Genome Editing Strategies Towards Enhancement of Rice Disease Resistance
Rukmini Mishra, Wei Zheng, Raj Kumar Joshi, Zhao Kaijun
Review
Genome Editing Strategies Towards Enhancement of Rice Disease Resistance
Rukmini Mishra1, Wei Zheng2, Raj Kumar Joshi3, Zhao Kaijun2
()
The emerging pests and phytopathogens have reduced the crop yield and quality, which has threatened the global food security. Traditional breeding methods, molecular marker-based breeding approaches and use of genetically modified crops have played a crucial role in strengthening the food security worldwide. However, their usages in crop improvement have been highly limited due to multiple caveats.Genome editing tools like transcriptional activator-like effector nucleases and clustered regularly interspaced short palindromic repeats (CRISPR)-associated endonuclease Cas9 (CRISPR/Cas9) have effectively overcome limitations of the conventional breeding methods and are being widely accepted for improvement of crops. Among thegenome editing tools, the CRISPR/Cas9 system has emerged as the most powerful tool of genome editing because of its efficiency, amicability, flexibility, low cost and adaptability. Accumulatedevidencesindicatethatgenomeeditinghasgreatpotential in improving the disease resistance in crop plants. In this review, we offered a brief introduction to the mechanisms of differentgenome editing systems and then discussed recent developments in CRISPR/Cas9 system-based genomeediting towards enhancement of rice disease resistance by different strategies. This review also discussed the possible applications of recently developed genome editing approaches like CRISPR/Cas12a (formerly known as Cpf1) and base editors for enhancement of rice disease resistance.
genome editing technology;rice improvement; CRISPR/Cas9; CRISPR/Cas12a;base editor; disease resistance
Rice (L.) serves as the significant source of carbohydrate for more than twothirds of the world’s population. The burgeoning population coupled with climatic change and emergence of new phytopathogens have a drastic effect on global rice productivity. During the last few decades, the traditional breeding approaches including mutation breeding and molecular breeding have significantly contributed to the development of effective disease resistance strategies in rice as required for strengthening global food security. However, these methods are time-consuming and labor-intensive. Gene transfer techniques by way of-based transformation and other approaches have significantly improved the yield, quality and disease resistance in plants. However, the biosafety regulations, and social and ethical issues related to transgenic crops have always been a major hindrance to public acceptance of these genetically modified (GM) crops (Lusser et al, 2012). Afterwards, better strategies for high-yielding andstress-resistant rice varieties are the need of the hour for increasing rice productivity and ensuring global food security.
Plant diseases are the major constraints that have threaten the agricultural development and global food security. Crops are susceptible to a wide range of pathogens including bacteria, viruses and fungi, which cause significant economic loss (FAO, 2017). The pathogens affect the growth and development of crops,which results in huge yield loss thereby creating hurdlesfor sustainable agriculture. Multiple disease management strategies and risk assessment tools including the usage of disease forecasting protocols, combinatorial usage of chemicals, fungicides and biological control agents, crop rotations and host resistance breeding have been under practice for a very long time (Ul Haq and Ijaz, 2020). However, most of these strategies are inadequate to achieve successes in many pathosystems due to diversity in host range of the phytopathogens, environmental toxicity, discrepancy in the assessment of disease resistance reaction and inefficient disease forecasting systems. Therefore, understanding the molecular mechanism governing the interaction between host and the pathogen is central to the development of an effective disease resistance strategy.
Plants utilize multiple resistance responses towards preventing the colonization by pathogenic microorganisms. On one hand, plant resistance to phytopathogens is typically regulated by the resistance () genes encoding nucleotide-binding leucine-rich repeat (NB-LRRs) proteins, which neutralize the molecular activity of pathogen effector proteins in the plant cell (Cui et al, 2015). On the other hand, targeted mutation or knockout of susceptibility () gene(s) that acts as a host-pathogen compatibility factor also induces recessive immunity against the adapted phytopathogens (van Schie and Takken, 2014). As majority of plant diseases arise due to compatible interaction between host and the pathogen, altering the-gene(s) that promotes compatibility could be highly significant in the development of broad spectrum and durable molecular breeding strategies for disease resistance (van Schie and Takken, 2014).
Recently, the emergence of multiple new breeding techniques including speed breeding platforms, high throughput genotyping and precise genome editing coupled with genetic engineering have successfully generated multiple disease-free high-yielding crop varieties (Li et al, 2020). Among them, the genome editing approaches have emerged as the revolutionary tools for crop improvement (Voytas and Gao, 2014). The genome editing tools are represented by sequence specific nucleases (SSNs) that introduce a double stranded DNA break at a specific genomic region thereby inducing the host DNA repair pathways either by homologous recombination (HR) or through non-homologous end joining (NHEJ) (Sander and Joung, 2014). While NHEJ is an error prone method that creates random mutations leading to frame shift and target gene knockout, HR pathway is much more precise resulting in gene replacement or gene knock-in when donor DNA templates are available (Baltes et al, 2015). Nevertheless, these natural processes of DNA repair result in mutation leading to alteration of specific trait. Multiple genome editing tools including zinc finger nucleases (ZFNs), transcriptional activator-like effector nucleases (TALENs) and more recently developedclustered regularly interspaced short palindromic repeats (CRISPR)-associated endonuclease Cas9 (CRISPR/Cas9) have facilitated the introduction of agronomically important traits in many plant species (Zaidi et al, 2018). Among these platforms, CRISPR/Cas9 system has greater acceptance by the scientific community for its simplicity, high specificity for target cleavage, involvement of no complex protein chemistry and universal applicability (Zaidi et al, 2018). What’s more, CRISPR/Cas-mediated disease resistance has already been reported against multiple phytopathogens including bacteria (Peng et al, 2017), fungi (Wang et al, 2016) and viruses (Chandrasekaran et al, 2016; Zaidi et al, 2016) in different crops including rice (Sun et al, 2017; Li S Y et al, 2018; Tomlinson et al, 2019; He et al, 2020) either by target specific modification of the host genes or by precise alteration of the pathogen genomes. In thisreview, we summarized the different strategies associated with genome editing and their subsequent applications in the improvement of crops with focus on highlighting the advancements of the CRISPR/Cas9 system and its role in conferring disease resistance in rice.
Overview of genome editing technologies
Genome editing is a novel approach wherein SSNs are used to make precise modifications in the genomic DNA. Creation of a double stranded break (DSB) activates the cell’s DNA repair mechanism, either through NHEJ or HR method. NHEJ incorporates insertion and deletion (InDel) mutations whereas HR method results in gene insertion or replacement thatis much more precise. Mega nucleases such as the I-I endonuclease enzyme from yeast constituted the earliest known genome editing system (Paques and Duchateau, 2007). However, these are the least efficientamong the editing toolbox due to unclear communicationbetween mega nuclease protein residues and corresponding specific target DNA sequence (Hsu et al, 2014). ZFNs are the SSNs that bind DNA through an engineered array of zinc finger motifs (Carroll, 2011). A specific zinc-finger entails about 30 amino acids in a conserved ββα configuration. The non-specific cleavage domainIis dimeric in nature and as such a pair of ZFNs are designed to bring theI monomers close to the specific DNA target for creation of a DSB(Bogdanoveand Voytas,2011)(Fig.1-A).ZFNs have been successfully used as a genome editing tool in a wide range of crops and model plants including, tobacco and maize (Cai et al, 2009; Shukla et al,2009; Osakabe et al,2010). Yet, their usage as an editing tool is highly limited due to several constraints including off-target binding of the zinc-fingermotifs and multifaceted interactions between aminoacid residues and the target sequence (Carrol, 2011; Voytas, 2013).
