Antibody Therapies Targeting Complex Membrane Proteins
Georgin To’ Slzr,Ziyi Hung,Ningyn Zhng,Xue-Gung Zhng,e,*,Zhiqing An,*
a Texas Therapeutics Institute, Brown Foundation Institute of Molecular Medicine, The University of Texas Health Science Center at Houston, Houston, TX 77030, USA
b Jiangsu Institute of Clinical Immunology, The First Affiliated Hospital of Soochow University, Suzhou 215123, China
c Jiangsu Key Laboratory of Clinical Immunology, Soochow University, Suzhou 215123, China
d Jiangsu Key Laboratory of Gastrointestinal Tumor Immunology, The First Affiliation Hospital of Soochow University, Suzhou 215123, China
e State Key Laboratory of Radiation Medicine and Protection, Soochow University, Suzhou 215123, China
Keywords:Antibody therapy Complex membrane protein Ion channels Transporters Membrane-bound enzymes GPCRs Drug discovery
ABSTRACT In analyses of protein families that may serve as drug targets, membrane-associated G-protein-coupled receptors (GPCRs) dominate, followed by ion channels, transporters, and—to a lesser extent—membrane-bound enzymes. However, various challenges put such membrane proteins among key groups of underutilized opportunities for the application of therapeutic antibodies. Antibodies hold the promise of exquisite specificity, as they are able to target even specific conformations of a particular membrane protein, as well as adaptability through engineering into various antibody formats.However, the ease of raising and isolating specific, effective antibodies targeting membrane proteins depends on many factors.In particular,the generation of specific antibodies is easier when targeting larger,simpler,extracellular domains with greater uniqueness of amino acid sequence.The rareness of such ideal conditions is illustrated by the limited number of approved biologics for targeting GPCRs and other complex membrane proteins. Challenges in developing antibodies to complex membrane proteins such as GPCRs, ion channels, transporters, and membrane-bound enzymes can be addressed by the design of the antigen, antibody-generation strategies, lead optimization technologies, and antibody modalities.A better understanding of the membrane proteins being targeted would facilitate mechanism-based drug discovery.This review describes the advantages and challenges of targeting complex membrane proteins with antibodies and discusses the preparation of membrane protein antigens and antibody generation,illustrated by select examples of success.
1. Introduction
As of September 2021,there are 127 antibodies marketed in the United States or European Union,according to a list maintained by the Antibody Society [1]. Of these, over half—77 of 127—target membrane proteins.However,the majority of these antibodies target membrane proteins with a simple transmembrane domain and a large extracellular domain (ECD), such as tyrosine kinase receptors. Only three target complex membrane proteins, in particular G-protein-coupled receptors (GPCRs). It is notable that approximately 40% of all approved drugs work through GPCRs; this discrepancy highlights the opportunity for therapeutic antibodies targeting complex membrane proteins [2]. In fact, ion channels,transporters, GPCRs, and membrane-bound enzymes are among the largest classes of membrane proteins for drug discovery [3].
Several excellent reviews have been published on antibodies targeting membrane proteins by the industry and by academic groups focused on this topic [2-7]. The current review focuses on the ways in which therapeutic antibody discovery and development technologies are evolving the possibilities for antibodybased targeting of difficult membrane proteins. This category of proteins includes GPCRs, ion channels, transporters, and membrane-bound enzymes.
GPCRs and ion channels are among the protein families that are most targeted by small molecule therapeutics; a 2016 analysis of the US Food and Drug Administration(FDA)-approved drug targets showed that GPCRs and ion channels are responsible for 33% and 18% of small molecule therapeutic effects, respectively [8]. Yet GPCRs and ion channels have been highlighted as understudied opportunities for biomedical research and clinical development[9]. The limitations that have been encountered in the development of small molecule drugs against complex membrane proteins include problems with selectivity. Although small molecules can reach small pockets more easily than antibodies can, antibodies can readily target complex membrane proteins with sufficiently large ECDs. One example of such complex membrane proteins is C-C chemokine receptor type 4 (CCR4), the GPCR targeted by the approved therapeutic mogamulizumab for the treatment of relapsed or refractory mycosis fungoides and Sézary disease [10].Erenumab, the GPCR targeting calcitonin gene-related peptide receptor (CGRPR) approved for the prevention of migraine, is another example [11]. Engineerability is a key feature of several advantages of targeting complex membrane proteins with antibodies. The superior selectivity and specificity of antibodies are especially advantageous when a particular conformation of a protein is associated with a disease condition.One example is monoclonal antibody (mAb) BIL010t, which targets the nonfunctional conformation of the ion channel P2X7 (nfP2X7), and which is currently in clinical trials for the treatment of basal cell carcinoma[12].Conjugation to antibodies can improve the biodistribution and pharmacokinetics of candidate therapeutics. This was illustrated in a study of an ion channel inhibitor that achieved a greatly extended half-life and better distribution to nerve fibers [13]. In addition to the direct effects of binding, antibodies can act through effector functions such as antibody-dependent cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). The success of mogamulizumab demonstrates the promise of effector function modifications for empowering therapeutic antibodies [14].
