Stabilization of heavy metals in sediments: A bioavailability-based assessment of carbon adsorbent efficacy using diffusive gradients in thin films
Yiqin Chen, Yueyue Wang, Ruyi Zheng, Ju Wen, Juan-Ying Li, Qian Wang, Jie Yin
College of Marine Ecology and Environment, Shanghai Ocean University, Shanghai, 201306, China
ABSTRACT
This study investigated the effectiveness of carbon adsorbents as remediation material for sediments contaminated with heavy metals and the feasibility of utilizing diffusive gradients in thin films (DGT) as a biomimetic tool to estimate the accumulation of heavy metals in Venerupis philippinaram (Manila clam). The results showed that carbon materials had significant inhibitory effects (14.0–53.0%) on the enrichment of heavy metals in organisms and the order of increasing overall inhibitory effect was: charcoal, peat, activated carbon, and biochar.There were significant correlations (P <0.0001) between the four heavy metals accumulated in Venerupis philippinaram and those accumulated in DGT devices after 28 days in the laboratory. Observed concentrations of heavy metals enriched in DGT (5.4–42.0%) were less than accumulations in Venerupis philippinaram. The results of in situ DGT applications showed significant correlations between the amount accumulated in DGT and the acid-soluble portion in sediments for both Cu and Pb, with positive linear correlations and R-squares of 0.97 and 0.92, respectively. These results supported the notion that a DGT device can be used as a biomimetic tool that predicts and monitors the accumulation of heavy metals in aquaculture ponds. Future studies should focus on improving the stabilization of heavy metals in sediments using different types of carbon sorbents, as well as minimizing simulation deviations using DGT.
ARTICLEINFO
Keywords:
Carbon amendment sediment remediation
DGT
Venerupis philippinaram
Heavy metals
Bioavailability
Bioaccumulation
1.Introduction
Aquaculture has undergone unprecedented growth to meet the demand of protein for human consumption, creating significant growth in high-protein food production (Taylor, 2009). The aquaculture industry in China has contributed more than 60% of global production in recent years (Wang, Ding, & Gong, 2016). Meanwhile, there has been a growing concern over the health-related risks caused by toxic-chemical contamination of aquaculture products (Gu et al., 2017; Guo et al., 2008;Liang et al., 2016). Toxic chemicals, including heavy metals and hydrophobic organic pollutants (HOCs; such as pyrethroids, organochlorine pesticides, and polycyclic aromatic hydrocarbon), are likely to accumulate in sediments and organisms (Wu et al., 1999; Yang et al.,2012). These toxic chemicals can be readily transferred to aquaculture organisms from the culturing environment and associated sediments(Kalanzi et al., 2013; Kwok et al., 2014; Tao et al., 2012; Turkmen et al.,2009). Bioavailable toxic chemicals are the proportion of the toxic chemicals that can be transferred to aquatic organisms. When these bioavailable toxic chemicals are ingested via aquatic products, they can accumulate and cause associated adverse effects in humans (Fu et al.,2019; Järup, 2003). Therefore, it is important to reduce the bioavailable portion of toxic chemicals in sediments to ensure the safety of aquaculture products to consumers (Li et al., 2018).
There are a variety of techniques used for reducing toxic chemicals in sediments, including chelation by plant roots (Sarwar et al., 2016) and physiological metabolism of actinomycetes (Alvarez et al., 2017). Carbon sorption is the most cost- and time-effective technique (Knox et al.,2014, 2016; Liu et al., 2018; Mamindy-Pajany et al., 2013; Pedersen et al., 2015) and has been widely used for sediment remediation, especially for HOC contamination (Ghosh et al., 2011). However, there is a lack of effective procedures for measuring the reductions of bioavailable HOCs and heavy metals after carbon-sorption remediation. The bulk of sediment-associated contaminants are usually defined as the accumulation of pollutants in sediments, which cannot be reliably used to predict their bioavailability and toxicity (Hu et al., 2018). Recently, the bioavailability of sediment-associated pyrethroids has been successfully evaluated via equilibrium passive sampling (Li et al., 2015), indicating that passive sampling was a more reliable alternative means for the measurement of reduced bioavailable contaminants in sediments.