Unlike ZFNs, TALENs are designed by combining aI nuclease domain with a transcription activator-like effector (TALE) DNA binding domain (Fig.1-A). TALEs are secretory proteins frombacteria that are characterized by the presence of a C-terminal acidic activation domain and nuclear localization signal sequence, central DNA binding domain (DBD) and N-terminal translocation signal sequence (Bogdanoveetal,2010). TALEs are primarily responsible for the transcriptional activation of the disease susceptibility genes in host plants.TALENshavebeen well establishedinmanyplant species,including rice, wheat, tobacco and barley (Wang et al,2014; Li T et al,2016; Blanvillain-Baufume et al,2017). Although TALENs are qualitatively more effective over ZFNs in terms of high target specificity and low off-target activity, the TALE DBDs are represented by extensive repeat structure which acts as a limitation for their comprehensive use in editing of multiple genomes. In contrast, the type II CRISPR/Cas9 system is the utmost innovative genome editing method that has superseded ZFNs and TALENs due to its efficiency and robustness. CRISPR/Cas9 makes use of a DNA endonucleaseCas9alongwithasmallRNAmoleculecalled single guideRNA (sgRNA)that regulatestheCas9 mediated site specific DSB at specific targets (Fig. 1-A). The attachment of the Cas9-sgRNA structure to the target DNA and the ensuing cleavage depends on the presence of a protospacer adjacent motif (PAM) sequence (5′-NGG-3′) at the 3′-end of the target site. Hence, the requisite of only different spacer sequences makes CRISPR/Cas9 a very simple and highly effective editing tool that has been greatly exploited in recent years for improvement of model plants and major crops (Ma et al,2015; Xu et al,2016; Zhang Y et al, 2020).
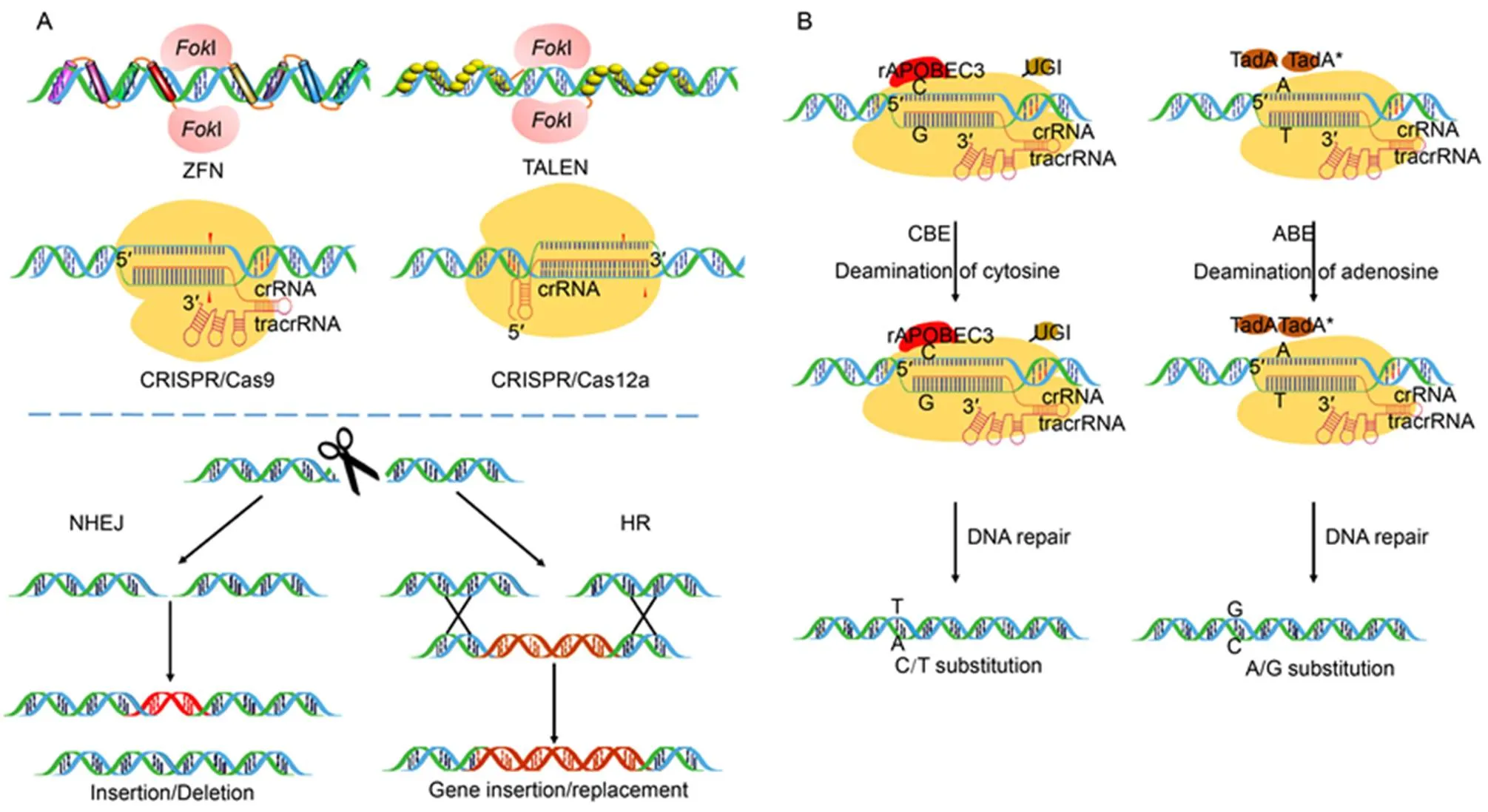
Fig. 1. Sequence specific nucleases used for genome editing.
A,Sequence specific nucleases including zinc finger nucleases(ZFNs), transcriptional activator-like effector nucleases (TALENs) and clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9 or CRISPR/Cas12a create double-stranded breaks (DSBs) at the target site which is subsequently repaired either by non-homologous end joining (NHEJ) or homologous recombination (HR) by cellular system leading to gene disruption by insertion/deletion, gene addition or replacement, respectively.The cleavage of the targeted DNA is facilitated byI endonuclease in ZFNs and TALENs and Cas endonucleases in CRISPR/Cas9 and CRISPR/Cas12a. CRISPR/Cas9 makes use of a 100 nt single guide (sgRNA) comprising of crisprRNA (crRNA) and trans-acting crispr RNA (tracrRNA) while CRISPR/Cas12a requires only a 40–45 nt long crRNA to facilitate gene editing.
B, Base editing platforms used for target specific single base modification. Cytidine base editor (CBE) uses Cas9 nickase together with a cytidine deaminase rAPOBEC3 and an uracil DNA glycosylase inhibitor (UGI) to facilitate cytosine to thymine conversions. Adenine base editor (ABE) consists of Cas9 nickase fused withderived ecTadA(WT)-ecTadA* heterodimer to facilitate adenosine to guanine conversions.
While trait improvement has been highly fruitful using CRISPR/Cas9, the prerequisite of an NGG PAM sequence has restricted its usage to potential target sites. Lately, several Cas9 variants and homologous proteins, such as Cas9-VRER, Cas9-VQR, Cas9-EQR, Cpf1-RVR, Cpf1-RR and SaCas9, have improved the feasibility of engineering a wide range of Cas9s with improved PAM specificities for genome editing in eukaryotic cells (Kleinstiver et al, 2015; Gao et al, 2017). Hu et al (2018) used a phage-assisted continuous evolution process to develop a SpCas9 variant, called xCas9, whichhas broader range of PAM compatibility including NG, GAA and GAT, and at the same time greater DNA specificity and lower genome-wide off-target activity compared to SpCas9. More recently, novel cytosine and adenine base editors with engineeredCas9 (SaCas9-NG), SpCas9-NG variants, have substantially expanded the targetable sites in the rice genome (Hua et al, 2019). Additionally, SpCas9-NG variants have greatly expanded the scope of genome editing in potato and tomato by targeting the non-canonical NGA and NGT PAMs (Veillet et al, 2020). Taken together, an efficient usage of these Cas9 variants would be critical to accelerate rice improvementthrough enhanced resistance to multiple phytopathogens.
