睡眠障碍中心的认证标准Standards for Accreditation of Sleep Disorders Centers
Introduction
Accreditation by the American Academy of Sleep Medicine (AASM) is a voluntary program offered to Sleep Disorders Centers and Laboratories for Sleep Related Breathing Disorders that meet the standards contained in this document. These standards have been developed for the primary purpose of ensuring the highest quality care be delivered to patients with a sleep disorder.
For the purpose of clarity and brevity the remainder of this document will use the term “sleep facility” when referring to sleep disorders centers and/or laboratories for sleep-related breathing disorders. Your application must indicate the type of facility for which you are applying for accreditation.
In broad terms, the Standards for Accreditation describe the required structural, professional and human resource, clinical and technical standards, emergency and quality assurance methods required for accreditation by the AASM. Sleep facilities achieving accreditation should be recognized in the community as a resource for expertise in sleep medicine.
The AASM uses a rigorous evidence-based process to establish Practice Parameters on a variety of topics that are relevant to the practice of Sleep Medicine. Accredited sleep facilities must adopt and follow the standards in all active AASM Practice Parameter papers. Standards can easily be identified as they are all in bolded print in every practice parameter paper. In addition, it is recommended that accredited sleep facilities adopt and follow all active AASM Clinical Guidelines.
The AASM recognizes that the practice of Sleep Medicine, like all other medical disciplines, is dynamic, complex and requires clinical judgment. AASM Practice Parameters are not designed to limit physicians from using their medical judgment, which, in individual patients, may require deviation from AASM Practice Parameters. AASM accredited sleep facilities are expected to document instances requiring deviation from AASM Practice Parameters.
The AASM reserves the right to modify, add, or remove accreditation standards at its own discretion without notice.
American Academy of Sleep Medicine
2510 North Frontage Road
Darien, IL 60561-1511
Voice: 630.737.9700
Facsimile: 630.737.9790
Preamble
AASM accredited sleep facilities must be in compliance with all accreditation standards at the time of application. If it is determined in the application review process that a sleep facility is not in compliance with the required standards, the application will be returned and the sleep facility will need to resubmit its application once the required standards are met.
It is the responsibility of the AASM accredited facility to ensure that all sleep facility clinical and technical personnel, regardless of their role, work as regulated by their primary field′s scope of practice, and as consistent with their education, expertise, and state licensure. Only an appropriately licensed MD, DO, PA, or NP may make a medical diagnosis and prescribe medical treatment.
Denial of accreditation will be recommended by the site visitor, reviewers, accreditation committee, or staff when one or more of the following conditions are identified:
1.The sleep facility fails to meet any of the accreditation standards that are indicated as “MANDATORY.” Sleep facilities will not be issued provisos for accreditation standards indicated as MANDATORY.
2.The sleep facility is determined to be non-compliant with more than thirteen (13) accreditation standards.
3.The sleep facility fails to resolve provisos within the period of time allotted to correct the proviso(s).
4.The AASM has evidence that the sleep facility submitted falsified documents or misrepresented information in seeking to achieve or retain accreditation.
5.The sleep facility fails to notify the AASM within 30 days of initiation of any government, local, state or federal investigation or adverse action taken against the facility that impacts the ability to meet any standards.