Innovative crystallization and other structural biology techniques have empowered researchers to better understand membrane proteins [15]. Peptides made cyclic to mimic the native structure of membrane proteins have enhanced the production of antigens for raising antibodies targeting these proteins [16]. The isolation of therapeutic antibodies targeting membrane proteins also benefits from technological advances,such as DNA immunization with Escherichia coli (E. coli) GroEL as a molecular adjuvant to produce strong and specific antibody responses [17].
Four key challenges have historically impeded the development of antibodies targeting complex membrane proteins. A first key challenge is the isolation of pure protein stabilized in the desired conformation to serve as an antigen to isolate antibodies.A second key challenge is overcoming immunological tolerance—that is,the tendency of an organism’s immune system to be unresponsive to substances that resemble the organism’s own normal proteins.Therefore, immune tolerance can inhibit the generation of a large and diverse panel of antibodies against these proteins. A third key challenge for the development of therapeutic mAbs against complex membrane proteins is that the extracellular epitope areas of these targets—the area most available to antibodies—can differ substantially. Efficiently screening isolated antibodies for effectively altering the function of their target membrane proteins is a fourth key challenge of targeting complex membrane proteins with therapeutic antibodies.
Diverse strategies have been applied as technological solutions to the challenges of generating antibodies targeting complex membrane proteins. These solutions have achieved some success at unlocking the advantages of these potential therapies. Examples of antibody therapies in clinical and preclinical development against complex membrane proteins are listed in Table 1 [12,18-40]. The developing pipeline of antibodies targeting complex membrane proteins can be meaningfully grouped according to target class, antigen format, antibody format, and antibody production platform. Varying degrees of information are available about their ligands, binding, mechanisms of action, effectiveness, epitopes, therapeutic indications, and side effects (Table 1).
The penetration challenge might be addressed by antibody engineering.If the half-life in circulation is a challenge,engineering the fragment crystallizable region(Fc)portion of antibodies can be used to modify the half-life in either direction. Furthermore, Fc effector mechanisms such as ADCC can be increased or decreased by antibody engineering. Complex membrane proteins selectively activate specific signaling pathways based on a number of factors.These factors include ligands and other interacting proteins. Antibodies have shown the capacity to recognize and stabilize specific states or conformations of GPCRs and ion channels [12,41-43].Accordingly, these antibodies can be used to bias the function of a complex membrane protein toward a preferred pathway.
By searching ClinicalTrials.gov, PubMed, and lists prepared by the Antibody Society, we identified complex-membrane-proteintargeting antibodies currently in preclinical and clinical development. Using representative examples, we herein discuss the mechanisms of action of antibodies targeting these complex membrane proteins, relevant sources and preparation of membrane protein antigens, and antibody generation.
2. Mechanisms of action of antibodies targeting complex membrane proteins
GPCRs function by being activated by an extracellular ligand at an ECD and transmitting a signal into a cell using an intracellular domain [44]. Ion channels function by forming a pore through which ions can cross the hydrophobic core of cellular membranes[45].Ion channels,GPCRs,and other membrane-bound proteins are active in many important cellular processes and have identified roles in diverse diseases as well.As such,they are attractive targets for pharmacological manipulation to promote healthy function or prevent pathological function [8,9]. Since they are so commonly used for biological processes and since their structures are highly conserved,it is of particular importance that therapeutic interventions against these targets have highly tuned selectivity.This is one criterion that makes antibodies especially promising therapeutic options. The diversity of the structures of membrane-bound proteins, such as ion channels and GPCRs, and of membrane-bound enzymes, such as matrix metalloproteases, creates an abundance of opportunities for interventions regulating specific functions of these proteins. This abundance also contributes to the promise of therapeutic antibodies against complex membrane proteins; the complex structures of antibodies can be engineered in many ways to match a wide variety of targets.
