Lipophilic extractives of the inner and outer barks from six different Pinus species grown in Indonesia
Masendra•Tatsuya Ashitani•Koetsu Takahashi•Ganis Lukmandaru
Abstract The chemical compositions of the dichloromethane extracts of inner and outer barks from six Pinus species(P.elliotii,P.oocarpa,P.caribeae,P.merkusii,P.montezumae,and P.insularis)grown in Indonesia were investigated by GC and GC–MS.Generally,the amounts of extractive contents were higher in the inner bark than in the outer bark except for P.merksuii.Fatty acids,monoterpenes,sesquiterpenes, resin acids,triterpenoids,and steroids were detected and quantified.Inner and outer barks differed not only in content of these compounds but also in their composition.Fatty acids and alcohols were the major classes of lipophilic compounds in the outer bark of P.caribeae,P.insularis,and P.montezumae.Steroids and triterpenoids were the dominant compounds identified in the inner bark of P.elliotii,P.insularis,and P.merkusii.Resin acids were the most abundant group in the inner bark of P.oocarpa whereas monoterpenes and sesquiterpenes were recorded in minor quantities in both bark layers of all species.
Keywords Bark extractive·Lipophilic·Inner bark·Outer bark·Pinus spp.
Introduction
In Indonesian forests,especially in Java,Pinus merkusiihas been planted in large areas(476,126 ha)by Perhutani,a state-owned enterprise for building material production.Coverage ofP.merkusiiis expanding due to the choice of this species for many afforestation projects.Pines are grown not only for timber,but also for rosin and turpentine.P.merkusiiis the only pine present in Indonesia and is native to South East Asia(Wijayanto et al.2015).Pine species from other countries have been planted by Indonesian Government through the Centre for Forest Biotechnology and Tree Improvement Research to supply the demand for long fiber sources for paper production.
Pine bark is an important biomass resource,amounting 10–15%of total tree weight.Bark is mostly used as boiler fuel,but a huge surplus is still discarded as waste residue(Ku et al.2007).The utilization of pine bark has been reported before;for antioxidant activities(Ku et al.2007),anti-cancer and radical scavenging activities(Yesil-Celiktas et al.2009).The famous commercially successfully product that incorporates bark is pycnogenol,the patented trade name ofP.maritimebark extract(Silliman et al.2003).
Pine wood is commonly used for producing pulps and boards,but all bark is removed prior to the chipping process due to the high lignin/polyphenol content,which causes difficulties in processing.Normally,in the pulping process,bark enters in minor amounts with wood chips.Pine wood and bark contain lipophilic extractives of fatty acids,terpenoids,and steroids(Wijayanto et al.2015;Yamamoto et al.2011;Willför et al.2003,2009).Lipophilic extracts in minor amounts can affect and be responsible for the formation and deposition of pitch on pulp(Kilulya et al.2012;Bikovens et al.2013;Zule et al.2015).Thus,knowledge of the chemical composition of the lipophilic extracts is important to predict pitch problems that might arise during pulp and paper making.
Most studies of wood extracts have been made on total bark composite samples(Willför et al.2009;Sato et al.2009;Valentín et al.2010),although inner and outer bark differ considerably both in morphology and chemical composition(Fengel and Wegener 1989;Ha fizog˘lu et al.2002;Krogell et al.2012;Seki et al.2012).In this study,we used GC and GC–MS to describe the chemical composition of lipophilic extracts of the inner and outer bark of Indonesian pine(P.merkusii)and compared those with extracts of pines from other countries grown in Indonesia(P.elliotii,P.caribeae,P.oocarpa,P.montezumae,andP.insularis).
Materials and methods
Sample collection and extraction
Bark samples(2 individual trees of each species)were obtained from the Centre for Forest Biotechnology and Tree Improvement Research,Situbondo,East Java.The site is at 7°42′N 113°56′E,with temperature range of 24.7–27.9 °C,annual rainfall of 1300 mm,and elevation of 13 m a.s.l.The species wereP.elliotii,P.caribeae,P.oocarpa,P.merkusii,P.montezumae,andP.insularis(36–88 years old).Of these six species,onlyP.oocarpaandP.merkusiihave been planted for their oleoresins.The inner bark was characterized by its thin-layer and brighter color than the outer bark.After careful separation,each portion was milled to produce bark powder(100 mesh)and then was extracted(2 g)with dichloromethane for 6 h in a soxhlet apparatus(Freire et al.2002).The solvent was evaporated to dryness and the extracts were weighed.The results were expressed in percent of dry bark.The age,thickness,and seed source of samples are described in Table 1.