Table of Contents of Accreditation Standards
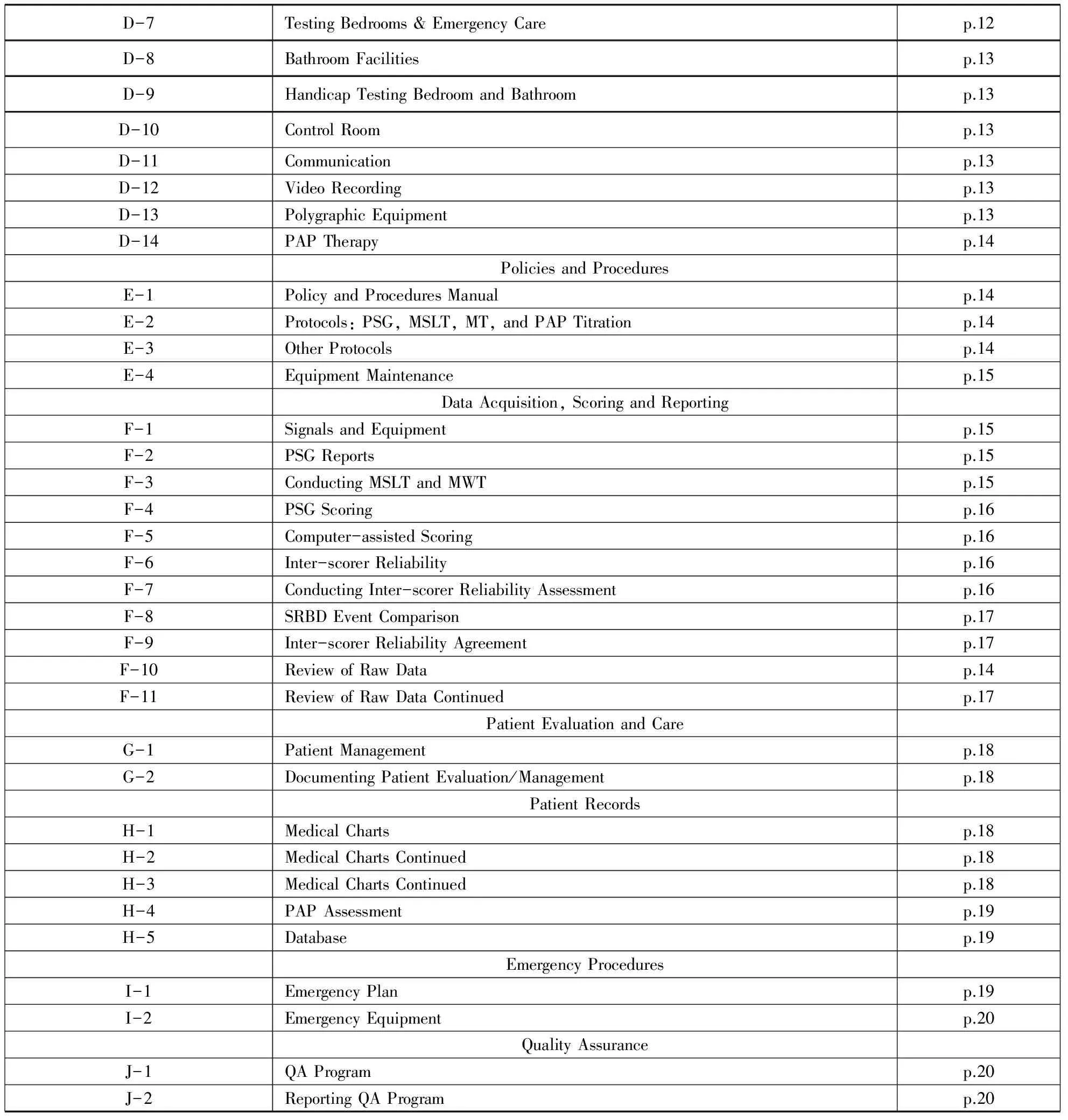
Continued Table of Contents of Accreditation Standards
A.General Standards
Standard
A-1-Facility License(MANDATORY)
AASM accredited sleep facilities must maintain a valid license,certificate of occupancy,and/or permit,when required by applicable law and regulation,to provide health care services.It is the responsibility of AASM accredited facilities to maintain compliance with all licensing acts,local building codes and any other laws relevant to the facility′s operation.
Failure to comply with the stipulations in this paragraph is sufficient for denial and/or revocation of accreditation.A facility′s valid healthcare license,certificate of occupancy or business permit fulfills this standard.If applicable law does not require a sleep facility to have a healthcare license,certificate of occupancy or business permit,written attestation of such by the Medical Director is required.
Standard
A-2-Medical Code of Ethics(MANDATORY)
AASM accredited sleep facilities are required to follow the Code of Medical Ethics of the American Medical Association which the AASM adopted as official policy in 1998.The sleep facility must either have on hand or have the ability to access easily the Code of Medical Ethics of the American Medical Association Council on Ethical and Judicial Affairs Current Opinions.
