Mangiferin ameliorates hyperglycemia by inhibiting oxidation and α-glucosidase activity
Chi-Chi He,Zhuo Luo,Lu-Lu Wang,Xu-Xian Xiao,Jian-An Hu,Yi-Fang Li,Hiroshi Kurihara,Rong-Rong He*
1XiangYa School of Public Health,Central South University,Changsha,China.2Anti-Stress and Health Research Center,College of Pharmacy,Jinan University,Guangzhou,China.3College of Chemistry and Chemical Engineering,Central South University,Changsha,China.
Abstract Objective:Mangiferin(MF)is a polyphenol isolated from the root of Anemarrhena asphodeloides Bge..This study was aimed to investigate the effects of MF on hyperglycemia in animal models of insulin resistance and streptozotocin(STZ)-induced diabetes.Methods:The diabetes mellitus model was established in mice by receiving a multiple hypodermic injection of hydrocortisone sodium succinate(HCSS)(70 mg/kg)or a single intravenous injection of STZ(130 mg/kg).Meanwhile MF at different dosage(50,100 and 200 mg/kg)were oral administrated for consecutive 10 days.Data of blood glucose were collected at different time after intraperitoneal injection of insulin(0.5 U/kg)to investigate the insulin resistant.As well as the oxygen radical absorbance capacity(ORAC)and superoxide dismutase(SOD)activity of kidney were measured.The in vitro experiment was established to investigate the inhibitory capacity of MF to α-glucosidase.Results:Oral administration of MF significantly prevented insulin resistance caused by HCSS injection.STZ-induced diabetic symptoms were also improved,including fasting blood glucose,glycated hemoglobin,plasma triglycerides,hepatic glycogen,kidney SOD and ORAC level.The in vitro experiment demonstrated that MF had potent α-glucosidase inhibitory activity.Conclusion:The obtained results demonstrate that MF ameliorates insulin resistance and STZ-induced glucose metabolism disturbance.The MF exerts the protective effects through improving the antioxidant ability,promoting hepatic glycogen synthesis and inhibiting α-glucosidase activity.
Keywords:Mangiferin,Streptozotocin,Diabetes,Insulin resistance,α-glucosidase
Background
Diabetesmellitusisametabolicdisease which is characterized by sustained high blood glucose level and metabolic disturbance.As of 2014,an estimated 387 million people have diabetes worldwide.This number will continue to rise to 592 million by 2035[1].Developing countries account for 75%of the global burden for diabetes,and diabetes deaths occurred in lowand middle-income countries is over 80%[2].Blood glucose monitoring is valuable for the therapy of diabetes,and glycated haemoglobin(HbA1c)concentration gives an indication of average blood glucose levels.The excessive glycation of haemoglobin and other proteins could easily induce oxidative stress[3,4].Therefore,regulating blood glucose homeostasis is the main therapy strategy,however developing drugs with activities of reducing protein glycation and oxidation is more significant.
Anemarrhenae Rhizoma,the dried root of Anemarrhena asphodeloides Bge.,is a medicinal plant commonly found in the northwestern and northern China.It is often used in traditional Chinese medicine as an anti-diabetic,antipyretic,anti-inflammatory and antidepressant drug[6].Mangiferin(MF),a natural polyphenol isolated from Anemarrhenae Rhizoma(Figure 1),has been suggested to improve diabetic symptoms and its complications[7,8].However,the pharmacological activities and mechanism of MF is needing to be further researched.The current study was aimed to evaluate the activities and mechanism of MF in insulin resistance and diabetic mice model,which will provide insights of using MF in the treatment of diabetes related metabolic disorders.
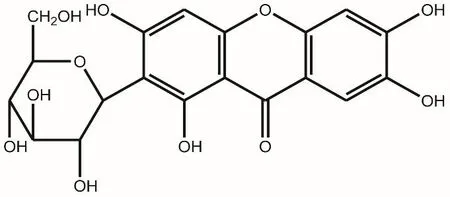
Figure 1 Chemical structure of mangiferin
Materials and Methods
Chemicals and Reagents
MF was provided by Guangzhou LifeTech Pharmaceutical Co.,Ltd.(Guangzhou,China).Hydrocortisone sodium succinate and insulin injection were purchased from Tianjin Biochem Pharmaceutical Co.,Ltd.(Tianjin China).Metformin(Met),streptozotocin(STZ),p-nitrophenol glucoside,acarbose and α-glucosidase were purchased from Sigma(St.Louis,MO,USA).2,2’-azobis(2-amidinopropane)dihydrochloride(AAPH),6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid(Trolox,a water-soluble vitamin E analogue)and sodium fluorescein were purchased from Wako Pure Chemical Industries,Ltd.(Osaka,Japan).Pierce BCA protein assay kit was obtained from Thermo Fisher Scientific(Waltham,MA,USA).HbA1c,plasma triglycerides(TG)and Superoxide dismutase(SOD)kit were purchased from Jiancheng Bioengineering Institute(Nanjing,China).