The advent of another class II CRISPR-associated endonuclease, CRISPR/Cas12a or Cpf1, has broadened the horizon for genome editing with greater accuracy and competence (Endo et al, 2016) (Fig. 1-A). Comparedto CRISPR/Cas9 system, the CRISPR/Cas12a recognizes T-rich (5′-TTTN-3′ and 5′-TTN-3′) PAMs at the 5′-end of the target site, resulting in high cleavage efficiency (Zetsche et al, 2015). Unlike CRISPR/Cas9 which requires a 100 nt sgRNA, the CRISPR/Cas12a complex doesn’t require a tracrRNA and therefore can facilitate gene editing with only a crRNA of 40–45 nt consisting the repeat and the spacer. And the RuvC and nuclease domains of Cas12a cleave the target and the secondary strand of the DNA at 23and 17 bp downstream of the PAM sequence, respectively, resulting in staggered ends with 5 bp overhangs (Zetsche et al, 2015). By itself, the development of bigger mutations using CRISPR/Cas12a upsurges the efficiency of HR mediated donor gene insertion at the specific genomic location. Moreover, Cas12a simultaneously acts as an RNase to convert pre-crRNA to crRNA and a nuclease to cleave the dsDNA, demonstrating dual enzymatic activity (Zetsche et al, 2015). Therefore, CRISPR/Cas12a has the potential to generate multiple crRNA driven by a single promoter, making it simpler than CRISPR/Cas9 system. Moreover, the off-target cleavage by Cas12a is relatively lower than Cas9. These features make CRISPR/Cas12a more advanced over Cas9, making it a potentially important genome editing tool in the future (Zaidi et al, 2017). The Cas12a from(FnCas12a)and its ortholog from(LbCas12a) andsp. BV3L6 (AsCas12a) have been used to introduce targeted mutations in many crops (Endo et al, 2016). Emerging reports have already demonstrated the successful adaptation of CRISPR/Cas12a system in rice improvement (Yin et al, 2017; Li S Y et al, 2018). Yin et al (2017) utilized the CRISPR-LbCpf1 to target the early developmental gene(), a positive regulator of stomatal development in rice. The homozygous mutant plants show 8-fold reduction in the stomatal density on the abaxial leaf surface. Likewise, Li S Y et al (2018) reported that the donor repair template with only the left homologous arm is good enough for precise targeted allelic replacement in the wild type() gene resulting in herbicide resistant rice plants. Most recently, a comparative assessment of three Cas9 (Cas9 D10A nickase, HiFi Cas9 nuclease, and WT Cas9 nuclease) and two Cas12a nucleases (LbCas12a and AsCas12a) was carried out to determine their mutation efficiency on a single target site of the rice() gene (Banakar et al, 2020). The study showed that LbCas12a results in deletion between 2 to 20 bp without the loss of PAM site, leading to higher editing efficiency over the Cas9 variants,which suggests the potential of Cas12a to generate specific and heritable targeted mutations in rice and thus can be used as a precise genome editing tool for future rice improvement programmes including in the development of disease resistant rice varieties.
Although the HR-based gene replacement is a feasible approach with CRISPR/Cas9 and CRISPR/Cas12a, the efficiency of template DNA delivery and target insertion is significantly low. To overcome this problem, the base editor technology has emerged as a new and advanced approach for precise nucleotide substitutions in a programmable manner without the requirement of a DSB or donor template (Fig. 1-B) (Komor et al, 2016). The base editors comprise of a catalytically inactive CRISPR/Cas9 domain (dCas9 or Cas9 nickase) and a cytosine or adenosine deaminase domain which converts one base to another. These are capable of making single-base variations or substitutions without creating a DSB in the target DNA, thereby limiting the frequency of InDels. Recently, base editing system has been efficiently used in creating targeted point mutations at multiple endogenous loci in rice and wheat (Li C et al, 2018). Most recently, rBE5 ()base editor has been used to target, an agriculturally important rice gene that harbours a point mutation modulating the defense response to rice blast fungus (Ren et al, 2018). Cytosine and adenine base editors have also been efficiently used for precise base modification (C to T or A to G) in eukaryotic genomes (Zong et al, 2017; Hua et al, 2018; Qin et al, 2019). Base editing tool boxes have been efficiently optimized and demonstrated in several crops including rice, wheat, maize and tomato (Lu and Zhu, 2017; Zong et al, 2017;Li C et al, 2018; Tang et al, 2019). In course of time, a wide range of adenine base editors (ABEs) and cytidine base editors (CBEs) variants have been developed for efficient target specific base modification (Mishra et al, 2020). Furthermore, precise base editing in RNA has been realized using a catalytically-inactive Cas13 (dCas13) in association with adenosine deaminase acting on RNA (ADAR) to direct adenosine to inosine conversion (Cox et al, 2017). Cas13 is a type IV CRISPR-linked RNA guided RNase while ADARs mediate precise editing of the transcripts (Nishikura, 2010). Together, they have been used to develop an RNA editing system named as RNA Editing for Programmable A to I Replacement (REPAIR) that has significant applicability for research in therapeutics and biotechnology (Stafforst and Schneider, 2012).However, it is yet to be utilized in plant system.
Traditional genome editing including CRISPR/Cas9 system involves the delivery of DNA cassettes encoding editing machineries into the host genome. Often, the random integration of the editing cassette results in undesirable genetic changes and off-target effects (Zhang et al, 2018). Further, the introduction of editing cassette into the host genome invokes ethical and regulatory concerns (Jones, 2015). Therefore, scientistsare increasing getting aligned to the usage of DNA-freegenome editing technologies to minimize the probability of off-target mutations. DNA-free genome editing makes use of the CRISPR/Cas ribonucleoprotein (RNP) complexes that is delivered into the cell by protoplast transformation or particle bombardment leading to target DNA modification. CRISPR-RNP is an assembly of CRISPR specific guide RNA and Cas9 protein together forming an active enzyme complex (Zhang et al, 2018). The first DNA-free genome edited plants were obtained by transfecting the CRISPR/Cas RNPs into the protoplast of, tobacco, lettuce and rice (Woo et al, 2015). Likewise, particle bombardment mediated DNA free editing using the CRISPR-Cas9 RNPs has been successfully demonstrated in maize (Svitashev et al, 2016), wheat (Liang et al, 2017) and rice (Toda et al, 2019). In rice, Cas9-gRNA RNPs have been directly delivered into the zygotic tissues with a mutation efficiency of 14% to 64% (Toda et al, 2019). In maize, CRISPR/Cas9 RNPs have been used to generate both knockout as well as knockin mutants (Svitashev et al, 2016).
Moreover, the CRISPR/Cas-RNPs have relatively high editing efficiency and low off-target mutations compared to CRISPR/Cas system (Liang et al, 2017; Toda et al, 2019). In another study, the base editing has been combined with the DNA-free editing system to facilitate higher frequency of C to T conversion (1.8%) in wheat (Zong et al, 2018). Overall, this transgene free precise editing system has enormous potential for the improvement of rice as well as other important crop species. Although the usage of CRISPR/Cas RNPs is still at its infancy, it can be effectively explored to accelerate rice crop breeding and the edited products can get wider acceptance in public overcoming the biosafety regulatory hurdles.