Some structures and potential points of interaction for antibodies and GPCRs are shown in Fig. 1(a). One path for antibody interaction with a GPCR is through stabilizing an active state; for example,antibodies acting as agonistic ligands were raised against human GPR56 to analyze its function in human glioma cells[46].A second path for antibody interaction with a GPCR is agonist activation by promoting dimerization; antibodies against metabotropic glutamate receptor 7 and β1-adrenergic receptor have been shown to be effective at activating GPCRs this way [47,48]. Alternatively,antibodies may act as antagonists inactivating a GPCR. One example is the case of a diverse array of antagonistic antibodies that were raised against the glucose-dependent insulinotropic polypeptide (GIP) receptor [49]. These GIP-receptor-targeting antibodies were presented as having potential as candidate therapies for the treatment of people affected by diabetes or obesity. Mogamulizumab was approved in 2018 for the treatment of lymphomas Sézary syndrome and mycosis fungoides [50]. Mogamulizumab targets the GPCR CCR4, but has not been shown to promote its activity as an agonist or inhibit its activity as an antagonist.Instead, mogamulizumab functions by depleting cells expressing CCR4 in the tumor environment,including tumor cells and regula-tory T cells inhibiting the host’s anti-tumor response [18,51,52].The cell-depleting effect of mogamulizumab has been shown to function through ADCC enhanced by Fc engineering,namely,defucosylation[18].Erenumab is another GPCR-targeting antibody that was approved in 2018 for the prevention of migraine in adults[11]. Erenumab acts as an antagonist against the GPCR known as CGRPR by competing with ligand binding [25,26]. One path of interaction between an antibody and an ion channel is illustrated in Fig. 1(b); an antibody can promote inhibition through internalization.An example of an antibody shown to promote internalization and inhibition of ion channel function targets is calcium release-activated calcium channel protein 1 (Orai-1) [37]. Orai-1 is an autoimmune disorder target that forms pores for the calcium influx necessary for T cell activation [53]. Targeting specific states is yet another path by which antibodies may modify the function of a complex membrane protein.This path is illustrated in Fig.1(c).Antibodies have been generated to target both the inward-open and outward-open states of the glucose transporter GLUT4 [38].Membrane-bound enzymes are another category of complex membrane proteins whose function has been effectively modified by antibodies, as illustrated in Fig. 1(d). Using newly available information about the structures of membrane proteins, antibodies have been engineered with corresponding structures designed to reach into deep catalytic sites and ligand binding sites. These antibodies have long complementarity determining region (CDR)loops on the heavy chain (CDR-H3s) encoding convex paratopes.These CDR-H3s are either taken from antibodies isolated from immunized camelids [40,54] or designed to mimic the CDR-H3 regions of such antibodies [39]. The proteinase matrix metalloproteinase-14 (MMP-14), a cancer drug target, is one
example of a membrane protein that has been successfully targeted by antibodies via a long CDR-H3 [39]. Another example is the human apelin receptor (APJ), a target for therapies to treat patients with chronic heart failure [40]. JN241-9, the antibody reported to target the human apelin receptor,is especially remarkable for two reasons:First,APJ is a class A GPCR,which has a smaller N-terminal ECD than more frequently targeted class B GPCRs;and second,JN241-9 functions as an agonist to replace the natural APJ ligand[40].The therapeutic antibody MEDI9447 is reported to antagonize the enzyme CD73 (ecto-5’-nucleotidase) by stabilizing the inactive conformation and by crosslinking inter-CD73 dimers(Fig. 1(d)) [54].
