Phyllanthus acidus (L.) Skeels and Rhinacanthus nasutus (L.) Kurz leaf extracts suppress melanogenesis in normal human epidermal melanocytes and reconstitutive skin culture
Moragot Chatatikun, Takeshi Yamauchi, Kenshi Yamasaki, Anchalee Chiabchalard,Setsuya Aiba
1Department of Medical Technology, School of Allied Health Sciences, Walailak University, Thailand
2Department of Dermatology, Tohoku University Graduate School of Medicine, Sendai, Miyagi, Japan
3Deparment of Clinical Chemistry, Faculty of Allied Health Sciences, Chulalongkorn University, Thailand
Keywords:Phyllanthus acidus (L.) Skeels Rhinacanthus nasutus (L.) Kurz Melanin Tyrosinase MITF
ABSTRACT Objective: To determine the effect of extracts from Phyllanthus acidus (P. acidus) (L.) Skeels and Rhinacanthus nasutus (R. nasutus) (L.) Kurz leaves on melanogenesis and the underlying mechanism in normal human epidermal melanocytes (NHEM) and a reconstitutive skin model.Methods: NHEM and a reconstitutive skin model were stimulated with ethanol extracts of P. acidus (L.) Skeels and R. nasutus (L.) Kurz leaves. mRNA expression of microphthalmiaassociated transcription factor (MITF), tyrosinase (TYR), tyrosinase-related protein 1 (TYRP1)and dopachrome tautomerase (DCT) were examined by real-time PCR. The melanin content in NHEM was also measured. Moreover, protein levels of tyrosinase were determined using western blot analysis.Results: In NHEM and the reconstitutive skin model, ethanol extracts from P. acidus (at 12.5 and 25.0 µg/mL) and R. nasutus (at 6.25 and 12.50 µg/mL) significantly diminished mRNA expression of MITF, TYR, TYRP1 and DCT in a concentration-dependent manner. P. acidus and R. nasutus extracts also reduced the amount of melanin in α-MSH-stimulated NHEM.Moreover, P. acidus and R. nasutus extracts markedly suppressed tyrosinase at the translational level in the reconstitutive skin model.Conclusions: P. acidus and R. nasutus extracts significantly reduced melanogenesis in NHEM and the reconstitutive skin model, suggesting that P. acidus and R. nasutus extracts can inhibit melanin synthesis through downregulation of MITF, TYR, TYRP1 and DCT. Therefore, the ethanol extracts of P. acidus and R. nasutus contain compounds that have the potential for development as a skin lightening agent for the treatment of hyperpigmentation disorder or melasma.
1. Introduction
Melasma are gray-brown symmetrical patches in the sun-exposed areas of the facial skin[1]. In Southeast Asian countries with Fitzpatrick skin types Ⅲ-Ⅴ, control of melasma is particularly challenging, and melasma is frequently related to a physiological impact[2]. The effect of ultraviolet radiation (UVR) on melanin synthesis is well established, and repeated exposure to suberythemal doses of UVR induces melanogenesis by increasing the level of melanin[3]. α-melanocyte stimulating hormone (α-MSH) is released from keratinocytes through ultraviolet exposure. Binding to melanocortin 1 receptor (MC1R) on melanocytes, α-MSH stimulates the activities of adenylate cyclase via G-proteins, and increases the intracellular levels of cyclic adenosine monophosphate (cAMP)[4,5].Protein kinase A is activated by the increased level of cAMP and adds a phosphate group to the cAMP-response element binding protein which up-regulates the expression of the microthalmia associated transcription factor (MITF)[6]. Then, MITF activates transcription of pigmentation enzyme genes such as tyrosinase(TYR), tyrosinase-related protein 1 (TYRP1) and dopachrome tautomerase (DCT)[7]. Decrease in expression of melanogenic genes TYR, TYRP1 and DCT , leads to reduction in melanin synthesis[8].