Diffusive gradient in thin films (DGT), a dynamic passive sampling technique, was initially used to measure labile heavy metals in natural water sources (Davison & Zhang, 1994). This technique is also suitable for measuring the flux of heavy metals between sediments to pore water(Zhang et al., 1995). It is worth noting that heavy metals exist in various forms with distinct bioavailability and toxicity (Hu et al., 2018; Liang et al., 2017; Thinh et al., 2018). According to the Community Bureau of Reference (BCR), heavy metals can be divided into four species, namely:exchangeable and carbonate bound (acid soluble, F1); Fe/Mn oxide or hydrous oxide bound (reducible, F2); organic substance or sulfide bound(oxidable, F3); and residual (F4) forms (Jiang et al., 2012). One of the disadvantages of conventional and ex situ methods (i.e. sequential extraction) is that the characteristics of sediments and the heavy metal speciation is changed after the extraction (Gao et al., 2016; Parker et al.,2017). By contrast, DGT eliminates the risk of speciation changes during the transportation, storage, and analysis of samples (Tian et al., 2018; Xu et al., 2018; Yin et al., 2014).
DGT measurements reflect the dynamic supply of metals from solid phase, regardless of binding ligands, and can provide valuable insights into the microscale biogeochemical heterogeneity of sediments (Gao et al., 2016; Harper et al., 1998; Mangal et al., 2016; Parker et al., 2017).The DGT technique has been widely used to measure metal bioavailability in soils and sediments, with results interpreted as free-dissolved(labile) concentrations in pore water (C, μg/cm) (Amato et al.,2014, 2015, 2018; Amirbahman et al., 2013; He et al., 2018; Roulier et al., 2008). However, systematic studies on the correlations among the DGT measurements, different heavy-metal species measurements, and the bioaccumulation measurements are very limited, except for a few focused on copper (Ferreira et al., 2008; Lin et al., 2015; Martin &Goldblatt, 2007) and cadmium (Dabrin et al., 2012).
Therefore, following upon the assessment of the bioavailability and bioaccumulation of pyrethroids to Venerupis philippinaram (V. philippinaram) after carbon sorbents amendment (Li et al., 2015), this study uses DGT devices as passive samplers along with adsorbent materials mixed by three types of black carbon (BC)-based materials (charcoal,biochar, and activated carbon) and one humic substance-based (peat soil) carbon in sediments. The aims of this study are to explore the effect of adsorbent materials in sediment on reducing the bioavailability of heavy metals in sediments and bioaccumulation of heavy metals in V. philippinaram and the feasibility and reliability of using DGT to predict the bioaccumulation of heavy metals in benthic organisms.
2.Materials and methods
2.1.Sediments and adsorbents
Surface sediment samples were collected using a Peterso n sampler(PSC-1/16) from the intertidal zone in Yangshan Port, Shanghai, China(N303835.59, E1220322.72). The samples were transferred to glass bottles, packed with ice, and delivered to the laboratory where they were stored at −20C until analysis.
Four types of carbon sorbents, including peat soil, activated carbon,charcoal, and biochar, were used in this study, representing different source materials (the peat soil represents humic substances and the other three types of sorbents were examples of black carbon) and combustion procedures (Hua et al., 2017; Jia & Gan, 2014). The coal-based activated carbon was purchased from Shanghai Zhanyun Chemical Co., Ltd. (Shanghai, China). The peat soil was purchased from Pindstrup Mosebrug AS (Pindstrup, Danmark). The charcoal was derived after the combustion of bamboo and the biochar was produced through incomplete combustion of peanut shells at 400C (Jia & Gan, 2014). All carbon sorbents were ground in a mortar and sieved through a 2-mm mesh before use.
Sediment samples were acidified with 1 M HCl and then dried at 60C for 24 h to remove the inorganic carbon before elemental analysis.The measured carbon content was assumed to be the sum of organic carbon (OC) and black carbon (BC). The total content of OC and BC were measured by Elemental Vario max CNS Analyzer (Elementar Company,GER). BC content was also measured after baking in a muffle furnace at 400C for 24 h to remove thermally labile organic carbon (Gustafsson,Haghseta et al., 1996). The total carbon in the activated carbon, charcoal, and biochar was deemed BC. Specific surface area (SSA) and microporosity were determined by Brunauer-Emmett-Teller (BET) nitrogen isotherms using Tristar II 3020 Surface Area Analyzer (Micromeritics, Norcross, GA). All parameters were summarized in Table S1.
2.2.Subject organism and DGT device
V. philippinaram, adopted as the test organisms for heavy metal bioaccumulation, were purchased from Shanghai Luchao aquatic market and acclimated under laboratory conditions for more than three weeks prior to the experiments. V. philippinaram is a common commercial shellfish cultured in freshwater sediments, which are widely distributed in China, North Korea, and Japan. There are V. philippinaram along the coast of China from south to north, of which Liaoning Province and coastal Jiangsu Province are particularly rich. During acclimation, the seawater was aerated continuously and the salinity and temperature of the seawater were 25 ± 2C and 20 ± 1C, respectively. The light cycle was 16:8 h (light:dark) and V. philippinaram were fed daily with 2 g of Phaeodactylum per tank (Ngo et al., 2016).