Animals
Male Kunming mice,weighing from 18.0 to 22.0 g,were purchased from the Guangdong Medicinal Laboratory Animal Centre,Guangzhou,China.The animals were kept in a specific pathogen-free animal room at 23 ± 2°C with a 12-hour dark-light cycle and fed with standard laboratory diet and tap water.The animals were allowed to acclimatize to the environment for 1 week before the experiment.
Intraperitoneal insulin tolerance test
For induction ofinsulin resistance,animals were randomly divided into six groups,including normal control,model group,Met group(300 mg/kg,),and three MF groups(50,100 and 200 mg/kg,MF-L,MF-M and MF-H).All mice except normal control and model group were orally administrated with the specific drug for 10 d.Normal control and model group were administrated with same volume of CMC-Na solution.Meanwhile,hydrocortisone sodium succinate(70mg/kg)was subcutaneously injected for 10 d in all mice except normal control to induce insulin resistance.After the last injection,all mice were fasted for 12 h for intraperitoneal insulin tolerance test.Insulin(0.5U/kg)was intraperitoneally administered to allanimals.Blood glucose was measured at 0,30,60 and 120 min after insulin administration.
STZ-induced diabetic mice model
For induction of experimental diabetes,animals were randomly divided into six groups as stated above.All mice except control group were intravenously injected with the β-cell toxin STZ for one week(130 mg/kg/day in 20 mM sodium citrate,pH 4.5).The development of hyperglycemia in rats was confirmed by plasma glucose estimation 12 h post STZ injection.animals with elevated blood glucose(>12 mmol/L)were considered diabetic and used for the following experiments.
Measurement of blood glucose,HbA1c and plasma TG in STZ-induced diabetic mice
After induction of experimental diabetes,all mice except normal control and model group were orally administrated with the specific drug for 10 d.Normal control and model groups were administrated with same volume of CMC-Na solution.On the last day of drug administration,all mice were fasted for 12 h to determine blood glucose.To obtain plasma,liver and kidney,all mice were anesthetized by ether and sacrificed on ice.Blood was collected by heart puncture and transferred to centrifuge tubes with sodium heparin.After centrifugation at 4°C,10000 rpm for 8 min,plasma was used to determine thecontentofTG,whilethe precipitated red blood cells were used to determ ine the level of HbA1c using commercial kits.
M easurement of hepatic glycogen
Liver tissue was obtained from anesthetized mice and washed with normal saline.A ll tissues were dried,weighed,and put into test tubes with lye solution in a ratio of 1:3(mg:μl).The test tubes were placed in a boiling water bath for 20 min and then cooled by running water.Hydrolyzed hepatic(1%)were mixed with distilled water to obtain sample solutions.The prepared sample solutions were then combined with chromogenic reagent and placed in the boiling water bath for 5 min further.Finally,Hepatic glycogen content was determ ined with an MK3 microplate reader at 620 nm(Thermo Scientific,Waltham,MA,USA)according to previous report[15].
M easurement of kidney SOD activity and Oxygen Radical Absorbance Capacity(ORAC)level
Kidney homogenate was prepared with normal saline and diluted to suitable concentrations before SOD and ORAC assay.The total SOD activity was detected by commercial kits according to the manufacturer’s protocol.The automated ORAC assay was carried out on a GENios luciferase-based microplate reader(TECAN,Männedorf,Switzerland)with an excitation/em ission filter pair of 485/527 nm as previously described[16].
