activity of different solvents extracts of Acacia cyanophylla.Pak J Weed Sci Res 2017;23:79-90.
3.Iqbal A,Siraj-ud-Din I,Ali G,Rahman ZU,Rabi F,Khan MS.Pharmacological evaluation of tamarix aphylla for antiinflammatory,anti-pyretic and anti-nociceptive activities in standard animal models.West Ind Med J 2017;1-22.
4.Cragg GM,Newman DJ.Natural products:a continuing source of novel drug leads.Biochim Biophys Acta 2013;1830:3670-95.
5.Iqbal A,Din S,Khan I,Jan S.Antimicrobial activities of selected weed plants.Pak J Weed Sci Res 2015;21:229-38.
6.Orisakeye O,Olugbade T.Epicatechin and procyanidin B2 in the stem and root bark of Sterculia tragacantha Lindl (Sterculiaceae).Med Chem 2014;4:334-7.
7.Ouédraogo M,Konate K,Zerbo P,Barro N,Sawadogo LL.Phytochemical analysis and "in vitro" antifungal profile of bioactive fractions from" Sterculia setigera" (Sterculiaceae).Curr Res J Biol Sci 2013;5:75-80.
8.Khatiashvili N,Gogilashvili L,Yarosh E,Kemertelidze E.Lipids from Sterculia platanifolia and Hamamelis virginiana seeds.Chem Nat Compd 2007;43:315-6.
9.Rabbi F,Zada A,Nisar A,Sohail M,Khalil SK,Shah WA.Bioassay-guided isolation,identification of compounds from sterculia diversifolia and investigation of their anti-glycation and antioxidant activities.Pharm Chem J 2020;53:1137-44.
10.Rabbi F,Zada A,Nisar A.Larvicidal,leishmanicidal,insecticidal and anthelmintic effects of Sterculia diversifolia stem bark and leaf.Bangladesh J Pharmacol 2020;15:32-8.
11.Rabbi F,Zada A,Adhikari A,Jabeen A,Nisar A,Ullah I.Sterculia diversifolia bears anti-cancer and immunomodulatory activities.Bangladesh J Pharmacol 2017;12:51-5.
12.Rabbi F,Zada A,Nisar A,et al.Evaluation ofin-vivobiological activities of Sterculia diversifolia (G.Don) in relevance to the isolated secondary metabolites.J Anim Plant Sci 2021;31:296-306.
13.Rabbi F,Zada A,Nisar A,Sohail M,Khalil SK,Ahmad AA.In vivolaxative,anti-diarrheal,hepatoprotective and diuretic investigations of Sterculia diversifolia and its isolated compounds.J Tradit Chin Med 2021;41:717-24.
14.Rabbi F,Zada A,Adhikari A,Nisar A,Khalil SK.Chemical constituent with cytotoxicity from Sterculia diversifolia.J Asian Nat Prod Res 2020;23:899-905.
15.Salem MZM,Ali HM,Mansour MM.Fatty acid methyl esters from air-dried wood,bark,and leaves of Brachychiton diversifolius R.Br:antibacterial,antifungal,and antioxidant activities.BioResources 2014;9:3835-45.
16.Rabbi F,Zada A,Adhikari A,et al.GC-MS analysis,metal analysis and antimicrobial investigation of sterculia diversifolia.Pharm Chem J 2020;54:943-53.
17.Kolawole O,Makinde J.Central nervous system depressant activity of Russelia equisetiformis.Niger J Physiol Sci 2007;22:59-63.
18.Fatma A,Küçükboyaci N,Duman H,Şener B,Choudhary MI.Cytotoxic,phytotoxic and insecticidal activities of chrysophthalmum montanum (DC.) Boiss.Turkish J Pharm Sci 2017;14:290-3.
19.Saleem U,Mahmood S,Ahmad B,Saleem M,Anjum AA.Estimation of genotoxic and mutagenic potential of latex and methanolic leaves extract of Euphorbia helioscopia by comet assay and Ames test.Asian Pac J Trop Dis 2015;5:S145-50.
20.Benamar H,Marouf A,Bennaceur M.Phytochemical composition,antioxidant and acetylcholinesterase inhibitory activities of aqueous extract and fractions of Pistacia atlantica subsp.atlantica from Algeria.J Herbs,Spices Med Plants 2018;24:229-44.
21.Babatunde JO,Kayode OK.Inhibitory action of dried leaf of Cassia alata (Linn.) Roxb against lipoxygenase activity and nitric oxide generation.Sci Agropecu 2019;10:185-90.
22.Medina-Pérez G,Peralta-Adauto L,Afanador-Barajas L,et al.Inhibition of urease,elastase,and β-glucuronidase enzymatic activity by applying aqueous extracts of opuntia oligacantha CF först acid fruits:in vitroessay under simulated digestive conditions.Appl Sci 2021;11:7705-17.
23.Samaha H,Ali NAA,Mansi I,Abu-El-Halawa R.Antimicrobial,antiradical and xanthine oxidase inhibitory activities of Juniperus procera plant extracts from Albaha.World J Pharmacy Pharm Sci 2017;6:232-42.
24.Ibrahim S,Ameh D,Atawodi S,Umar I,Jajere U,Mohammed S.In vitroinhibitory effect of methanol leaf extract of cadaba farinosa on carbonic anhydrase activity.Int J Biochem Res Rev 2016;11;1-8.