B.Personnel
Standards B-1 through B-6 relate to the appointment,responsibilities and continuing education of a physician medical director and/or the appointment of a physician or PhD board certified sleep specialist.
A physician meeting all the requirements of B-1 through B-6 can fulfill both designations.A PhD can only fulfill the board certified sleep specialist designation.
Standard
B-1-Medical Director(MANDATORY)
AASM accredited sleep facilities must designate a single medical director who is a physician with a license valid in the state of the facility and in all states in which patients are seen.A copy of each state medical license must be submitted with the application.
An individual can serve as medical director of three(3)sleep facilities,regardless of their accreditation status.If an individual is medical director of more than three facilities,the total number of beds shall not exceed 16 beds.
Standard
B-2-Medical Director Responsibilities(MANDATORY)
The medical director:
is responsible for the direct and ongoing oversight of testing,
is responsible for the qualifications of all medical and technical personnel,
must be present in the sleep facility on a regular basis and not less than 8 hours each month.
Standard
B-3-Medical Director Continuing Education(MANDATORY)
The medical director must participate in at least 10 credits per year averaged over three years of AMA PRA Category 1 CME credit in sleep medicine.Compliance with CME requirements must be documented.
Standard
B-4-Board Certified Sleep Specialist(MANDATORY)
AASM accredited sleep facilities must designate a single professional sleep specialist defined by at least one of the following:
6.An individual who is board-certified in sleep medicine by the American Board of Sleep Medicine or an individual certified in sleep medicine by either a member board of the American Board of Medical Specialties or a member board of the American Osteopathic Association.
7.An individual who has been accepted by an ABMS or AOA approved board to sit for the examination in sleep medicine.To retain the accreditation,the examination in sleep medicine must be passed within 2 examination cycles.To meet this requirement,the individual must provide,in the application packet,a letter of acceptance to sit for the examination by the ABMS or AOA approved board.Upon completion of the examination,the individual must provide a copy of the official notification from the ABMS or AOA board indicating final status.
8.An individual who has completed a 12 month fellowship in sleep medicine and is awaiting the first available opportunity to apply to an ABMS board to sit for the sleep medicine examination.To retain accreditation,the ABMS examination in sleep medicine must be passed within 2 examination cycles.
Standard
B-5-Board Certified Sleep Specialist Responsibilities(MANDATORY)
The designated board certified sleep specialist:
a.must provide direct and ongoing oversight of the testing protocols and the quality of testing including the proper operation and calibration of the equipment,
b.must review,report,and modify as necessary the facility′s quality assurance program on a quarterly basis,
c.must be on-site in the sleep facility on a regular basis and not less than 8 hours monthly,
d.cannot serve as the board certified sleep specialist of more than three(3)sleep facilities,regardless of their accreditation status.If an individual is the designated board certified sleep specialist of more than three facilities,the total number of beds shall not exceed 16 beds.
Standard
B-6-Board Certified Sleep Specialist Continuing Education(MANDATORY)
The board certified sleep specialist must participate in at least 10 credits per year averaged over 3 years of AMA PRA Category 1 CME(for a physician),CE(for a PhD)in sleep medicine.Compliance with CME/CE requirements must be documented.If a single individual serves as both the designated medical director and board certified sleep specialist,the individual must meet only the CME requirements of the medical director.
The AASM has adopted job descriptions which delineate education,training,and responsibilities of sleep technologists,sleep technicians,and sleep technician trainees.Standards B-7 through B-13 address requirements for sleep technologists,technicians and trainees.All sleep technologists/technicians must be able to perform the duties listed in the AASM approved job descriptions.These standards do not differentiate between the various job descriptions or titles that sleep facilities may use for the employment of sleep technicians and technologists.For example,a sleep technologist or technician whose primary responsibility is to score a sleep study is subject to the same accreditation standards as a sleep technologist or technician whose primary responsibility is sleep test monitoring.Specifically,CPR certification is required for all technical staff members,regardless of their duties.