Analytical conditions
Before GC and GC–MS,each sample of dichloromethane soluble extract was prepared for direct injection and silylation.Briefly,40 μL sample(1 mg mL-1)were evaporated then were mixed with TMS-IH and BSA each 20 μL for derivatization.The aliquot was injected after the mixture had stood for 30 min.GC- flame ionization detection(GC-FID)was performed with GC-2014(Shimadzu,Japan)under the following conditions:DB-5 capillary column(30 m × 0.25 mm I.D.and 0.25 μm;GL Sciences,Tokyo,Japan);a column temperature from 100°C(1 min)to 320 °C at 5 °C min-1;injection temperature of 250 °C;detection temperature of 320°C and helium was used as the carrier gas.GC-mass spectrometry(GC–MS)data were collected with a GCMS-QP 2010(Shimadzu,Japan)under the following conditions: DB-1 capillary column(30 m × 0.25 mm I.D.and 0.25 μm;GL Sciences,Tokyo,Japan);column temperature from 50 °C(1 min)to 320 °C at 5 °C min-1;injection temperature of 250 °C;detection temperature of 320°C;acquisition mass range from of 50–800 AMU using helium as the carries gas.For direct injection,GC and GC–MS condition was similar to derivatization but different in GC column temperature(from 50°C).Components were identified by comparison with authentic compounds,the NIST MS library,and the literature(Fang et al.1991).
Quantification of extracts
Calibration curves of palmitic acid and α-pinene were prepared for fatty acids and monoterpenes,respectively.Abietic acid was used as standard solution for resin acids as well as stigmast-3-ene-4-one for steroids.Other components were quantified by comparison with internal standards i.e.veratric acid and heneicosane.Stigmast-3-ene-4-one was isolated fromP.merkusiibark in the previous study(Masendra et al.2017).
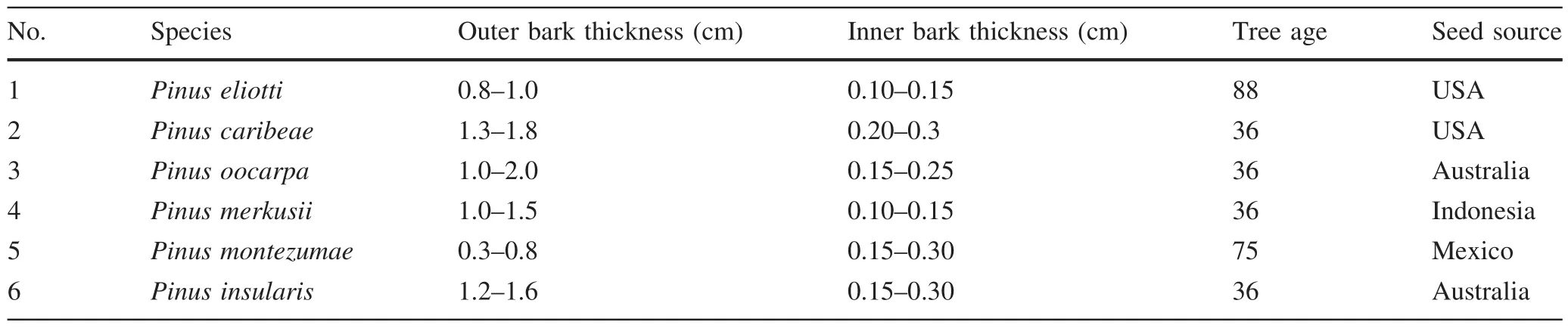
Table 1 Description of the bark samples
Chemicals
Heneicosane,veratric acid,palmitic acid,linoleic acid,linolenic acid,stearic acid,oleic acid,arachidic acid,pimaric acid,dehydroabietic acid,abietic acid,α-pinene,βmyrcene,and d-limonene were obtained from Kanto Chemical Co.,Inc.(Tokyo,Japan),and Wako Pure Chemical Industries,Ltd.(Osaka Japan)as component standards.
Results and discussion
Determination of extract contents
Dichloromethane extract contents of the outer bark were much higher than those of inner bark except forP.merkusii(Fig.1).Previous studies also showed that the outer bark had higher extractive content than inner bark onP.taeda(Fengel and Wegener 1989)and Norway spruce(Krogell et al.2012).The extractive content values of outer and inner bark ofP.oocarpawere the highest(4.5%)and the lowest(0.7%),respectively,compared to other species.These contents were within the ranges reported for the bark ofP.nigra(3.3%)(Hafizog˘lu et al.2002),P.pinea(2.1%)(Nunes et al.1999),P.elliotii(2.1%),P.taeda(1.7%),P.virginiana(1.5%),andP.echinata(2.6%)(Fengel and Wegener 1989).
GC and GC–MS analysis
Figures 2 and 3 show chromatograms from the inner bark extract ofP.merkusiiby direct injection and derivatization method,respectively.The lipophilic constituents in the bark samples can be grouped into monoterpenes and sesquiterpenes(ret.time 4–21 min),fatty acids and alcohols (ret.time 17–41 min),resin acids (ret.time 25–27 min),and steroids and triterpenoids(ret.time 49–55 min).Lipophilic constituents from the sixPinusspecies are listed in Table 2.