Genome editing strategies for rice disease resistance
During the latter half of the 19th and early 20th centuries, host resistance breeding has played a pivotal role in the improvement of yield and other agronomic traits of rice, thereby addressing the challenges of feeding the world’s growing population. However, traditional methods of resistance breeding are expensive and time-consuming, and sometimes the resistance allele influences plant growth and development (Miah et al, 2013). Extensive genetic and genomic studies have revealed significant molecular details about rice innate immunity, including a large number of targets for control and inhibition of pathogenic infection. While plants have evolved resistance () genes that can neutralize pathogen derived virulence proteins or effectors and activate effector triggered immunity, they also have susceptibility () genes that are essentially involved in successful pathogenic infection. In context of genome editing, the primary strategy for disease resistance involves knocking out of thesegenes through the error prone NHEJ pathway-based repair of target DNA (Fig. 2-A). Alternatively, knocking ingene allele to the target site of interest via the HR mechanism has the potential for resistance development in widely accepted susceptible genotypes (Fig. 2-B and-C). Besides, alleles of certainandgenes vary at the single nucleotide level. Thus, precise editing by the way of targeted replacement or base editing can generate allelic variants for disease resistance (Fig. 2-B and-D). Then again, editing platforms can be used to induce disease resistance by altering the-regulatory regions of target genes and quantitative trait loci. More than hundred regulatory mutations have been realized in the tomatopromoter using a CRISPR/Cas9 system (Rodriguez-Leal et al, 2017). Such modifications can result in systematic assessment of-regulatory regions with resistant traits,which can enhance rice breeding (Fig. 2-D). Simultaneous editing of severalgenes or regulatory elements via a multiplex editing platform (Fig. 2-E) can essentially result in broad spectrum disease resistance. Multiple sgRNAs driven by independent promoters can be multiplexed into single Cas9 or Cpf1/sgRNA expression vector using the Golden Gate cloning or the Gibson assembly method (Silva and Patron, 2017). Wang et al (2017)demonstrated the feasibility of multiplex gene editing using CRISPR/Cpf1 system. The advent of multiple editing platforms has facilitated one or more of these strategies towards resistance development against biotic stress in crops particularly in rice (Mishra et al, 2018; Yin and Qiu, 2019). The application of CRISPR/Cas tools has mainly been explored in rice against viral infection, followed by efforts to improve fungal and bacterial disease resistance (Table1). Recent studies demonstrating the power of the genome editing technology in establishing resistance to these pathogen categories are discussed below.

Fig. 2. Genome editing strategies towards disease resistance in plants.
Multiple genome editing platforms can facilitate disease resistance through knock-out of susceptibility () genes (A), homology directed replacement of novel alleles(B), knock-in of resistance () genes (C), regulatory modification ofgene expression (D) and multiplex editing of resistance and susceptibility factors (E).
HR, Homologous recombination; ZFN, Zinc finger nuclease; NHEJ, Non-homologous end joining; ABE, Adenine base editor; CBE, Cytidine base editor; UGI, Uracil DNA glycosylase.

Table 1. List of genes targeted by genome editing tools for rice disease resistance.
TALENs, Transcription activator-like effector nucleases; CRISPR/Cas9, Cluster regularly interspersed short palindromic repeat/Cas9; BLB, Bacterial leaf blight.
Resistance to bacterial pathogen
Bacterial leaf blight (BLB), caused by γ-proteobacteriumpv(), is one of the most destructive vascular diseases of rice, especially in the major rice growing regions of Southeast Asia and sub-Saharan Africa. It is singly responsible for more than 75% of yield loss with millions of hectares of rice affected annually (Varshney et al, 2019). Identification of genetically resistant rice plants and their usage in genomic-assisted breeding have been the most effective method for developingresistant varieties. However, the emergence of new and noveltypes has been the greatest challenge in controlling the disease. Thepathogenicity in rice is primarily established through the injection of DNA-binding proteins called TALEs that bind to the effector-binding elements (EBEs) in the promoters of thegenes in rice (Cohn et al, 2014). TALEs usually target the sugar transporting SWEET gene family in rice which are primarily responsible for releasing the sugar into the apoplast as nutrition of thepathogens (Cohn et al, 2014). Multiple TALEs including AvrXa7, TalC, Tal5 and PthXo3 found in differentstrains all target the(also known as) gene in rice and were therefore considered as the major susceptibility factors associated with bacterial blight infection (Antony et al, 2010; Li et al, 2012; Hutin et al, 2015). TALEN-mediated editing results in the development of desired mutations within thepromoter region containing an EBE for AvrXa7, which is overlapped with another EBE for PthXo3. The edited rice plants with desired homozygous mutation for the 4 or 9 bp deletion at the target site are highly resistant to bacterial blight (Li et al, 2012). Zhou et al (2015) also reported the identification of another sucrose transporter geneas the disease susceptibility factor for PthXo2 TALE (transcription activator-like effector) dependentstrain,and also identified that the PthXo2-dependent strain inducesexpression specifically inrice IR24 due to the presence of a mysterious effector binding site, which is not present in the alleles ofrice Nipponbare and Kitaake. In another study, a naturally occurring 18 bp deletion inandwild rice species is predicted to prevent the binding of TALEs known to targetand confer resistance to BLB (Hutin et al, 2015).The allele, named as(t), is resistant against half of the testedstrains. More recently, Blanvillain-Baufume et al (2017) used the TALEN constructs to develop an allele library of thepromoter region in rice to assess the susceptibility level of edited rice lines carrying mutations in the EBEs of AvrXa7, Tal5 and TalC. And, the rice lines with disruption of the AvrXa7 and Tal5 EBEs results in resistance tostrains that are dependent on the corresponding TALEs.
CRISPR/Cas9-mediated disruption of the TALE EBEs of two susceptibility genes,and, in rice variety Kitaake was attempted to facilitate broad spectrum BLB resistance in rice (Xu et al, 2019). One of the rice mutantdemonstrates broad spectrum resistance to majority of thestrains. Further, two PthXo2-like TALEs are identified as major virulence factors in the compatiblestrains. As the PthXo2 TALEs primarily target the susceptibility gene, an analysis of EBE variants in thepromoter across 3000 rice varieties revealed the presence of at least 10-like haplotypes. CRISPR/Cas9 strategy was further used to introduce InDels in the EBE of thepromoter of theline, and a novel rice line with three edited OsSWEET EBEs resulted in broad spectrum resistance against all the testedstrains (Xu et al, 2019). This suggests that genome engineering of TALE-susceptibility factor co-evolved loci is crucial for preventing effector triggered susceptibility, thereby realizing broad spectrum BLB resistance.
Recently, Kim et al (2019) employed the CRISPR/Cas9 system to knockout rice(also known as) that acts a susceptibility factor for thestrains carrying the TAL effector PthXo1. Desired modifications inare stably transferred into T0, T1, T2and T3generations without the transferred DNA (T-DNA) through genetic segregation. Not only had the homozygous mutants displayed significantly enhanced resistance tostrain, the editing ofalso did not affect the agronomic traits and pollen viability in the mutant lines. Olivia et al (2019) sequenced 856 TALEs across multipleisolates from Asia and Africa distinctively targeting promoters of multiple OsSWEET genes (,and) and found the complexity involved in the development of TALE insensitive rice lines. To overcome this difficulty, a CRISPR/Cas9-mediated genome editing system is used to introduce mutations at the promoters of all the three SWEET genes at EBEs recognized by variousTALEs. Sequence analyses reveal multiple TALE variants forandalleles. Introduction of as many as five SWEET promoter mutations into the rice accessions Kitaake, IR64 and Ciherang-Sub1 resulted in vigorous and broad spectrum BLB resistance. Most recently, Zhang Q W et al (2020) facilitated significant improvement in rice response to BLB pathogen through usage of a T5 exonuclease (T5exo)-Cas fusion tool box. Compared with CRISPR/Cas9, T5exo-Cas9 or -Cas12a results in larger deletionmutations at the targeted site of UPTPthXo1(up-regulated by transcription activator-like effector PthXo1) box of thegene promoter in edited rice plants. Phenotypic analysis revealed that the rice plants edited by T5exo-Cas9 or -Cas12a have significant reduction in lesion length and enhancementin resistance to BLB as compared to those edited by CRISPR/Cas9 or CRISPR/Cas12a, which might be associated with the factor that T5exo-Cas9 induces larger deletions at the-regulatory element (Zhang Q Wet al, 2020). All these studies clearly indicate that the genome editing of rice especially with the CRISPR/Cas system has significantly contributed in conferring broad spectrum BLB resistance, and these strategies can be effectively used in non-transgenic genomic assisted crop improvement programmes.