An increasing number of studies have sought to develop natural compounds from plants as potential therapeutic agents to treat melasma[9]. The current mainstay treatment of melasma is topical hydroquinone application and hydroquinone affects the formation,melanization and degradation of melanosomes[10]. Given the side effects of hydroquinone therapy, researchers have been searching for alternative treatments, and increasingly interest has focused on the development of natural products as an alternative treatment for melasma.
Phyllanthus acidus (P. acidus) (L.) Skeels belongs to the Phyllanthaceae family, commonly known as a gooseberry tree and bears edible yellow fruits. P. acidus is a rich resource of secondary metabolites and various terpenes, tanins, anthraquinones, flavonoids,alkaloids and saponins[11]. A 50% ethanol extract of P. acidus leaves(as used in this study) is known to contain derivatives of quercetin,kaempferol, epicatechin, coumaric, and cinnamic acids[12]. Recently,new cleistanthane diterpenoids and phyllaciduloids A-D which showed cytotoxic effects against human cancer cell lines were identified from the roots and stems of P. acidus[13]. P. acidus leaf extracts can inhibit α-glucosidase, and show hepatoprotective effects, hypoglycemic and hypolipidemic effects, inhibition of α-amylase, anti-microbial activity,inhibition of lipid peroxidation, analgestic activity, anti-in flammatory activity, and neuprotective effects[12,14-19]. Moreover, aqueous leaf extracts of P. acidus can generate stable silver nanoparticles with activity against bacterial pathogens because of their flavonoids and quercitin[20].
Rhinacanthus nasutus (R. nasutus) (L.) Kurz contains alkaloids,anthraquinones, flavonoids (quercetin, rutin), saponins, triterpenoids,napthoquinones (Rhinacanthin-A, -B, -C, -D, -E, -F, -G, -N, -M,-O, -Q), carotenoids, and polyphenols[21-25]. In a recent study, the new naphthoquinones racemate, rhinacasutone, rhinacanthone,rhinacanthins-C, -D, -E, -N, and -Q), and heliobuphthalmin were identified from the root of R. nasutus[26]. Moreover, lignans are the main chemical compounds of the aerial parts (including leaves and branches) of R. nasutus. R. nasutus leaves have skin treatment, antiviral, neurominidase, anti-microbial, anti-allergic, and anti-diabetic activities, as well as hypolipidemic effects[25-31]. In addition, a methanol extract of R. nasutus leaves can restore the kidney damage occurring as a consequence of diet induced obesity in rats[32].
To date, no study has investigated effects of P. acidus (L.) Skeels and R. nasutus (L.) Kurz extracts on melanogenesis. To address this,we investigated the effect of ethanol extracts of P. acidus and R.nasutus leaves on the mRNA expression of MITF, TYR, TYRP1 and DCT in α-MSH-stimulated moderately pigmented normal human epidermal melanocyte (NHEM) and in a reconstitutive skin model.We also examined the expression of tyrosinase enzyme, which is a target for a whitening agent.
2. Materials and methods
2.1. Chemicals and reagents
α-MSH, RPMI 1640 medium, DMSO, CaCl2and kojic acid, were purchased from Sigma (St. Louis, MO). Dichloromethane, absolute ethanol, absolute methanol and petroleum ether were purchased from Merck (Darmstadt, Germany). Collagen type 1a matrix was purchased from Nitta gelatin (Osaka, Japan). ISOGEN reagent was obtained from Nippon Gene (Tokyo, Japan). Penicillin/streptomycin,phosphate buffer, trypsin/EDTA, MEM medium, Medium254,DMEM medium, RIPA buffer, an anti-tyrosinase antibody (T311), an anti-GAPDH antibody (PA1-987) and HRP-linked antibodies were obtained from Thermo Fisher Scientific (Waltham, MA). MTT was purchased from Roche (Basel, Swiss). Humedia-KG2 was purchased from Kurabo (Osaka, Japan). Fetal calf serum (FCS) was obtained from Hyclone (Chicago, IL). Mayer’s hematoxylin solution was obtained from Muto pure chemicals (Tokyo, Japan).