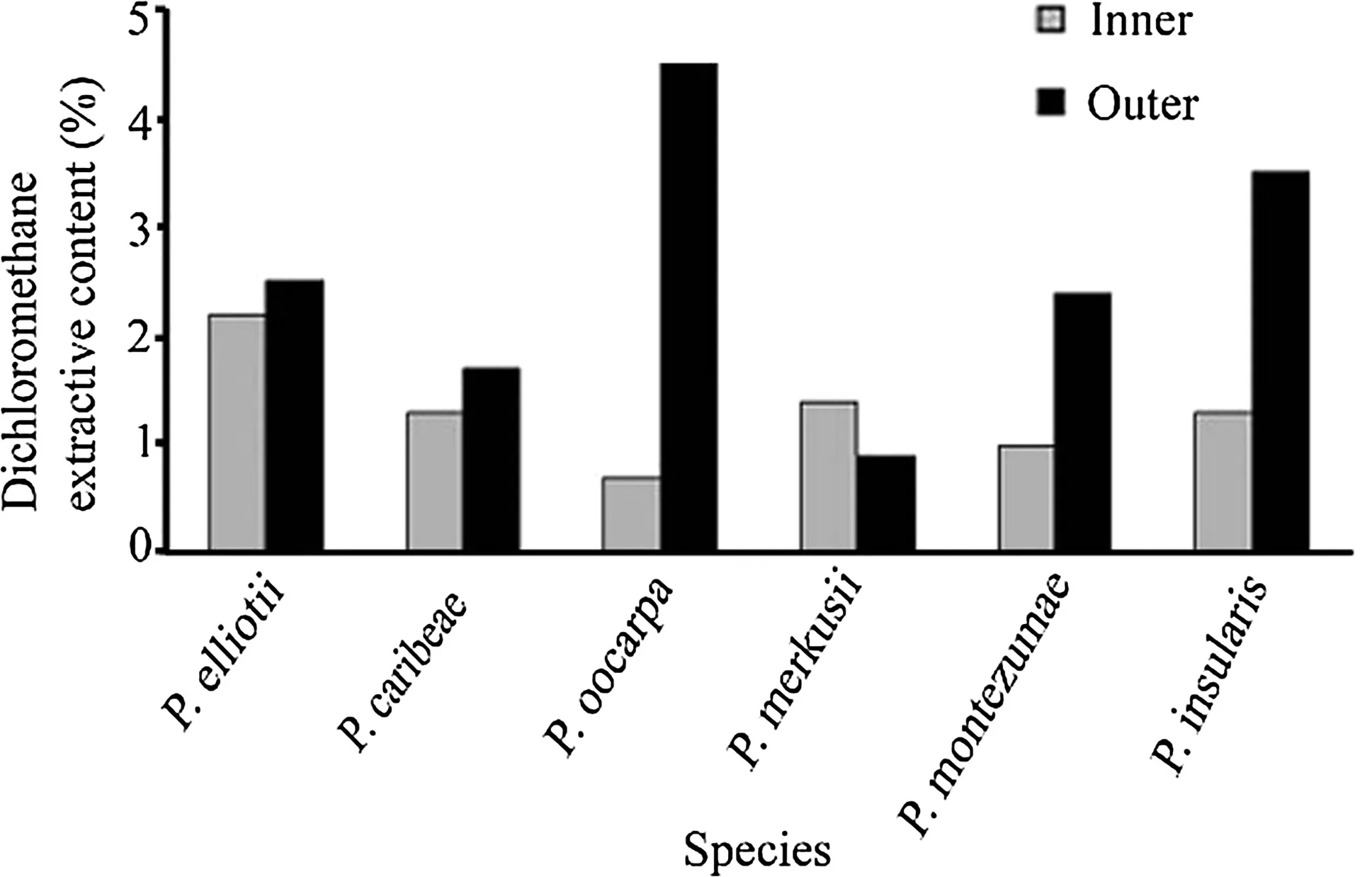
Fig.1 Dichloromethane extractive content(%,wood dry weight,average of two measurements)
Fatty acids and alcohols composition
In the inner bark,palmitic acid(C-16)and oleic acid(C-18)were the most predominant fatty acids among the sixPinusspecies,except forP.merkusii.Arachidic acid(C-20)was present in considerable amounts inP.merkusiiandP.caribeae.Unlike the other acids,palmitic acid and oleic acid were detected in a low concentrations in the bark ofP.oocarpaandP.merkusii.In the outer bark region,tetracosanoic acid(C-24)and n-tetracosanol-1 were most abundant.The fatty acids in our sixPinusspecies were quite common in some annual plants(Evdokimova et al.2013;Rahman and Mohammad 2011;Oliveira et al.2006).The fatty acids and alcohols in this experiment ranged from 4 to 27%on the basis of extract weight(Table 2).Large amounts of fatty acids and alcohols were detected in the bark ofP.nigra(Hafizog˘lu et al.2002),P.wallichianaandP.gerardiana(Willför et al.2009).The common fatty acids in the inner bark of six species(palmitic and oleic acids)also were reported inP.wallichiana,P.roxburghii,andP.gerardiana(Willför et al.2009).Arachidic acid,a major constituent inP.merkusii,was found in small amounts inP.taedabark(Fengel and Wegener 1989).Large concentrations of tetracosanoic acid were also reported for outer bark samples of six species,and these were similar to reports forP.taedaandP.sylvestris(Fengel and Wegener 1989;Valentín et al.2010).Outer bark samples had higher amounts of fatty acids than inner bark except inP.merkusii(Table 2).This was also reported for the bark ofP.taeda(Fengel and Wegener 1989)and Norway spruce(Krogell et al.2012).