Resistance to fungal pathogens
More than 30% of the plant diseases are caused by fungal phytopathogens (Giraud et al, 2010; Sharma et al, 2012). Rice blast caused by the hemibiotrophic filamentous fungusis the most widespread and devastating disease of rice. It is a recurrent difficulty for both upland and lowland rice which is highly vulnerable to this fungal phytopathogen from the seedling to the adult stage. The frequent occurrence of new races of blast pathogen has resulted in more than 30% of losses in global rice production,which is enough to feed 60 million people (Nalley et al, 2016). Utilization of blast resistant cultivars has being the most effective measure for the management of blast pathogen. As of now, more than 100 major blastgenes have been identified and 30 of them have been molecularly cloned (Xiao et al, 2019). Major blastgenes including/,,and///have been deployed in combination with genomic-assisted breeding and transgenic programmes for the development of resistant genotypes across the rice producing regions (Li Y et al, 2016). For instance, the recently identifiedricelocus consists of cluster of genes encoding nucleotide-binding site-leucine-rich repeat (NBS-LRR) receptors that confers resistance to blast funguswithout compromising the yield trait. The locus is characterized by a pair of epigenetically regulated antagonistic pair of NBS-LRR receptors,and. Whilefacilitates broad spectrum resistance,preventshomodimerization to suppress resistance and in turn increases seed production (Deng et al, 2017). Likewise, a single base modification in the regulatory region ofinduces blast resistance in rice (Li et al, 2017). However, the current challenge lies in the development of a collection of blast resistance genes that can be used against the incessantly evolving and varied strains of. Therefore, precise genome editing tools are the need of the hour for implementation of effective plant resistance in rice.
Enhanced resistance to blast disease has been demonstrated in rice by targeting the ethylene response transcription factor genevia CRISPR/Cas9-targeted knockout (Wang et al,2016). CRISPR/Cas9 induced InDel mutations are reported in the targeted gene with a mutation frequency up to 42% in the T0generation. The allelic mutations are stably transmitted to succeeding generations. Blast resistance screening in six homozygous mutated T2lines reveals significantly lower lesions as compared to wildtype plants at both the seedling and tillering stages. Further, no significant difference is observed in the agronomic performance of the mutants, suggesting that precise editing does not alter other important traits of the plants. Furthermore, the utilization of multiple CRISPR/Cas9 constructs (Cas9/multi-target-sgRNA) for targeting several sites withinlocus results in higher number of mutants. These suggest that CRISPR/Cas9 mediated targeting of multiple sites has the potential to increase the mutation efficiency, thereby enhancing blast resistance in rice. CRISPR/Cas9 SSN has been used to disrupt, a gene encoding exocyst subunit protein to explore its functional role in plant immunity (Ma et al, 2018).has been previously implicated in root hair elongation, pollen germination and defense response in other plant species (Bloch et al, 2016). Two sgRNAs are designed to target the third and tenth exons of thegene. CRISPR/Cas9 inducedmutant displays improved immune responses coupled with up-regulated expression of pathogenesis-related proteins, salicylic acid synthesis genes, increased levels of salicylic acid, and improved resistance to the rice blast pathogen. However, the mutant lines also show altered growth and agronomic traits including dwarf stature, smaller seedlings, shorter main roots and decreased or impaired plant height, panicle length, tiller number, 1000-grain weight and spikelet fertility as compared to the wild type. Multiple approaches to blast resistance in rice can be fructified through precise editing of multifunctional gene(s) associated with defense signaling in rice. Recently, CRISPR/Cas9 has also been adopted to induce mutations in the proline-rich motif offor rice resistance against(Li et al, 2019). CRISPR/Cas9 vector has been designed to facilitate simultaneous targeted mutation of the thermosensitive male sterile 5 (), rice blastgeneand BLBgenein the rice variety Pinzhan (Li et al, 2019). Three of the generated mutants with homozygous frameshift mutations at all the three genes () display characteristics of thermosensitive genic male sterility with enhanced resistance to infections ofandstrain PXO99 (Li et al, 2019), thereby significantly accelerating the process of hybrid rice breeding.
Rice sheath blight, caused byKuhn [Teleomorph stage,(Frank) Donk], is the second most important fungal disease of rice after blast that amounts of yield loss in the range of 8%–50% across the tropical rice growing countries of the world (Savary et al,2000). CRISPR/Cas9 editing has been recently attempted to modify() gene to understand its functional role in rice sheath blight resistance (Shah et al, 2019).is critically implicated in inducing disease susceptibility by acting as a universal adaptor between RNA polymerase II and DNA-bound transcription factors (Bäckström et al, 2007; Thatcher et al, 2009). CRISPR/Cas9 constructs have been mobilized into therice variety ASD16 via-mediated transformation that facilitated mutation in thelocus. However, the mutation efficiency and disease resistance affinity of the edited rice lines are yet to be ascertained.
Resistance to viral pathogens
Viral diseases also act as major global constraint in the effort to increase rice productivity. Among the different viral infections, rice tungro disease severely affects rice production in about 3.5 million hectares throughout the major rice producing Asian nations (Chancellor et al, 2006). The disease is basically caused by the interaction of two different viruses namelyrice tungro spherical virus (RTSV) with a single stranded RNA genome, and rice tungro bacilliform virus (RTBV), with a double stranded DNA genome (Hull, 1996). RTBV develops the primary disease symptoms, and spreads the disease by assisting the transmission of RTBV through the green leaf hopper species such asand(Hibino and Kabauatan, 1987). Host resistance breedingprogrammes through the screening of huge rice germplasm collections have resulted in the identification of multiple rice varieties with specific resistance to either or both of the viruses (Khush et al, 2004). Concentrated molecular research in anrice variety Utri Merah demonstrated that the RTSV resistance is controlled by the presence of single-nucleotide polymorphism or deletion affecting the YVV amino acid residues in the translation initiation factor four gamma () gene (Lee et al, 2010). As such, it is essential to develop RTSV resistant variety to prevent the secondary spread of this disease. Recently, CRISPR/Cas9 genome editing has been successfully used to mutategene in the RTSV susceptible rice variety, IR64, to develop new sources of resistance to RTSV (Macovei et al, 2018). Mutation frequency ranging from 36% to 86% is realized, and induced mutations are transmitted into the subsequent generation with no detectable modifications in the closest off-target sites. Sequence analysis and pathogen infection assay revealed that in-frame mutation in the amino acid residues adjacent to the YVV residues confers enhanced RTSV resistance together with improved agronomic parameters such as plant height and grain yield. The stable mutants can be released in the rice tungro disease prone areas as alternative source of RTSV resistance for controlling the infection and improving the rice productivity.
Future prospects for enhancing rice disease resistance
Although genome editing approaches depend upon the targeted mutation ofgenes towards introduction of disease resistance in plants, it might come with a fitness cost. Knocking outgenes,which encode proteins responsible for pre-penetration structures, defense suppression and replication machinery, may lead to phenotypic abnormalities and nutritional deficiency that can affect plant growth and development (van Schie and Takken, 2014). Whereas OsSWEETrice mutants induce resistance through restricted sugar availability for BLB pathogen, it might also lead to reduced plant stature and pollen abortion (Chu et al, 2006). This could be possibly mitigated through targeted editing of different promoter regions of thegenes has been demonstrated for SWEET genes in rice (Blanvillain-Baufume et al, 2017). Alternatively, instead of fully knocking out angene, disease resistance can also be developed by introducing synthetic variants ofgene identical to the allele that naturally occurs in resistant genotypes (Bastet et al, 2017). Such new allele can induce plant resistance and at the same time demonstrate normal protein functions with no developmental cost. Besides, the recently developed base editing tools can also be used for precise introduction of single base transitions in somegenes,which vary only at the single nucleotide polymorphism level. Moreover, the availability of multiple pathogen inducible promoters and regulatory elements in plants can be targeted using a pathogen induced CRISPR system for transient switch off thegene with no compromise in their fitness roles. Such a CRISPR vector system that can exploit the pathogen inducible promoter could be conceptualized to demonstrate their effectiveness towards disease resistance in field crops. On the contrary, the introduction of a custom design sequence into the genome will be more appropriate when specific allelic variants are involved in resistance response. A CRISPR system coupled with the HR mechanism can indefinitely expand the possibility of knocking ingene allele into the target site of choice for developing disease resistance. Although HR is still technically challenging in plants due to low efficiency and lack of multiplexing protocols, CRISPR toolboxes need to be designed and analyzed to expand their applications in resistance breeding for rice improvement.