2.2. Preparation of P. acidus (L.) Skeels and R. nasutus (L.)Kurz
Leaves of P. acidus (L.) Skeels and R. nasutus (L.) Kurz were obtained from HRH Princess Maha Chakri Sirindhorn Herb Garden,Rayong city, Thailand. Identification was confirmed and voucher specimens were placed at the Department of Botany, Faculty of Science, Chulalongkorn University, Thailand. The voucher specimen numbers of P. acidus (L.) Skeels and R. nasutus (L.) Kurz are A015128 (BCU) and A 015129 (BCU), respectively. The ethanol extracts were prepared by the Soxhlet method as described in our previous study[33]. All ethanol extracts were stored at -20 ℃ until use.
2.3. Cells and tissue culture
NHEM from moderately pigmented skin and normal human epidermal keratinocytes (NHEK) were obtained from Thermo Fisher Scientific. NHEM were cultured in Medium 254 containing human melanocyte growth supplement at 37 ℃ in a humidified chamber with 5% CO2[34].
A reconstitutive cultured human skin model (the reconstitutive skin model), which has a similar morphology and physiology to human skin, was reconstituted with NHEM, NHEK, and human dermal fibroblasts as previously described[35,36]. Brie fly, a collagen type 1a matrix containing fibroblasts (3.5×105cells/well) was placed on a transwell clear polycarbonate membrane with a 0.4 µm pore size and the membrane was immersed into DMEM medium,supplemented with 2 mM L-glutamine, 100 UI/mL penicillin, 100 µg/mL streptomycin and 10% FCS in the well of a 6-well plate[37]. The plate was cultured at 37 ℃ in a 5% CO2incubator for 3 d. Then, the plate was incubated with fresh culture medium and incubated for 3 d.On day 7, mixed epidermal cells (4.2×105cells) were added onto the plate at a melanocyte to keratinocytes ratio of 1: 2.5 (1.2×105melanocytes and 3×105keratinocytes) and cells were cultured in keratinocyte culture medium (HuMedia-KG2) for 2 d in a 5% CO2incubator. Subsequently the reconstitutive reconstructed skin model was treated with 0.5 mM Ca2+(day 9), 1.0 mM Ca2+(day 11) and 1.5 mM Ca2+(day 13 and 15) in keratinocyte culture medium. On day 17, DMEM medium/high glucose supplemented with 10% FCS was used to replace the medium and cells were incubated for 3 d in a 5% CO2incubator. On day 20, the upper layer of plate (the reconstructed skin compartment) was exposed to air and lower layer was incubated with DMEM medium/high glucose and 10% FCS for 3 d. The culture medium in the lower layer was changed every day. On day 27, the reconstitutive skin model was used for other experiments.
2.4. Cell viability assay for NHEM
Cell viability was established through the MTT assay essentially as described by others[38]. Brie fly, NHEM were seeded at a concentration of 3×103cells/well and incubated for 24 h. After 24 h, the cells were incubated with 1 µM of α-MSH medium containing DMSO as a control or with each ethanol extract at doses of 3.125-100.000 µg/mL for 48 h. Then, the cells were incubated with 10 µL of yellow tetrazole for 4 h at 37 ℃. Solubilizing agent (100 µL/well) was added into each well and incubated overnight at 37 ℃ after which the absorbance of formazan was measured at 550 nm using a microplate reader (Molecular Devices, USA). Values are expressed as a percentage of control group (no treatment).
2.5. Real-time quantitative RT-PCR for MITF, TYR,TYRP1, DCT mRNA
NHEM were seeded at a density of 3×105cells/well and incubated for 24 h in a 5% CO2incubator. After 24 h, the cells were incubated with 1 µM of α-MSH with each ethanol extract for 48 h[38].Extraction of total RNA was performed with ISOGEN reagent.