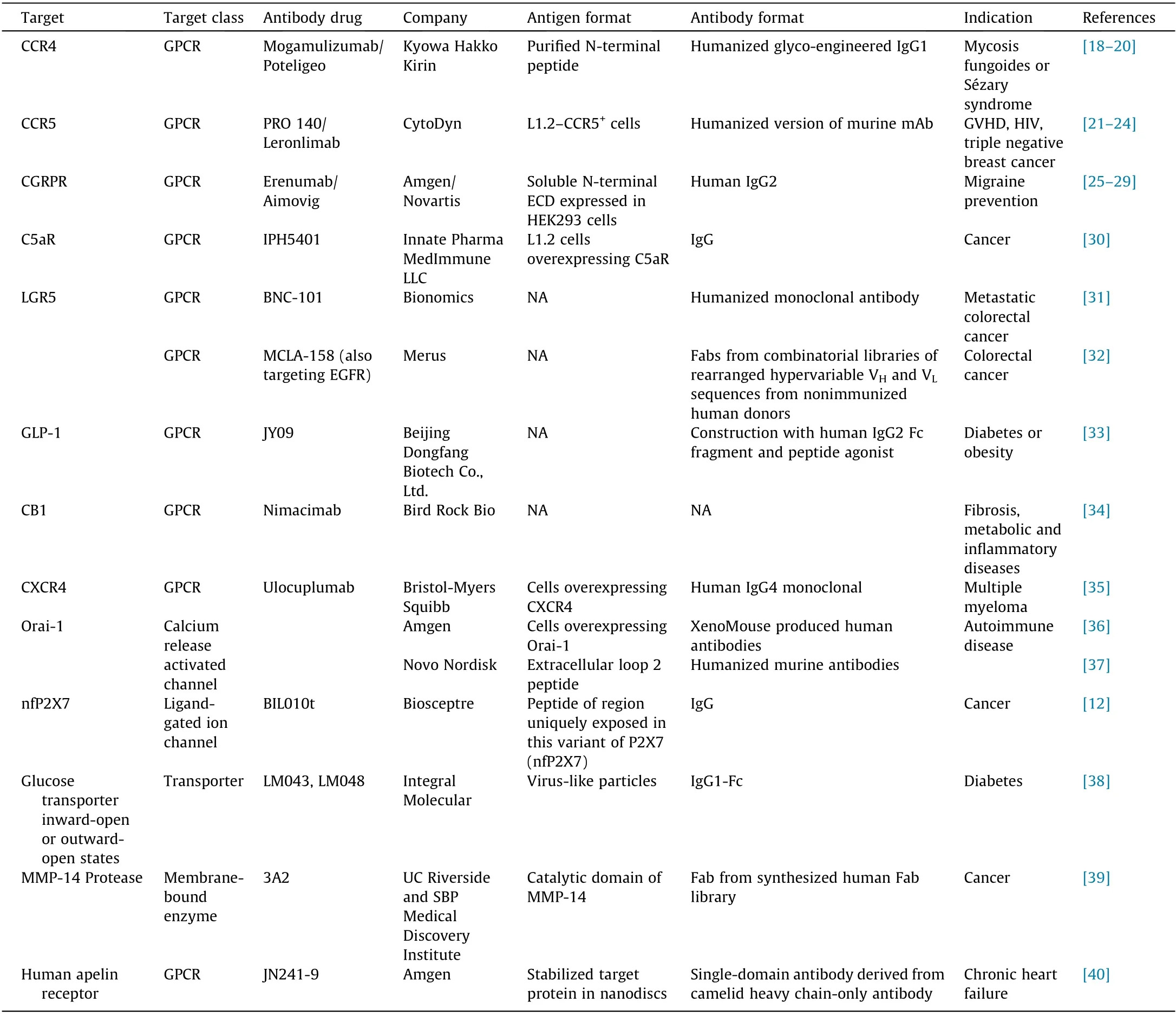
Table 1 Antibodies targeting membrane proteins in development with key technologies employed to overcome challenges.

Fig. 1. Select paths for antibodies targeting membrane proteins. (a) Antibodies can activate GPCRs, for example by acting as agonistic ligands or promoting dimerization.Alternatively, antibodies can inactivate GPCRs, for example by competitively displacing the ligand. (b) Promoting internalization is one way in which antibodies target ion channels. (c) Antibodies have been raised to be specific against a particular functional state, in this case targeting inward-open and outward-open states for the uptake of glucose by cells. (d)Antibodies have been designed to specifically to reach into the active site of enzymes such as MMP-14. They can also inactivate enzymes by stabilizing inactive conformations, such as the CD73 dimer.
3. Antigen preparation
Obtaining specific, functional antibodies to modulate the function of membrane proteins begins with the preparation of antigens.The antigens used to generate therapeutic antibodies ideally present all epitopes that are relevant for the desired effect on the target protein. The ideal complex membrane antigen therefore presents conformation and post-translational modifications relevant to the desired effect.The entire protein need not be presented,but relevant domains that may contain epitopes for targeting by antibodies should not be omitted. Alterations may be imposed to generate pure, functionally active antigen in large amounts. These alterations should preserve overall conformation, ligand binding,cell surface expression, processing, and relevant epitopes. Strategies to select epitopes for therapeutic antibodies vary for different classes of complex membrane proteins. For most, the epitopes should be at ECD loops and N-terminal sites[36-38,40].Intracellular epitopes might be targeted as tools to reveal structure and function, such as LM043 against the intracellular loops of the inwardopen conformation of GLUT4[38].For membrane-bound enzymes,the epitope may be at a site where antibody binding would compete with substrate binding [39]. An epitope might be at a ligand binding site, as is the case for JN241-9-targeting apelin receptor[40]. In some cases, an antigen used to generate therapeutic antibodies has been validated for small-molecule therapeutics with the antibodies and small molecules interacting at different epitopes, enabling synergistic effects [21]. Given the size and complexity of complex membrane proteins, the effective discovery of antibodies against these proteins must cover a broad range of diverse epitopes. The selection of antigen format is guided by factors that include an analysis of the structure and function of the target,and information about antigen formats that were previously successful in isolating antibodies against similar targets. Subclasses of membrane protein targets can be defined based on the ways in which the proteins perform their function. For example,the physiological processes involving ion channels can be grouped into categories according to function,such as the coupling of stimulation and secretion.Common systems of classification for GPCRs group these proteins according to sequence,function,and phylogenetic analyses [55,56]. Fig. 2 illustrates select innovative antigen formats that have enabled the successful development of therapeutic mAbs against complex membrane proteins. Developments in antigen generation, including novel detergent calixarenes and lipid nanodisc innovation of saposine-lipoprotein (Salipro) particles, have recently been reviewed more comprehensively elsewhere [4].