The ZrO-Chelex DGT device was purchased from Nanjing Intelligent Environment Technology Co., Ltd. The thickness of the fixed film and diffusion layers in DGT devices were 0.40 and 0.90 mm, respectively.The DGT device window area was 150 mm × 20 mm (length × width).One DGT device product was cut into six equal pieces and each piece(0.0065 g) represented a DGT device in this study.
2.3.Bioaccumulation experiment
In each of four 5 L glass tanks, approximately 2 kg (wet weight) of the surface sediments (30% water content) was placed at the bottom. The carbon sorbents mentioned above were added individually at a dry weight ratio of 3% before homogenization (Li et al., 2018). We previously examined the influence of the amount of the carbon sorbents on both the adsorption rate and the survival of benthic organisms,finding that the adsorption rate was not significantly improved when carbon-sorbent doses were of more than 3% for pyrethroids (Li et al.,2015). Meanwhile, the survival rate was above 95% when carbon-sorbent dosages were of less than 3%. The tanks containing samples were then sealed and aged at 4C for 28 days before the following bioaccumulation experiment. The bioaccumulation experiment was carried out according to EPA Method 600/R-99/064 and divided into a carbon-amended group and a carbon-free group. Each group had two replicates.
For the carbon-amended group, the surface of the sediments was then covered with 4 L artificial seawater (Method ISO-10253) after the aged sediment samples in the tanks were adjusted to 20C. Thirty V. philippinarams of similar size (length 32 ± 2 mm and height 12 ± 2 mm) and 14 DGT devices were added to each tank for the bioaccumulation experiment, which lasted for 28 days, as outlined by the EPA recommended method. Throughout the experiment, two V. philippinaram and ~2 g of sediments were sampled from each tank,freeze-dried, and analyzed every four days (Li et al., 2018).
Duplicate DGT devices were sampled from the sediments and the surface of each device was immediately rinsed with deionized water.Afterwards, the fixed film was removed from the cases and kept for further extraction, while the membrane was discarded. Before extraction, the fixed film was wiped to remove excess water, before it was placed into a 30 mL glass tube, then immersed in 10 mL NaOH (1 mol/L)for 24 h and in 10 mL HNOfor another 24 h. The extracts were passed through a 0.22 μm glass filter before analysis. The same procedures were used for the carbon-free group.
2.4.Calculating labile concentrations of heavy metals in the sediment using DGT
After introducing a fixed-film extraction factor (f), the extraction rate of target contaminants (Wang, Ding, & Gong, 2016), the accumulation of heavy metals in the fixed film of the DGT, M (ng), was calculated using the equation:

where Cis the measured concentration of heavy metals (μg/L) in the fixed film, Vis the volume of the extraction liquid (15 mL), and Vis the volume of the fixed film (1 mL). The values of fis 1.03, 0.96, 0.94, 0.94,and 0.74 for Cu, Pb, Cd, Cr, and As, respectively (Wang, Ding, & Gong,2016). Within a given period, the concentration of heavy metals within the interface between the DGT device and the pore water (C) was defined as (Ding et al., 2011; Ding et al., 2010):

where M is the accumulated mass of heavy metals over the deployment time (ng), Δg is the thickness of the diffusive film (0.09 cm), and D is the diffusion coefficient of heavy metals in the diffusion layer (cm/s).These parameters were obtained from a DGT website (www.dgtresearch.com). A is the area of the film (5 cm) and t (s) is the time that the DGT applied in the sediments.
2.5.Analytical methods
Agilent 7700x ICP-MS was used for sample analysis. The analytical method was adopted from S´anchez L´opez et al. (2003), with the operating conditions as follows: Radio Frequency Power: 1500W; Carrier Gas Flow: 0.9 L/min; Auxiliary Gas Flow: 0.8 L/min; Nebulizer Gas Flow:0.5 L/min; and Plasma Gas Flow: 15 L/min; Lens Voltage: 7.25 V. The calibration ranges were 0.001–50.000 pg/mL for the heavy metals analyzed. More details on the analytical method for heavy metal species are provided in Section S1.
Four single-species intensive-mode freshwater aquaculture ponds were selected for a DGT-in-situ application. Specifically, they were a grass carp pond (S1) (380 m), a crab pond (S2) (410 m), a soft-shelled turtle pond (S3) (360 m), and a shrimp pond (S4) (380 m), all located in coastal Shanghai near the East China Sea. At the last culturing stage of each pond (S1, S2, S3, and S4) before harvest, two DGT devices were deployed. When withdrawing the DGT devices, two sediment samples were also simultaneously taken and analyzed for exchangeable and carbonate-bound (acid soluble, F1) heavy metals.