The reconstitutive skin model was treated with each ethanol extract for 7 d. The medium containing ethanol extract was changed every 2 d. After treatment, the medium in each well was removed after which the reconstructed skin model was crushed by a Cryo-press(Microtec, Japan) and samples were kept in a (-80 ℃) freezer until RNA isolation. Total RNA was extracted as above.
Total RNA was quantified by Nanodrop at 260 and 280 nm[39]. For cDNA synthesis, one 10 µL reaction comprised of 1 µL of 2.5 µM oligo dT primer, 8 µL of 1 µg extracted RNA and 1 µL of 10 mM dNTP. The reactions were incubated for 5 min at 65 ℃. Then, PCR nucleotide mixture, 5× PrimeScript buffer, RNAse inhibitor (40 U/µL), and PrimeScript RTase were added in a new PCR tube[40]. This reaction was incubated for 10 min at 30 ℃, for 60 min at 42 ℃, and for 15 min at 72 ℃ to synthesize cDNA[41].
Real time PCR was undertaken using a M×3000P qPCR system (Aligent Technologies, United States). The reaction was undertaken for GAPDH, MITF, TYR, TYRP1 and DCT with a starting denaturation at 95 ℃ for 3 min, followed by 40 cycles of at 95 ℃ for 5 s and at 60 ℃ for 20 s. The primers used for qPCR were as follows: for GAPDH 5’-CTTAGCACCCCTGGCCAAG-3’(forward) and 5’-TGGTCATGAGTCCTTCCACG-3’ (reverse);for TYR 5’-GACGTCAGCACCCCACAAAT-3’ (forward) and 5’-GCAGCTTTATCCATGGAACCA-3’ (reverse); TYRP1 5’-TGCACACCTTCACAGATGCA-3’ (forward) and 5’-AAGCGCCAACTACTGCTATGG-3’ (reverse); DCT 5’-TCCTTCCTTGAACGGGACAAA-3’ (forward) and 5’-TGGCATAGCTGTAGCCAAGTTG-3’, (reverse); MITF 5’-CGGGAACAGGACCATGGTTA-3’ (forward) and 5’-AGCTAGCCCCTGAAATGAATCC-3’ (reverse). From the results,the relative changes were compared to GAPDH mRNA. All data were shown as fold change against control group, determined using the 2–ΔΔctmethod.
2.6. Measurement of melanin
Melanin levels were determined as described elsewhere[38]. In short, NHEM (3×105cells/well) were seeded onto a 6-well culture plate and left for 24 h. Then, the cells were treated with each ethanol extract and 1 µM of α-MSH in DMEM medium for 48 h. After treatment with each ethanol extract, the cells were trypsinized,then transferred into a tube and washed twice with PBS[42]. The cell pellets were dissolved in 1 N NaOH at 95 ℃ for 30 min[43].Absorbance of melanin was measured at 475 nm. The amount of melanin was calculated from synthetic melanin, and the results were shown as a percentage of control group.
2.7. Western blots for the reconstitutive skin model
The reconstitutive skin model was treated with extract for 7 d, after which the culture medium was discarded and cells were crushed using a Cryo-press (Microtec, Japan) and the resultant samples were transferred into a new tube. Total protein was extracted by adding 1 mL of RIPA buffer and protein concentrations in sample were determined by a BCA protein assay kit (Thermo Fisher Scientific). Western blot analysis was performed as previously described[38]. Samples with equal protein content (20 µg/sample)were run on a 10% SDS-PAGE and proteins were transferred onto a PVDF membrane. The PVDF membranes were blocked with 5% BSA. After blocking, the membranes were incubated with a mouse monoclonal anti-tyrosinase antibody (1: 50) (Thermo Fisher Scientific, Waltham, MA) or a rabbit polyclonal anti-GAPDH antibody (1: 1 000) (Thermo Fisher Scientific). Signal was developed using an HRP-conjugated anti-rabbit IgG antibody, or an HRP-conjugated anti-mouse IgG antibody (Cell Signaling Technologies,Danvers, MA) and a LumiGLO® Peroxidase Substrate kit (Seracare,Milford, MA). Signal was recorded with an ImageQuant™ LAS 4000 (GE Healthcare Life Sciences, Marlborough, MA). GAPDH serves to normalize the results.