Standard
B-7-Sleep Technicians and Technologists
AASM accredited sleep facilities must maintain appropriately trained,supervised,and,where required by state law,licensed sleep technologists.Technologist staffing must be adequate to address the workload of the sleep facility and assure the safety of patients.The AASM recommends a patient to technologist ratio of 2∶1 under usual circumstances for attended polysomnography.
Standard
B-8-Registered Sleep Technologist
A minimum of one sleep technologist must be registered in sleep technology or accepted to sit for the registry examination through one of the following organizations:
a.The American Board of Sleep Medicine(ABSM)
b.The National Board of Respiratory Care(NBRC)
c.The Board of Registered Polysomnographic Technologists(BRPT)
d.An equivalent examination accepted by AASM
The individual(s)fulfilling this standard must be present at the facility a minimum total of 30 hours a week.If the facility is open fewer than 40 hours per week,the registered technologist must be present at the facility 75% of the time.
Note:An accredited facility that loses its sole registered technologist will have 120 days to fulfill this standard.
Standard
B-9-Non-registered Sleep Technologist
All technologists and technicians conducting sleep testing who are not registered by the ABSM,NBRC,BRPT,or other accepted certification body:
a.must be enrolled in or have completed the A-STEP Online Self Study Modules,
b.or must be enrolled in or have completed a CAAHEP accredited sleep technology training program,or electroneurodiagnostic(END)program,or respiratory therapy add-on track for sleep technology.
Standard
B-10-Sleep Technician and Technologist Continuing Education
The sleep facility′s technical staff must each participate in an average of 10 hours per year of AMA PRA Category 1 CME or CEC sleep-related educational activities over a three year period.This must be documented for each technical staff member.Education sessions conducted at the facility are acceptable for fulfilling this standard provided the session has defined educational objective(s)and attendance is documented by a roster signed by the sleep facility′s medical director.
Standard
B-11-Sleep Technician and Technologist Additional Certification
Each sleep technician and technologist must have valid cardiopulmonary resuscitation.Valid certification in cardiopulmonary resuscitation includes skills training.
C.Patient Policies
Standard
C-1-Patient Acceptance
The sleep facility′s Policy and Procedures Manual must address patient acceptance policies.Written policies for patient acceptance must include:
a.age limitations,
b.a mechanism for acceptance,
c.criteria for exclusion,
d.information required from a physician prior to all sleep testing in the sleep facility.
The AASM recognizes that concern for patient safety,clinical judgment,or other appropriate reasons may limit a sleep facility from accepting all patients.
Standard
C-2-Direct Referral(MANDATORY)
Directly referred patients cannot exceed 80% of the patients tested by the sleep facility.A direct referral is defined as,all patients not seen in consultation by a sleep facility staff physician prior to or within three months following an in-laboratory sleep study originally ordered by the patient′s referring physician.
Exceeding the direct referral threshold in any single calendar year is sufficient for denial of reaccreditation.
Standard
C-3-Record Review of Direct Referrals
For patients directly referred,the medical director or a designated sleep facility staff physician or the designated board certified sleep specialist must review the information provided for each patient and determine if the proposed evaluation conforms to established AASM Practice Parameters,or,if not,whether the evaluation is indicated for other reasons.Evidence of compliance with this standard must be included in the medical chart.
Standard
C-4-Practice Parameter Requirements
The clinical evaluation of patients accepted for sleep testing conducted in the sleep facility must comply with the AASM “Practice Parameters for the Indications for Polysomnography and Related Procedures”.Evidence of compliance with this standard must be included in the medical chart.
D.Facility and Equipment
Standard
D-1-Permanent Address
AASM accredited sleep facilities must have a permanent,physical address recognized by the United States Postal Service; such address may not include a P.O.Box.This standard is not intended to address the issue of mail delivery,rather identification and location of the sleep facility for patient access and convenience.
Standard
D-2-Phone Line
The program must have a separate phone line(s)/direct dial access into the sleep facility.
Standard
D-3-Signage
There must be appropriate signage identifying the program as “sleep facility.”
Standard
D-4-Stationery
AASM accredited sleep facilities must have stationery identifying the sleep facility and,at a minimum,include the sleep facility address and phone number.For hospital-based sleep facilities this standard will be met provided the sleep facility is located in the building carrying the primary address listed on the hospital′s stationery.