2.6.Quality control
One blank sample (Milli-Q water) was included for every 5 samples to check for laboratory contamination. The average proportion of recovered spiked heavy metals ranged from 92 to 105% for sediments,89–103% for organisms, and 87–99% for DGTs. Replication of recovery showed high reproducibility with a coefficient of variation (CV) of less than 20% for all the samples (Table S2).
2.7.Statistics
Origin Pro 8 (OriginLab Corp. USA) was used to calculate the adsorption rate constants. The relationship between the concentrations of heavy metals in DGT and V. philippinaram was evaluated by t-test and linear-regression analyses using GraphPad Prism 6 (GraphPad Software,Inc., USA).
3.Results
3.1.Effectiveness of carbon sorbents in reducing bioaccumulated heavy metals in V. philippinaram
The effectiveness of carbon sorbents on the bioaccumulation of heavy metals in V. philippinaram was assessed in an in-vivo experiment.The contents of the target heavy metals in V. philippinaram from the carbon-amended groups decreased over various extents over 28 days compared with the carbon-free groups (control) (Fig. 1). This indicated that the carbon materials inhibited the enrichment of heavy metals in organisms to a certain degree. The order of the increasing overall inhibitory effect was: charcoal, peat, activated carbon, and biochar.
Specifically, the reductions in accumulation of heavy metals over 28 days ranged from 13.6 to 20.7%, 24.7–34.6%, 35.7–52.8%, and 26.6–40.2% for the charcoal group, peat soil group, biochar group, and activated carbon group, respectively. The most significant bioaccumulation reduction for all four heavy metals was observed in the biochar group and the order of increasing inhibitory effects on the absorption of heavy metals was: Cr, Cd, Cu, and Pb (Table S3).