2.8. Statistical analysis
Results are shown as the mean±SEM of three independent experiments. Statistical analysis was undertaken by ANOVA with Dunnett’s post-hoc test using SPSS version 20.0. P<0.05 indicated statistical significance.
3. Results
3.1. Effect of P. acidus and R. nasutus extracts on viability of NHEM
To determine the potential toxicity of P. acidus and R. nasutus extracts, NHEM were treated with different concentrations (3.125 to 100.000 µg/mL) of each extract and 1 µM of α-MSH for 48 h and cell viability were determined by the MTT assay. Low concentrations of P. acidus extract (3.125-25.000 µg/mL) did not affect NHEM cell viability though higher concentrations (50.000-100.000 µg/mL)significantly suppressed viability (Figure 1A). The R. nasutus extract between concentrations of 3.125- 12.500 µg/mL also did not affect NHEM cell viability (Figure 1B). Therefore, we used the two highest non-cytotoxic concentrations; 12.50 and 25.00 µg/mL of P. acidus and 6.250 and 12.50 µg/mL of R. nasutus, in further experiments.

Figure 1. Effects of P. acidus (PA) and R. nasuthus (RN) extracts on cell viability in NHEM.
3.2. P. acidus and R. nasutus extracts attenuate MITF, TYR,TYRP1 and DCT mRNA induced by α-MSH in NHEM
We examined whether P. acidus and R. nasutus extracts affected the transcription of melanogenic enzymes such as MITF, TYR, TYRP1 and DCT. α-MSH increased mRNA expression of these melanogenic enzymes, and P. acidus and R. nasutus extracts significantly suppressed the α-MSH-dependent increase in MITF, TYR and DCT expression in a concentration-dependent manner (Figure 2A, 2B and 2D). P. acidus extract also significantly reduced TYRP1, while R. nasutus extract at the concentration of 12.5 µg/mL but not 6.25 µg/mL decreased TYRP1 (Figure 2C). These results suggest that the P. acidus and R.nasutus extracts modify melanogenesis enhanced by α-MSH through suppression of MITF, TYR, TYRP1 and DCT expression.

Figure 2. Effects of PA and RN extracts on mRNA levels of MITF, TYR, TYRP1 and DCT in NHEM.
3.3. P. acidus and R. nasutus extracts reduce melanin contents in NHEM stimulated by α-MSH
To determine the effects of P. acidus and R. nasutus extracts on melanin synthesis, we stimulated NHEM with 1 µM of α-MSH for 48 h together with each ethanol extract. As shown in Figure 3, α-MSH markly enhanced the melanin content in NHEM by about 18% when compared with control cells (without α-MSH treatment). The P.acidus extract at a dose of 25 µg/mL, and the R. nasutus extract at a dose of 12.5 µg/mL significantly diminished the amount of melanin as compared to the α-MSH-treated cells. These results indicate that P. acidus and R. nasutus extracts exert an anti-melanogenic effect on NHEM.
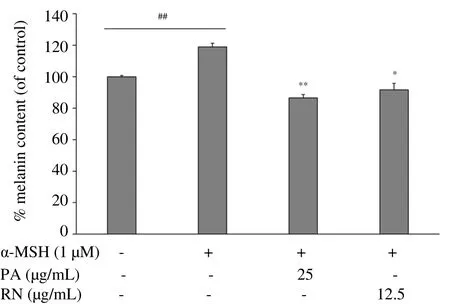
Figure 3. Effects of PA and RN extracts on melanin synthesis in NHEM.