25.Pimentel D,McNair S,Janecka J,et al.Economic and environmental threats of alien plant,animal,and microbe invasions.Agric Ecosyst Environ 2001;84:1-20.
26.Khan I,Nisar M,Ebad F,et al.Anti-inflammatory activities of Sieboldogenin from Smilax china Linn:experimental and computational studies.J Ethnopharmacol 2009;121:175-7.
27.Batish DR,Kaur M,Singh HP,Kohli RK.Phytotoxicity of a medicinal plant,anisomeles indica,against phalaris minor and its potential use as natural herbicide in wheat fields.Crop Prot 2007;26:948-52.
28.Saeed M,Khan H,Khan MA,Simjee SU,Muhammad N,Khan SA.Phytotoxic,insecticidal and leishmanicidal activities of aerial parts of Polygonatum verticillatum.Afr J Biotechnol 2010;9:1241-4.
29.Moreira V,Dias A,Martins A,et al.Evaluation of genotoxic and cytotoxic effects of antileishmanial extract from Julocroton triqueter (Euphorbiaceae).Genet Mol Res 2013;12:1132-42.
30.Labieniec M,Gabryelak T.Effects of tannins on Chinese hamster cell line B14.Mutat Res Genet Toxicol Environ Mutagen 2003;539:127-35.
31.Da Silva J,Herrmann S,Heuser V,et al.Evaluation of the genotoxic effect of rutin and quercetin by comet assay and micronucleus test.Food Chem Toxicol 2002;40:941-7.
32.Kleinsasser NH,Sassen AW,Semmler MP,Harréus UA,Licht AK,Richter E.The tobacco alkaloid nicotine demonstrates genotoxicity in human tonsillar tissue and lymphocytes.Toxicol Sci 2005;86:309-17.
33.Fabini E,Tramarin A,Bartolini M.Combination of human acetylcholinesterase and serum albumin sensing surfaces as highly informative analytical tool for inhibitor screening.J Pharm Biomed Anal 2018;155:177-84.
34.Nor MNM,Rahim NS,Mohamad S,Hashim SN,Razali Z,Muhammad NA.Phytochemical screening,antioxidant and enzyme inhibition activity of phoenix dactylifera ajwa cultivar.In:Saian R,Abbas M,editors.Future of ASEAN (ICoFA) 2017:Proceedings of the Second International Conference on the Future of ASEAN (ICoFA);2017 May 4;Singapore:Spirnger,2018:561-9.
35.Ahmed T,Zahid S,Mahboob A,Mehpara Farhat S.Cholinergic system and post-translational modifications:an insight on the role in Alzheimer's disease.Curr Neuropharmacol 2017;15:480-94.
36.Guo S,Song Z,Ma R,Yang Y,Yu M.Genome-wide identification and expression analysis of the lipoxygenase gene family during peach fruit ripening under different postharvest treatments.Acta Physiol Plant 2017;39:111.
37.Choudhary MI,Mohammad MY,Musharraf SG,Parvez M,Al-Aboudi A.New oxandrolone derivatives by biotransformation using Rhizopus stolonifer.Steroids 2009;74:1040-4.
38.Kavak DD,Altıok E,Bayraktar O,Ülkü S.Pistacia terebinthus extract:as a potential antioxidant,antimicrobial and possible βglucuronidase inhibitor.J Mol Catal B Enzym 2010;64:167-171.
39.Abu-Izneid T,Rauf A,Saleem M,et al.Urease inhibitory potential of extracts and active phytochemicals of Hypochaeris radicata(Asteraceae).Nat Prod Res 2020;34:553-7.
40.Rauf A,Uddin G,Raza M,et al.Urease inhibition potential of Dinaphthodiospyrol from Diospyros lotus roots.Nat Prod Res 2017;31:1214-8.
41.Ristic B,Sikder MOF,Bhutia YD,Ganapathy V.Pharmacologic inducers of the uric acid exporter ABCG2 as potential drugs for treatment of gouty arthritis.Asian J Pharm Sci 2020;15:173-80.
42.Vargas-Santos AB,Peloquin CE,Zhang Y,Neogi T.Association of chronic kidney disease with allopurinol use in gout treatment.JAMA Intern Med 2018;178:1526-33.
43.Saleem M,Saeed A,Khan A,et al.Benzamide sulfonamide derivatives:potent inhibitors of carbonic anhydrase-II.Med Chem Res 2016;25:438-48.
 Journal of Traditional Chinese Medicine2022年4期
Journal of Traditional Chinese Medicine2022年4期
- Journal of Traditional Chinese Medicine的其它文章
- Effectiveness of redcore lotion in patients with vulvovaginal candidiasis:a systematic review and Meta-analysis
- Efficacy and safety of external application of Chinese herbal medicine for psoriasis vulgaris:a systematic review of randomized controlled trials
- Effectiveness and safety of electroacupuncture for the treatment of pain after laparoscopic surgery:a systematic review
- Effect of astragaloside IV on the immunoregulatory function of adipose-derived mesenchymal stem cells from patients with psoriasis vulgaris
- Shenqihuatan formula (参七化痰方) reduces inflammation by inhibiting transforming growth factor-beta-stimulated signaling pathway in airway smooth muscle cells
- Drug response biomarkers of Pien Tze Huang (片仔癀) treatment for hepatic fibrosis induced by carbon tetrachloride