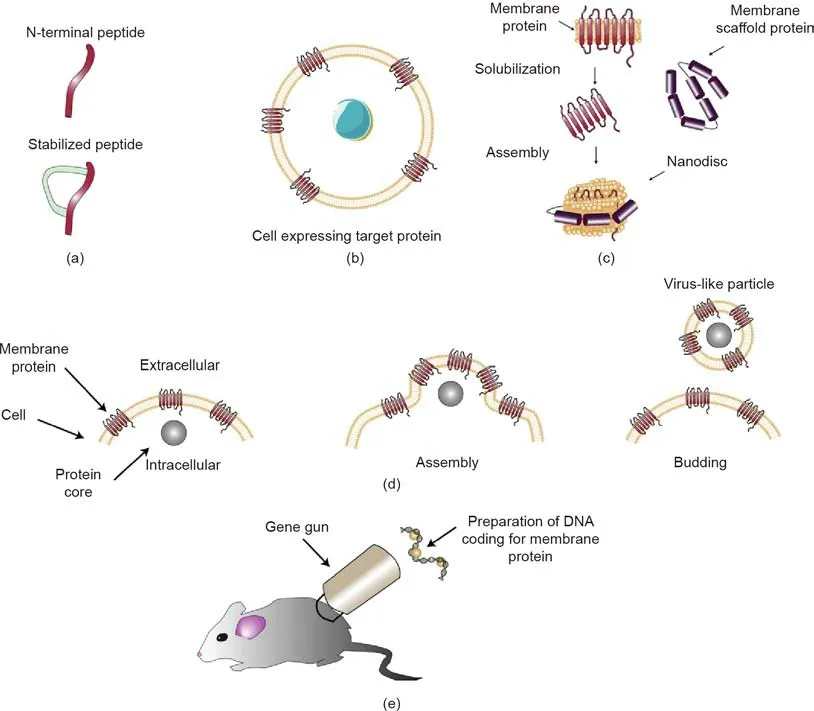
Fig. 2. Select antigen formats. (a) The simplest form of antigen is the peptide that is intended as the target. If necessary, a peptide can be stabilized into a relevant conformation,for example,by the chemical addition of a linker or by cyclization.(b) For more native stabilization of the desired conformation of a membrane protein,cells can be made to express the target membrane protein.(c)Purified membrane proteins may be prepared as antigens in nanodiscs with membrane scaffold proteins supporting nanodisc mimics of cell membrane lipid environments. These mimics preserve the relevant conformation of a membrane protein without the components of a real cell membrane that might generate non-specific antibodies. (d) Virus-like particles are another way to preserve the native conformation of a purified membrane protein for antigenic purposes. (e) DNA immunization of animals with DNA encoding the membrane protein of interest can be part of a strategy to effectively raise immune responses specific to that protein.
3.1. Peptides
Peptides are the simplest antigen format, and are the easiest and most economical to make in large quantities. Three cases in which the peptide antigen format may be preferred are: ①cases in which the required antibody only needs to bind to a receptor in order to deplete a particular population of cells; ②cases in which the extracellular N-terminus of the receptor is directly involved in its function;and ③cases in which the membrane protein has a large ECD.One successful application of peptides to generate antibodies against GPCRs involved CCR4 [19]. Peptides were one of the antigen formats used to generate antibodies against the ion channel Orai-1 [37]. Peptides can be stabilized by being constrained, or made cyclic, to mimic the native structure [57](Fig. 2(a)). One example of this strategy is the chemical linkage of peptides onto scaffolds (CLIPS), a technique that has been successfully applied to generate antibodies against the GPCR CXC chemokine receptor-2 (CXCR2) [16]. Fig. 2(a) illustrates extracellular N-terminal peptides,with and without stabilizing constraints,used as an antigen to generate antibodies targeting complex membrane proteins.
3.2. Cells expressing membrane peptides or proteins
Lipid environments remain important for the correct conformation and activity of complex membrane proteins. More complex antigen formats incorporating native structures are used with the objective of improving the chances of isolating antibodies with the desired biological functions. mAbs with therapeutic potential have been created by means of immunization using cells expressing membrane peptides or proteins [36,58], membrane proteins with structures such as nanodiscs simulating the native membrane environment [59], antigen formats in which virus-like particles take the place of the native membrane [38,60], and DNA encoding the membrane protein of interest [61,62].