3.4. P. acidus and R. nasutus extracts decrease melanogenic gene expressions in the reconstitutive skin model
To gain further understanding of the effects of P. acidus and R.nasutus extracts on melanogenic mRNA expression in human skin,we employed a reconstitutive skin model. The reconstitutive skin model has an epidermal layer consisting of NHEK and NHEM on top of a dermal layer containing dermal human dermal fibroblasts in a collagen Ⅰ matrix, mimicking normal human skin[35,36]. The reconstitutive skin model was treated with each extract for 7 d, and mRNA levels of melanogenic enzymes were examined by real-time PCR analysis. As shown in Figure 4A-4D, P. acidus (at 25.0 µg/mL)and R. nasutus (at 12.5 µg/mL) significantly suppressed mRNA levels of MITF, TYR, TYRP1 and DCT when compared with untreated control. It is noteworthy that the suppression of melanogenetic gene by P. acidus and R. nasutus extracts was more than kojic acid (at 125 µg/mL). We also confirmed that P. acidus and R. nasutus extracts significantly decreased tyrosinase protein expression compared to the control model (Figure 5). Thus, in agreement with the antimelanogenic effect of P. acidus and R. nasutus in monoculture of NHEM, we observed P. acidus and R. nasutus extracts decreased the transcriptional and translational levels of melanogenic enzymes in the reconstitutive skin culture model.

Figure 5. Effects of PA and RN extracts on the expression of tyrosinase protein in the reconstitutive skin model.
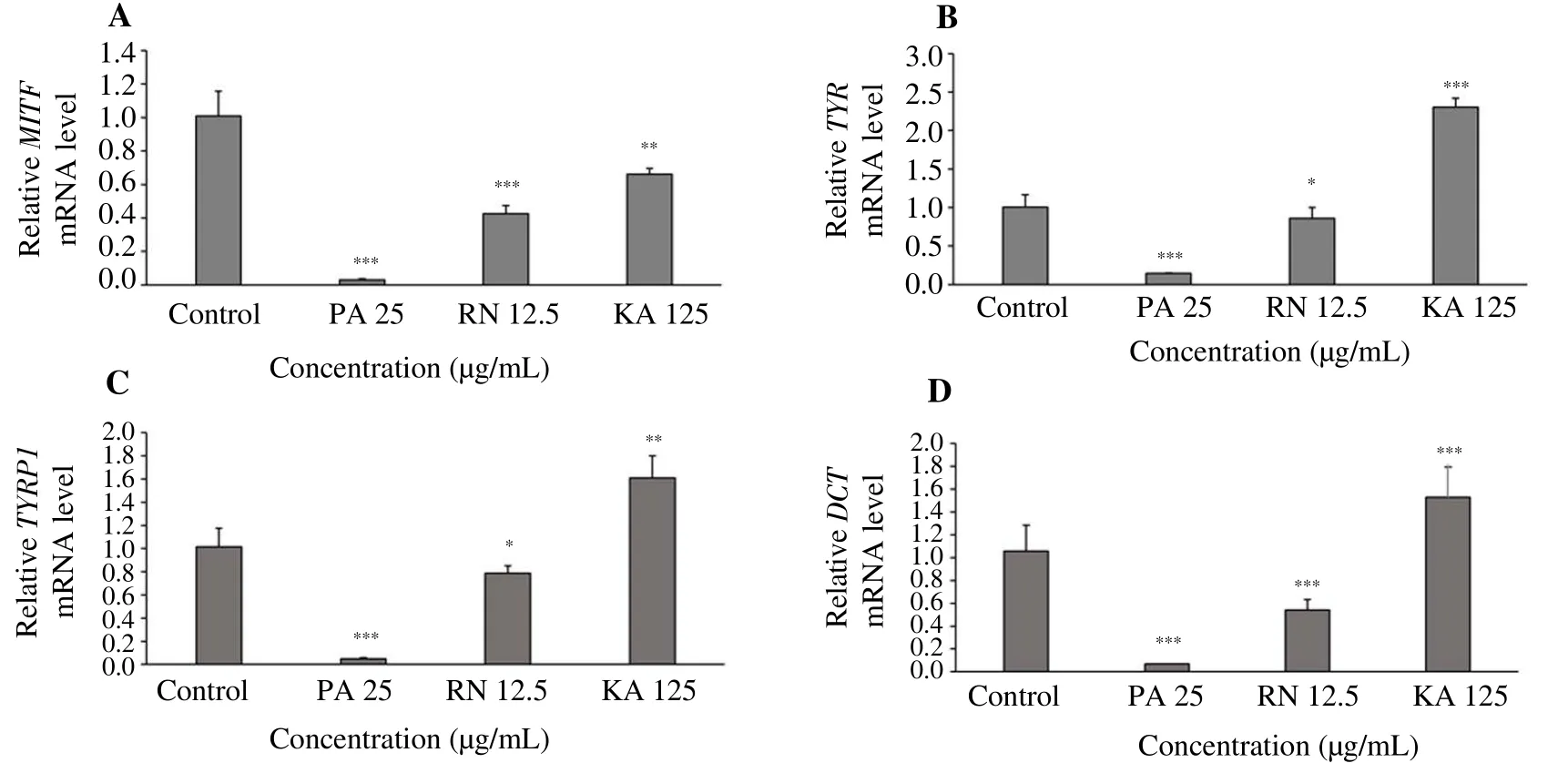
Figure 4. Effects of PA and RN extracts on mRNA levels of melanogenic enzymes in the reconstitutive skin model.
4. Discussion
Natural drugs from plants are a popular developmental route because of many advantages, such as fewer side effects, being less expensive and being based on a long history of traditional use[44].In this study, we sought to determine if herbal extracts have the potential to regulate melanogenic gene expression. We observed the inhibitory effects of P. acidus and R. nasutus extracts on the melanogenic enzymes mRNA expression in medium