Monoterpene and sesquiterpene composition
The identified monoterpenes and sesquiterpenes represented 0.1–1.14%of extract weight(Table 2).Further,αpinene,β-myrcene,and d-limonene,caryophyllenyl alcohol,caryophyllene oxide,6-epi-shyobunol,caryophyllene oxide isomer were the monoterpenes and sesquiterpenes identified in both inner and outer barks.Monoterpenes and sesquiterpenes have been reported in other morphological parts such as needles ofP.caribeae(Chowdhury et al.2008),turpentine oils,gum oleoresins,and rosins ofP.merkusii(Coppen et al.1998;Wiyono et al.2006).Monoterpenes and sesquiterpenes as constituents of lipophilic extracts in the bark were isolated together with diterpenes from the bark ofP.silvestris(Norin and Winell 1972).
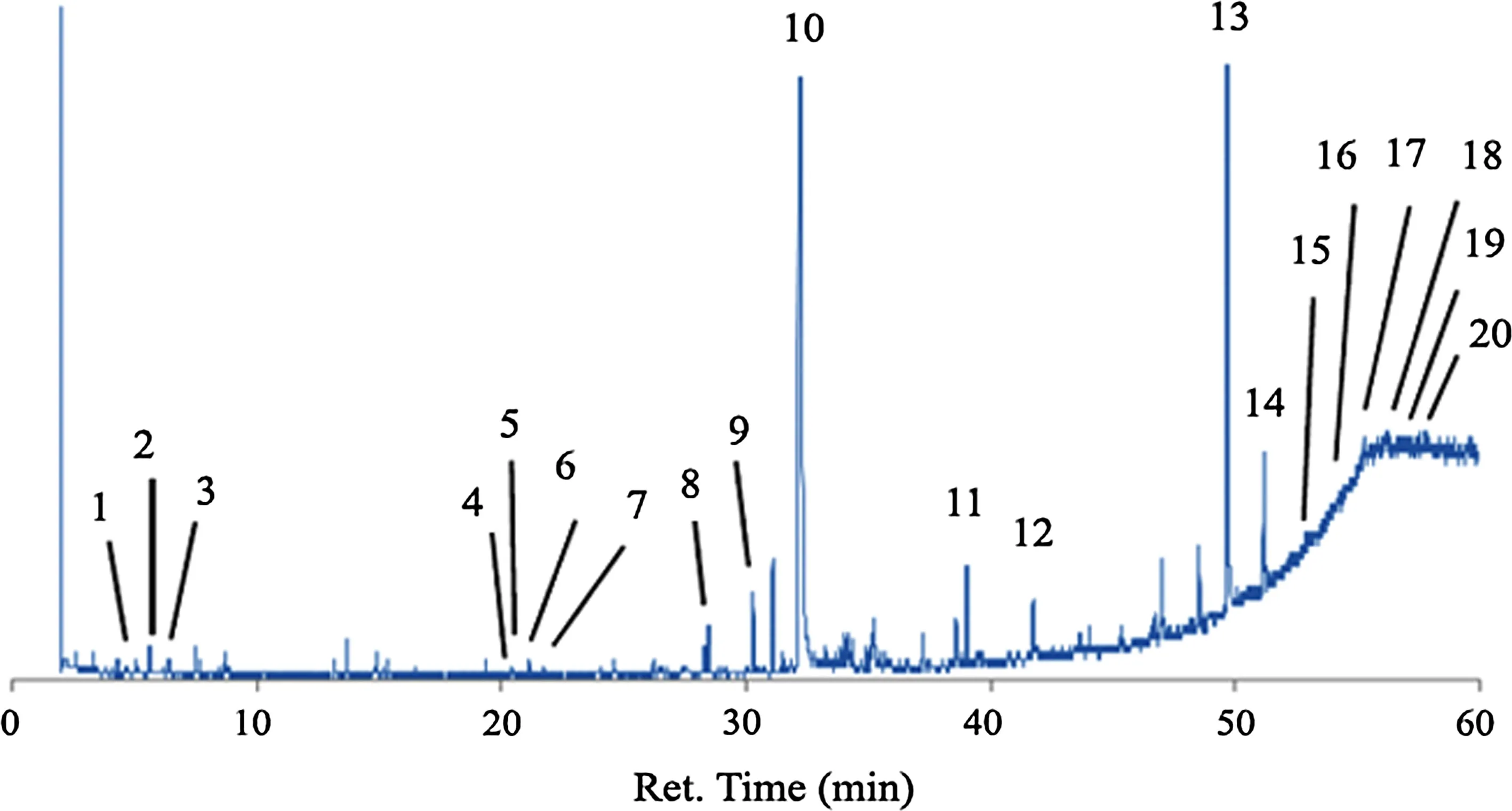
Fig.2 Chromatogram of dichloromethane extract from P.merkusii(Direct injection method),peak 1 α-pinene(4.3 min),2 β-myrcene(5.5 min),3 d-limonene(6.4 min),4 Caryophynllenyl alcohol(20.3 min),5 Caryophyllene oxide(20.5 min),6 6-epi-shyobunol(21.7 min),7 Caryophyllene oxide isomer(21.8 min),8 1-hexadecanol(27.5 min),9 Octadecane(30.2 min),10 internal standard:heneicosane(32.2),11 1-heneicosanol(38.5 min),12 n-tetracosanol-1(41.7 min),13 β-sitosterol(49.6 min),14 stigmast-4-en-3-one(51.1 min),15 2,4-cholestadien-1-one(51.7),16 25-hydroxycholesterol(52.6 min),17 acetic acid,octamethyl docosahydropicen-3-yl ester(53.4 min),18 betulin(53.6 min),19 3β-methoxyserrat-14-one(54.1 min),20 Serrat-14-en-3β,21β-diol(55.3 min)
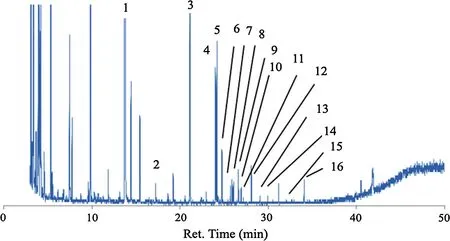