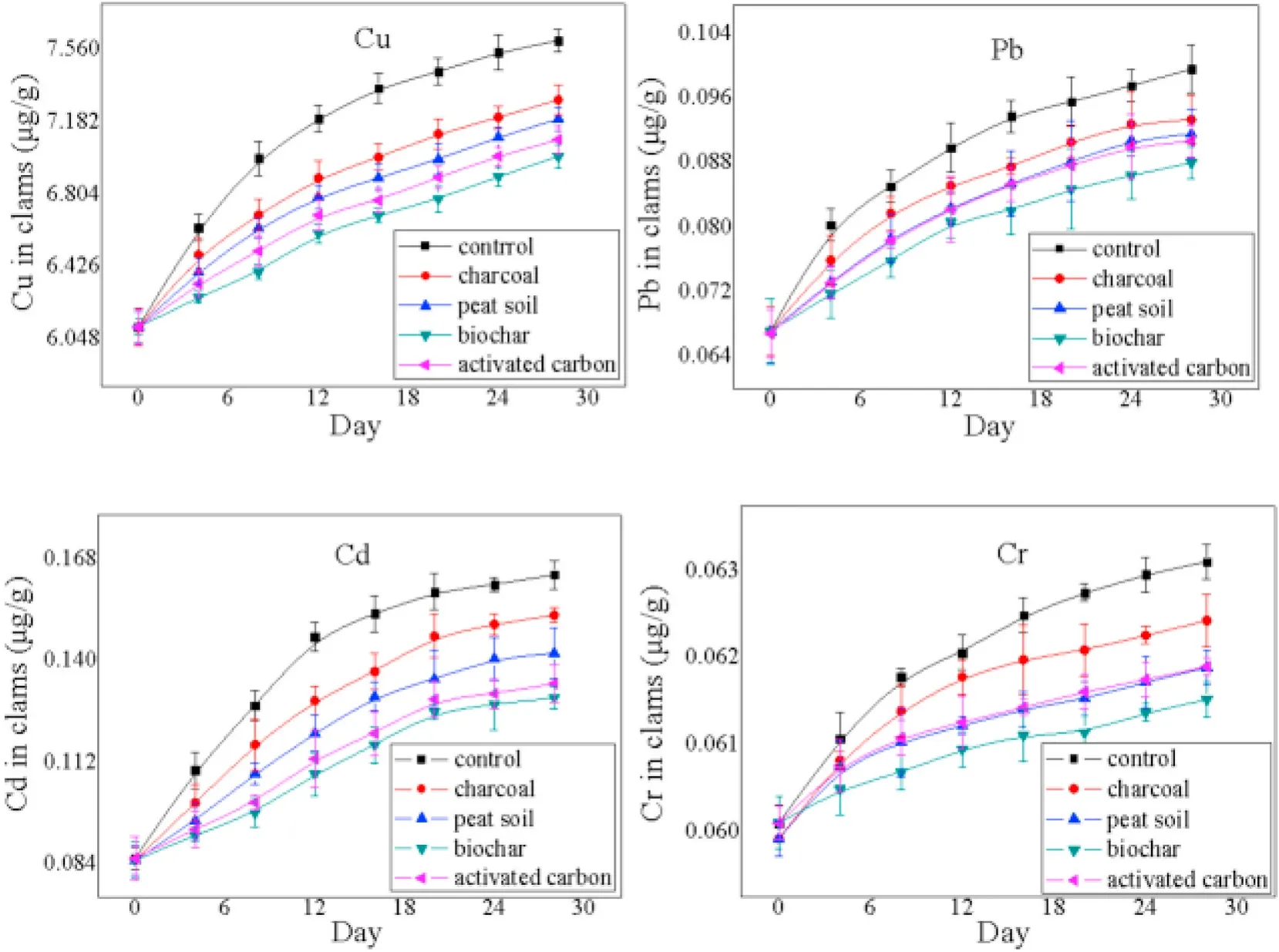
Fig. 1.Concentrations of heavy metals in V. philippinaram during bioaccumulation experiments (error bars represent the standard error for n =3 measurements).
3.2.Effectiveness of carbon sorbents in reducing CDGT
The influence of carbon sorbents on the bioavailability of heavy metals in sediments (Caccording to Eq. (2)) was further analyzed using DGT devices, where the DGT was used to measure the labile heavy metals in sediment pore water (Fig. 2). The dry weight of DGT used was 0.0065 g.

Fig. 2.CDGT values during bioaccumulation experiments (error bars represent the standard error for n =3 measurements).
The Ccontent of four heavy metals decreased by 5.4–13.0%,12.0–25.0%, 34.0–42.0%, and 24.0–33.0% for the charcoal carbon group, peat soil carbon group, biochar group, and activated carbon group, respectively. The highest inhibitory effect on Cwas observed in the biochar group and the order of the increasing effects was: Cu, Cr,Cd, and Pb, with a reduction of Cof 42%, 38%, 35%, and 34%,respectively (Table S3).
3.3.Correlation between the reduction rates of bioaccumulation of heavy metals in V. philippinaram and in CDGT
There were significant correlations (P <0.005) between the reduction rates of heavy metals bioaccumulated in V. philippinaram and that of Cover 28 days of the experiment (Fig. 3).
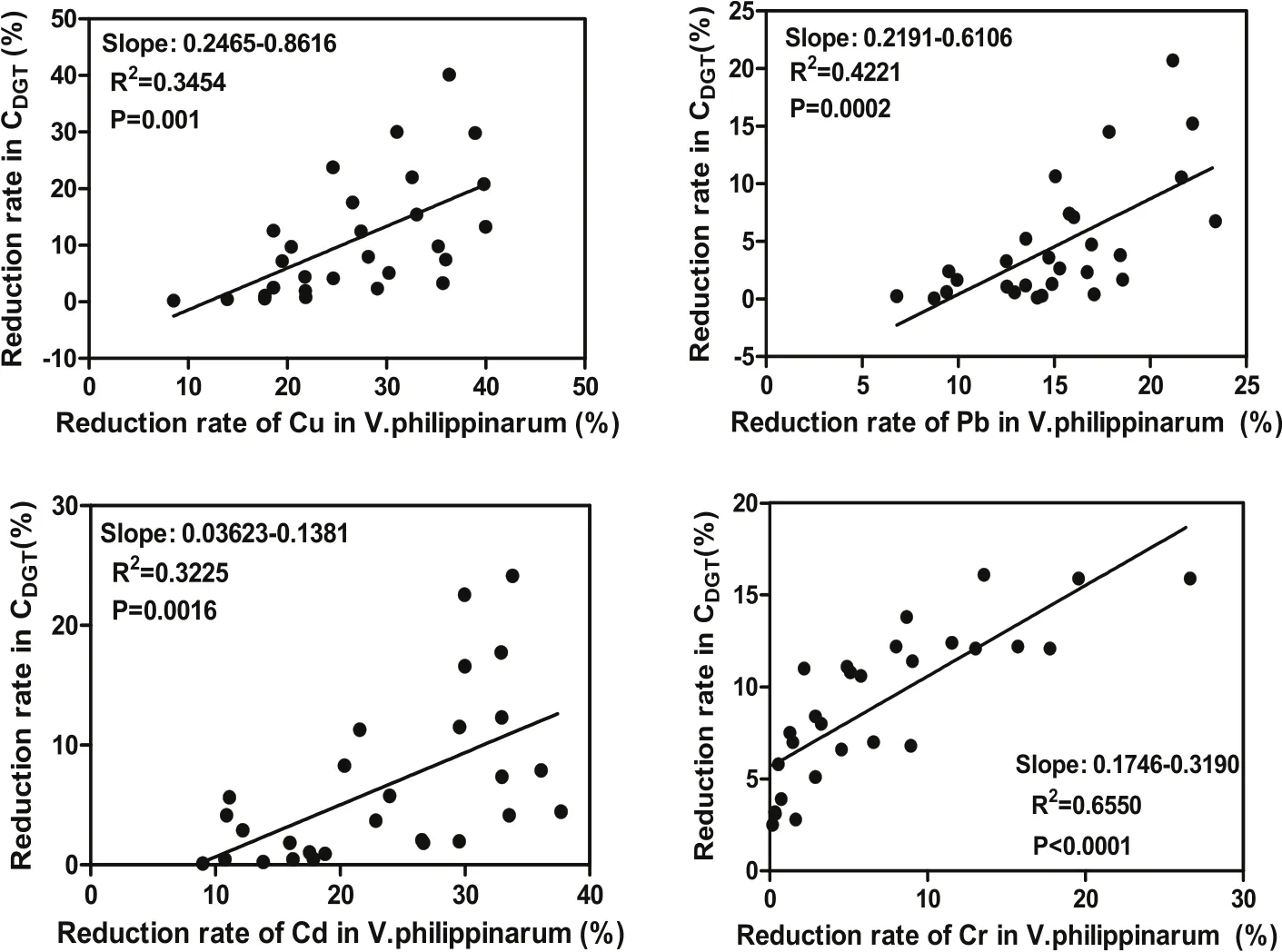