Standard
D-5-Use of Space
Accreditation is granted to a single sleep facility,generally defined by a physical space used primarily for conducting sleep testing.All of the elements required to conduct sleep tests must be available within the defined testing space.The administrative office(s)and/or staff clinician office(s)of the sleep facility may be separate from the laboratory testing site.In such circumstances,the administrative and/or staff clinician office(s)must also meet the AASM Standards for Accreditation,as they function as a part of the broader sleep disorders center.
In circumstances of mixed use,defined as testing rooms being used for other medical testing or examination during non-sleep testing(daytime)hours,the testing room(s)must meet all of the space and equipment standards of a single use sleep testing room.
Standard
D-6-Testing Bedrooms-Physical Characteristics
All testing bedrooms must be single occupancy,private and comfortable,have hard walls,floor-to-ceiling,and a privacy door that opens directly to a corridor or common use area such that the patient can access the testing bedroom without having to pass through another testing bedroom.
Standard
D-7-Testing Bedrooms & Emergency Care
Patient testing bedrooms must not have any impediments to the delivery of emergency care.The patient testing rooms:
a.must be of sufficient size to accommodate emergency personnel access with a minimum of 24 inches of available clear space on 3 sides of the bed,
b.must include a testing bed with a mattress not smaller than a standard hospital bed.
Standard
D-8-Bathroom Facilities
The sleep facility must have clean bathrooms with a minimum ratio of one bathroom for every three testing rooms; these bathrooms must each contain a toilet and a sink.Each bathroom will have a working privacy door.Sole access to a shared bathroom shall not be through a testing bedroom.
Standard
D-9-Handicap Testing Bedroom and Bathroom
At least one testing bedroom and bathroom must be handicap accessible as defined either by local building regulations or section 6.3 and 6.4 of the American with Disabilities Act(http://www.access-board.gov/adaag/html/adaag.htm).
Standard
D-10-Control Room
The dimensions of the control room must not be less than 40 square feet total or 20 square feet per testing bedroom,whichever is larger.
Standard
D-11-Communication
The facility must maintain a two-way communication system between the patient bedroom and the control room and/or sleep facility personnel.
Standard
D-12-Video Recording
Each testing bedroom in the facility must have a mechanism for visual monitoring and video recording of patients during testing.Time delayed photographs will not be considered compliant with this standard.
Standard
D-13-Polygraphic Equipment
The facility must maintain polygraphic equipment capable of recording and storing physiologic parameters using sensors,and recommended or alternative derivations as described in The AASM Manual for the Scoring of Sleep and Associated Events:Rules,Terminology,and Technical Specifications.The equipment must meet all of the “RECOMMENDED” minimal technical and digital specifications described in section III of the manual.
Standard
D-14-PAP Therapy
The facility must maintain equipment for the delivery of positive airway pressure therapy for sleep apnea,including remote control of the device(pressure output,device mode).
E.Policies and Procedures
The sleep facility must maintain written protocols,in paper or electronic form,for all testing procedures conducted in the facility.There are additional standards in sections F and I that are required to be included in the Policy and Procedures Manual.
Standard
E-1-Policy and Procedures Manual
AASM accredited sleep facilities must maintain a Policy and Procedures Manual that is easily accessible,in paper form or electronically,from the control room.The manual must contain all policies,procedures,protocols specific to the sleep facility,and all current AASM Practice Parameters and Clinical Guidelines.
Standard
E-2-Protocols:PSG,MSLT,MWT,and PAP Titration
The sleep facility must maintain written,paper or electronic format,protocols for comprehensive polysomnography,multiple sleep latency test,maintenance of wakefulness test,and titration of positive pressure therapy.
Sleep facilities are encouraged to use “Clinical Guidelines for the Manual Titration of Positive Airway Pressure in Patients with Obstructive Sleep Apnea” in constructing PAP titration protocols.
Standard
E-3-Other Protocols
AASM accredited sleep facilities that conduct esophageal pressure monitoring and actigraphy must maintain protocols for these procedures.