Fig.3 Chromatogram of dichloromethane extract from P.merkusii(derivatization method).Peak 1 internal standard:veratric acid(13.7 min),2 tetradecanoic acid (17.2 min),3 palmiticacid(21.1 min),4 linoleic acid(24.0 min),5 oleic acid(24.2),6 stearic acid(24.7),7 pimaric acid(25.4 min),8 pimaric acid isomer(25.7 min),9 isopimaric acid(25.9 min),10 dehydroabietic acid(26.6 min),11 cis-5,8,11-Eicosatrienoic acid(26.9 min),12 abietic acid(27.2 min),13 Arachidic acid(28.1 min),14 Docosanoic acid(31.1 min),15 Tetracosanoic acid,(34.0 min),16 hexacosanoic acid(36.7 min)
Resin acids composition
Pimaric acid and dehydroabetic acid were the most abundant components in the resin acid family detected by GC–MS,followed by isopimaric acid,especially inP.montezumae.Isopimaric and pimaric acid ranked second in quantity of resin acids among the sixPinusspecies.Baeza and Juanita(2001)reported that the major resin acid was often dehydroabietic acid,although this depended on tissue type,age and species.In an earlier report,dehydroabietic acid was isolated fromP.luchuensisand accounted for 14.7 mg or 17.5%of the bark extracts(Yamamoto et al.2011).Dehydroabietic,isopimaric,and pimaric acid were extracted from the bark ofP.taeda(Fengel and Wegener 1989),P.nigra(Hafizog˘lu et al.2002),P.sylvestris(Valentín et al.2010),and from the knotwood,heartwood,and sapwood ofP.merkusii(Wijayanto et al.2015).Concentrations of resin acids in the inner bark ofP.elliotii,P.caribea,andP.insulariswere lower than those in the outer bark.In contrast,resin acids were identified in greater amounts in the inner bark of other pine samples.Thequantity of resin acids in the sixPinuswas 2–13%of extract weight(Table 2)and was quite similar to the levels of resin acids ofP.nigra,ca.7.5%(Hafizog˘lu et al.2002).