Acknowledgements
This research was supported by the China Priority Program-Breeding of Seven Major Crops (Grant No. 2017YFD01100100), the Innovation Program of Chinese Academy of Agricultural Sciences (Grant No. 01-ICS) and the Talented Young Scientist Program of China (Grant No. India-17-01). We thank Dr. Muktikanta Mishra, President, Centurion University of Technology and Management for his encouragement and support.
Antony G, Zhou J H, Huang S, Li T, Liu B, White F, Yang B. 2010. Ricerecessive resistance to bacterial blight is defeated by induction of the disease susceptibility gene., 22(11): 3864–3876.
Bäckström S, Elfving N, Nilsson R, Wingsle G, Björklund S. 2007. Purification of a plant mediator fromidentifies PFT1 as the Med25 subunit., 26(5): 717–729.
Baltes NJ, Hummel AW, Konecna E, Cegan R, Bruns AN, Bisaro DM, Voytas DF. 2015. Conferring resistance to Gemini viruses with the CRISPR-Cas prokaryotic immune system., 1: 15145.
Banakar R, Schubert M, Collingwood M, Vakulskas C, Eggenberger AL, Wang K. 2020. Comparison of CRISPR-Cas9/Cas12a ribonucleoprotein complexes for genome editing efficiency in the rice() gene., 13(1):4.
Bastet A, Robaglia C, Gallois JL. 2017. eIF4E resistance: Natural variation should guide gene editing., 22(5): 411–419.
Blanvillain-Baufume S, Reschke M, Sole M, Auguy F, Doucoure H, Szurek B, Meynard D, Portefaix M, Cunnac S, Guiderdoni E, Boch J, Koebnik R. 2017. Targeted promoter editing for rice resistance topv.reveals differential activities for SWEET14-inducing TAL effectors.,15(3): 306–317.
Bloch D, Pleskot R, Pejchar P, Potocký M, Trpkošová P, Cwiklik L, Vukašinović N, Sternberg H, Yalovsky S, Žárský V. 2016. Exocyst SEC3 and phosphoinositides define sites of exocytosis in pollen tube initiation and growth., 172(2): 980–1002.
Bogdanove AJ, Schornack S, Lahaye T. 2010. TAL effectors: Finding plant genes for disease and defense., 13(4):394–401.
Bogdanove AJ, Voytas D F. 2011. TAL Effectors: Customizable proteins for DNA targeting., 333:1843–1846.
Cai CQ, Doyon Y, Ainley WM, Miller JC, Dekelver RC, Moehle EA, Rock JM, Lee YL, Garrison R, Schulenberg L, Blue R, Worden A, Baker L, Faraji F, Zhang L, Holmes MC, Rebar EJ, Collingwood TN, Rubin-Wilson B, Gregory PD, Urnov FD, Petolino JF. 2009. Targeted transgene integration in plant cells using designed zinc finger nucleases., 69:699–709.
Cai L L, Cao Y Y, Xu Z Y, Ma W X, Zakria M, Zou L F, Cheng Z Q, Chen G Y. 2017. A transcription activator-like effector Tal7 ofpv.activates rice geneto suppress rice immunity., 7(1): 5089.
Carroll D. 2011. Genome engineering with zinc finger nucleases., 188(4): 773–782.
Chancellor TCB, Holt J, Villareal S, Tiongco ER, Venn J. 2006. Spread of plant virus disease to new plantings: A case study of rice tungro disease., 66:1–29.
Chandrasekaran J, Brumin M, Wolf D, Leibman D, Klap C, Pearlsman M, Sherman A, Arazi T, Gal-On A. 2016. Development of broad virus resistance in non-transgenic cucumber using CRISPR/Cas9 technology., 17(7):1140–1153.
Chu ZH, Yuan M, Yao LL, Ge XJ, Yuan B, Xu C G, Li X H, Fu B Y, Li Z K, Bennetzen J L, Zhang Q F, Wang S P. 2006. Promoter mutations of an essential gene for pollen development result in disease resistance in rice.,20(10):1250–1255.
Cohn M, Bart RS, Shybut M, Dahlbeck D, Gomez M, Morbitzer R, Hou BH, Frommer W B, Lahaye T, Staskawicz B J. 2014.virulence is promoted by a transcription activator-like effector-mediated induction of a SWEET sugar transporter in cassava.,27(11):1186–1198.
Cox DBT, Gootenberg JS, Abudayyeh OO, Franklin B, Kellner MJ, Joung J, Zhang F. 2017. RNA editing with CRISPR-Cas13., 358:1019–1027.
Cui H T, Tsuda K, Parker JE. 2015.Effector-triggered immunity: From pathogen perception to robust defense.,66: 487–511.
Deng Y W, Zhai K R, Xie Z, Yang D Y, Zhu X D, Liu J Z, Wang X, Qin P, Yang Y Z, Zhang G M, Li Q, Zhang J F, Wu S Q, Milazzo J, Mao B Z, Wang E, Xie H, Tharreau D, He Z H. 2017. Epigenetic regulation of antagonistic receptors confers rice blast resistance with yield balance.,355: 962–965.
Endo A, Masafumi M, Kaya H, Toki S. 2016. Efficient targeted mutagenesis of rice and tobacco genomes using Cpf1 from., 6:38169.
FAO. 2017. The Future of Food and Agriculture: Trends and Challenges. Rome: Food and Agriculture Organization of the United Nations.
Gao L Y, Cox DBT, Yan WX, Manteiga JC, Schneider MW, Yamano T, Nishimasu H, Nureki O, Crosetto N, Zhang F. 2017. Engineered Cpf1 variants with altered PAM specificities., 35: 789–792.
Giraud T, Gladieux P, Gavrilets S. 2010. Linking the emergence of fungal plant diseases with ecological speciation., 25(7): 387–395.
He L, Li X F, Xu Y, Liu H L, He M L, Tian X J, Wang Z Y, Wu X J, Bu Q Y, Yang J. 2020. High-efficiency reduction of rice amylose content via CRISPR/Cas9-mediated base editing., 27(16): 445–448.
Hibino H, Cabauatan PQ. 1987. Infectivity neutralization of rice tungro associated viruses acquired by vector leafhoppers., 77: 473–476.
Hsu PD, Lander ES, Zhang F. 2014. Development and applications of CRISPR-Cas9 for genome engineering, 157(6): 1262–1278.
Hu JH, Miller SM, Geurts MH, Tang W X, Chen L W, Sun N, Zeina C M, Gao X, Rees H A, Liu D R. 2018. Evolved Cas9 variants with broad PAM compatibility and high DNA specificity., 556:57–63.
Hua K, Tao X P, Yuan F T, Wang D, Zhu JK. 2018. Precise AT to GC base editing in therice genome., 11(4): 627–630.
Hua K, Tao X P, Zhu JK. 2019. Expanding the base editing scope in rice by using Cas9 variants., 17(2):499–504.
Hull R. 1996. Molecular biology of rice tungro viruses., 34: 275–297.
Hutin M, Sabot F, Ghesquière A, Koebnik R, Szurek B. 2015. A knowledge-based molecular screen uncovers a broad-spectrumresistance allele to bacterial blight from wild rice., 84(4): 694–703.
Jones HD. 2015. Regulatory uncertainty over genome editing., 1:14011.
Khush GS, Angeles E, Virk PS, Brar DS. 2004. Breeding rice for resistance to tungro virus at IRRI., 36(2): 101–106.
Kim Y A, Moon H, Park C J. 2019. CRISPR/Cas9-targeted mutagenesis ofin rice to confer resistance topv.., 12: 67.
Kleinstiver BP, Prew MS, Tsai SQ, Topkar V V, Nguyen NT, Zheng Z L, Gonzales A P W, Li Z Y, Peterson R T, Yeh J R J, Aryee M J, Joung J K. 2015. Engineered CRISPR-Cas9 nucleases with altered PAM specificities., 523: 481–485.
Komor AC, Kim YB, Packer MS, Zuris JA, Liu DR. 2016. Programmable editing of a target base in genomic DNA without double stranded DNA cleavage.,533: 420–424.