Cells can be used to express membrane proteins, as illustrated in Fig. 2(b). Ideally, expression hosts should produce functional,correctly folded protein at high levels, with proper posttranslational modifications. They should also be adaptable to large-scale culture. Large-scale transient transfection methods,such as the MaxCyte electroporation system[63],facilitate the isolation of cells expressing membrane proteins, even when the successfully transfected cell is rare.An mAb targeting the leucine-rich repeat-containing G-protein-coupled receptor 5 (LGR5) is one example of an antibody that was successfully generated using the cells expressing the format of the membrane proteins [64]. In this case, the receptor peptide was co-transfected with receptor activity-modifying protein (RAMP). The strategy of immunization with cells expressing membrane peptides or proteins has also been effectively employed to produce antibodies modulating the function of ion channels. Antibodies inhibiting ion channel function have been generated by the immunization of animals expressing the calcium-release activated channel Orai-1 [36] and the ligandgated ion channel P2X7 [58]. Orai-1 is targeted for the treatment of patients with autoimmune disease [36,37]. Blocking P2X7 can be helpful in the treatment of patients with inflammatory disease[65].
3.3. Purified membrane proteins: Nanodiscs and SMALPs
Historically,detergents were used to purify membrane proteins from their membranes. Mutations can be used to stabilize membrane proteins in detergent [66,67]. One solution to the requirement of providing a lipid environment in order to enhance the native quality of the membrane protein folding is nanodiscs or lipid discs [68]. Functional apelin receptor APJ-agonist antibodies were generated and isolated using an engineered version of the APJ receptor expressed in insect cells and reconstituted in nanodiscs and liposomes [40]. Another such solution is styrene maleic acid lipid particles (SMALPs) [69,70]. These are two examples of alternatives to detergents for isolating membrane proteins from membranes while preserving their structure, function, and other features that are important for generating antibodies against complex membrane proteins. The process of preparing antigens from purified membrane proteins,lipid environments,and scaffold proteins is illustrated in Fig. 2(c).
3.4. Virus-like particles and proteoliposomes
Other antigen formats that mimic native membranes include virus-like particles (VLPs), a lipid-bilayer-based antigen format that resembles native cellular membranes more than nanodiscs.This form of antigen has been employed, along with strategies to optimize immunization, for the discovery of antibodies targeting complex membrane proteins including GPCRs, ion channels, and transporters [38,71,72]. VLP antigens were used along with the immunization of a divergent species—namely, chicken—in order to isolate state-dependent GLUT4 antibodies useful for characterizing GLUT4 and for studies toward the development of new therapeutics against diabetes [38]. The process of generating a viruslike lipoparticle is illustrated in Fig. 2(d).
3.5. DNA immunization
DNA coding for a receptor can be delivered into a host that will both express the membrane protein and produce antibodies against that protein. Fig. 2(e) illustrates this strategy of antigen preparation.A gene gun can be used to introduce coated DNA particles so that they will be expressed by cells presenting the antigen[73,74]. Choosing adjuvants to boost the immune response presents another opportunity for optimization. DNA immunization has been employed to generate antibodies activating the β1-adrenergic receptor with stabilized receptor (StaR) proteins injected following complementary DNA (cDNA) delivery in order to boost the immune response [47]. The co-injection of plasmids that expressed the GPCR endothelin A receptor (ETAR) and the adjuvant E. coli GroEL generated an enhanced immune response to the GPCR [17].
4. Antibody generation
When researchers study complex membrane proteins as targets of therapeutic antibodies, they focus on topics such as the structure and function of the targeted proteins,the process of preparing antigens in different formats,and strategies for antibody isolation.Limited access to enabling technologies is an additional obstacle that impedes the advancement of lead antibodies from academic laboratories to the preclinical development stage.
Technological platforms for antibody generation involve tools and expertise for generating and maintaining diverse sources of antibodies;different methods for isolating antibodies that perform a desired function; and optimizing isolated antibodies through affinity maturation, humanization, ADCC enhancement, presentation in various formats,and other strategies.Fig.3 illustrates select platform options for the generation of optimized antibodies.
Ease of access to advanced antibody discovery and generation technology can help improve the efficiency of the antibody isolation process. This efficiency, in turn, enables the isolation of rare antibodies that are effective against difficult targets such as complex membrane proteins.

Fig. 3. Select platforms used to generate antibodies targeting membrane proteins. Once a membrane protein antigen is prepared, it can be used to generate antibodies targeting the membrane protein. From this point, three stages can be defined for the generation of an optimized antibody. (a) Creating a source of diverse candidate antibodies is a first stage.This might be done through the immunization of animals or construction of natural or synthetic antibody libraries.Wild-type mice,mice deficient in the target protein,and immunoglobulin-humanized mice can be immunized.Sheep,chickens,camels,and llamas have also been immunized for the successful generation of candidate therapeutic antibodies targeting membrane proteins.Non-human primates are another option.Human donors and structural and functional analyses of antibodies can be used to create natural or synthetic antibody libraries that can be cloned into vectors for antibody expression.(b)Isolation is a second stage in the antibody-generation process. From a large number of candidates, the most promising lead antibodies can be isolated for larger scale production through screening phage libraries or antibodies produced by hybridomas or cloning antibody genes from individual plasma or memory B cells. (c) Once preliminary assays produce a further refined list of candidate therapeutic antibodies, these antibodies can be further optimized by means of engineering techniques including afucosylation and construction as different modalities,including IgG,Fab,single chain antibody fragment(scFv),domain antibodies,bispecific antibodies,and antibodies with‘‘payloads”—that is,drugs,oligonucleotides,or radioconjugates. RT-PCR: reverse transcription-polymerase chain reaction;α-1,6-fucosyltransferase (FUT8).