pigmented NHEM stimulated by α-MSH. We also used a reconstitutive skin model which is similar to human skin structure containing epidermis and dermis layers. This model allowed us to examine the ability of P. acidus and R. nasutus extracts to permeate through the epidermal layer and to have idea if P. acidus and R. nasutus extracts exert inhibitory effects on melanogenesis in human skin[45].We observed that P. acidus and R. nasutus extracts significantly decreased tyrosinase protein expression when compared with the control model, and P. acidus and R. nasutus extracts suppressed melanogenetic genes more than kojic acid (at 125 µg/mL). P. acidus and R. nasutus extracts inhibited melanogenesis through suppression of melanogenetic enzymes in α-MSH-stimulated NHEM and in the reconstitutive skin model.
DNA damage in keratinocytes is results from UVR exposure,and this results in the up-regulation of the p53 protein[46]. The p53 protein promotes the activation of proopiomelanocortin, which is further cleaved to generate α-MSH[47]. Keratinocytes secrete α-MSH which binds to MC1R on melanocytes[48,49]. MC1R stimulation by α-MSH induces an increased cAMP within the melanocytes which upregulate the transcription of MITF through the CRE binding protein[50]. Binding of MITF to the M box sequences in the gene promoters activates the transcription of TYR, TYRP1 and DCT genes. Our previous study found that P. acidus and R.nasutus extracts inhibit melanogenesis through inhibiting mushroom tyrosinase activity by about 43% and 65%, respectively[33]. In this study, P. acidus and R. nasutus extracts significantly reduced MITF,TYR, TYRP1 and DCT at the transcriptional level in α-MSH-induced NHEM. P. acidus (at 25 µg/mL) and R. nasutus (at 12.5 µg/mL)extracts significantly suppressed melanin content without affecting NHEM cell viability. Given that UVR increases α-MSH production from keratinocytes, our data indicates that P. acidus and R. nasutus extracts inhibit melanin synthesis through downregulation of MITF,TYR, TYRP1 and DCT in α-MSH-stimulated NHEM and in the reconstitutive skin model.