Lee JH, Muhsin M, Atienza GA, Kwak DY, Kim SM, De Leon TB, Angeles E R, Coloquio E, Kondoh H, Satoh K, Cabunagan R C, Cabauatan P Q, Kikuchi S, Leung H, Choi I R. 2010. Single nucleotide polymorphisms in a gene for translation initiation factor (eIF4G) of rice () associated with resistance to rice tungro spherical virus., 23(1): 29–38.
Li C, Zong Y, Wang YP, Jin S, Zhang DB, Song QN, Zhang R, Gao C X. 2018. Expanded base editing in rice and wheat using a Cas9-adenosine deaminase fusion., 19: 59.
Li S F, Shen L, Hu P, Liu Q, Zhu X D, Qian Q, Wang K J, Wang Y X. 2019. Developing disease-resistant thermosensitive male sterilerice by multiplex gene editing.,61(12):1201–1205.
Li S Y, Zhang X, Wang W S, Guo X P, Wu Z C, Du W M, Zhao Y D, Xia L Q. 2018. Expanding the scope of CRISPR/Cpf1 mediated genome editing in rice., 11(7): 995–998.
Li T, Liu B, Spalding MH, Weeks DP, Yang B. 2012. High-efficiency TALEN-based gene editing produces disease-resistant rice., 30: 390–392.
Li T, Liu B, Chen CY, Yang B. 2016. TALEN-mediated homologous recombination produces site-directed DNA base change and herbicide-resistant rice.,43(5): 297–305.
Li W, Deng Y W, Ning Y S, He Z H, Wang GL. 2020. Exploiting broad-spectrum disease resistance in crops: From molecular dissection to breeding., 29: 575–603.
Li W T, Zhu Z W, Chern M, Yin J J, Yang C, Ran L, Cheng M P, He M, Wang K, Wang J, Zhou X G, Zhu X B, Chen Z X, Wang J C, Zhao W, Ma B T, Qin P, Chen L H, Wang Y P, Liu J L, Wang W M, Wu X J, Li P, Wang J R, Zhu L H, Li S G, Chen X W. 2017. A natural allele of a transcription factor in rice confers broad-spectrum blast resistance., 170(1):114–126.
Li Y, Xiao J H, Chen L L, Huang X H, Cheng Z K, Han B, Zhang Q F, Wu C Y. 2016. Rice functional genomics research: Past decade and future., 11(3):359–380.
Liang Z, Chen K L, Li T D, Zhang Y, Wang Y P, Zhao Q, Liu J X, Zhang H W, Liu C M, Ran Y D, Gao C X. 2017. Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9ribonucleoprotein complexes., 8:14261.
Lu Y M, Zhu JK. 2017. Precise editing of a target base in the rice genome using a modified CRISPR/Cas9 system., 10(3):523–525.
Lusser M, Parisi C, Plan D, Rodriguez-Cerezo E. 2012. Deployment of new biotechnologies in plant breeding., 30: 231–239.
Ma J, Chen J, Wang M, Ren Y L, Wang S, Lei C L, Cheng Z J, Sodmergen. 2018. Disruption ofincreases the content of salicylic acid and induces plant defense responses in rice., 69(5):1051–1064.
Ma XL, Zhang Q Y, Zhu Q L, Liu W, Chen Y, Qiu R, Wang B, Yang Z F, Li H Y, Lin Y R, Xie Y Y, Shen R X, Chen S F, Wang Z, Chen Y L, Guo J X, Chen L T, Zhao X C, Dong Z C, Liu Y G. 2015. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants., 8(8):1274–1284.
Macovei A, Sevilla NR, Cantos C, Jonson GB, Slamet-Loedin I, Cermak T, Voytas D F, Choi I R, Chadha-Mohanty P. 2018. Novel alleles of rice eIF4G generated by CRISPR/Cas9-targeted mutagenesis confer resistance to rice tungro spherical virus.,16(11): 1918–1927.
Miah G, Rafii MY, Ismail MR, Puteh AB, Rahim HA, Asfaliza R, Latif MA. 2013. Blast resistance in rice: A review of conventionalbreeding to molecular approaches., 40:2369–2388.
Mishra R, Joshi RK, Zhao K J. 2018. Genome editing in rice: Recent advances, challenges, and future implications., 9:1361.
Mishra R, Joshi RK, Zhao K J. 2020. Base editing in crops: Current advances, limitations and future implications.,18(1):20–31.
Nalley L, Tsiboe F, Durand-Morat A, Shew A, Thoma G. 2016. Economic and environmental impact of rice blast pathogen () alleviation in the United States., 11(12): e0167295.
Nishikura K. 2010. Functions and regulation of RNA editing by ADAR deaminases., 79: 321–349.
Olivia R, Ji C H, Atienza-Grande G, Huguet-Tapia JC, Perez-Quintero A, Li T, Eom J S, Li C H, Nguyen H, Liu B, Auguy F, Sciallano C, Luu V T, Dossa G S, Cunnac S, Schmidt S M, Samet-Loedin I H, Cruz C V, Szurek B, Frommer W B, White F F, Yang B. 2019. Broad-spectrum resistance to bacterial blight in rice using genome editing.,37(11):1344–1350.
Osakabe K, Osakabe Y, Toki S. 2010. Site-directed mutagenesis inusing custom-designed zinc finger nucleases., 107(26):12034–1239.
Paques F, Duchateau P. 2007. Meganucleases and DNA double-strand break-induced recombination: Perspectives for gene therapy., 7(1): 49–66.
Peng A H, Chen S C, Lei T G, Xu L Z, He Y R, Wu L, Yao L X, Zou X P. 2017. Engineering canker-resistant plants through CRISPR/Cas9-targeted editing of the susceptibility genepromoter in citrus., 15(12):1509–1519.
Qin L, Li J Y, Wang Q Q, Xu Z P, Sun L, Alariqi M, Manghwar H, Wang G Y, Li B, Ding X, Rui H P, Huang H M, Lu T L, Lindsey K, Daniell H, Zhang X L, Jin S X. 2019. High efficient and precise base editing of C∙G to T∙A in the allotetrapoid cotton () genome using a modified CRISPR/Cas9 system., 1: 12.
Ren B, Yan F, Kuang Y J, Li N, Zhang D W. Zhou X P, Lin H H, Zhou H B. 2018. Improved base editor for efficiently inducing genetic variations in rice with CRISPR/Cas9-guided hyperactive hAID mutant., 11(4): 623–626.
Rodriguez-Leal D, Lemmon ZH, Man J, Bartlett ME, Lippman ZB. 2017. Engineering quantitative trait variation for crop improvement by genome editing., 171(2):470–480.
Sander JD, Joung JK. 2014. CRISPR-Cas systems for editing, regulating and targeting genomes., 32(4): 347–355.
Savary S, Willocquet L, Elazegui FA, Castilla NP, Teng PS. 2000. Rice pest constraints in tropical Asia: Quantification of yield losses due to rice pests in a range of production situations., 84(3):357–369.
Shah PR, Varanavasiappan S, Kokiladevi E, Ramanathan A, Kumar KK. 2019. Genome editing of ricegene to study its role in rice sheath blight disease resistance., 8(6): 2356–2364.
Sharma TR, Rai AK, Gupta SK, Vijayan J, Devanna BN, Ray S. 2012. Rice blast management through host plant resistance: Retrospect and prospects., 1(1): 37–52.
Shukla VK, Doyon Y, Miller JC, Dekelver RC, Moehle EA, Worden SE, Mitchell J C, Arnold N L, Gopalan S, Meng X D, Choi V M, Rock J M, Wu Y Y, Katibah G E, Gao Z F, McCaskill D, Simpson M A, Blakeslee B, Greenwalt S A, Butler H J, Hinkley S J, Zhang L, Rebar E J, Gregory P D, Urnov F D. 2009. Precise genome modification in the crop speciesusing zinc finger nucleases., 459:437–441.
Silva NV, Patron NJ. 2017. CRISPR-based tools for plant genome engineering.,1(2): 135–149.
Stafforst T, Schneider MF. 2012. An RNA-deaminase conjugate selectively repairs point mutations., 51: 11166–11169.