M easurement of α-glucosidase inhibitory activity
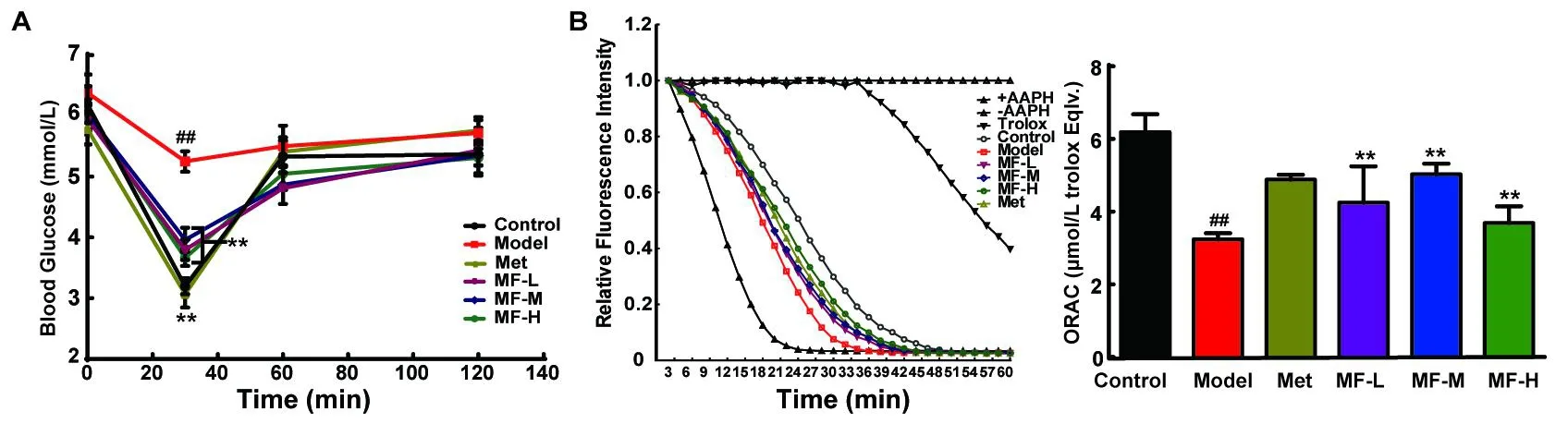
Figure 2 Effects of mangiferin on HCSS-induced insulin resistance.Blood glucose level(A)and kidney ORAC(B)was determined.The data were expressed as mean±S.D.(n=10).Data were regarded as statistically significant with##P<0.01 vs control and*P<0.05;**P<0.01 vs model mice.
Theα-glucosidase inhibitory assay was performed spectrophotometrically on 96-well microplate reader at 400 nm according to a reported method[17].In brief,the sample solution(2.2 ml)and 1 U/m l α-glucosidase(0.01 ml)were added to 20 mM p-nitrophenol glucoside in 0.01 Mphosphate buffer(pH 7,0.21 ml)to start the reaction.Each reaction was carried out at 37°C for 15 min and stopped by adding 0.2 MNa2CO3(0.01 ml).Acarbose was used as positive control.Enzymatic activity was quantified by measuring absorbance at 400 nm.The assay was performed in triplicates.
Statistical analysis
All numeric numbers are represented as the mean±SD.The data obtained were analyzed by ANOVA followed by Dunnett’s significant post-hoc test using the SPSS 18.0 statistical software.A P<0.05 was considered statistically significant.
Results
Effects of MF on intraperitoneal insulin tolerance test
As illustrated in Figure 2A,glucose concentrations before insulin(0.5 U/kg)injection was around 5.78~6.38 mmol/L.Blood glucose of normal control mice dropped to 3.20±0.13 mmol/L at 30 min after insulin injection.When comparing to normal control,significant higher blood glucose was noticed in model mice(5.25±0.17 mmol/L),the HCSS-induced insulin resistance mice.However,the oral administration of three different doses of MF could decrease blood glucose to about 66%of baseline level in model mice.The effect of high-dose MF(200 mg/kg)was the most significant.This effect of MF on improving blood glucose might be related with its antioxidant activity.Because,MF group got different degree of recovery of ORAC compared to model group(Figure 2B).
Effects of MF on fasting blood glucose in diabetic mice induced by STZ
Figure 3A shows that the 12 h fasting blood glucose in the animals was elevated to above 20 mmol/L after streptozotocin(STZ)injection.After 10 d of oral administration,the fasting blood glucose of all three MF groups was remarkably lowered.This finding showed that MF had blood glucose lowering ability sim ilar to Met.
Effects of MF on HbA1c and plasma TG in STZ-induced diabetic mice
Glycated HbA1chas been proposed for the determination of glucose control owing to its higher repeatability and being assessed under non-fasting state.As showin Figure 3B,the level of glycated HbA1c was significantly elevated in model mice compared with normal control.Oral administration of Met could prevent the elevation of HbA1c by 12.70%.Meanwhile,high dose of MF had the most prom ising effect,with 10.63%lowering effect on HbA1c.For plasma TG,STZ caused a significant increase in model mice(Figure 3C).Intriguingly,the effect of MF was more notable than the positive control(Met).Moreover,all three doses could inhibit the increase of plasma TG level,even lower than those in normal control mice.