AASM accredited sleep facilities that test sleep patients under age 13 years must maintain population specific protocols in the Policy and Procedures Manual for comprehensive polysomnography,titration of positive pressure therapy and capnography.
Standard
E-4-Equipment Maintenance
A written plan for monitoring of all patient-related equipment for electrical and mechanical safety is required.The written plan must include specific instructions regarding documentation of compliance.The plan must address:monthly visual inspection of equipment by staff for apparent defects; adhering to manufacturer′s recommendations for monitoring and maintenance of recording equipment; and electrical safety testing by a certified electrician or biomedical engineer to include at least annual testing for ground fault(resistance should be less than 0.5 ohms from chassis to ground)and chassis leakage current(amperage should be less than 100 microamps from equipment that will be in direct contact with the patient,and less than 300 microamps from equipment that will not be in direct contact with the patient,including the recording PCs).
F.Data Acquisition,Scoring and Reporting
Standard
F-1-Signals and Equipment
The signals collected and the equipment used for comprehensive polysomnography must be in compliance with The AASM Manual for the Scoring of Sleep and Associated Events:Rules,Terminology,and Technical Specifications.
Standard
F-2-PSG Reports
Reports of polysomnography must include all the “RECOMMENDED” parameters from section II of The AASM Manual for the Scoring of Sleep and Associated Events:Rules,Terminology,and Technical Specifications.
Standard
F-3-Conducting MSLT and MWT
The multiple sleep latency test and maintenance of wakefulness test must be conducted using the protocol described in Box 1 and Box 2 of the “Standards of Practice Committee of the American Academy of Sleep Medicine:Practice Parameters for Clinical Use of the Multiple Sleep Latency Test and the Maintenance of Wakefulness Test.”
Standard
F-4-PSG Scoring
Each epoch of each polysomnogram must be scored for sleep staging,arousals,respiratory events and limb movement in accordance with The AASM Manual for the Scoring of Sleep and Associated Events:Rules,Terminology,and Technical Specifications.
Standard
F-5-Computer-assisted Scoring
If used,computer-assisted scoring of polysomnography must be reviewed and edited for accuracy.
Standard
F-6-Inter-scorer Reliability
Inter-scorer reliability must be determined between each scorer and a reference sleep specialist as defined in standard B-4 or a corporate appointed board certified sleep specialist; such individual must be an employee of the corporation and serves as the reference sleep specialist.Inter-scorer reliability assessment must be conducted for each sleep facility.In cases where a corporate sleep specialist serves as the reference standard for inter-scorer reliability,the sleep facility′s designated board certified sleep specialist must attest in writing that he/she has reviewed the results of the inter-scorer reliability assessment,and will take corrective action when results fall below the sleep facility′s level of acceptable agreement as defined in its quality assurance program.
Use of the AASM Inter-Scorer Reliability program fulfills the requirements of this standard.
Standard
F-7-Conducting Inter-scorer Reliability Assessment
For comprehensive polysomnography,the following parameters must be compared:sleep staging epoch-by-epoch agreement,respiratory events,leg movements and arousals.Sleep technologists must be blinded to the scoring of the sleep facility designated reference specialist and to all other scoring technicians.Comparisons between each scorer and the facility′s designated reference sleep specialist must be made on 200 consecutive 30-second epochs in each of three polysomnograms per quarter,for a total of 12 polysomnograms per year.
Use of the AASM Inter-Scorer Reliability program fulfills the requirements of this standard.
Standard
F-8-SRBD Event Comparison
Sleep related breathing event comparisons for laboratory polysomnography must at minimum include analysis by total number of events and by the following event types:Obstructive apnea,central apnea and hypopnea.If the sleep facility reports respiratory effort related arousals,this event must be included in the comparison.
Use of the AASM Inter-Scorer Reliability program fulfills the requirements of this standard.
Standard
F-9-Inter-scorer Reliability Agreement
Comprehensive polysomnography studies must report agreement between scorer and the facility′s designated reference sleep specialist as percent concordance defined as the quotient of the total number of epochs of agreement for a given parameter and the total number of epochs in the analysis sample multiplied by 100.
Use of the AASM Inter-Scorer Reliability program fulfills the requirements of this standard.