Sun Y W, Jiao G A, Liu Z P, Zhang X, Li J Y, Guo X P, Du W M, Du J L, Francis F, Zhao Y D, Xia L Q. 2017. Generation of high-amylose rice through CRISPR/Cas9-mediated targeted mutagenesis of starch branching enzymes., 8:298.
Svitashev S, Schwartz C, Lenderts B, Young JK, Cigan MA. 2016. Genome editing in maize directed by CRISPR-Cas9 ribonucleoproteins complexes., 7:13274.
Tang X, Ren Q R, Yang L J, Bao Y, Zhong Z H, He Y,Liu S S, Qi C Y, Liu B L, Wang Y, Sretenovic S, Zhang Y X, Zheng X L, Zhang T, Qi Y P, Zhang Y. 2019. Single transcript unit CRISPR 2.0 systems for robust Cas9 and Cas12a mediated plant genome editing., 17(7):1431–1445.
Thatcher LF, Manners JM, Kazan K. 2009.hijacks COI1-mediated jasmonate signaling to promote disease development in.,58(6):927–939.
Toda E, Koiso N, Takebayashi A, Ichikawa M, Kiba T, Osakabe K, Osakabe Y, Sakakibara H, Kato N, Okamoto T. 2019. An efficient DNA- and selectable-marker-free genome-editing system using zygotes in rice.,5:363–368.
Tomlinson L, Yang Y, Emenecker R, Smoker M, Taylor J, Perkins S, Smith J, MacLean D, Olszewski N E, Jones J D G. 2019. Using CRISPR/Cas9 genome editing in tomato to create a gibberellin-responsive dominant dwarf DELLA allele., 17: 132–140.
Ul Haq I, Ijaz S. 2020. Plant Disease Management Strategies for Sustainable Agriculture Through Traditional and Modern Approached.Springer.
van Schie CC N, Takken FL W. 2014. Susceptibility genes 101: How to be a good host., 52: 551–581.
Varshney RK, Godwin ID, Mohapatra T, Jones JDG, McCouch SR. 2019. A SWEET solution to rice blight., 37: 1280–1282.
Veillet F, Perrot L, Guyon-Debast A, Kermarrec MP, Chauvin L, Chauvin JE, Gallois J L, Mazier M, Nogue F. 2020. Expanding the CRISPR toolbox inusing SpCAS-NG variant and application for gene and base editing incrops., 21: 1024.
Voytas DF. 2013. Plant genome engineering with sequence-specific nucleases., 64:327–350.
Voytas DF, Gao C X. 2014. Precision genome engineering and agriculture: Opportunities and regulatory challenges.,12(6): e1001877.
Wang F J, Wang C L, Liu P Q, Lei P L, Hao W, Gao Y, Liu Y G, Zhao K J. 2016. Enhanced rice blast resistance by CRISPR/Cas9-targeted mutagenesis of the ERF transcription factor gene., 11(4): e0154027.
Wang M G, Mao Y F, Lu Y M, Tao X P, Zhu JK. 2017. Multiplex gene editing in rice using the CRISPR-Cpf1 system.,10(7):1011–1013.
Wang Y P, Cheng X, Shan Q W, Zhang Y, Liu J X, Gao C X, Qiu J L. 2014. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew., 32:947–951.
Woo J W, Kim J, Kwon S I, Corvalán C, Cho S W, Kim H, Kim S G, Kim S T, Choe S, Kim J S. 2015. DNA-free genome editing in plants with preassembled CRISPR-Cas9 ribonucleoproteins.,33(11): 1162–1164.
Xiao W M, Yang Q Y, Huang M, Guo T, Liu Y Z, Wang J F, Yang G L, Zhou J Y, Yang J Y, Zhu X Y, Chen Z Q, Wang H. 2019. Improvement of rice blast resistance by developing monogenic lines, two-gene pyramids and three-gene pyramid through MAS., 12(1):78.
Xu R F, Yang Y C, Qin R Y, Li H, Qiu C H, Li L, Wei P C. 2016. Rapid improvement of grain weight via highly efficient CRISPR/Cas9-mediated multiplex genome editing in rice, 43(8): 529–532.
Xu Z Y, Xu X M, Gong Q, Li Z Y, Li Y, Wang S, Yang Y Y, Ma W X, Liu L Y, Zhu B, Zou L F, Chen G Y. 2019. Engineering broad-spectrum bacterial blight resistance by simultaneously disrupting variable TALE-binding elements of multiple susceptibility genes in rice., 12(11): 1434–1446.
Yin K Q, Qiu JL. 2019. Genome editing for plant disease resistance:Applications and perspectives., 374: 20180322.
Yin X J, Biswal AK, Dionora J, Perdigon KM, Balahadia CP, Mazumdar S, Chater C, Lin H C, Coe R A, Kretzschmar T, Gray J E, Quick P W, Bandyopadhyay A. 2017. CRISPR-Cas9 and CRISPR-Cpf1 mediated targeting of a stomatal developmental genein rice.,36(5):745–757.
Zaidi SS E A, Tashkandi M, Mansoor S, Mahfouz MM. 2016. Engineering plant immunity: Using CRISPR/Cas9 to generate virus resistance., 7:1673.
Zaidi SS E A, Mahfouz MM, Mansoor S. 2017. CRISPR-Cpf1: A new tool for plant genome editing., 22(7):550–553.
Zaidi SS E A, Mukhtar MS, Mansoor S. 2018. Genome editing: Targeting susceptibility genes for plant disease resistance., 36(9): 898–906.
Zetsche B, Gootenberg JS, Abudayyeh OO, Slaymaker IM, Makarova KS, Essletzbichler P, Volz S E, Joung J, van der Oost J, Regev A, Koonin E V, Zhang F. 2015. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system.,163(3):759–771.
Zhang Q W, Yin K Q, Liu G W, Li S N, Li M G, Qiu JL. 2020. Fusing T5 exonuclease with Cas9 and Cas12a increases the frequency and size of deletion at target sites., 63: 1–10.
Zhang Y, Massel K, Godwin ID, Gao C X. 2018. Applications and potential of genome editing in crop improvement., 19(1):210.
Zhang Y, Pribil M, Palmgren M, Gao C X. 2020. A CRISPR way for accelerating improvement of food crops., 1: 200–205.
Zhou J H, Peng Z, Long J Y, Sosso D, Liu B, Eom JS, Huang S, Liu S Z, Cruz C V, Frommer W B, White F F, Yang B. 2015. Gene targeting by the TAL effector PthXo2 reveals cryptic resistance gene for bacterial blight of rice., 82(4):632–643.
Zong Y, Wang Y P, Li C, Zhang R, Chen K L, Ran Y D, Qiu J L, Wang D W, Gao C X. 2017. Precise base editing in rice, wheat and maize with a Cas9-cytidine deaminase fusion., 35: 438–440.
Zong Y, Song Q N, Li C, Jin S, Zhang D B, Wang Y P, Qiu J L, Gao C X. 2018. Efficient C to T base editing in plants using a fusion of nCas9 and human APOBEC3A.,36: 950–953.
10 April 2020;
4 August 2020
Zhao Kaijun(zhaokaijun@caas.cn)
Copyright © 2021, China National Rice Research Institute. Hosting by Elsevier B V
This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/)
Peer review under responsibility of China National Rice Research Institute
http://dx.doi.org/10.1016/j.rsci.2021.01.003
(Managing Editor: Wang Caihong)
- Rice Science的其它文章
- Genetic Interaction of Hd1 with Ghd7, DTH8 and Hd2 Largely Determines Eco-Geographical Adaption of Rice Varieties in Southern China
- Drought Tolerance in Rice: Focus on Recent Mechanisms and Approaches
- RAVL1 Activates IDD3 to Negatively Regulate Rice Resistance to Sheath Blight Disease
- Osa-miR439 Negatively Regulates Rice Immunity Against Magnaporthe oryzae
- Exogenous Peroxidase Mitigates Cadmium Toxicity, Enhances Rhizobial Population and Lowers Root Knot Formation in Rice Seedlings
- Effects of Early- and Late-Sowing on Starch Accumulation and Associated Enzyme Activities During Grain Filling Stage in Rice