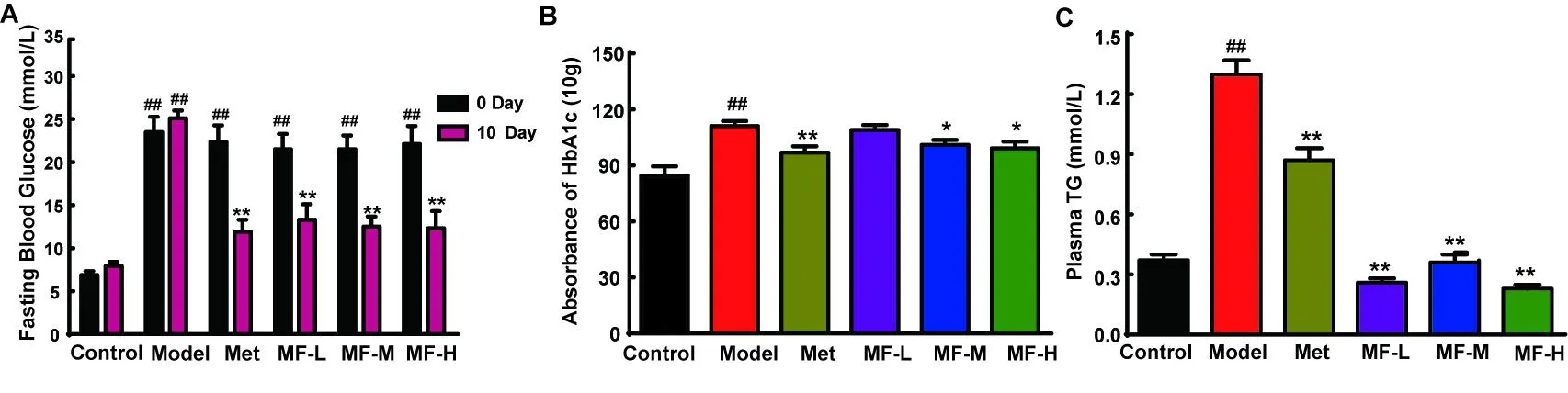
Figure 3 Effects of mangiferin on STZ-induced diabetic mice.Fasting blood glucose level(A)was determined.The level of HbA1c(B))and plasma TG(C))were also measured by commercial kits.The data were expressed as mean±S.D.(n=10).Data were regarded as statistically significant with##P<0.01 vs control and*P<0.05;**P<0.01 vs model mice.
Effects of M F on hepatic glycogen level in STZ-induced diabetic mice
To confirm whether MF has effect on liver function of regulating blood glucose,hepatic glycogen content was measured.STZ-induced diabetic mice were obviously lower than those of normal control(Figure4A).Adm inistration of Met improved hepatic glycogen content to 67.77%of normal level.However,only high dose of MF could recover hepatic glycogen after 10 d.
Effects of MF on kidney oxidation in STZ-induced diabetic mice
The administration of STZ induced a 41.81%decrease of kidney superoxide dismutase(SOD)activity in model mice,when compared with normal control(Figure 4B).Oral adm inistration of high dose MF significantly recovered SOD activity by 34.74%.There was no significant effect of Met on SOD activity.However,Met could improve ORAC from 3.00±0.16 to 4.24±0.24 μmol/L Trolox equiv.(Figure 4C).Oral adm inistration of midle and high dose of MF also recovered ORAC value to 4.21 ± 0.31 and 4.50 ± 0.32 μmol/L Trolox equiv.,respectively.
Inhibitory effect of MF on α-glucosidase activity
The α-glucosidase inhibitory activity of MF was in a dose-dependent manner(Figure5).Under the concentration of 5.0 mg/m l,the effect of MF was even better than the positive control acarbose.
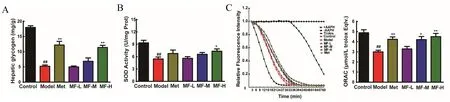
Figure 4 Effects of mangiferin on hepatic glycogen and kidney antioxidant capacity in STZ-induced diabetic mice.Hepatic glycogen(A),kidney SOD(B)and ORAC(C&D)were determined respectively to evaluate the protective mechanism of mangiferin.The data were expressed as mean±S.D.(n=10).Data were regarded statistically significant with##P<0.01 vs control and*P<0.05;**P<0.01 vs model mice.