Standard
F-10-Review of Raw Data
The physician or individual meeting B-4 interpreting a polysomnogram,multiple sleep latency test or maintenance of wakefulness test must conduct an epoch by epoch review the entire raw data recording for every study interpreted.Signed attestation of this review must be kept either in the patient record in the form of signature on the report of the test or in a separate journal.
Standard
F-11-Review of Raw Data Continued
The board certified sleep specialist must conduct an epoch-by-epoch review of the raw data of each record to assure that the quality of the recording and the scoring of sleep and associated events is sufficient to allow for interpretation.Specifically,this includes over-reading the raw data and interpretations of staff physicians who do not meet Standard B-4.
Signed attestation of this review must be kept either in the patient record in the form of signature on the report of the test or in a separate journal.
G.Patient Evaluation and Care
Standard
G-1-Patient Management
In accordance with the sleep facility′s patient acceptance policies,its physicians must demonstrate the capability and experience diagnosing and managing the full range of sleep disorders.This includes availability(within the sleep facility or by referral)of recognized and effective treatments for these disorders.Appropriate follow-up for patients who require continued management must be offered and available.
Standard
G-2-Documenting Patient Evaluation/Management
The sleep facility medical staff must document ongoing evaluation and management of patients with sleep disorders.
H.Patient Records
Standard
H-1-Medical Charts
AASM accredited sleep facilities must maintain appropriate medical charts for each patient evaluated by the sleep facility medical staff and for directly referred patients.
Standard
H-2-Medical Charts Continued
Medical charts of patients seen by sleep facility medical staff must document all patient interactions in the sleep facility,including initial evaluation,testing(if any),diagnosis,treatment,and follow-up.
Standard
H-3-Medical Charts Continued
Prior to testing,all patient medical charts must include:history and physical examination or patient questionnaires,or other screening assessment.Written indication that either a physician or the designated sleep specialist has reviewed and approved the proposed evaluation must be noted in the chart.
Standard
H-4-PAP Assessment
Patients prescribed positive airway pressure treatment by the sleep facility medical staff must be offered a follow-up positive airway pressure assessment within 12 weeks of treatment initiation.Positive airway pressure assessment must minimally include a measurement of treatment use and clinical response to the therapy as determined by:
a.direct patient inquiry,
b.office encounter with sleep facility technical or medical staff,
c.the referring physician,
d.questionnaires,
e.telephone inquiry to the referring physician or the patient,
f.an informatic system capable of obtaining positive airway pressure use and a metric of clinical response.
The patient medical chart must contain documentation of the assessment as described above,or written evidence of follow-up attempts to obtain the positive airway pressure treatment assessment.
Standard
H-5-Database
The sleep facility must maintain a cumulative database of the final diagnosis,using the most recent diagnostic and coding manual of the AASM,and procedures performed for each patient evaluated.This database must include cases seen by the facility′s medical staff that did not require polysomnography.
I.Emergency Procedures
Standard
I-1-Emergency Plan
AASM accredited sleep facilities must have a written emergency plan accessible in paper or electronic format that delineates the following:
a.mechanisms and specific details for contacting emergency personnel,
b.the sleep facility personnel to be contacted in an emergency,
c.outline the specific responsibilities of the technical staff.
At a minimum,emergency policies must include procedures for the following:
a.Cardiac emergencies
b.Neurologic emergencies,particularly seizures
c.Psychiatric emergencies,particularly suicidal ideation
d.Environmental emergencies such as fire,weather,belligerent patients,and bomb threats
Standard
I-2-Emergency Equipment
The sleep facility must have accessible all appropriate emergency equipment to address all possible emergencies outlined in the plan.
J.Quality Assurance
Standard
J-1-QA Program
AASM accredited sleep facilities must have a quality assurance program that addresses inter-scorer reliability as outlined in standard F and at least three other quality assurance indicators.
Standard
J-2-Reporting QA Program
All quality assurance metrics must be reported and reviewed by the sleep facility′s medical director or the designated board certified sleep specialist a minimum of once each quarter.The reviewer of the report must sign and date the report; a copy of the signed report must be kept on file for a minimum of one year.