Fig. 3.Correlation between the reduction rates of heavy metals bioaccumulated in V. philippinaram and the reduction rates of CDGT over 28 days.
3.4.Correlation between the reduction rate in CDGT and the reduction rate of acid soluble heavy metals in sediment
The correlations between the reduction rates of Cand the four species of heavy metals (acid soluble, F1; reducible, F2; oxidable, F3;and residual, F4) in sediments were investigated separately. Only a significant correlation between Cand the acid-soluble heavy metals was found in sediments. As the four species of heavy metals were only assessed at the beginning and the end of the 28-day experiment, there were only four data points available for the correlation analysis (Fig. 4).The increasing degree of correlations was: Cu, Pb, Cd, and Cr, with each P-value less than 0.05.

Fig. 4.Correlation between the reduction rate in CDGT and the reduction rate of the amount of acid soluble heavy metals in sediments.
3.5.DGT in situ application for predicting the amount of acid soluble heavy metals
The correlation between the amount of heavy metals accumulated in DGT and the amount of acid-soluble heavy metals in sediments were further investigated. Since the amount of acid-soluble Cr and Cd were below detection limits, only the correlations for Cd and Pb were obtained (Fig. 5).
Significant correlations between the accumulations in DGT and the acid-soluble portion accumulated in sediments were found for both Cu and Pb, with R-square values of the linear regressions equaling 0.97 and 0.92, respectively (Fig. 5).