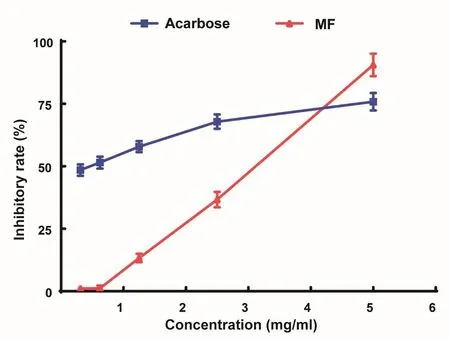
Figure 5 Inhibitory effect of mangiferin(MF)on α-glucosidase activity.The data were expressed as mean±S.D.The results are representative of three separate experiments.
Discussion
The pharmacological mechanism of traditional Chinese medicine on diabetes mellitus can be divided into several categories,such as promoting insulin secretion,increasing insulin sensitivity,prolonging the absorption of glucose,inhibiting degradation of hepatic glycogen and boosting antioxidant capacity[9].The present study employed HCSS-induced insulin resistance and STZ-induced diabetes experimental model to study the effects of MF on glucose metabolism in mice.Results showed that MF could prevent HCSS-induced insulin resistance and lower fasting blood glucose level in STZ-induced diabetic mice.These data revealed that MF possessed potent hypoglycem ic activity in diabetic animals.
To further understand the mechanism of MF on experimental diabetes,several parameters were studied,including the level of HbA1c,plasma TG,hepatic glycogen,kidney SOD and ORAC.Result showed that MF could significantly reduce the level of HbA1c and plasma TG in STZ-induced diabetic mice.The formation of HbA1c is through a non-reversible glycation of HbA1c under plasma glucose exposure.Its level can be used to predict the average blood glucose levels over the previous 8 weeks prior to the measurement[10].Meanwhile,the change in plasma TG level is closely related to diabetic complications[11].These results demonstrated that MF could protect STZ-induced diabetic mice from prolonged high blood glucose level and may prevent the incidence of diabetic complications.In our experiments,we also observed that hepatic glycogen content,kidney SOD activity and ORAC level were improved after MF administration.These results were sim ilar with previous studies[12,13],suggesting that the mechanism of MF might also include the inhibition of glycogen degradation and promotion of anti-oxidant capacity inside the diabetic animal.Another strategy to lower blood glucose concentration is to inhibit the activity of α-glucosidase,which is responsible for the breakdown of starch and disaccharides to glucose in the small intestine[14].In the current study,we detected the in vitro α-glucosidase inhibitory activity of MF.When compared to the positive control acarbose,a high dose(5.0 mg/m l)of MF showed a better inhibitory effect on α-glucosidase activity than acarbose.This suggested that the anti-diabetic effect of MF could be connected with its inhibition on α-glucosidase.
In conclusion,we found that MF possessed potent anti-diabetic activity.The underlying mechanism of MF was possibly due to the promotion of anti-oxidant capacity,which protected hepatic cells from β-cell toxicity.Meanwhile,MF could lower plasma TG level,promote hepatic glycogen synthesis and inhibit the activity of α-glucosidase.These findings were in line with the integral medication of multi-factorial,multi-target action of traditional Chinese medicine and could provide newinsights on the use of traditional Chinese medicine on the treatment of diabetes mellitus.
 TMR Modern Herbal Medicine2018年1期
TMR Modern Herbal Medicine2018年1期
- TMR Modern Herbal Medicine的其它文章
- Go with the flow,emerge as the times require and propel the modernization of traditional Chinese medicine to a new era
- HPLC Fingerprinting and Spectrum-antitumor Effect Relationship for Discrim ination between Mylabris phalerata Pallas and Mylabris cichorii Linnaeus
- Explore the direct and/or the synergistic antihypertensive effects of wind-dispelling herbs involving Fangfeng and Baizhi on hypertensive rats with liver-yang hyperactivity based on vasoactive substances
- External application of Chinese medicine formula combined with analgesic drugs to treat lung squamous cell carcinoma pain:A case study with mixed methods
- Perspectives in app lication of biosensors for the modernization of traditional Chinese medicine
- Swallow anything and everything,seek difference and truth-The consideration on development of post-modern Chinese medicine