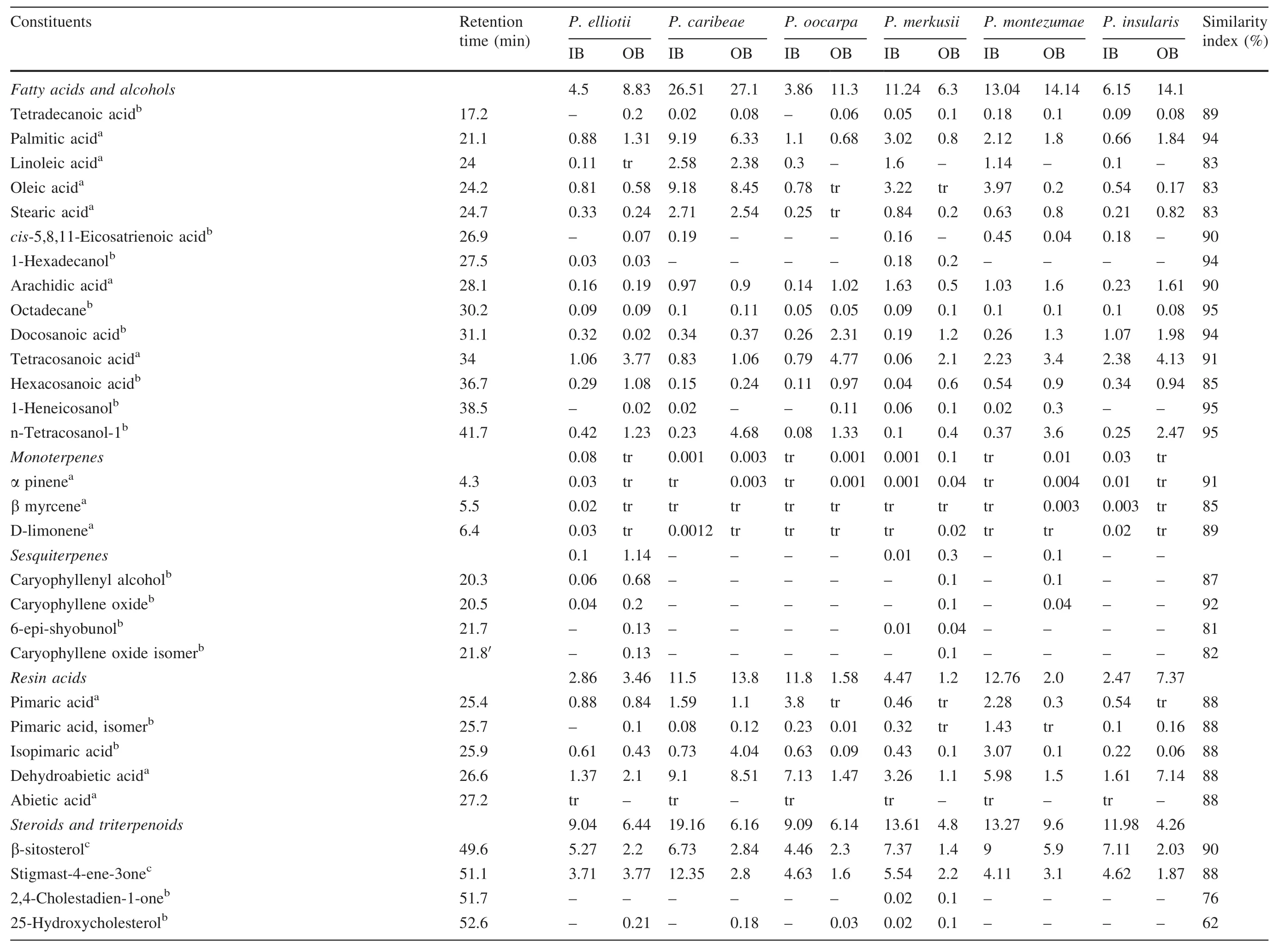
Table 2 Lipophilic constituents of the inner and outer barks identified by GC and GC–MS(%dryextract,average of two measurements)
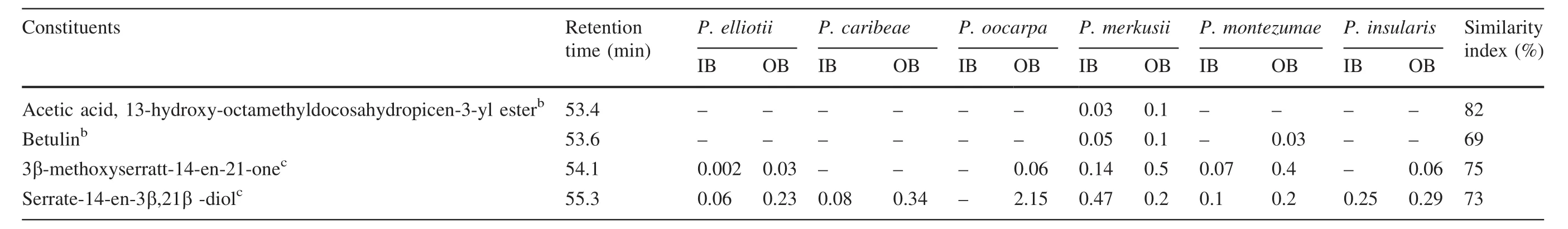
Table 2 continued
Steroids and triterpenoids composition
The last group identified in the bark of all testedPinusspecies were steroids and triterpenoids.β-sitosterol and stigmast-4-ene-3-one were among the most predominant compounds in all samples.Other steroids and triterpenoids identified in smaller amounts were 2,4-cholestadien-1-one,25-hydroxycholesterol, acetic acid, 13-hydroxy-4,4,6a,6b,8a,11,11,14b-octamethyl docosahydropicen-3-yl ester,betulin,3β-methoxyserratt-14-en-21-one,and serrate-14-en-3β,21β -diol.Sitosterol was mostly found in the bark ofP.wallichiana,P.roxburghii,P.gerardiana(Willför et al.2009),P.nigra(Hafizog˘lu et al.2002),andP.luchuensis(Yamamoto et al.2011).The inner bark ofP.caribeae,P.merkusii,andP.montezumaecontained total steroids and triterpenoids in high amounts.Further,sitosterol(7.37%)and stigmast-4-ene-3-one(5.54%)were at high concentrations in the inner bark ofP.merkusii.Sitosterol accounted for 3–9%of total extract weight.These results were similar to those reported forP.luchuensisbark,around 8.9%(Yamamoto et al.2011)andP.nigrabark,around 8%(Hafizog˘lu et al.2002).