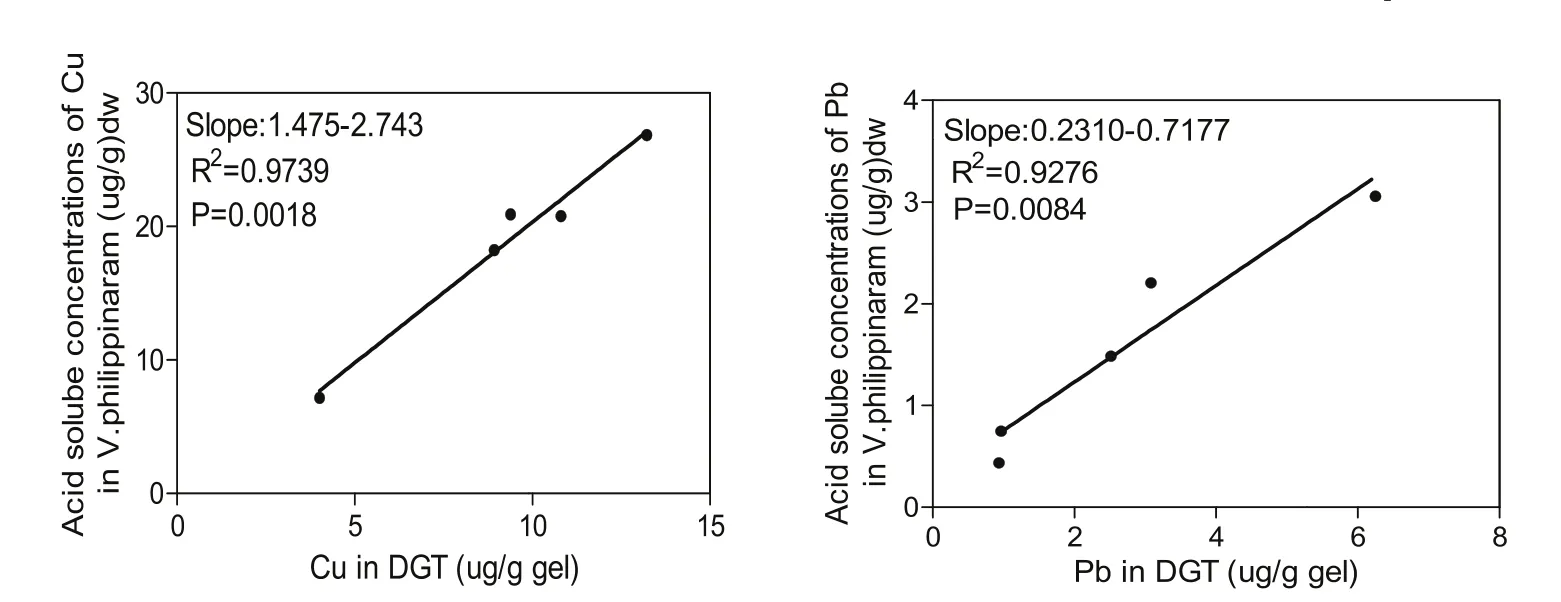
Fig. 5.Correlation between the amount of Cu and Pb accumulated with in situ DGT and the amount of acid-soluble heavy metals in corresponding sediments.
4.Discussion
The significant inhibitory effects (14–53%) of using adsorbents on the enrichment of heavy metals in organisms suggested efficient remediation for the in situ application, especially with biochar. The sorption efficacies of three materials (biochar, activated carbon, and peat soil)towards heavy metals were found to be similar and higher than that of charcoal. It is probably because that the dominant parameters for the sorption of heavy metals are specific surface area (SSA) and black carbon(BC) content (Nejadshafiee & Islami, 2019). In this study, biochar and activated carbon showed similar SSA values, while biochar had the highest amount of BC (Table S1). The presence of BC, which has a high sorption capacity, would largely modify the bioavailable fraction of sediment-associated heavy metals (Keiluweit et al., 2010).
After adding carbon sorbents into the sediments, the enrichment of heavy metals in V. philippinaram decreased notably. Meanwhile, there was no significant correlation between the rate of reduction for heavy metals in sediments and V. philippinaram (Fig. S1). It suggested that the decreased level of heavy metals in V. philippinaram might be due to the decreased amount of labile heavy metals in sediment pore water, as it is the most important driving force for bioaccumulation (Amato et al.,2018; Li et al., 2018). Thus, the influence of carbon sorbents on the bioavailability of heavy metals in sediment (Caccording to Eq. (2))was further analyzed, where the DGT was used to measure the labile heavy metals in sediment pore water.
Cdecreased with time for both the control group and carbonadded groups (Fig. 2). Added carbon sorbents reduced the content of heavy metals that enriched on the DGT film more than that of the carbon-free groups. The order of the sorption capacity of carbon sorbents was as same as the order for bioaccumulation in V. philippinaram.To further explore the feasibility of DGT as a biomimetic prediction tool,the correlation between reduction rates of bioaccumulation of heavy metals in V. philippinaram and in Cwas further investigated. The results indicated that it is possible to use DGT as a biomimetic tool to predict the bioavailability-based stabilization efficacy of carbon adsorbents on heavy metals in sediments (Cheng et al., 2019; Dabrin et al.,2012). However, the deviation of biomimetic prediction should not be overlooked, with the highest R value of 0.66 for Cr (Fig. 3). The accuracy of the bioaccumulation prediction by DGT was not as high as found by Amato et al. (2014; 2015), but higher than Zhang et al. (2019), which focused on freshwater benthic organisms. The process of heavy metal enrichment is quite complicated for living organisms, including absorption and in-vivo transformation processes, while the enrichment in the DGT device is relatively easy and relies more on the active capacity of heavy metals in sediments during a purely physical adsorption process (Cheng et al., 2019). Therefore, when simulating heavy metal bioaccumulation from sediments via DGT, it is necessary to consider the major biases caused by in-vitro disturbances, in-vivo transformation,and indiscrimination of heavy metal speciation.