Our previous study[33] showed that P. acidus and R. nasutus extracts have phenolic compounds corresponding to approximately 50 and 17 mg gallic acid equivalent/g dry weight. The flavonoid content in the P. acidus extract was around 11 mg quercetin equivalents/g dry weight, while that of the R. nasutus extract was around 9 mg quercetin equivalents/g dry weight. The reactive oxygen species(ROS) scavenging activities of the two extracts were examined by an ABTS assay, and it was shown that P. acidus and R. nasutus extracts had ROS scavenging activities of around 99% and 56%,respectively. These antioxidant properties are important for regulating melanogenesis and skin treatments[51]. Human skin cells are normally exposed to ROS and oxidative stress as a result of exposure to UVR[52]. So, P. acidus and R. nasutus extracts may have phytochemical compounds that scavenge ROS generated as a consequence of UVR exposure. Our results suggest that P. acidus and R. nasutus extracts have phytochemical compounds which decrease melanin synthesis through the suppression of melanogenetic genes in α-MSH-stimulated NHEM and in the reconstitutive skin model.Thus, P. acidus and R. nasutus extracts seem to have the potential for development as ingredient for anti-melasma agents or skin whitening agents. In this study, we further suggest the exact compounds of P.acidus and R. nasutus that have the anti-melanogenic effects.
In conclusion, the present study showed that ethanol extracts of P. acidus and R. nasutus suppressed cellular melanin content in α-MSH-stimulated NHEM by decreasing mRNA expression of MITF, TYR, TYRP1, DCT. P. acidus and R. nasutus extracts also suppressed melanogenic enzymes, especially tyrosinase expression at the transcriptional and translational levels in the reconstitutive skin model. Therefore, this study suggests that ethanol extracts of P.acidus and R. nasutus inhibit melanogenesis through the suppression of MITF, TYR, TYRP1 and DCT. These extracts have the potential to be ingredients of skin whitening agents against hyperpigmentation by UVR. In future studies, we aim to isolate the bioactive compounds of P. acidus and R. nasutus extracts using high performance liquid chromatography. We will also develop these extracts as a nanoemulsion cream for treating hyperpigmentation. The limitation of this study is the use of these extracts in model systems, and the P.acidus and R. nasutus extracts should be investigated for toxicity and anti-melanogenic effects on human skin.
Conflicts of interest statement
The authors declare that there was no conflict of interest.
Acknowledgements
This work was financially supported by by research grant from Kao Melanin Workshop (KY), by Grant-in-Aid for Challenging Exploratory Research 16K15542 (KY) and a Grant-in-aid for Scientific Research C 24591622 (KY) from the Ministry of Education, Culture, Sports, Science and Technology, Japan, by Novartis Pharma Research Grants (KY), and by grants from the Department of Dermatology, Tohoku University Graduate School of Medicine, Japan. We thank Chanat Kumtornrut for helpful discussions about the results. Finally, we greatly appreciate Professor Dr. Duncan R. Smith (Institute of Molecular Biosciences, Mahidol University) for his reviewing of this manuscript and was grateful manuscript writing camp from School of Allied Health Sciences,Walailak University.
 Asian Pacific Journal of Tropical Medicine2019年3期
Asian Pacific Journal of Tropical Medicine2019年3期
- Asian Pacific Journal of Tropical Medicine的其它文章
- SARS and its treatment strategies
- Antihydatic and immunomodulatory effects of Algerian propolis ethanolic extract: In vitro and in vivo study
- Chemical composition of Mentha suaveolens and Pinus halepensis essential oils and their antibacterial and antioxidant activities
- Isolation and structural elucidation of antifungal compounds from Curcuma amada
- Status of intestinal parasitic infections among rural and urban populations, southwestern Iran
- X-linked Toll-like receptor 7 polymorphism associated with susceptibility to Chikungunya Fever