The most abundant of triterpenoids detected in all specimens were 3β-methoxyserratt-14-en-21-one and serrate-14-en-3β,21β–diol. 3β-methoxyserratt-14-en-21-one was also detected in the bark ofP.armandii(Fang et al.1991;Fang and Cheng 1992),andP.luchuensis(Yamamoto et al.2011),while serrate-14-en-3β,21β–diol was reported from the bark ofP.lambertiana(Rowe 1964).Serrate-14-en-3β,21β–diol was not detected in the inner bark ofP.oocarpaonly,while 3β-methoxyserratt-14-en-21-one was not detected in both inner and outer bark ofP.caribeae.The triterpenoids in the six samples were in the range of 0.1–1%.This was lower than reported forP.luchuensis, in which 3β-methoxyserratt-14-en-21-one accounted for 11.0%(Yamamoto et al.2011).In general,concentrations of steroids and triterpenoids were higher in inner than outer bark.These results were generally consistent with the chemical composition of lipophilic extracts ofP.taeda(Fengel and Wegener 1989),and Norway spruce(Krogell et al.2012).
Inner and outer bark composition
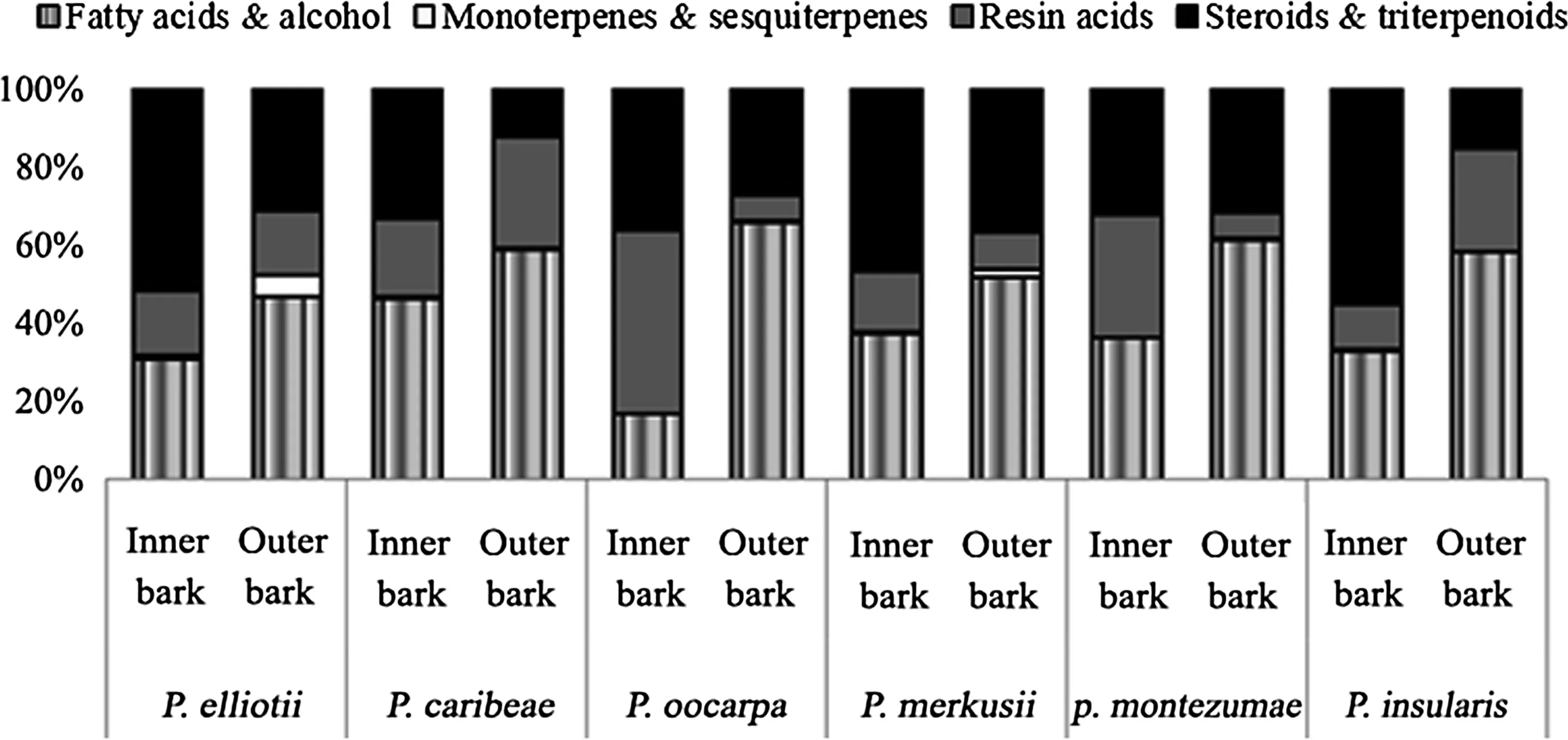
Fig.4 Major families of compounds identified in the bark of the six Pinus species(percentage of extract weight,average two measurements)
In general,the major group of the lipophilic components in both the inner and outer bark was found to be varied and dependent on species and radial position(Fig.4).In contrast,the monoterpenes and sesquiterpenes occurred in low concentrations in all species.The levels of fatty acids and alcohols increased,while the steroids and triterpenoids decreased from the inner bark to the outer bark.Another factor accounting for variation was tree age;the age range was 36–88 years.Previous studies reported that extracts from the trees were depended on tree age(Baeza and Juanita 2001;Berrocal et al.2004;Aspeánd Fernández 2011;Seki et al.2012).Hence,it is important to account for the effect of tree age in future works.
Smaller concentrations of lipophilic extracts in tree wood ease the pulping process.In addition to content,the composition strongly affects the pitch deposition during pulping and papermaking (Back and Allen 2000).Unsaponifiable constituents such as steroids and terpenoids are the main causes for pitch problems in the kraft pulping of some hardwoods(Gutiěrrez et al.1999,2001;Martínez-Iñigo et al.2001).The smaller amounts of unsaponifiable constitutent extracts recorded in the outer bark in our study could be an advantage in terms of pitch formation.On the other hand,the high proportions of neutral compounds in inner bark ofP.elliotii,P.merkusii,andP.insulariswould suggest tendency to deposit large amounts of pitch.From an ecological perspective,large amounts of steroids and terpenoids might be important as barriers against herbivores(Gershenzon 1994;Wittstock and Gershenzon 2002)and might defend against natural enemies,thereby preventing damage and protecting from chemical deterioration(Seki et al.2012).