4.1. Animals and libraries as antibody sources: The isolation of rare antibodies
The immunization of animals with a prepared antigen is an important source of antibodies.The choice of animal is key.Several different animals that have been used for immunization as antibody sources are illustrated in Fig. 3(a). Wild-type mice are one choice. An antibody-blocking lipid-activated GPCR S1P3, called EDD 7H9, was generated by immunizing wild-type mice with a synthetic peptide [75]. The objectives for the development of this therapeutic antibody included inhibition of breast cancer tumor growth and management of sepsis.As mentioned in the discussion of antigen formats, Orai-1 is a calcium-release activated channel that is targeted for the management of autoimmune disease. This is another example of a protein that has been effectively targeted with antibodies generated through the immunization of wildtype mice. Antibodies blocking T cell function by targeting Orai-1 have been generated through the immunization of wild-type BALB/c mice with a peptide spanning the second extracellular loop(ECL2) of Orai-1 [37]. Anti-Orai-1 antibodies targeting the ECL2 of Orai-1 have also been generated by means of the immunization of a mouse that was genetically engineered to produce human antibodies[36].In this case,the antigen format was cells overexpressing the Orai-1 protein.
Another option for an immunization animal is mice deficient in the target protein. One example of antibodies generated by the immunization of mice deficient in the target protein are GMA102 and GMA105, which target the GPCR glucagon-like peptide-1 receptor (GLP1R). These antibodies have entered phase I clinical trials for the treatment of patients with type 2 diabetes mellitus(T2DM)or obesity.In this case,GLP1R knockout mice were immunized with purified and refolded human GLP1R N-terminal ECD[76]. The variable heavy chain (VH) and variable light chain (VL)regions of the murine antibodies were subsequently cloned into human embryonic kidney suspension cells (HEK293-6E) for production for structural characterization [77].
In addition to wild-type mice and mice that are deficient in the target protein, immunoglobulin-humanized mice are a choice of animal for immunization as a source of fully human antibodies.This genetic engineering enables human antibody responses while maintaining the function of the mouse immune system [78].Douthwaite et al. [79] developed a therapeutic mAb against GPCR formyl peptide receptor 1 (FPR1) using the VelocImmune mouse,along with affinity maturation employing phage display libraries[79]. FPR1 is targeted for the treatment of patients with inflammation-related diseases[80].The GPCR-targeting antibodies erenumab [25] and REMD-477 [81] were isolated using a mouse with human immunoglobulin loci incorporated into a mouse germline:the XenoMouse platform.Another example of the application of this platform is the isolation of antibodies targeting the calcium release activated channel Orai-1 [36]. Another type of immunoglobulin-humanized mice, Medarex KM, were employed to yield antibodies modifying the function of the GPCR CXCR4;one, called ulocuplumab, entered phase I clinical trials [35,82].
It is especially important to consider the immunization of animals other than mice when generating sources of antibodies against difficult membrane proteins. The company Biosceptre moved through phase I clinical trials for the treatment of patients with basal cell carcinoma with an antibody against nfP2X7,a variant of adenosine triphosphate (ATP)-gated calcium channel P2X7,raised by the immunization of sheep with a peptide uniquely exposed in this variant[12].Chickens,as animals that are phylogenetically distant from humans, have been reported as a valuable host for the generation of therapeutic antibody candidates [49].Camelids including llamas are sources of nanobodies, such as an antibody targeting CX3CR1, a chemokine receptor involved in the recruitment of CD8+T cells,for the treatment of patients with renal disease [83]. Camels were immunized as sources for the variable regions of a phage display antibody library from which APJagonist antibodies were isolated [40]. Fig. 3(a) also illustrates libraries of human and synthetic antibodies; these are other options that have been employed as sources of antibodies targeting difficult membrane proteins [38,39]. For human libraries, genetic material is extracted from immune cells isolated from human donors. For synthetic libraries, structural analysis informs the design for the synthesis of genetic material. Then, from extracted or synthesized genetic material, antibody genes are expanded and cloned into a vector for transfection into an expression host.
Whatever the animal source of antibodies may be,the isolation of rare antibodies with the desired function may be accomplished by hybridomas or B cell cloning,as illustrated in Fig.3(b).A hybridoma is produced by the fusion of a myeloma cell with a B cell producing antibodies with the desired antigen specificity [84].