In an attempt to explain the feasibility of using DGT as a tool for predicting the liable part of heavy metals in sediments, the correlations between the reduction rate of Cand the reduction rates of four species of heavy metals (acid-soluble, F1; reducible, F2; oxidable, F3;and residual, F4) in sediment, respectively, were investigated. Significantly positive correlations were found between Cand the acidsoluble heavy metals in sediment. This result suggested that DGT could reflect the change of bioavailable (i.e. acid-soluble portion) heavy metals in sediments. This further explained why the DGT device reliably corresponded with the accumulated bioavailable heavy metal portion in organisms. Similarly, Kong et al. (2003) used the MetPLATE™ model to show that the amount of acid-soluble heavy metals (i.e. exchangeable and carbonate-bound heavy metals) had strong linear relationships to their toxicities. Although Dabrin et al. (2012) found that Cd bound to the carbonate phase was weakly reactive to the supply of DGT demand, the authors did not investigate the relationship between the DGT demand and any other phases of Cd. The addition of carbon sorbents led to different proportions of different heavy metals spices in sediment, which might further lead to differences in the bioavailability of heavy metals.Therefore, in situ monitoring of DGT can be used to reliably reflect the effectiveness of the sediment remediation.
In order to further verify the precision of using DGT for predicting the accumulation of heavy metals in benthic organisms, four singlespecies intensive-mode freshwater aquaculture ponds were selected for a DGT-in-situ application. The correlation between the amount of heavy metals accumulated in DGT and the amount of acid-soluble heavy metals in sediments were further assessed. The results suggested that the DGT device can be used as a reliable tool for assessing heavy metal bioavailability in practical applications, such as in the culture ponds, at least for Cd and Pb (Fig. 5).
5.Conclusions
In general, the results demonstrated that a DGT-based passive sampling technique effectively senses the changing bioavailability of several heavy metals in amended sediments and evaluates the effectiveness of remediation measures. As a renewable energy-focused product of pyrolysis at a relatively low cost, biochar would be an ideal material for the remediation of heavy metal pollution in sediments, improving the quality and safety of the aquatic products in aquaculture. The significant correlations between the reduction rates of heavy metals bioaccumulated in V. philippinaram and the reduction rates of Cover 28 days demonstrated that DGT could be used as a biomimetic tool to predict the enrichment of heavy metals in benthic organisms from the sediments amended by carbon adsorbents.
Moreover, the correlations between the reduction rates in Cand four species of heavy metals (acid-soluble, reducible, oxidable, and residual) suggested that only the positive correlation between Cand the acid-soluble heavy metals in sediments was significant. This further explained that why the DGT device can reliably indicate the accumulated bioavailable heavy metal levels in organisms. Further studies should focus on other potential major simulation biases using DGT, such as in-vitro disturbances and in-vivo transformation with different species of organisms.
CRediT authorship contribution statement
Yiqin Chen: Conceptualization, Methodology, Software, Writing -original draft, Funding acquisition, Project administration. Yueyue Wang: Investigation, Data curation, Software, Writing - original draft.Ruyi Zheng: Investigation, Data curation, Writing - original draft. Ju Wen: Validation, Writing - original draft. Juan-Ying Li: Conceptualization, Writing - review & editing, Supervision, Funding acquisition.Qian Wang: Writing - review & editing. Jie Yin: Investigation,Validation.
Declaration of competing interest
The authors declare that there is no conflicts of interest.
Acknowledgements
This research was supported by Science and Technology Commission of Shanghai Municipality (18050502100) and Shanghai Ocean University (A1-2037-16-0001-12, A2-0203-00-100223 and A2-0203-00-100352).
Appendix A.Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.aaf.2020.07.007.
 Aquaculture and Fisheries2021年6期
Aquaculture and Fisheries2021年6期
- Aquaculture and Fisheries的其它文章
- Overview of aquaculture systems in Egypt and Nigeria, prospects,potentials, and constraints☆
- microRNA expression profile offish erythrocytes
- Medaka gcnf is a component of chromatoid body during spermiogenesis
- Environmental and energy requirements for different production biomass of Nile tilapia (Oreochromis niloticus) in recirculating aquaculture systems(RAS) in Kenya
- Some reproductive and biometric features of the endangered Gangetic Leaf Fish, Nandus nandus (Hamilton, 1822): Implication to the baor fisheries management in Bangladesh
- The role of nitric oxide and neuronal nitric oxide synthase in zebrafish(Danio rerio) shoaling.