Conclusion
The inner bark contained more lipophilic extracts than did the outer bark except in the case ofP.merkusii.The lipophilic extracts identified by GC and GC–MS were composed of four component families i.e.fatty acids and alcohols;monoterpenes and sesquiterpenes;resin acids;steroids and triterpenoids.Fatty acids and alcohols were the main group of compound in the outer bark ofP.caribeae,P.insularis,andP.montezumae.Steroids and triterpenoids represented the major pro-portion in the inner bark ofP.elliotii,P.insularis,andP.merkusii.Resin acids were most abundant in the inner bark ofP.oocarpawhereas monoterpenes and sesquiterpenes were minor compounds in all species.Palmitic acid(C-20),oleic acid(C-18),and tetracosanoic acid(C-24)were the major fatty acids whereas dehydroabietic acid was the most abundant component in resin acids.Sitosterol and stigmast-4-ene-3-one were the main steroids identified in all extracts.
As a minor fraction of bark normally enters the pulp mill with wood chips,the comparatively high amount of steroids and triterpenoids in the lipophilic fractions from the inner bark ofP elliottii,P.merkusii,andP.insularissuggests that pitch formation problems will be more severe for those species than for others.On the other hand,the lower abundance of unsaponifible fractions in the outer bark of all six pines seems to be clearly beneficial as far as pitch formation is concerned.Thus,further work should be conducted to determine the effects of the bark lipophilic fractions during pulping and bleaching stages.
AcknowledgementsWe thank the Centre for Forest Biotechnology and Tree Improvement Research for providing research materials.
 Journal of Forestry Research2018年5期
Journal of Forestry Research2018年5期
- Journal of Forestry Research的其它文章
- Environmental load of solid wood floor production from larch grown at different planting densities based on a life cycle assessment
- An overview of proven Climate Change Vulnerability Assessment tools for forests and forest-dependent communities across the globe:a literature analysis
- Assessing the vulnerability of a forest ecosystem to climate change and variability in the western Mediterranean sub-region of Turkey:future evaluation
- Effects of management regimes on carbon sequestration under the Natural Forest Protection Program in northeast China
- Mixed-effects modeling for tree height prediction models of Oriental beech in the Hyrcanian forests
- Estimation of a basal area growth model for individual trees in uneven-aged Caspian mixed species forests