4.2. Modifications to antibody format
Following the isolation of antibodies against difficult membrane proteins,additional obstacles might need to be overcome in order to fine-tune those antibodies as powerful therapeutic tools;some finetuning methods are illustrated in Fig. 3(c). ‘‘Glyco-engineering,”in this case,involves the manipulation of antibodies for reduced fucosylation, called afucosylation or defucosylation, which can give these‘‘nonfucosylated”antibodies a higher affinity for Fc receptors and can overcome the impediment of insufficient ADCC [14]. The nonfucosylated antibody mogamulizumab was produced using the POTELLIGENT®technology platform produced by the generation of a cell line with knockout α-1,6-fucosyltransferase (FUT8) gene[14,85].
The use of different formats of antibodies is another area for fine-tuning antibodies to overcome challenges for generating mAbs against complex or difficult membrane proteins.Various formats include IgG, antigen-binding fragment (Fab), single chain antibody fragments (scFvs) [57,71], domain antibodies [86],nanobodies [49], bispecific [87], and antibody conjugates [88](Fig. 3(c)).
5. Summary
This review discussed ways in which technological solutions facilitate the overcoming of obstacles to the development of therapeutic antibodies targeting complex membrane proteins.These solutions primarily exist in two areas. The preparation of complex membrane proteins to serve as antigens is one area of key technological solutions enabling the development of antibodies that effectively target such proteins. The second area of key technological solutions in this field involves antibody optimization and production technology platforms. Antibodies have many unique advantages over small-molecule and peptide drugs; complex membrane proteins play a broad range of roles in normal and disease states. Therefore, many research groups and companies are working toward developing therapeutic antibodies targeting complex membrane proteins.One limitation to this overview is that there are too many new developments in this field for everything to be covered here.The selection provided in this review is a representative snapshot of the state of this field based on our understanding and experience of therapeutic antibody development.One area for future improvement includes the development of guidelines for informing the best choices of antigen format and antibody-generation platforms for a given target. Another area for improvement is the development of guidelines for selecting target complex membrane proteins. Screening is another step in the development of therapeutic antibodies in which access to technology can play an important role. Binding assays and affinity measurement are usually the first steps, and the validation of the antibody in vitro and in vivo are the next steps. Functional assays depend on the target, the desired change for the target, and the mechanism by which the antibody is intended to make that change. For example, to achieve the inhibition of calcium-release activated channel Orai-1, which is targeted for reducing autoimmunity, the in vitro antibody functional assays used in screening included calcium flux assays,current measurement,an assessment of the antibody effect on T cell activation, and cytokine release assays [36,37]. Assays for anti-Orai-1 antibody inhibition function in vivo involved the characterization of graft-versus-host disease(GVHD) symptoms in a humanized GVHD mouse model [37].Insights from structure were key in the design of a strategy that enabled the discovery of the human apelin receptor agonist antibody JN241-9. Functional screening for antibody activation of this receptor involved the detection of intracellular cyclic adenosine monophosphate (cAMP) levels and an assay of β-arrestin recruitment [40]. Functional screening for agonist antibodies targeting β1-adrenergic receptor involved cell-based assays used to evaluate cAMP stimulation and an enzyme fragment complementation assay to measure signaling through β-arrestin. Screening for the function of a cannabinoid receptor type 1 (CB1) receptor agonist antibody also used a cAMP functional assay [89]. Agonist activity against bradykinin B2 receptor (BKB2R) is detected by functional screening assessing the ability to stimulate phosphorylation of glycogen synthase kinase 3 (GSK-3) [90]. Advances in available technology and expertise have already enabled significant progress toward the generation, isolation, screening, and engineering of therapeutic antibodies targeting complex membrane proteins.Application of the products of these and upcoming advances will enable far more targeting of complex membrane proteins in the near future.
Acknowledgments
This work was partly supported by the Cancer Prevention and Research Institute of Texas, USA (PR150551 and RP190561), and the Welch Foundation (AU-0042-20030616). The work was also supported by the National Natural Science Foundation of China(31700778 and 31320103918),and Jiangsu Province’s Key Laboratory of Medicine (XK201135).
Compliance with ethical guidelines
Georgina To’a Salazar, Ziyi Huang, Ningyan Zhang, Xue-Guang Zhang,and Zhiqiang An declare that they have no conflicts of interest or financial conflicts to disclose.
- Engineering的其它文章
- PVC Formulation of Anastrepha suspensa Pheromones Suitable for Field Studies
- Handheld Ultrasound Advances Diagnosis
- Nuclear Energy Seeks Revival with Advanced Fuel Options
- Mars Helicopter Exceeds Expectations
- Toward Systemic Thinking in Managing Environmental Risks
- The Experimental Advanced Superconducting Tokamak

